Mus spretus Lataste, 1883
|
publication ID |
https://doi.org/10.1644/840.1 |
|
persistent identifier |
https://treatment.plazi.org/id/BE7F87F2-1A67-FFB0-DA39-F565446FF908 |
|
treatment provided by |
Carolina |
|
scientific name |
Mus spretus Lataste, 1883 |
| status |
|
Mus spretus Lataste, 1883 View in CoL
Western Mediterranean Mouse
Mus spretus Lataste, 1883:17 View in CoL . Type locality ‘‘l’oued Magra, entre M’Sila et Barika, au nord du chott du Hodna, Hauts-Plateaux,’’ Algeria.
Mus spicilegus hispanicus Miller, 1909:421 View in CoL . Type locality ‘‘ Silos , Burgos, Spain.’’
Mus spicilegus lusitanicus Miller, 1909:422 View in CoL . Type locality ‘‘ Cintra , Portugal.’’
Mus spicilegus mogrebinus Cabrera, 1911:555 View in CoL . Type locality ‘‘Tagu¨ idert, provincia de Haha´,’’ Morocco.
Mus spicilegus caoccii Krausse, 1919:95 View in CoL . Type locality ‘‘ Sardegna,’’ Italy. Toschi (1965:230) determined that this is a synonym of Mus musculus Linnaeus, 1758 View in CoL .
Mus spicilegus lynesi Cabrera, 1923:430 View in CoL . Type locality, ‘‘ Tazarot , kabila de Beni-Aro´s, Yebala,’’ Morocco.
Mus spicilegus rifensis Cabrera, 1923:431 View in CoL . Type locality ‘‘alrededores de Melilla, Rif oriental,’’ Morocco.
Mus musculus spretus: Schwarz and Schwarz, 1943:69 View in CoL . Name combination.
Mus hispanicus: Sage, 1978:550 View in CoL . Name combination.
Mus spretus hispanicus: Marshall and Sage, 1981:24 View in CoL . Name combination.
Mus spicilegus spretus: Marshall, 1986:17 View in CoL . Name combination.
CONTEXT AND CONTENT. Order Rodentia View in CoL , suborder Myomorpha View in CoL , superfamily Muroidea View in CoL , family Muridae View in CoL , subfamily Murinae View in CoL . Two subspecies are recognized ( Palomo et al. 1985):
M. s. parvus Alcover, Gosa`lbez, and Orsini, 1985:6. Type locality ‘‘St Rafel (Eivissa)’’, Balearic Islands, Spain. Preoccupied by the same name proposed by Bechstein (1800), now allocated as a junior synonym of Apodemus sylvaticus ; current usage of the primary homonyms parvus Bechstein, 1800, and parvus Alcover, Gosa`lbez, and Orsini, 1985, may be maintained because they are associated with genera considered to be distinct since 1899 (Article 23.9.5—International Commission on Zoological Nomenclature 1999).
M. s. spretus Lataste, 1883 . See above. Includes M. spicilegus hispanicus , M. spicilegus lusitanicus , M. spicilegus mogrebinus , M. spicilegus lynesi , and M. spicilegus rifensis .
NOMENCLATURAL NOTES. For many years, the attempt to understand the number of valid species in the genus Mus has proven difficult. Schwarz and Schwarz (1943) tried to simplify the taxonomy, condensing more than 130 known scientific names of wild and commensal stocks of Mus into a single species: Mus musculus . They recognized 15 subspecies, including M. m. spretus , and proposed the evolutionary scenario that the commensal subspecies of Mus underwent multiple evolutionary events from aboriginal subspecies. In the wild stock, 4 different types of Mus musculus were distinguished: M. m. wagneri, M. m. spicilegus , M. m. manchu, and M. m. spretus , the last being found in the wild state only. The other 3 developed into commensals connected with human habitation and cultivation and followed human migration, but under suitable conditions could become feral and return to more or less wild habits. This revision was followed with minor changes by Ellerman and Morrison-Scott (1951). All the wild forms are of medium size and the tail is always shorter than the length of the head and body (Schwarz and Schwarz 1943).
At the end of the 1970s, the advent of biochemical genetic methods, such as enzyme and protein electrophoresis, shed light on this problem, confirming M. spretus as a valid species ( Britton et al. 1976; Britton and Thaler 1978; Marshall and Sage 1981; Thaler et al. 1981). Currently, there is agreement that in Europe there are 3 taxa of aboriginal ‘‘outdoor’’ mice ( M. spretus , M. spicilegus , and M. macedonicus ) that are specifically different from commensal mice. However, there is no consensus on the best way to classify the commensal forms. One approach is to give species status to the major genetic lineages ( M. domesticus and M. musculus ), whereas the alternate approach is to classify them as members of the species M. musculus : M. m. musculus and M. m. domesticus (e.g., Auffray et al. 1990a; Boursot et al. 1993; Gerasimov et al. 1990; Machola´ n 1996a; Marshall 1998; Musser and Carleton 1993, 2005; Sage et al. 1993). In any case, M. spretus seems to form a sister group to all other West Palearctic mice (Gue´net and Bonhomme 2003; Lundrigan et al. 2002; Lundrigan and Tucker 1994; Machola´ n 2006; Prager et al. 1996, 1998; Tucker et al. 2005).
DIAGNOSIS
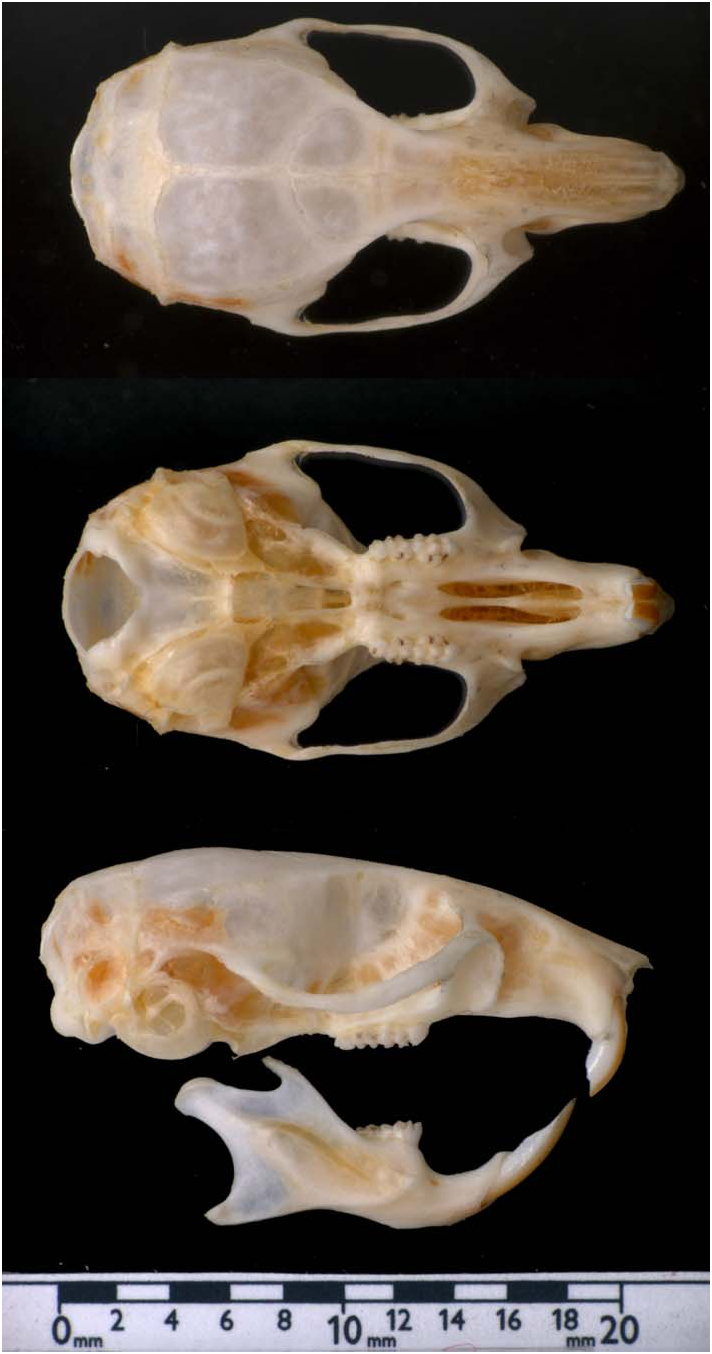
Mus spretus is a small, wild (noncommensal) murine with a rounded muzzle and small eyes and ears ( Fig. 1 View Fig ). Fur on the ventral surface is white or buff with a slate-gray base and the dorsum is ochraceous brown, with a clean lateral line separating the 2 areas. Throughout almost its entire range, M. spretus is sympatric (but not syntopic) with M. musculus and some criteria can be used to distinguish between them. In M. spretus length of tail is always shorter than length of head and body, the ratio between the widths of the upper ramus of the zygomatic process of the maxilla and the zygomatic arch is always greater than 0.8, M1 has 4 very distinctive anterior tubercles, and there are only 4 intermolar palatal rugae ( Fig. 2 View Fig ; Darviche and Orsini 1982; Machola´n 1996b; Orsini et al. 2001). Juvenile forms of Apodemus sylvaticus have similar coloration to adult M. spretus , but both forms can be easily differentiated by eye and pinna size and length of hind foot.
GENERAL CHARACTERS
External and skull measurements (average and range, mm) of Mus spretus adults (age.12 weeks) from Ma´ laga, southern Spain ( n 5 90— Palomo 1986), were: length of head and body, 85.80 (79.0–93.0); length of tail, 65.34 (59.0–73.0); length of hind foot, 16.44 (15.0–21.0); mass, 16.44 g (15.0– 19.0); condylobasal length, 20.20 (19.1–21.9); cranium height, 6.74 (6.4–7.1); and length of mandible, 11.92 (10.9– 13.0). There are no morphological or karyological differences in populations in the Iberian Peninsula, southern France, and Morocco (Britton-Davidian et al. 1978; Palomo et al. 1983, 1985). M. s. parvus (Ibiza, Spain) is significantly smaller, and has a paler coloration than the nominal subspecies ( Alcover et al. 1985).
DISTRIBUTION
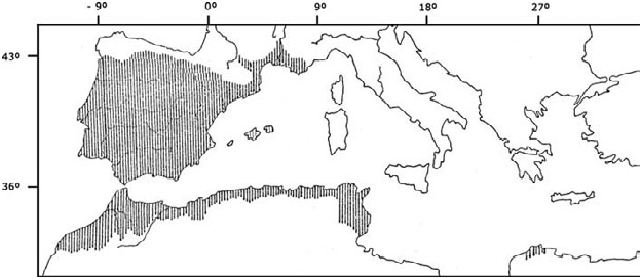
In Europe, Mus spretus occupies southern France ( Fig. 3 View Fig ), from the Fre´jus region to the Toulouse area, and along the Rhone Valley up to Valence, but is absent from Corsica (Khammes and Aulagnier 2003). M. spretus is found throughout practically the entire Iberian Peninsula, including the whole of continental Portugal ( Mathias 1999). In Spain, its distribution correlates with the Mediterranean climate, but it does not appear in the northern fringe of the country (i.e., from Galicia to the Pyrenees). It occurs in the Balearic Islands of Mallorca, Menorca, and Ibiza, but is absent from the Canary Islands ( Palomo 2007). In North Africa, it occupies the Maghreb area, from Morocco up to Algeria and Tunisia. In Morocco, the Atlas Mountains and the Sahara Desert limit its distribution toward the south (Orsini 1982). In Algeria, M. spretus appears in the Mediterranean belt and to the south it reaches the northern border of the Aures Mountains (Kowalski and Rzebik- Kowalska 1991; Thomas 1913). In Tunisia, it appears in the Mediterranean region ( Gharaibeh 1997) and in the south it
FOSSIL RECORD
The North African origin of Mus spretus , suggested by Schwarz and Schwarz (1943), is supported by the absence of mice in Europe before the Neolithic, and is confirmed by morphologic evolution ( Engels 1983) and mitochondrial DNA polymorphism analysis ( Boursot et al. 1985). The oldest fossils exhibiting attributes of M. spretus in North Africa occur in middle Pleistocene strata in Morocco (Amani and Geraads 1993; Thaler 1986) and Tunisia (Mein and Pickford 1992). In the Doukkala II site in Morocco, Darviche et al. (2006) reported 10,000- to 40,000-year-old fossils of M. spretus , but they did not find evidence that M. musculus occurred at that site at that time. M. spretus probably inhabited North Africa before M. musculus arrived in the area ( Michaux et al. 1990). The present distribution of M. spretus could be explained by ancestral colonization throughout North Africa, at a time when the Sahara offered a hospitable environment. After desertification, an isolated population could have remained as a refuge in the Cyrenaican Plateau ( Libya). M. spretus differentiated in North Africa before spreading to southwestern Europe via Neolithic navigators and its close relationship with agricultural environments ( Auffray et al. 1990b; Dobson 1998; Dobson and Wright 2000; Gippoliti and Amori 2006).
seems to prefer oases ( Bernard 1969; Bernard and Gannoun 1965). An isolated population exists on the Libyan Cyrenaican Plateau ( Marshall 1998; Orsini 1982) but it was not cited by Hufnagl (1972) or Gharaibeh (1997). According to Marshall and Sage (1981), M. spretus also occurs in Daghalia ( Egypt) but this has not been completely established.
FORM AND FUNCTION
Dental formula is 1/1, 0/0, 0/0, 3/3, total 16 ( Fig. 2 View Fig ). The coat of Mus spretus is composed of 2 kinds of hair, the overhair and underhair. There are 3 types of overhair (together making up about 20% of the total amount of hair): the guard hair (or monotriches), the awls (without constrictions), and the auchenes (with a single constriction). The underhair (zigzags) is shorter and usually has 3 flat constrictions, constituting the basic predominant hair. Because of the small size of the underhairs and their location, they play a minor role in determining the overall color of the animal ( Palomo 1986). The hair cortex is surrounded by a thin layer of overlapping scales, the cuticle, which in M. spretus may appear differently depending on the type and parts of hair studied: petals, regular mosaic, or transitional (Palomo 1988).
First (juvenile) pelage is acquired in the nest, 17–21 days after birth. The first 2 postnatal molts are complete and show regular topography, starting from the ventral surface. Such molts are called juvenile and intermediate and occur, within margins of variability, between 38 and 52 and 64 and 81 days, respectively. Superposition between both molting processes has not been observed (Espan˜ a et al. 1985; Palomo and Vargas 1988a). Once the 3rd (adult) coat is acquired, the animals undergo new molting processes in which temporary and topographic manifestations have an irregular pattern. These molts affect either the whole coat or restricted areas, independently of season, age, or physiological condition (Palomo and Vargas 1988b). Hair density decreases in successive coats, but hair length and breadth increase. The result of both sets of changes is that the 2nd (intermediate) coat has greater coverage, which may be related to fur requirements when the animals leave the nest ( Vargas et al. 1987b).
Data on metabolic level and energy requirements of M. spretus have been compiled by Khammes and Aulagnier (2003). In severe dry conditions individuals reduce their body fluid loss to balance their water requirements ( Sicard et al. 1985). The temperature for optimal metabolic conditions for M. spretus is 19.94 u C, and during experiments the animals spend about 67% of their time at 8–14 u C ( Metcheva et al. 1994a). In the thermoneutral zone (assumed to reflect the basal metabolic rate) of 27–28 u C, the lowest values of resting metabolic rates were 3.3 ml O 2 g 21 h 21 in males and 2.7 ml O 2 g 21 h 21 in females. At 0 u C, oxygen consumption was higher (7 ml O 2 g 21 h 21), without differences between sexes ( Metcheva et al. 1994b). According to Go´recki et al. (1990), the thermoneutral zone is 32–33 u C and usual rectal temperature is close to 34 u C. Mira and Mathias (1994) analyzed seasonal effects on blood variables and blood plasma proteins throughout the year.
ONTOGENY AND REPRODUCTION
Ontogeny.— Development is very similar to that described for Mus musculus ( Berry et al. 2008; Espan˜ a et al. 1985; Silver 2005; Theiler 1989). Gestation is about 19–20 days. Newborns are naked, blind, and bright pink (in the 1st few days of life, when suckling, milk can be seen in the stomach through their translucent bodies). The young are born without teeth, the toes are not separated, and the eyes, ears, and vagina are closed. Hair begins to appear at 2–4 days, ears open at 3–5 days, skin pigmentation becomes visible at 5–7 days, and the eyes begin to open at 12–14 days. As soon as the eyes and ears are fully functional, the pups begin to eat solid food. However, nursing can continue to at least the end of the 3rd week and sometimes a week or more longer. By the end of the 3rd week of life, the young resemble adults in every aspect other than size and sexual differentiation. When they leave the nest, at 17–21 days, the fur is fully grown, and the incisor and 3rd molar have fully erupted. Finally, the vagina opens at 24–28 days (e.g., Berry et al. 2008; Espan˜ a et al. 1985; Silver 2005; Theiler 1989).
Postnatal growth in M. spretus was studied by Prudeˆncio and Ramalhinho (1998), who observed that adult size is reached at 8–9 weeks. Even though spermatogenesis and 1st ovulations and pregnancies may occur within 4–5 weeks of birth (Dura´ n and Sans-Coma 1986), females reach sexual maturity between 6 and 7 weeks, whereas males mature from 8 weeks onward ( Vargas et al. 1991). However, the season of birth seems to influence when maturity is reached in both sexes.
Reproduction.— Spermatozoa are quite different than those of Mus musculus . The head is scythe-shaped with a terminal hook, which is a prolongation of the acrosome. The middle part of the sperm tail is larger than in M. musculus and the transition to the main part is well marked ( Vargas et al. 1984b).
Reproductive characteristics of M. spretus in the southern Iberian Peninsula were analyzed by Vargas et al. (1991) at monthly intervals over a 6-year period. M. spretus exhibits seasonal reproductive cycles with 2 well-marked phases: a period of sexual inactivity in winter (November– January), with a reduction in the size of the testicles and seminal vesicles, and a period of sexual activity during the remaining months, with 2 phases of maximum activity: April–May and August–September. The interannual variations observed in the duration and intensity of the reproductive cycles appear to be mainly due to environmental conditions, especially temperature. Sexual activity positively correlates with both temperature and length of photoperiod. The average litter size was 5.53 6 1.37 SD ( n 5 193, range: 2–10, mode: 5). There was a correlation between litter size and mass, and size and age class of female. The embryo resorption rate was 1.57%. The population structure varies throughout the year, according to reproductive cycle. During spring, the population is composed of adults, which are responsible of the 1st peak of reproduction. Young animals are the dominant fraction during the summer and autumn, whereas subadults and newborns from the 2nd peak of reproduction form the winter stock (Antu´ nez et al. 1990; Cassaing 1982; Vargas et al. 1984 a, 1986). M. spretus has an average life expectancy of,4 months, with a longevity of 14–15 months, and the animals do not usually survive their 2nd winter ( Cassaing 1982; Cassaing and Croset 1985).
ECOLOGY
Space use.— Mus spretus is not a commensal species although it occasionally occupies abandoned buildings. It inhabits a variety of agroecosystems, including crops, orchards, grasslands, scrubland, or forests, although it prefers open habitats. During the regeneration process following fires in the Mediterranean ecosystem, it is a very frequent species in exposed zones, irregular in scrubland, and very scarce in mature forests ( Fons et al. 1988). In general, M. spretus avoids open woodlands or pathways, preferring grassland sites with tall vegetation where it can create a system of grass tunnels and sites where shrubs, brambles, or dead wood provide additional cover ( Gray et al. 1998). In the Maghreb, it is mainly associated with sparse woody vegetation and a high percentage of bare ground. The highest abundance has been observed in agricultural land and no mice have been found in mature forests, dense maquis and scrubland, and tree plantations ( Khidas et al. 2002). Their low water requirements allow survival where other rodents are eliminated ( Orsini et al. 1982).
In southern Spain, M. spretus is mainly a nocturnal species, except in winter, when it also is diurnal. The activity curves have a peak in summer, are bimodal in spring and autumn (reaching a maximum immediately after dusk, and another before dawn), and are multiphasic during winter. Total length of the daily activity period correlates with the length of the night ( Vargas et al. 1987a). These results were confirmed by radiotracking 10 adult males captured in Portugal during spring ( Gray et al. 1998).
Diet.— The diet of Mus spretus basically reflects the availability of resources more than the preferences of the mice. Analyses of stomach contents show that M. spretus feeds mainly on grass seeds, cultivated or uncultivated plants, and fruit. Insects, mainly in the form of larvae, seem to constitute a substantial part of the diet ( Khidas et al. 2002; Orsini 1982; Palomo 1990, 2007). Damage to crops has been reported in Tunisia (Bernard and Gannoun 1965). Under laboratory conditions M. spretus consumes 1.5 times less water than feral M. musculus and 2 times less than laboratory mice ( Sicard et al. 1985). Orsini (1982) reported a daily average consumption of 1.35 g of dry matter per hour.
Diseases and parasites.— Ectoparasites are usually associated with Mus spretus . The presence of 8 different species of fleas (Siphonaptera) has been recorded in populations in France (Beaucournu and Launay 1990). A high prevalence of both lice (Anoplura) and mites (Acari) was reported in specimens from northern Spain ( Carrió et al. 1997). Endoparasites (helminths sensu lato) including nematode and cestode species also have been reported ( Behnke et al. 1993). The behavior of picking up and carrying feces in the mouth is particularly favorable to infestation via parasite eggs in the feces (Hurst and Smith 1995). Protozoa infections include Cryptosporidium ( Torres et al. 2000) and Rickettsia (Lledo´ et al. 2003). M. spretus is considered an important reservoir of arbovirus transmitted by hematophagous vectors ( Chastel et al. 1984).
Interspecific interactions.— Mus spretus constitutes an important fraction of the diet of more than a dozen predators, including carnivores, owls, and snakes ( Palomo 2007). It forms between 6% and 95% of the diet of these predators and there are striking fluctuations depending on predator, habitat, and season (Khammes and Aulagnier 2003). Throughout the entire Mediterranean area the abundance of M. spretus in the diet of the barn owl ( Tyto alba ) is constant (e.g., Aulagnier et al. 1999; Barbosa et al. 1992; Brunet-Lecomte and Delibes 1984; Cheylan 1976; Herrera 1974; Herrera and Jaksic 1980; Orsini 1982; Saint- Girons and Thouy 1978; Temme 2002; Torre et al. 2004; Vargas et al. 1988). Vargas et al. (1988) analyzed the diet of the barn owl in the Iberian Peninsula and concluded that the proportion of M. spretus captured by these birds increased in parallel with the overall number of small mammals included in their diet, showing strong dependence on a bioclimatic or latitudinal gradient. Moreno and Barbosa (1992) confirmed these results in central Spain, noting that bioclimatic and biogeographical factors, such as altitude, latitude, and longitude, have a greater relevance than habitat factors, such as vegetation mosaics. The barn owl is a relatively opportunistic predator that consumes the more abundant species of local prey (including birds), but in the case of M. spretus it seems to select adult mice during spring, when juveniles are the most abundant fraction although difficult to access. The hunting success of the barn owl on M. spretus has been estimated to be close to 16% ( Vargas et al. 1988).
Miscellaneous.— Recently, Mus spretus has been established as a suitable bioindicator of genetic risk induced by environmental pollution in natural areas ( Ieradi et al. 1998; Nunes et al. 2001b) or those affected by environmental disasters ( Festa et al. 2003; Ruíz-Laguna et al. 2001; Tanzarella et al. 2001). The adverse effects of heavy metals (mostly chromium, manganese, iron, copper, zinc, and selenium) were appraised by Nunes et al. (2001a), who compared the fluctuating asymmetry of dental (molar) characteristics and confirmed that tooth size was reduced in the contaminated area, and that developmental instability increased in relation to stress, even when contamination levels were low.
BEHAVIOR
Male and female adults are significantly more often captured in the same place, suggesting a close relationship with space. The sedentary nature of older males, at the start of reproduction, suggests territorial organization ( Cassaing 1984; Cassaing and Croset 1985). Nevertheless, tests performed on captive animals show that Mus spretus does not fiercely attempt to exclude others from its territory, but suggest that mice establish a dominance relationship using stylized submission postures and are relatively tolerant ( Hurst et al. 1996, 1997). This behavior is quite different from that of resident M. musculus , which are highly intolerant and aggressive toward unfamiliar intruders, which readily take flight to avoid resident attacks (Gray and Hurst 1997). M. spretus appears to use odor cues to identify occupied areas and then competes for dominance over them ( Hurst et al. 1997). Both trapping and radiotelemetry studies show that the ranges of adult males are mutually exclusive, whereas adult females have similarly dispersed areas but that overlap with those of males (Cassaing and Croset 1985; Hurst et al. 1996, 1997). Lactating females are particularly aggressive ( Hurst et al. 1996). Gray et al. (1998) found that each male territory overlapped the territory of at least 2 females. The mean range of males was 343 m 2 6 95 SD and residents cover less than one-third of their total range over 24 h. Most fixes (70%) were located in 1–4 core areas, which represented only a very small proportion of each range (6.9%). Although the complete defense of a large complex range is likely to be impracticable, the defense of core areas seems much more feasible ( Gray et al. 1998).
Daily displacements have been analyzed using capture– recapture methods (Cassaing and Croset 1985) or marking animals with fluorescent powders ( Palomo 1990). The data obtained vary depending on the habitat, sex, age, and season, and the average ranged from 27.8 m to 112.0 m.
Mus spretus is sympatric with M. musculus over nearly the entire distribution range, but they usually do not share habitats, thus behaving like allopatric species. M. spretus is not a commensal species, whereas M. musculus lives inside or close to human constructions. In some areas of southern France, M. musculus inhabits wet habitats (riparian forest or irrigated land cultures) from which M. spretus is absent. Laboratory experiments and observations in open-air enclosures suggest that the presence of M. musculus in this optimal habitat can affect reproduction of M. spretus ( Orsini et al. 1982) . Cassaing (1984) analyzed the interactions between both species and verified that in captivity males of M. spretus dominate due to being highly aggressive. In the wild, however, examination of ecological data shows that this dominance is not sufficient to eliminate M. musculus from the most favorable biotopes. In shared areas, M. musculus numerically dominated M. spretus , which failed to reproduce. Cassaing (1984) suggested that in M. spretus , only breeding males may be aggressive, but because these make up only a small proportion of the population they cannot prevent massive colonization by M. musculus . Thus, females of M. spretus , which were never pregnant and obese at the end of the experiments, suffered from psychophysiologic stress that prevented them from breeding. The result of competition between the 2 types of mice seems to basically depend on habitat quality. Because of their low water requirements, M. spretus only dominates in the driest habitats ( Sicard et al. 1985). Competition with Apodemus sylvaticus is low because this species is not abundant in open habitats, and M. spretus avoids closed forests ( Fons et al. 1988; Khidas et al. 2002; Torre et al. 1996).
Droppings of M. spretus are small and cylindrical, usually 5–6 mm long and 2–2.5 mm thick. They are similar to those of M. musculus but appear to be more moist, probably reflecting a higher proportion of living plant and insect material in their diet rather than dry stored products. M. spretus shows a behavior pattern not reported in other rodents: feces are picked up and carried short distances in the mouth or rolled along the ground with the tip of the snout, often repeatedly. The simplest explanation for the function of fecal manipulation is that it is a hygienic response to remove sticky fresh feces away from pathways and resting sites (Hurst and Smith 1995).
GENETICS
All species in the Mus musculus complex and the closely related wild species, M. spretus , M. spicilegus , and M. macedonicus (included in subgenus Mus ), have the same standard karyotype (diploid number 5 40— Evans 1981; Gue´net and Bonhomme 2003; Silver 2005) composed of 20 pairs of acrocentric chromosomes, including 19 autosomal pairs and the X and Y sex chromosomes individually recognizable by banding techniques (Q- and G-bands— Nesbitt and Francke 1973). Surprisingly, all 19 autosomes and the X chromosome appear to be telocentric, with a centromere at one end and a telomere at the other ( Silver 2005). The relationship between telomere length and aging in M. spretus , and its relation to senescence in humans, were studied by Coviello-McLaughlin and Prowse (1997).
The standard karyotype is not constant in M. musculus and ranges from a diploid number (2n) of 22 to 39. This variation is a result of Robertsonian fusions, which involve the joining together of pairs of acrocentrics at their centromeres to form metacentric chromosomes. Mice captured from the Valle di Poschiavo in southeastern Switzerland have 13 sets of chromosomes (7 metacentric and 6 telocentric— Gropp et al. 1972). These mice were initially classified as belonging to a separate species named Mus poschiavinus . Further studies have led to the discovery of additional nonstandard karyotypes in M. musculus from other regions of Europe as well as South America and northern Africa (Adolph and Klein 1981; Castiglia et al. 2005; Gu¨ ndu¨ z et al. 2001; Nunes et al. 2005; Sadoyan et al. 2003; Said and Britton-Davidian 1991; Wallace 1981). Pialek et al. (2005) report 97 distinct ‘‘populations’’ characterized by various combinations of metacentric chromosomes, primarily in the western forms of mice. Robertsonian fusions like these have not been reported in populations of M. spretus .
Although M. spretus is sympatric with M. musculus , they do not produce hybrids in nature. Nevertheless, under laboratory conditions, individuals breed and produce viable hybrid offspring. This indicates that premating isolating mechanisms probably occur in nature ( Bonhomme et al. 1978). In captivity, the aggressiveness of male M. spretus toward female M. musculus means that mating only takes place between male M. musculus and female M. spretus . Hybrid males are sterile due to absence of spermatozoa (Pelz and Niethammer 1978), but females are fertile. There is a detectable horizontal flow of a unique sequence from chromosome 4 between the 2 species in laboratory strains (Greene-Till et al. 2000). Given the sterility of F 1 males and the known partial genetic incompatibility between the genomes of the 2 species, genetic exchanges are very limited even though they are possible ( Orth et al. 2002). The sterility of the hybrid males (consistent with Haldane’s rule) is a good reason to use inbred strains as a model for biological, genetic, and developmental research (Coviello-McLaughlin and Prowse 1997; Elliott et al. 2001; Gouyon et al. 1993; Gue´net and Bonhomme 2003; Mayer et al. 2000; Zhao et al. 1996). A variety of strains derived from the wild with welldefined taxonomical origins have been established in various laboratories in recent years and a list of the strains and stocks of M. spretus is available in Bonhomme and Gue´net (1996).
Recent taxonomic treatment of the genus Mus ( Marshall 1998; Musser and Carleton 2005) recognizes 4 subgenera: Mus , Pyromys , Coelomys , and Nannomys . Monophyly of the subgenus Mus is supported by a variety of data sets ( Bonhomme et al. 1984; Chevret et al. 2005; Ferris et al. 1983; Sage 1981; She et al. 1990). Within the subgenus Mus, Lundrigan et al. (2002) uncovered 3 major clades: a ‘‘House Mouse clade’’ that includes M. musculus ( M. m. musculus + M. m. domesticus ), M. molossinus , and M. castaneus ; a ‘‘Palearctic clade’’ that includes these taxa plus M. macedonicus , M. spicilegus , and M. spretus ; and an ‘‘Asian clade’’ that includes M. cervicolor , M. cookii , and M. caroli . These data are fully concordant with the classical tree presented by Boursot et al. (1993). The position of M. spretus as basal to the rest of the Palearctic taxa ( Lundrigan et al. 2002) or sister to the M. spicilegus – M. macedonicus clade within the Palearctic clade ( Tucker et al. 2005) is most likely due to the use of parsimony analysis versus maximumlikelihood analysis.
CONSERVATION
According to European Mammal Assessment and International Union for Conservation of Nature and Natural Resources Red List Categories and Criteria (Meinig and Amori 2007), Mus spretus is a common and widespread species within its range, with no major threats. It is classified as Least Concern (LC).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Mus spretus Lataste, 1883
| Palomo, L. Javier, Justo, Enrique R. & Vargas, J. Mario 2009 |
Mus spicilegus spretus :
| MARSHALL, J 1986: 17 |
Mus spretus hispanicus : Marshall and Sage, 1981:24
| SAGE, R 1981: 24 |
Mus hispanicus :
| SAGE, R 1978: 550 |
Mus musculus spretus : Schwarz and Schwarz, 1943:69
| SCHWARZ, W & AND H 1943: 69 |
Mus spicilegus lynesi
| CABRERA, A 1923: 430 |
Mus spicilegus rifensis
| CABRERA, A 1923: 431 |
Mus spicilegus caoccii
| TOSCHI, A 1965: 230 |
| KRAUSSE, A 1919: 95 |
Mus spicilegus mogrebinus
| CABRERA, A 1911: 555 |
Mus spicilegus hispanicus
| MILLER, G 1909: 421 |
Mus spicilegus lusitanicus
| MILLER, G 1909: 422 |
Mus spretus
| LATASTE, F 1883: 17 |