Cotoneaster majoricensis L. Sáez & Rosselló, 2012
|
publication ID |
https://doi.org/10.15553/c2012v672a5 |
|
DOI |
https://doi.org/10.5281/zenodo.5772933 |
|
persistent identifier |
https://treatment.plazi.org/id/03DA805D-AC25-A503-246C-FDB451E9F833 |
|
treatment provided by |
Carolina |
|
scientific name |
Cotoneaster majoricensis L. Sáez & Rosselló |
| status |
sp. nov. |
Cotoneaster majoricensis L. Sáez & Rosselló View in CoL , spec. nova ( Fig. 1 View Fig ).
Typus: SPAIN. BalearicIslands, Majorca: Puig Major de Son Torrella , Sa Coma Fosca , couloir nord, 31SDE8206, 1380 m, shady vertical limestone cliffs, 25.VI.2004, L.G. Valle & L.Sáez LS-6177 ( holo-: BC [ 903452 ]! ; iso-: BCB (Herb. L. Sáez)).
Species C. tomentoso et C. raboutense proxima, ab utroque fructo pyriforme, (1-)2 pyrenis praedito, distincta. Ab C. tomentoso foliissupra glabrescentibus vel sparse pilosis, petalisalbiset antherisminoribus differt.Ab. C. raboutense differt hypanthioplus minusque piloso et antherismajoribus 0,8- 1 mm longis (C. raboutense: 0,7-0,8 mm).
Shrub, erect or suberect, (0.5-) 1-2.7 m high, irregularly branched; young branches sparsely hairy, becoming graybrown and glabrous. Leaves deciduous, alternate, simple; stipules 3-4 1-1,5 mm, deciduous, subulate; petiole 4-8 mm; lamina (14-)25-50(-62) (12)-16-35(-50) mm, broadly elliptic to ovate-elliptic, obtuse, apex obtuse, truncate or emarginate; adaxial surface green, glabrescent, with few hairs, sometimes becoming glabrous; abaxial surface pilose or sparsely tomentose (hairs 0.5-1.5 mm long). Inflorescences terminal or axillary, corymbose, 2.1-2.7 2.5-3 cm, with 7- 14 flowers, with flowers opening in sequence over a longer period of time; bracts c. 7 2 mm, lanceolate; peduncle pilose; pedicels sparsely pilose. Hypanthium turbinate or campanulate, abaxially subglabrous or sparsely hairy (hairs 0.5-1.2 mm long), adaxially glabrous. Calyx lobes 1.2-2 1.3-1.9 mm, erect, triangular to broadly triangular, sparsely hairy. Petals 3.3-4 3.3-4 mm, suborbicular, erect, white, glabrous, imbricate in bud. Stamens 12-15, inserted in the mouth of hypanthium; filaments 3-3.5 mm long, subulate, erect, white; anthers 0.8-1 0.5 mm, white. Stigmas (1-)2 capitate. Fruit pyriform (5.5-)6.5-7.5 (4-) 5-6 mm, red to brownish-red, subglabrous or sparsely hairy at base, with persistent, incurved, fleshy calyx lobes; nutlets (1-)2. Pollen grains 3-colpate, prolate (37.68 ± 1.16 34.13 ± 1.50 µm).
Phenology.– Flowering time: (May-)June-August; fruiting time: July-August.
Taxonomical note. – Cotoneaster majoricensis is morphologically similar to C. tomentosus a widespread species in the mountains of Central and Southern Europe, but differs, by its adaxially glabrescent young leaves, white petals, smaller anthers, sparsely hairy hypanthium and calyx lobes, fruit size and number of nutlets ( Table 1 View Table 1 ). From C. raboutensis (restricted to southern and eastern France), the new species differs by its relatively larger anthers, and the higher number of flowers in the inflorescence, adaxially sparsely hairy calyx lobes at the abaxial surface, the fruit shape and the number of nutlets. The low number of nutlets, 1(-2) is a usual feature in sect. Cotoneaster Medik. , and together with fruit shape are important and useful markers for taxa delimitation (cf. FLINCK & al., 1998; HYLMÖ & FRYER, 1999). Cotoneaster integerrimus group seems more distantly related and differs by fewer flowered inflorescences [(1-)2-3(-4) flowers], reflexed pedicels, longer anthers and subglobose fruits.
Some 4-colpate pollen grains have been observed in anthers from both populations. The viability of the pollen, according to ALEXANDER (1980) technique, is 47.75 ± 5.63%.
The features shown by C. majoricensis are unique ( Table 1 View Table 1 ) and we failed to provide alternative taxonomic identifications following the key provided by DICKORÉ & KASPEREK (2010). In fact, the new species shows small flowers ( 3-6 mm diam.) and white petals, contradicting the key statements # 5 and 5’. Moreover, if this key is further followed, we are forced to choose between C. integerrimus and C. tomentosus (key statements # 25 and 25’). The Balearic plant has only two nutlets, 7-14 flowers per inflorescence, and elliptic to ovate-elliptic leaves, in contrast with C. integerrimus features: pyrenes 2-4, inflorescence 1-4-flowered, and leaves ± lanceolate-ovate. Furthermore, it cannot be included within the variation of the alternative choice, C. tomentosus characterized by pink or pale petals, nutlets 3-5, inflorescence 2-12-flowered, and leaves ovate or broadly ovate.
Ribosomal ITSsequences. – The ITS-1 was uniform in length across all accessions (230 bp), whereas the ITS-2 ranged between 213 and 217 bp. The variation in the ITS-2 length was due to three independent indels that were inferred after sequence alignment. Intragenomic polymorphisms (implying the presence of two overlapping bases at the electrophoretograms) ocurred at most sites. This ribosomal heterogeneity, apparently indicating a lack of concerted evolution of the ribosomal arrays, precluded the phylogenetic analysis of the ITS sequences. Twenty-two ITS-1 and 15 ITS-2 sites displayed point mutations between accessions. Cotoneaster majoricensis showed an unique 1-bp indel (position 253 not found in the other Cotoneaster accessions analyzed. Three accessions of C. intergerrimus (in broad sense) showed an exclusive 2-bp indel (positions 249-250 of the alignment) that was absent from the samples of Montseny ( Spain) and Mount Olympos ( Greece). The other 2-bp indel (at sites 255-256) was restricted to all C. intergerrimus samples (including C. scandinavicus ). Twenty-two ITS-1 and 15 ITS-2 sites showed point mutations between accessions ( Table 2 View Table 2 ), and intragenomic polymorphisms (implying the presence of two overlapping bases at the electrophoretograms) were found at most sites. This ribosomal heterogeneity, due to a lack of concerted evolution of the ribosomal arrays, precluded the phylogenetic analysis of the ITS sequences. However, the inspection of the nucleotide matrix revealed that sequences of C. majoricensis showed five unique mutations (including the 1-bp indel) not shared with the other Cotoneaster samples. Moreover, five other mutations were shared only with C. atlanticus from Jbel Tissouka ( Morocco) to which the ribosomal sequences of C. majoricensis appear to be most closely related. Moreover, we refrain to hypothesize an origin of C. majoricensis from C. atlanticus and C. tomentosus or C. integerrimus -like ancestors based on sharing specific polymorphic sites. Without cloning the ITS products it is not possible to know the sequences of the ITS copies present in the samples. Furthermore, biased concerted evolution of several sites towards a single parent might have occurred, obscuring the true relationships of the progenitors.
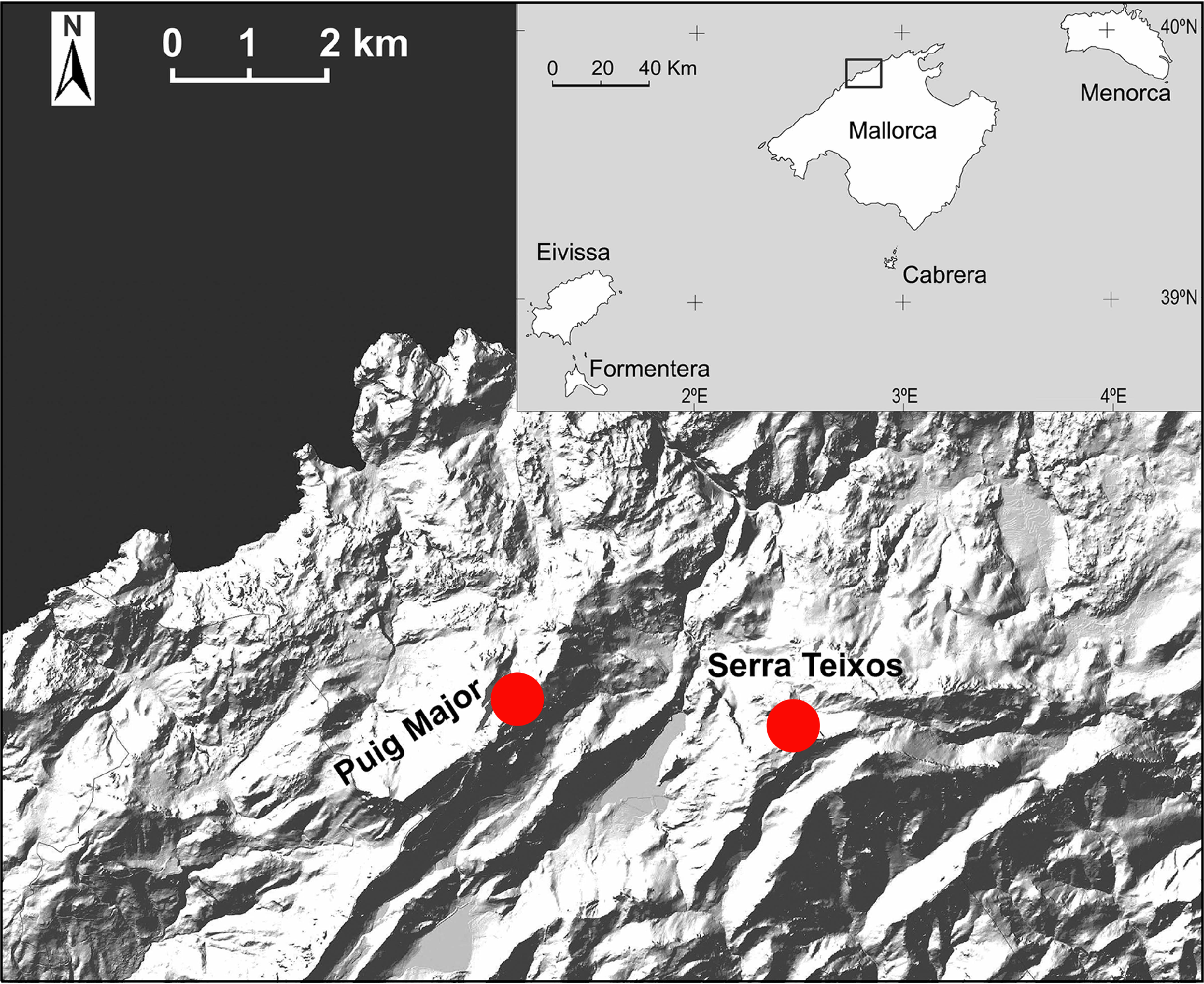
Habitat and distribution. – Currently, C. majoricensis is known from two mountains peaks of northern Majorca: Puig Major, the highest mountain of the Balearic archipelago, and Serra des Teixos ( Fig. 2 View Fig ). It grows in crevices of shady, almost vertical limestone cliffs on northern exposures, in a narrow altitudinal zone between 1180 and 1400 m. On the more exposed sites, the new species was mainly found growing together with several Balearic endemic taxa such as Agrostis barceloi L. Sáez & Rosselló , Crepistriasii (Cambess.) Nyman, Galium crespianum J. J. Rodr. , Galium balearicum Briq. , Lonicera pyrenaica subsp. majoricensis (Gand.) Gand. , and along with more widely distributed species such as Hieracium amplexicaule L. , Potentillacaulescens L., Sesleriainsularis Sommier subsp. insularis and Pimpinellatragium L. In more sheltered gullies and rock-bases several temperate trees and shrubs ( Taxus baccata L. , Ilexaquifolium L., Sorbusaria (L.) Crantz, Amelanchier ovalis Medik. , and higrophilous ferns ( Cystopterisfragilis (L.) Bernh. subsp. fragilis, Polystichum aculeatum (L.) Roth) were found associated.
Conservation assessment.– To date, we have identified two populations of Cotoneaster majoricensis , on the Puig Major de Son Torrella and Serra des Teixos (both populations are separated by a distance of 3,9 km). The summit of the Puig Major has been severely altered in the past (R UBÍ & MALAGARRIGA, 1971). The altitude of the mountain was lowered about 15 m for the building of military installations, including a radar station. The concomitant consequence of the permanent anthropic colonization of the mountain was the drastic change or disappearance of many of the north-facing microhabitats at the highest altitudes. Currently, several factors determine the decline of these habitats and their biota, including over-grazing, and damage by climbing activities. The main threat is due to predation by the introduced herbivores (goats and sheeps), likely being responsible for low local abundance and reduced colonization capacity of this narrow endemic species. In fact, our data indicate that natural recruitment is poor, since only five seedling plants were found in the period 2003-2009. Survival of C. majoricensis depends on intrinsic biological factors and stochastic events, as a consequence of its extremely small population size ( BARRETT & KOHN, 1991; PRIMACK & ROS, 2002).
The application of IUCN (2001) methodology to evaluate the vulnerability of C. majoricensis , produced the following results. The distribution of the species is highly restricted, with extremely low area of occupancy (c. 980 m 2). At present it is known to exist in 3 UTM 1 km 2 squares. The known populations of C. majoricensis are very small and only 94 individuals are known (29 of which in a reproductive state; Table 3 View Table 3 ). Decline of the population may be inferred by habitat quality and we have also registered a decrease of reproductive individuals in Puig Major population (from 19 specimens in 2006 to 14 specimens in 2009). Therefore C. majoricensis should be included in the “Critically Endangered” category according to the following criteria and subcriteria: CR C2a(i), D ( IUCN, 2001).
Other specimens studied. – SPAIN. BalearicIslands, Majorca: Puig Major de Son Torrella , 31SDE8206, shady vertical limestone cliffs, 16.VII.2002, L.Sáez LS-5965 ( BCB (herb. L. Sáez)) ; Puig Major de Son Torrella , 31SDE8206, 1350 m, 24.VII.2002, L.G.Valle & L.Sáez LS-5975 ( BCB (herb. L. Sáez)) ; Puig Major de Son Torrella , 31SDE8206, 1350 m, 24.V.2003, L.G. Valle & L.Sáez. LS-6106 ( BCB (herb. L. Sáez)) ; Puig Major de Son Torrella , Es Bufador , 31SDE8207, 1300 m, vertical cliffs, 19.VII.2010, L.Sáez LS-7230 (( BCB (herb. L. Sáez)) ; Majorca, Serra des Teixos , northern slope, Escorca , 31SDE8606, 1190 m, 13.VI.2006, L.G. Valle & L.Sáez LS-6433 ( BCB (herb. L. Sáez)).
Table 2. – Polymorphic sites for differents studied species of Cotoneaster Medik. in the ITS-1 (positions 31 to 214) and ITS-2 (249- 432) region of Cotoneaster accessions. the base with strongest signal in the electrophoretograms.
| 31 | 53 | 56 | 60 | 70 | 80 | 95 | 115 | 118 | 120 | 130 | 131 | 150 | 156 | 168 | 173 | 175 | 179 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C. integerrimus Medik. group [ C. pyrenaicus Gand. ], Spain: Cuberes (BCB) | C | Y[C] | A | G | Y[C] | C | C | R[G] | C | C | Y[T] | T | G | Y | C | Y | R[A] | Y |
| C. integerrimus Medik. group [ C. pyrenaicus Gand. ], Spain: Coma d Orient (BCB) | C | Y[C] | A | G | C | C | C | R | C | C | Y[T] | T | R[G] | Y | C | Y | A | Y |
| C. integerrimus Medik. group [ C. pyrenaicus Gand. ], Spain: Teruel (VAL 94401) | C | C | A | G | Y[C] | M | C | R | C | C | Y | T | G | Y[C] | C | Y | A | Y |
| C. integerrimus Medik. group [ C. pyrenaicus Gand. ], Spain: Montseny (BCB) | C | Y[C] | A | G | C | C | C | G | C | C | Y | T | G | C | C | Y | R | Y[C] |
| C. integerrimus Medik. group [ C. pyrenaicus Gand. ], Greece: Olympos (BCB) | C | Y | R[A] | G | Y | C | C | G | C | C | Y | T | G | Y | C | Y[C] | R | C |
| C. integerrimus Medik. group [ C. scandinavicus B. Hylmö ], Sweden (VAL [138524]) | C | C | A | G | Y[C] | C | C | G | C | C | Y | T | G | Y[C] | C | Y | R | Y[C] |
| C. tomentosus (Aiton) Lindl. , Spain: Cuenca (VAL 137454) | C | C | A | R | T | M[C] | C | R | C | C | T | T | G | C | C | Y[C] | R | Y[C] |
| C. tomentosus (Aiton) Lindl. , Spain: Cuberes (BCB) | Y | C | A | G | T | C | C | R | M | C | T | T | R[G] | C | C | C | R[G] | C |
| C. granatensis Boiss. , Spain: Alacant (VAL [39843]) | T | C | T | G | T | C | C | R[G] | A | C | T | T | G | C | G | C | G | C |
| C. atlanticus G. Klotz , Morocco: Tizi n’Test (BCB) | C | C | S | R[A] | T | C | M | R | C | M | T | Y | R | C | C | C | G | C |
| C. atlanticus G. Klotz , Morocco: Jbel Tissouka (BCB) | C | C | C | G | T | C | C | G | C | C | T | C | G | C | C | C | G | C |
| C. majoricensis L. Sáez & Rosselló, Spain: Balearic Islands, Mallorca (BCB) | C | C | G | G | T | C | C | R | C | C | T | C | G | Y | C | C | G | C |
Table 3. – Summary of data from 2009 survey.
| Locality | Puig Major | Serra des Teixos |
|---|---|---|
| Vegetative individuals | 47 | 18 |
| Reproductive individuals | 14 | 15 |
| Area of occupancy [m 2] | 30 | 21 |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |