Hyphessobrycon eilyos, Lima & Moreira, 2003
|
publication ID |
https://doi.org/10.1590/S1679-62252003000100003 |
|
DOI |
https://doi.org/10.5281/zenodo.5070224 |
|
persistent identifier |
https://treatment.plazi.org/id/512387A3-FFBF-FFD5-BA87-281FCF2EFADC |
|
treatment provided by |
Carolina |
|
scientific name |
Hyphessobrycon eilyos |
| status |
sp. nov. |
Hyphessobrycon eilyos View in CoL , new species
Holotype. MZUSP 75126 View Materials ( 22.3 mm SL): Brazil, Mato Grosso, município de Alto Araguaia, ribeirão do Sapo, km 464.04 of Ferronorte railroad, 17°31’11”S, 53°15’33”W; C. R. Moreira & F.C. T. Lima, 21 May 2001. GoogleMaps
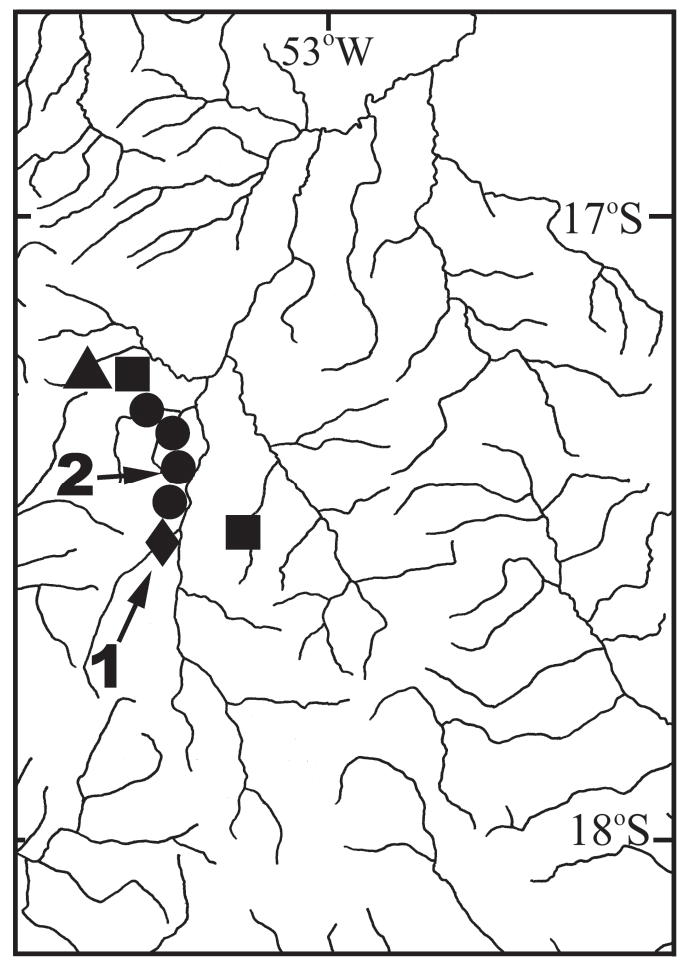
Geographic distribution. Hyphessobrycon langeanii is known from several streams in the upper rio Araguaia basin, states of Mato Grosso and Goiás, Brazil ( Fig. 5 View Fig ) .
Ecological notes. We observed Hyphessobrycon langeanii in schools, generally at midwater, sometimes associated with an unidentified Astyanax species. Habitats occupied by the species ranged from relatively large, deep, clearwater streams partially covered with aquatic vegetation (i.e., ribeirão do Sapo, córrego Gordura) to shallow, small streams with flooded areas (i.e., córrego Mosquito). Hyphessobrycon langeanii was collected syntopically with H. weitzmanorum at córrego Gordura, córrego Boiadeiro, córrego do Sapinho, and córrego do Mosquito, with H. eilyos at córrego do Rancho, and with both species at the ribeirão do Sapo. The occurrence of Hyphessobrycon langeanii in a broad number of sites, even in highly degraded, silted streams such as córrego Bandeira,
Paratypes. All localities in Brazil, Mato Grosso, município de Alto Araguaia: MZUSP 73344 View Materials (11, 15.0- 23.3 mm SL), same data as holotype. MZUSP 73363 View Materials (1, 22.1 mm SL), córrego do Rancho, below lagoa do Veado , 17°16’12”S, 53°24’52”W; C. R. Moreira & F.C. T. Lima, 22 May 2001 GoogleMaps . MZUSP 41406 View Materials (179, 5 cs, 14.6-24.7 mm SL); MCZ 162371 About MCZ (5, 20.4-23.6 mm SL); USNM 371923 About USNM (5, 19.5-23.1 mm SL); MNRJ 24781 View Materials (5, 21.6-22.1mm SL), córrego do Rancho (headwaters), swamp near the road, approx. 17°16’S, 53°24’W; L. P.S. Portugal & F. Langeani, 8 March 1989 GoogleMaps .
Diagnosis. Hyphessobrycon eilyos is distinguished from its congeners by the absence of humeral and caudal spots; by the presence of numerous dark chromatophores on the lateral surface of the body, with the chromatophores intensely concentrated on the ventral region from the pelvic-fin origin to the end of the caudal-fin base; dorsal, adipose, and caudal fins with carmine red pigmentation in life; the presence of 7- 11 maxillary teeth; 6 or 7 horizontal scale rows between the dorsal-fin origin and the lateral line; and 15 to 19 branched anal-fin rays. Detailed comparisons between H. eilyos and congeners sharing a general dark color pattern are presented in the “Discussion”, below.
Description. Morphometric data of the holotype and paratypes are presented in Table 2 View Table 2 . Body compressed, moderately deep, greatest body depth immediately anterior to dorsal-fin origin. Dorsal profile of head slightly convex from upper lip to vertical through middle of the orbit; straight to slightly concave from latter point to tip of supraoccipital spine. Predorsal profile of body convex, dorsal-fin base straight to slightly convex, posteroventrally inclined. Body profile straight to convex from end of dorsal-fin base to adipose fin; slightly concave between latter point and origin of dorsal most procurrent caudal-fin ray. Ventral profile of head and body convex from lower lip to pelvic-fin origin. Body profile straight from pelvic-fin insertion to anal-fin origin. Body profile along anal-fin base posterodorsally slanted and slightly concave to slightly convex. Ventral profile of caudal peduncle slightly concave.
Jaws unequal, mouth terminal, anteroventral end of dentary protruding slightly. Maxilla extends posteriorly to under middle of orbit. Premaxillary teeth in two rows ( Fig. 7 View Fig ). Outer row with 3(7) uni- to tricuspid teeth. Inner row with 6(7) tri- to pentacuspid teeth. Maxilla with 7(2), 8(3), 9(1), or 11(1), uni- to tricuspid teeth. Dentary with 4(3), 5(3), or 6(1) large tricuspid teeth followed by 9(1), 11(3), 12(2), or 14(1) smaller uni- to tricuspid teeth.
Scales cycloid, with few radii. Lateral line incompletely pored, with 3(1), 4(7), 5(18), 6*(13), or 8(1) perforated scales. Lateral series scales including perforated scales 28(1), 29(2), 30*(10), 31(5), 32(1), or 33(1). Horizontal scale rows between dorsal-fin origin and lateral line 6*(26) or 7(16), not including scale of predorsal series situated just anterior to first dorsalfin ray. Horizontal scale rows between lateral line and pelvicfin origin 4*(43) or 5(3). Predorsal scales 9(1), 10*(8), 11(7), 12(4), or 13(1). Circumpeduncular scales 11(1), 12(4), 13(7), or 15(1). Single row of 5-6 scales covering basis of anteriormost anal-fin rays.
Dorsal-fin rays ii,8(3), or ii,9*(61). Dorsal-fin origin at middle of standard length. Base of last dorsal-fin ray at vertical through just before anal-fin origin. First dorsal-fin pterygiophore inserting behind neural spine of 9th(7) vertebra. Adipose fin typically present, but reduced in some specimens, and absent in 52 of 215 specimens examined. Unbranched anal-fin rays iii*(52) or iv(12). Branched anal fin rays 15(1), 16(4), 17*(28), 18(23), or 19(8). First anal-fin pterygiophore inserting behind hemal spine of 15th(1), or 16th(6) vertebra. Pectoral-fin rays i,7(1), 9(4), 10(27), 11*(29), or 12(3). Specimens up to 14.9 mm SL retaining larval pectoral fin anatomy. Tip of pectoral fin reaching vertical through pelvic-fin origin. Pelvicfin rays i,5(7), 6*(56), or 9(1). Tip of pelvic fin reaching origin of anal fin. Caudal fin forked, upper and lower lobes rounded, and similar in size. Principal caudal-fin rays 10+9(4). Eight (1), 10(1), or 12(2) dorsal procurrent caudal-fin rays, and 9(1), 10(2), or 11(1) ventral procurrent caudal-fin rays. First gill arch with 6(7) epibranchial; 1(7) on cartilage between epibranchial and ceratobranchial, 10(7) ceratobranchial, and 2(7) hypobranchial gill-rakers. Four (7) branchiostegal rays, 3(7) on anterior ceratohyal, and 1(7) on posterior ceratohyal Vertebrae 32(4), 33(2), or 34(1). Supraneurals 4(5), or 5(2).
Color in alcohol. Ground color cream, tanner in specimens stored for long time in ethanol. Guanine present on opercle and infraorbitals. Dark chromatophores densely concentrated on dorsal surface of head, and anterior surface of lower jaw. Dark chromatophores scattered on remaining portions of head. Dorsal midline with dense concentration of small dark chromatophores. Dorso and ventrolateral portions of body with dense concentration of dark chromatophores, uniformly distributed. Humeral spot absent (apparent humeral spot in Fig. 6 View Fig is actually a result of the reduction of the musculature in that portion of the body wall). Dense concentration of deep-lying dark chromatophores along ventral midline, more concentrated from pelvic-fin insertion to posterior portion of anal-fin base. Chromatophores on caudal-fin base absent, resulting in a light area. Caudal fin with dark chromatophores concentrated on middle rays and distal margin. Dorsal fin with concentration of dark chromatophores along distal margin. Anal fin with heavy concentration of dark chromatophores, mainly on interradial fin membrane. Pectoral, pelvic, and adipose fins with dense concentration of dark chromatophores.
Color in life. Description based on photograph of the holotype (MZUSP 75126). Ground color dark gray. Sides of head, and body silvery. Dorsal and adipose fins bright red. Caudal fin bright red, except for distal portion of caudal-fin lobes.
Geographic distribution. Hyphessobrycon eilyos is known from córrego do Rancho and ribeirão do Sapo, both tributaries of the upper rio Araguaia basin in the state of Mato Grosso, Brazil ( Fig. 5 View Fig ).
Ecological notes. At the ribeirão do Sapo( Fig. 8 View Fig ), the type-locality, Hyphessobrycon eilyos was only collected in backwaters of the stream. These backwaters were characteristically tea-colored, contrasting with the clear water of the mainstream, and were choked with vegetable debris from the adjacent riparian forest.Our limited collecting activity at the Córrego do Rancho did not allow us to ascertain what microhabitat was occupied by Hyphessobrycon eilyos , but presumably the species is associated with large flooded areas with abundant aquatic vegetation present in that stream. For remarks on its syntopy with H. langeanii and H. weitzmanorum , see “Ecological notes” under H. langeanii .
Etymology. From the Greek eilyos , den, lurking-place, in allusion to the habitat (backwaters choked with vegetal matter) occupied by the new species (and according to local information, shared with anacondas, Eunectes murinus ). A noun in apposition.
Table 2. Morphometric data for Hyphessobrycon eilyos. Measurements based on the holotype (MZUSP 75126) and 61 paratypes (MZUSP 41406, 56 ex.; MZUSP 73344, 4 ex.; MZUSP 73363, 1 ex.).
| holotype n | range | mean | ||
|---|---|---|---|---|
| Standard length (mm) | 22.3 | 62 | 15.8-24.7 | - |
| Percents of standard length | ||||
| Depth at dorsal-fin origin | 40.0 | 62 | 32.0-40.7 | 37.2 |
| Snout to dorsal fin origin | 54.1 | 62 | 50.6-58.5 | 53.6 |
| Snout to pectoral-fin origin | 33.8 | 62 | 30.5-36.6 | 33.0 |
| Snout to pelvic-fin origin | 51.4 | 62 | 47.4-55.9 | 50.2 |
| Snout to anal-fin origin | 64.8 | 62 | 61.9-69.0 | 64.7 |
| Caudal peduncle depth | 12.2 | 62 | 9.2-13.0 | 11.3 |
| Caudal peduncle length | 12.6 | 62 | 11.7-17.4 | 15.1 |
| Pectoral-fin length | 21.0 | 62 | 14.0-22.6 | 20.1 |
| Pelvic-fin length | 18.3 | 62 | 14.1-20.0 | 17.3 |
| Dorsal-fin base length | 14.7 | 62 | 12.4-16.4 | 14.3 |
| Dorsal fin height | 28.7 | 62 | 23.7-31.5 | 28.4 |
| Anal-fin base length | 27.5 | 62 | 23.1-29.3 | 25.8 |
| Anal-fin lobe length | 19.5 | 62 | 14.2-22.5 | 19.6 |
| Eye to dorsal-fin origin | 36.4 | 62 | 32.7-38.9 | 35.8 |
| Dorsal-fin origin to caudal-fin | 51.9 | 62 | 47.8-55.5 | 52.4 |
| base | ||||
| Bony head length | 34.3 | 62 | 30.4-37.6 | 32.8 |
| Percents of head length | ||||
| Horizontal eye diameter | 35.4 | 62 | 35.2-44.4 | 39.9 |
| Snout length | 19.7 | 62 | 19.7-27.2 | 22.7 |
| Least interorbital width | 26.8 | 62 | 23.3-33.6 | 27.7 |
| Upper jaw length | 45.2 | 62 | 33.9-51.3 | 46.7 |
| R |
Departamento de Geologia, Universidad de Chile |
| T |
Tavera, Department of Geology and Geophysics |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |