Chauliogryllacris acaropenates Rentz, Su, Béthoux, 2018
|
publication ID |
https://doi.org/10.11646/zootaxa.4514.1.3 |
|
publication LSID |
lsid:zoobank.org:pub:6CF36E49-437B-4C69-A28E-F9A80CFA7945 |
|
DOI |
https://doi.org/10.5281/zenodo.5989628 |
|
persistent identifier |
https://treatment.plazi.org/id/03D487FA-DC41-FFBC-A1CC-099DDC1E8288 |
|
treatment provided by |
Plazi |
|
scientific name |
Chauliogryllacris acaropenates Rentz, Su, Béthoux |
| status |
sp. nov. |
Chauliogryllacris acaropenates Rentz, Su, Béthoux View in CoL sp. nov.
Figs. 1–5 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 View FIGURE 5 ; Table 1; Map 1
Holotype male. Label 1. “16.48’S 145.38’E ( GPS) Qld. Kuranda ( 335 m) (Top of the Range) 19 Butler Dr 15 JAN. 2008 DCF Rentz” Label 2 “ ANIC database #14008663” . Holotype in the Australian National Insect Collection ( ANIC), CSIRO, Canberra , ACT, Australia .
Type locality. The type locality is described in Wilson and Wilson (2018) “as Regional Ecosystem 7.11.1 Notophyll vine forest on metamorphics of moderate fertility of moist and wet lowlands and foothills ( REDD 2015) at 375 m in the McAlister Range near the village of Kuranda, Queensland. The forest is degraded with senescing emergent Acacia species and canopy thinning due to local clearing and the effects of wind tree falls during cyclones. The site has a monsoon climate with a Wet Season (mean ḵ 150 mm per month) in December through April, an Austral Winter Dry Season, and a mean±1 SD Annual Rainfall of 2120± 730 mm (100 years) (BOM 2015).”
Paratypes. Queensland ; Paratypes: 16.48’S 145.38’E ( GPS) Qld. Kuranda 335 m (Top of the Range) 19 Butler Dr DCF Rentz, collected on the following dates ; 1–15.i.2005, 1 female; 16–31.xii.2005, 1 male; 15–31.i.2006, 1 male, 1 female; 1–15.ii.2008, 2 males; 16–31.xii. 2008, 1 male; 16–30.xi.2010, 1 female; 16–28.ii.2011, 1 female; 16.31. i.2013, 1 female; 1–15.xii. 2016, 2 males; 1–15.i.2018, 1 male. 16 o 50’S 145 o 38’E. (GE) 3 Mason Rd. , Kuranda, ( 357 m) GoogleMaps 5 i.2018 B. Richardson: 1 male. 16 o 45'S. 146 o 37'E., Kuranda , 2 km SW on Kennedy Hwy, ("Arona") GoogleMaps , 3.i.2005, DCF Rentz, J Van Pelt, 1 female. 17 o 28'S. 16 o 50’S 145 o 24’E. (GE) Windy Hollow Rd (Kennedy Hwy) Kuranda, ( 453 m) GoogleMaps 3.iii.2011, DCF Rentz, 1 male; same locality GoogleMaps ; 2 ii. 2 0 18, DCF Rentz, SK Brannoch, Stop 4. 17.28 ’S 146.00'E., Polly Creek , Garradunga, nr Innisfail , 17.i.2006, J. Hasenpusch, 1 female. 16 o 44’S. 145 O 34’E., ( GPS) 15 km NW Jct Black Mtn Rd & Greene Lane on Black Mtn Rd ( 435 m) , 16.xi.2005, DCF Rentz, O & S Schmidt, Stop 14, 1 male. 16 o 06’11.53”S 145 o 27’13.08”E., James Cook University , Rainforest Site GoogleMaps , 5–7.x.2015 53m DCF Rentz, nr Crane, 1 female. James Cook University, Rainforest Site, 3–6.ii.2016 ( 46m), 1 male, DCF Rentz, B Richardson, Rainforest.
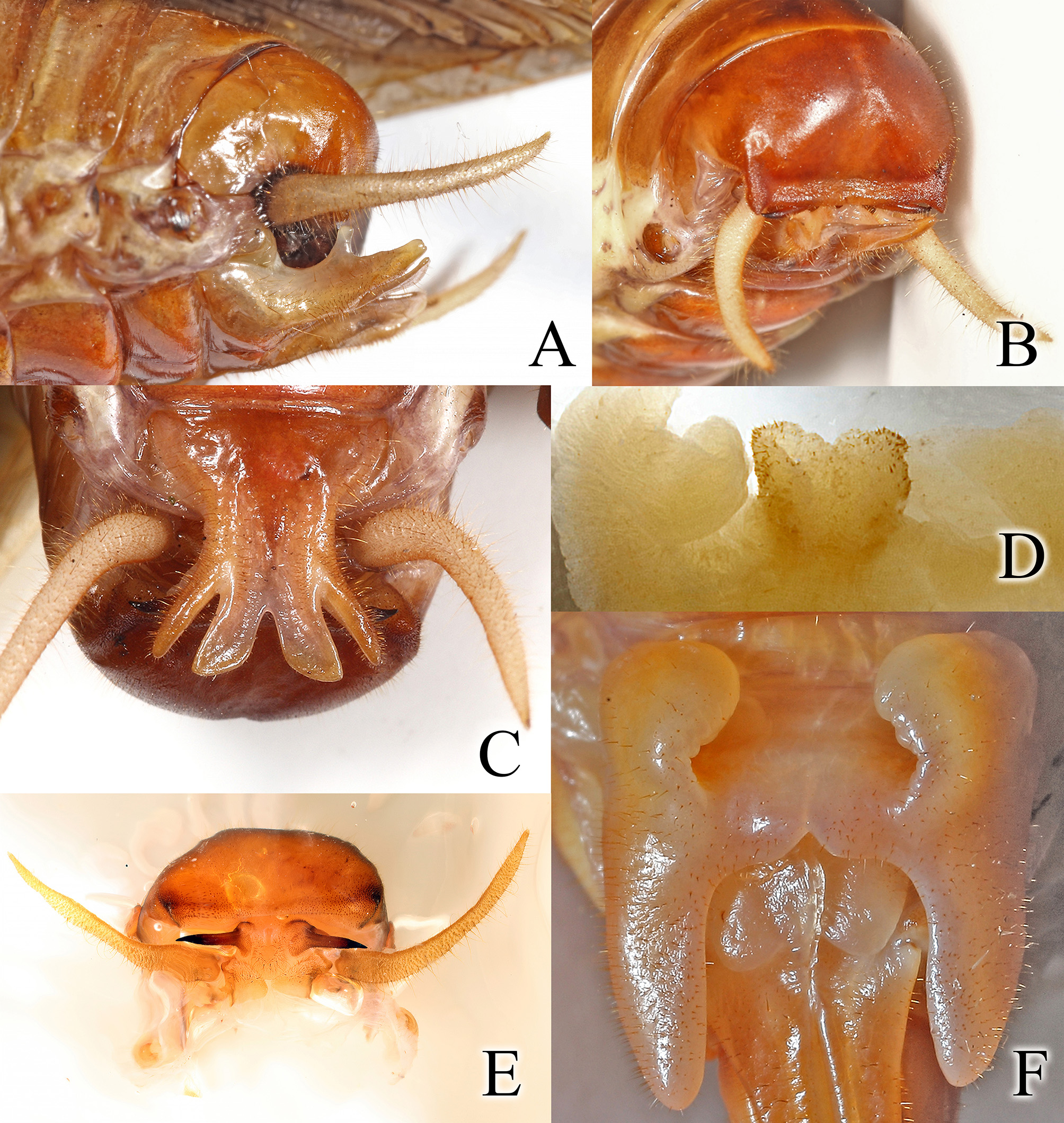
Description. Male. Size moderate to large for genus, form robust ( Table 1). Head very large ( Fig. 2A View FIGURE 2 ) protruding well in front of pronotum ( Fig. 1A, D View FIGURE 1 ). Fastigium of vertex very broad more than 5x as broad as antennal scape, margins not carinate or raised ( Fig. 2A View FIGURE 2 ); antennal scape elongate, about ¾ length of eye, pedicel short, stout, about 1/3 length of scape, flagellum 3–5 times length of body when intact. Frons sculptured, punctate ( Fig. 2A View FIGURE 2 ), the punctures irregular in shape, swollen below eye (belying large mandibles beneath; median ocellus relatively small ( Fig. 2A View FIGURE 2 ), about half the width of the antennal scape, lateral ocelli similarly small but very elongate. Eye elongate, narrow. Ventral surface of thorax unmodified ( Fig. 2E View FIGURE 2 ). Fore tibia unarmed dorsally ( Fig. 2C View FIGURE 2 ), ventral surface with 5 spines on each margin, in lateral profile slightly swollen. Middle tibia unarmed dorsally, ventral surface with 5 spines on each margin. Hind tibia armed dorsally with 4 stout teeth on each margin positioned in apical half ( Fig. 2B View FIGURE 2 ); ventral surface unarmed except apex with a single elongate spine on each side of dorsal surface, ventral surface with 4 apical spurs, each preceded by a single elongate spine, slightly shorter than the length of one of the spurs. Fore and middle femora unarmed; hind femur armed on ventral surface with 6 stout teeth on inner margin increasing in size to apex ( Fig. 2D View FIGURE 2 ), external margin with 1, 2 teeth but with a single apical spine; stridulatory area a single arched line of pegs ( Fig. 3B View FIGURE 3 ). Pronotum with deep furrows on surface ( Fig. 1A, D View FIGURE 1 ), “Z”-shaped on each side; anterior margin evenly convex, caudal margin truncate, humeral angles distinct, indented; lateral lobe shallow, ventral margin truncate, rimmed. Abdomen normal in shape for Gryllacrididae . Stridulatory teeth laterally on tergites 2, 3 ( Fig. 3A View FIGURE 3 ). Tenth tergite domed, apically indented, lateral angles acute ( Figs. 4A, B View FIGURE 4 ); subgenital plate ( Fig. 4C View FIGURE 4 ) dorsally with median flanges medially carinate, lateral flanges subconical with a small external tooth at base ( Fig. 4A View FIGURE 4 ). “Paraprocts” heavy, strongly divergent, well hidden beneath tenth tergite; cercus gracefully elongate, curved upward ( Figs. 4A, B, E View FIGURE 4 ). Concealed genitalia with phallus protruding, covered with stout setae ( Fig. 4D View FIGURE 4 ).
Female. Differs from male in following characters: size much larger, head much more pronounced, mandibles much more developed. Subgenital plate ( Fig. 4F View FIGURE 4 ) massive, lateral prongs parallel. Ovipositor straight, shorter than length of body. Pronotum ( Fig. 1A View FIGURE 1 ), abdomen ( Fig. 1D View FIGURE 1 ), ovipositor elongate, not thickened or modified apically.
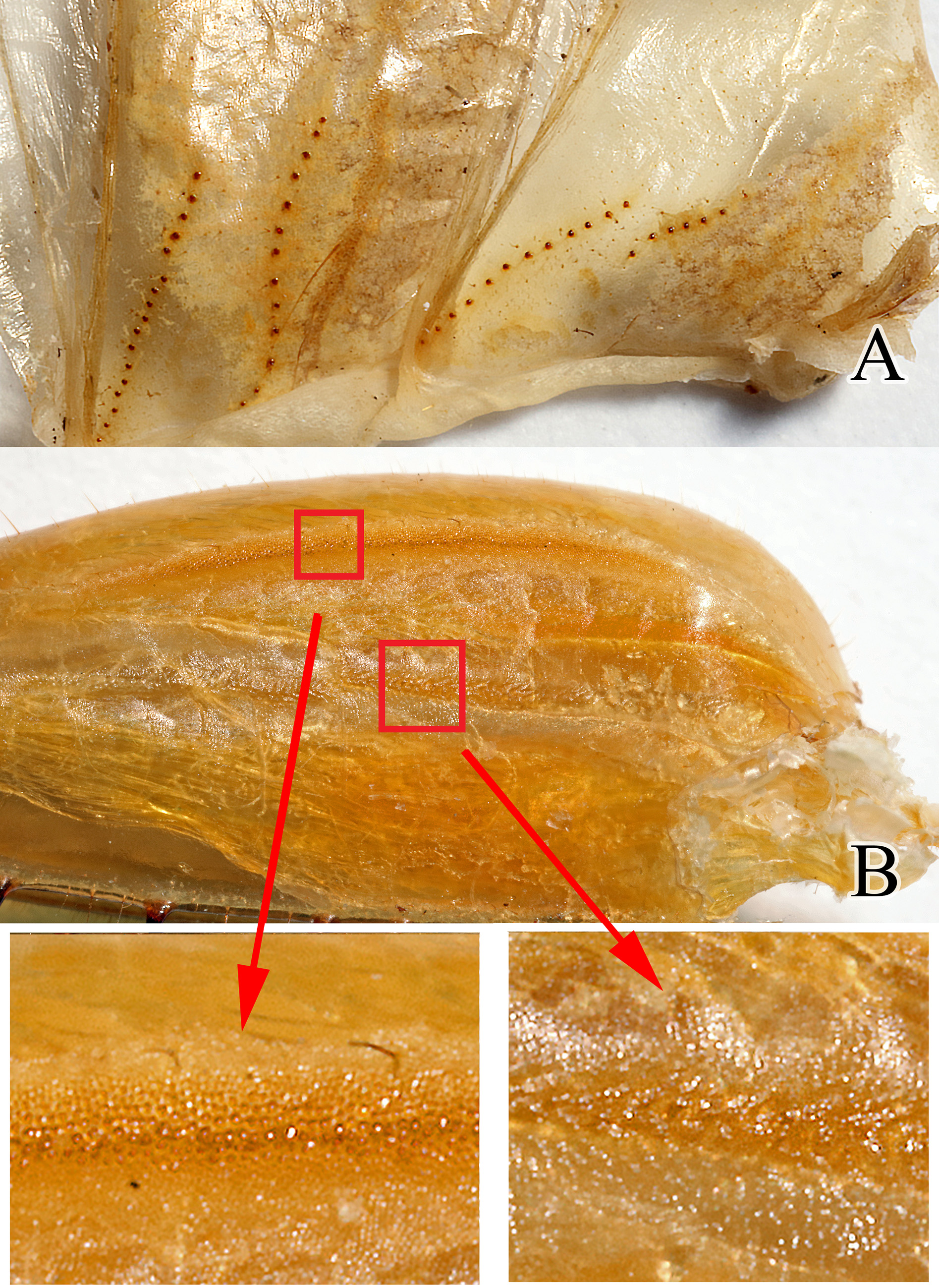
Tegmina and wing venation ( Fig. 5 View FIGURE 5 ). Generally speaking, the forewing venation pattern of Chauliogryllacris acaropenates sp. nov. corresponds to that represented on fig. 13A, G in Béthoux (2012). Specifically, MP+CuA (emerging from M+CuA) has a distinct free part (but of variable extent), and CuA+CuPaα is simple. The later state characterizes the ‘ Etagryllacris type’. In general appearance, forewings of Chauliogryllacris acaropenates sp. nov. resemble those represented on Fig. 16A, B in Béthoux (2012). Note, however, that the corresponding wings were selected because they displayed unusual venation patterns.
The occurrence of secondary intercalary veins is not extensively documented in hind wings of gryllacridids. Referring to cases documented in Béthoux (2012), the occurrence of secondary intercalary veins of 2nd order, as in Cha”uliogryllacris acaropenates , is unique, but might prove more widely distributed.
Colour. Overall colour of body reddish dark brown in life ( Figs 1A, B View FIGURE 1 ). Head black, this colour extending onto dorsal ¼ of clypeus, remainder of clypeus and labrum straw brown in life.
Remarks. This interesting Raspy cricket uses its mandibles to form burrows in tree holes initiated from broken branches. The resident cricket resides in the burrow indefinitely unless harassed or invaded by ants ( Fig. 6 View FIGURE 6 ). As it grows it uses the mandibles to enlarge the burrow as we have observed at night. We found no evidence of silk being incorporated into the burrow. A single observation was made of a cricket using silk to tie leaves together to form a shelter in which it passed the daylight hours ( Fig. 1B View FIGURE 1 ). The second night the cricket had abandoned the shelter. Perhaps, it was in the midst of finding a new tree hole site and the leaf shelter was just a temporary home. Nymphs may utilise leaf shelters if they cannot find suitable tree holes or perhaps, they are on the move to discover more productive habitats. But finding an adult in a leaf shelter during the day is unusual based on our observations.
We have no direct observations of feeding in nature but in observing the crickets after dark, we found them very actively searching vegetation, perhaps seeking other insects. In captivity they thrived on the Orthoptera Food Mixture ( Rentz, (2014)
Derivation of name. This species is named with reference to the species providing a home for a number of kinds of mites. Thanks to Dr E. Sidorchuk for suggesting the specific name of this species.
Research opportunities. Australian Gryllacrididae offer opportunities to resident biologists. The Australian Faunal Directory presently lists 116 described species in 29 genera. We know that there are many more than that. Hale and Rentz (2001) provided a world overview for the family which indicated that some 600 species were known at the time with about 200 species estimated to occur in Australia. Gryllacrididae can be encountered in almost all terrestrial habitats with fully winged, macropterous and apterous species being known. With the Australian species, all known stages in the life cycle are known to produce silk through the mouthparts. This they use to tie leaves together ( Fig. 1B View FIGURE 1 ), to form a shelter or line a burrow. In addition to the granivorous habits of the species noted above, some species are predaceous feeding on emerging cicadas or moulting orthopteroids at night. Others feed on flowers and seeds or fruits. Probably the majority of species are opportunists, taking advantage of what they come across in a night’s foraging. All known gryllacridids are nocturnal in their behavior. Copulation takes place after dark with males producing a large spermatophore that is carried by the female much in the manner of Tettigoniidae .
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |