Neognathophausia ingens ( Dohrn, 1870 )
|
publication ID |
https://doi.org/10.11646/zootaxa.4577.2.9 |
|
publication LSID |
lsid:zoobank.org:pub:36583309-8658-4D1D-B1AE-DF9E2DD7E1AA |
|
DOI |
https://doi.org/10.5281/zenodo.3510340 |
|
persistent identifier |
https://treatment.plazi.org/id/03C84A4C-0A18-1D4D-5ADA-DC0FFE06F9EC |
|
treatment provided by |
Plazi (2019-04-05 08:24:52, last updated 2024-11-29 08:53:27) |
|
scientific name |
Neognathophausia ingens ( Dohrn, 1870 ) |
| status |
|
Neognathophausia ingens ( Dohrn, 1870)
( Figs. 1–5 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 View FIGURE 5 )
Lophogaster ingens Dohrn, 1870: 610 , pl. 31, figs. 12–14.
Gnathophausia inflata Willemoes-Suhm, 1873 : in MS (nomem nudum).
Gnathophausia calcarata G. O. Sars, 1883: 5 –6; 1885: 35, pl. IV; Ortmann 1906: 30, pl. I (2a–f).
Gnathophausia bengalensis Wood-Mason & Alcock, 1891: 269 .
Gnathophausia doryphora Illig, 1906: 227 –228, fig. 1A–D.
Gnathophausia ingens— G. O. Sars, 1883: 4.— Illig 1930: 407 –408, figs. 11–12.— W.M. Tattersall 1939: 224 –225.— Fage 1941: 15 –24, figs. 20, 24–25; Nouvel 1943: 9–12, pl. I (4).— W.M. Tattersall 1951: 25 –26.— O. S. Tattersall 1955: 31 – 35.—Clarke 1961: figs. 1–8.— Pequegnat 1965: 402 –403, 406, 408, figs. 3–4, 8, tables 1–2.—Casanova 1977: 328–330.— Kathman et al. 1986: 160 –161.—Escobar-Briones & Soto 1991: table 1.— Casanova 1996: 129; Casanova et al. 1998: 60.— Serejo et al. 2007: Tabs. 3, 4.— Price et al. 2009: 923 –928.— Meland & Aas 2013: 203 View Cited Treatment , fig. 2.— Gunalan & Kumar 2014: 123, Figs. 1 View FIGURE 1 , 3–4 View FIGURE 3 View FIGURE 4 , tables 1 and 2.
Neognathophausia ingens — Petryashov, 1992: 47 –48.— Petryashov 2005: 959, 968–969, Fig. 3 View FIGURE 3 .— Petryashov 2007: Tab. 2; 2015: figs. 5, 6.— Wittmann & Wirtz 1998: 512.— Dürr & González 2002: 367, Tab. 3.— Haroun & Garrido 2003: 68.— Wittmann et al. 2004: 1261, table I.— Serejo et al. 2007: 142, Tab. 2.—Wittmann et al. 2007.—Wittmann & Riera 2012: 70–71.
Type locality. Off the west coast of Africa.
Material examined. One female (CL, 10.1 cm), Thiago EJ II, St. 23#1 (32°22.173’S, 50°13.829’W), 330 m, coll. Luiz F.R. Moro, 14/II/2018 ( UFRGS 6587 View Materials ); one female (CL, 8.7 cm), Thiago EJ GoogleMaps II, St. 24 (32°29.967’S, 50°16.941’W), 237 m, coll. Luiz F.R. Moro, 14/II/2018 ( UFRGS 6588 View Materials ) GoogleMaps ; one male (CL, 8.1 cm), REVIZEE Pesca, Bahia II, St. E0547 (21°46.57’S, 39°53.35’W), 1105 m, coll. N.O. Thalassa, 06/VII/2000 ( MNRJ 15042 View Materials ); one male (CL, 10 cm) and one US (3.5 cm), REVIZEE Pesca , Bahia II, St. 0 538 (20°27.667’S, 39°38.101’W to 20°32.771’S, 39°37.65’W), 1680 m, coll. N.O. Thalassa, 02/ VI GoogleMaps /2000 ( MNRJ 15041 View Materials ) ; one female (CL, 8.1 cm), REVIZEE Pesca, Bahia II, St. E0524 (19°43.663’S, 38°39.838’W to 19°42.684’S, 38°44.568’W), 925 m, coll. N.O. Thalassa, 27/VII/2000 ( MNRJ 15043 View Materials ) GoogleMaps .
Diagnosis. See Meland & Aas (2013).
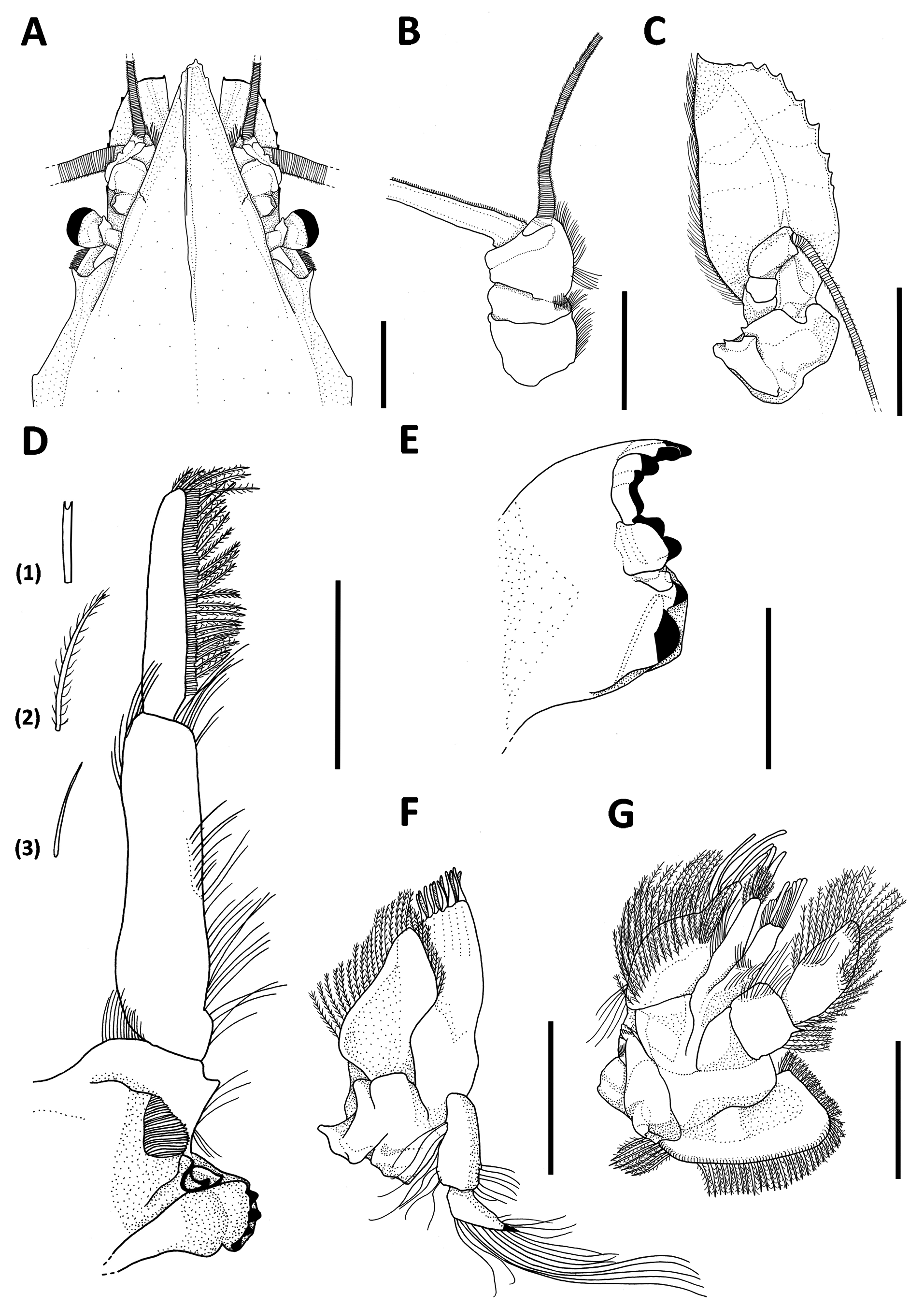
Description. Carapace: smooth; rostrum triangular, short and weakly denticulate, surpassing antennal scale laterally and covering eyestalks partially ( Figs. 1A, B View FIGURE 1 , 2A View FIGURE 2 ); dorsal ridge at the basis of rostrum unarmed ( Fig. 2A View FIGURE 2 ); dorsal keel interrupted in the middle region, extending anteriorly onto the rostrum and posteriorly onto the posterodorsal margin ( Fig. 1A View FIGURE 1 ); upper and lower lateral keels continuous ( Fig. 2A View FIGURE 2 ); cervical groove U-shaped ( Fig. 1A View FIGURE 1 ); posterodorsal spine reduced ( Fig. 1A, B View FIGURE 1 ); posterior margin of carapace emarginate with postero-lateral angles terminating in long and sharp spines ( Fig. 1A, B View FIGURE 1 ).
Eyes: large, reaching the end of the third antennular peduncle ( Fig. 1B View FIGURE 1 ); cornea distinctly semi-ellipsoidal, occupying about 1/3 of whole eye ( Figs. 1A, B View FIGURE 1 , 2A View FIGURE 2 ).
Antennule: antennular peduncle three-segmented; first and third segments about 2.3x as long as second; all segments bearing plumose setae ( Fig. 2B View FIGURE 2 ).
Antenna: antennal scale small and subovate, tapering into sharply pointed apex, unsegmented; external margin with 8 teeth and a short apical spine ( Fig. 2C View FIGURE 2 ).
Mandible: mandibular palp three-segmented; second segment about 2.9x as long as first, about 1.6x as long as third ( Fig. 2D View FIGURE 2 ); first and second segments bearing simple setae ( Fig. 2 View FIGURE 2 D-3), third segment bearing plumose setae ( Fig. 2 View FIGURE 2 D-2) and row of feathered setae ( Fig. 2 View FIGURE 2 D-1); incisive process bearing five teeth, lacinia mobilis bicuspidate ( Fig. 2E View FIGURE 2 ).
Maxillule: base with 11 spines, endite covered by plumose setae, retroverted palp bi-articulated bearing long simple setae ( Fig. 2F View FIGURE 2 ).
Maxilla: sympodite consisting of three few distinct segments, coxa with a simple endite, basis with a bilobate endite, all covered by plumose setae along their margins; endopod two-segmented, covered by plumose setae, distolateral margin of first segment with one spine; exopod rounded, outer margin with plumose setae ( Fig. 2G View FIGURE 2 ).
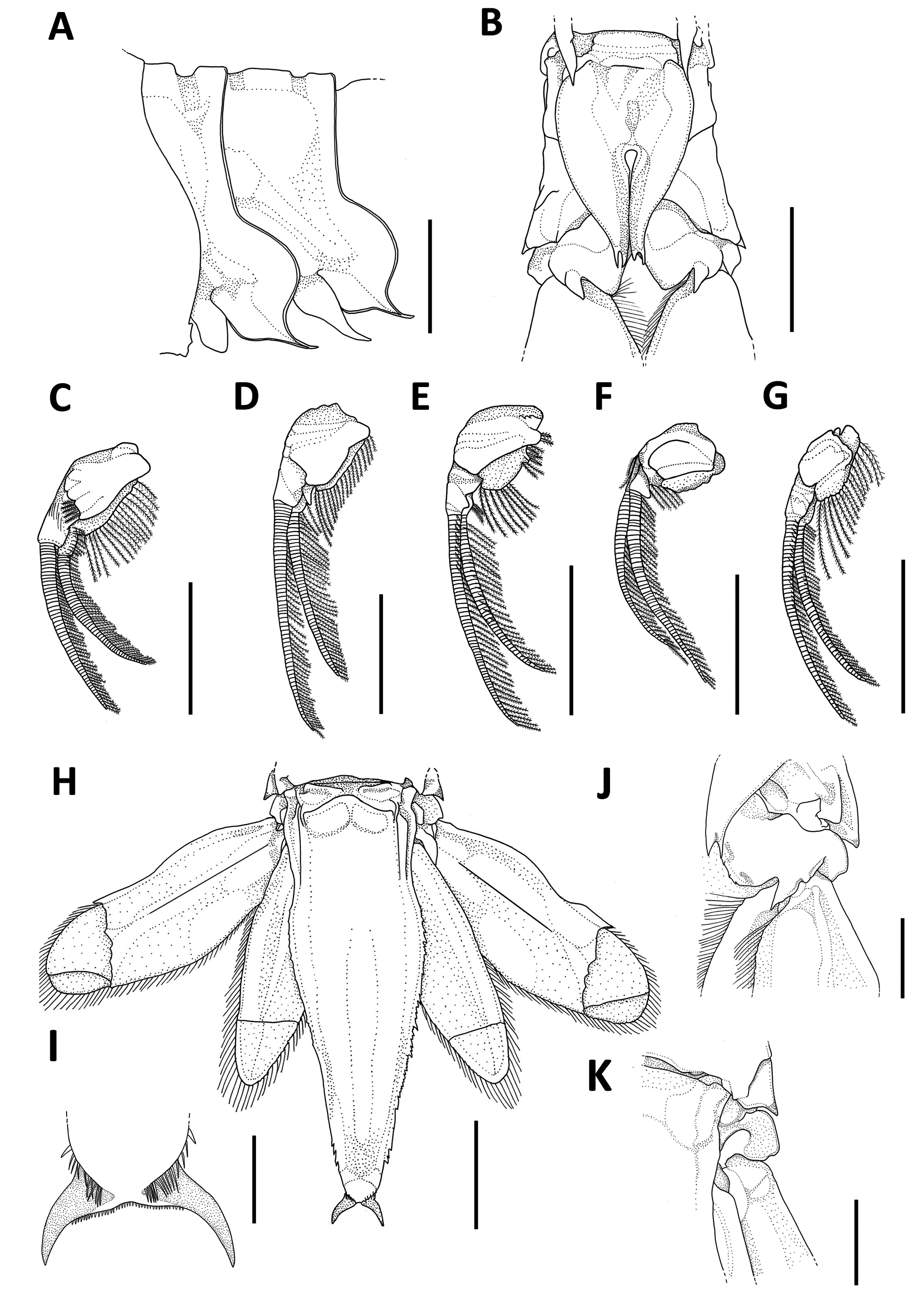
Thoracopods: first thoracopod with endopod five-segmented, short and robust, covered by simple setae, exopod small, ribbon-type, covered by simple setae, base bearing a long leaf-like epipod ( Fig. 3A View FIGURE 3 ); second thoracopod robust, exopod short, about 0.4x endopod length ( Fig. 3B View FIGURE 3 ); third thoracopod robust, exopod short, about 0.4x endopod length, inner margin of dactylus armed with nine spines ( Fig. 3C View FIGURE 3 ); fourth thoracopod slender, exopod short, about 0.5x endopod length, inner margin of dactylus armed with 11 spines ( Fig. 3D View FIGURE 3 ); fifth thoracopod slender, exopod short, about 0.4x endopod length ( Fig. 3E View FIGURE 3 ); sixth thoracopod slender, exopod short, about 0.5x endopod length ( Fig. 3F View FIGURE 3 ); seventh thoracopod slender, exopod short, about 0.6x endopod length ( Fig. 3G View FIGURE 3 ); eighth thoracopod slender ( Fig. 3H View FIGURE 3 ), exopod short, about 0.7x endopod length; all dactyli of endopods with one strong spine on apex, inner margin with 8–11 spines; oostegites leaf-like increasing in size posteriorly.
Pleon: pleural plates produced into two spines ( Fig. 4A View FIGURE 4 ); epimera of sixth somite fused in the mid-ventral line into heart-shaped plate ( Fig. 4B View FIGURE 4 ); all pleopods well developed, birramous in both sexes; exopods and endopods composed by 58–64 and 46–49 segments respectively, bearing compound setae ( Fig. 4 View FIGURE 4 C–G).
Tail fan: telson linguiform, about 3.5x as long as broad at its base, dorsal surface with two longitudinal keels ( Fig. 4H View FIGURE 4 ), lateral margins with irregularly distributed spines ( Fig. 4H View FIGURE 4 ), apex crescent-shaped ( Fig. 4H, I View FIGURE 4 ); uropods shorter than telson, inner margin of sympodite unarmed ( Fig. 4J, K View FIGURE 4 ).
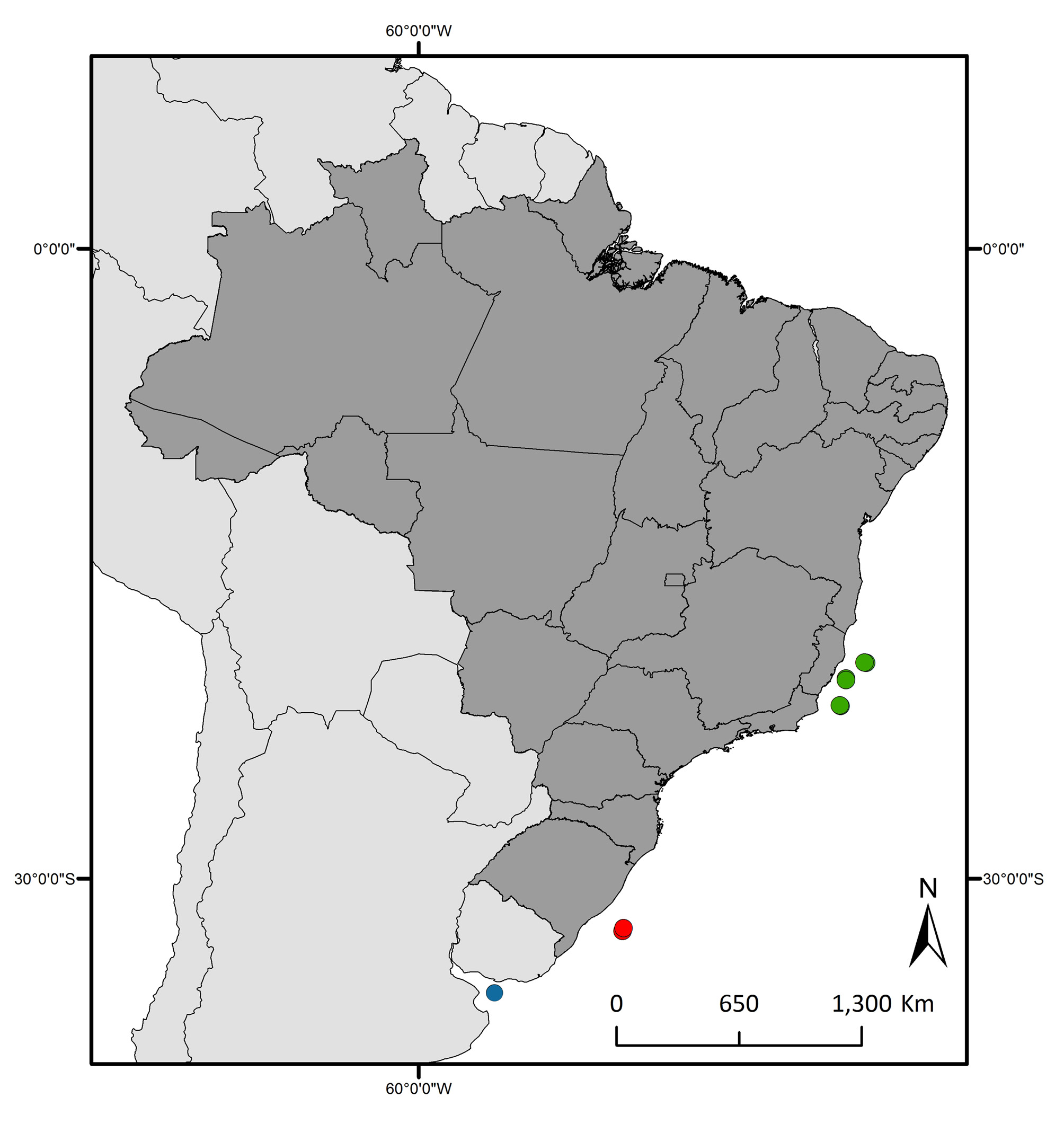
Distribution. Neognathophausia ingens is considered a cosmopolitan species, but does not occur in polar and sub-polar waters ( Meland & Aas 2013). Along the Atlantic coast of South America it has been recorded for the Brazilian coast and east of La Plata region in Argentina ( Fig. 5 View FIGURE 5 ) ( Tattersall 1951; 1955; Serejo et al. 2007; Meland & Aas 2013).
Bathymetric distribution. Occurring from 125 to 3391 m depth, being usually found between 600 to 1.500 m ( Meland & Aas 2013).
Ecology. In the marine food webs, N. ingens feeds mainly on fishes ( Hopkins et al. 1994) and is an important food source in the diet of fishes of the family Berycidae ( Dürr & Gonzalez 2002) and the sperm whale Physeter macrocephalus Linnaeus ( Best 1999) . Regarding reproductive ecology, this species is considered semelparous ( Childress & Price 1978).
Remarks. This species was first recorded from Brazil by Serejo et al. (2007) from the state of Bahia (BA) to Rio de Janeiro (RJ) (922–1815 m depth). However, a review of the material deposited in the MNRJ showed that there is no record for the state of Bahia. Therefore, the occurrence of N. ingens in Brazil has been limited to the states of Espirito Santo, Rio de Janeiro and Rio Grande do Sul ( Fig. 5 View FIGURE 5 ). The new records of N. ingens increases the southernmost limit for the species in Brazil about 1188 km. This contribution provides for the first time, illustrations and descriptions of the mandible ( Fig. 2D, E View FIGURE 2 ), thoracopods 3–8 and dactyli 2–8 ( Fig. 3 View FIGURE 3 B–H), pleopods 1–5 ( Fig. 4 View FIGURE 4 C–G), and sympodite of uropod in dorsal view ( Fig. 4K View FIGURE 4 ). The new illustrations could be useful for further taxonomic studies.
Best, P. B. (1999) Food and feeding of sperm whales Physeter macrocephalus off the west coast of South Africa. South African Journal of Marine Science, 21, 393 - 413. https: // doi. org / 10.2989 / 025776199784126033
Casanova, J. - P. (1996) Crustacea Mysidacea: Les Lophogastrides d'indonesie, de Nouvelle- Caledonie et des iles Wallis et Futuna. In: Crosnier, A. (Ed.), Resultats des Campagnes Musorstom. Vol. 15. Memoires du Museum National d'Histoire Naturelle, 168, pp. 125 - 146.
Casanova, J. - P., Joung, L. D. & Faure, E. (1998) Interrelationships of the two families constituting the Lophogastrida (Crustacea: Mysidacea) inferred from morphological and molecular data. Marine Biology, 132, 59 - 65. https: // doi. org / 10.1007 / s 002270050371
Childress, J. J. & Price, M. H. (1978) Growth rate of the bathypelagic crustacean Gnathophausia ingens (Mysidacea: Lophogastridae). I. Dimensional Growth and Population Structure. Marine Biology, 50, 47 - 62. https: // doi. org / 10.1007 / BF 00390541
Dohrn, A. (1870) Untersuchungen uber Bau und Entwicklung der Arthropoden. 8. Die Ueberreste des Zoea-Stadiums in der ontogenetischen Entwicklung der verschiedenen Crustaceen-Familien. Jenaische Zeitschrift fur Naturwissenschaft, 5, 471 - 491.
Durr, J. & Gonzalez, J. A. (2002) Feeding habits of Beryx splendens and Beryx decadactylus (Berycidae) off the Canary Islands. Fisheries Research, 54 (3), 363 - 374. https: // doi. org / 10.1016 / S 0165 - 7836 (01) 00269 - 7
Escobar-Priones, E. & Soto, L. A. (1991) Biogeografia de los Misidaceos (Crustacea: Peracarida) del Golfo de Mexico. Caribbean Journal of Science, 27 (1 - 2), 80 - 89.
Fage, L. (1941) Mysidacea Lophogastrida-I. The Carlsberg Foundation's oceanographical expedition round the world 1928 - 1930 and previous Dana-expeditions under the leadership of Prof. Johannes Schmidt. Dana Reports, 4 (19), 1 - 52.
Gunalan, B. & Kumar, C. P. (2014) Report of Gnathophausia ingens (Dohrn, 1870) from bathypelagic region of Bay of Bengal, corroborated by DNA barcoding and 18 S rRNA gene sequencing. Indian Journal of Fisheries, 61 (4), 123 - 126,
Haroun, R. & Garrido, M. J. (2003) Orden Mysidacea. In: Moro, L., Martin, J. L., Garrido, M. J. & Izquierdo, I. (Eds.), Lista de especies marinas de Canarias (algas, hongos, plantas y animales), pp. 68 - 69.
Hopkins, T. L., Flock, M. E., Gartner Jr, J. V. & Torres, J. J. (1994) Structure and trophic ecology of a low latitude midwater decapods and mysid assemblage. Marine Ecology Progress Series, 109, 143 - 156. https: // doi. org / 10.3354 / meps 109143
Illig G. (1906) Ein Weiterer Bericht ueber die Schizopoden der Deutschen Tiefsee-Expedition 1898 - 1899. Supplement 1. 2. Gnathophausien. Ibid, 30, 227 - 322.
Illig G. (1930) Die Schizopoden der Deutschen Tiefsee-Expedition. Wissenschaftliche Ergebnisse der Deutschen Tiefsee Expedition Faldivia , 22 (6), 397 - 625.
Kathman, R. D., Austin, W. C., Saltman, J. C. & Fulton, J. D. (1986) Identification manual to the Mysidacea and Euphausiacea of the northeast Pacific. Canadian Special Publication of Fisheries and Aquatic Sciences, 93, 1 - 411.
Meland, K. & Aas, P. O. (2013) A taxonomical review of the Gnathophausia (Crustacea, Lophogastrida), with new records from the northern mid-Atlantic ridge. Zootaxa, 3664 (2), 199 - 225. https: // doi. org / 10.11646 / zootaxa. 3664.2.5
Ortmann, A. E. (1906) Schizopod crustaceans in the U. S. National Museum. The families Lophogastridae and Eucopiidae. Proceedings of the United States National Museum, 31, 23 - 54. https: // doi. org / 10.5479 / si. 00963801.31 - 1480.23
Pequegnat, L. H. (1965) The bathypelagic mysid Gnathophausia (Crustacea) and its distribution in the eastern Pacific Ocean. Pacific Science, 9 (4), 399 - 421.
Petryashov, V. V. (1992) Notes on mysid systematics (Crustacea, Mysidacea) of Arctic and the North-Western Pacific. Zoologicheskii Zhurnal, 71 (10), 47 - 58.
Petryashov, V. V. (2005) Mysids (Crustacea, Mysidacea) collected by Soviet and Russian Antarctic expeditions. Lophogastrida, Petalophthalmida, and Mysida: Boreomysidae. Zoologicheskij Zhurnal, 84 (8), 957 - 973.
Petryashov, V. V. (2007) Biogeographical division of Antarctic and Subantarctic by Mysid (Crustacea: Mysidacea) fauna. Russian Journal of Marine Biology, 33 (1), 1 - 16. https: // doi. org / 10.1134 / S 1063074007010014
Price, W. W., Heard, R. W., Aas, P. & Meland, K. (2009) Lophogastrida of the Gulf of Mexico. In: Felder, D. L. & Camp, D. K. (Eds.), Gulf of Mexico Origins, Waters and Biota. Fol. 1. Biodiversity. Texas A & M University Press, College Station, pp. 923 - 928.
Sars, G. O. (1883) Preliminary notices on the Schizopoda of H. M. S. Challenger Expedition. Forhandlinger i Fidenskabsselskabet i Christiania, 7, 1 - 43.
Serejo, C. S., Young, P. S., Cardoso, I. C., Tavares, C., Rodrigues, C. & Almeida, T. C. (2007) Abundancia, diversidade e zonacao dos crustaceos no talude da costa central do Brasil (11 ° - 22 ° S) coletados pelo Programa REVIZEE / Score Central: prospeccao pesqueira. In: Costa, P. A. S., Olavo, G. & Martins, A. S. (Eds.), Biodiversidade da fauna marinha profunda na costa central Brasileira. Fol. 24. Museu Nacional, Rio de Janeiro, pp. 133 - 162.
Tattersall, W. M. (1939) The Euphausiacea and Mysidacea of the John Murray Expedition to the Indian Ocean. The John Murray Expedition 1933 - 34, Scientific Reports, 5, 203 - 246.
Tattersall, W. M. (1951) A review of the Mysidacea of the United States National Museum. Smithsonian Institution, United States National Museum Bulletin, 201, 1 - 292. Willemoes-Suhm, R. V. (1873) Notes from the Challenger VII, by W. Thomsen. Nature, 8, 400 - 403.
Tattersall, O. S. (1955) Mysidacea. Discovery Reports, 28, 1 - 190. https: // doi. org / 10.5962 / bhl. part. 16838
Wittmann, K. J. & Wirtz, P. (1998) A first inventory of the mysid fauna (Crustacea: Mysidacea) in coastal waters of the Madeira and Canary archipelagos. Boletim do Museu Municipal do Funchal, 5 (Supplement), 511 - 533.
Wittmann, K. J., Hernanez, F., Durr, J., Tejera, E., Gonzalez, J. A. & Jimenez, S. (2004) The epi- to bathypelagic Mysidacea (Peracarida) off the Selvagens, Canary, and Cape Verde Islands (NE Atlantic), with first description of the male of LongithoraxaliceiU. Nouvel, 1942. Crustaceana, 76 (10), 1257 - 1280.
FIGURE 1. Neognathophausia ingens (Dohrn, 1870). A, habitus lateral view (UFRGS 6587); B, habitus dorsal view (UFRGS 6587). Scale bars, 2.5 cm.
FIGURE 2. Neognathophausia ingens (Dohrn, 1870). A, rostrum (dorsal view); B, antenulle; C, antenna; D, mandible, (1) feathered seta, (2) plumose seta, (3) simple seta; E, mandible; F, maxillule; G, maxilla. Scale bars: A–C, 10 mm; D, F and G, 5 mm; E, 3.33 mm.
FIGURE 3. Neognathophausia ingens (Dohrn, 1870). A, first thoracopod; B, second thoracopod with detail of the dactylus; C, third thoracopod with detail of the dactylus; D, fourth thoracopod with detail of the dactylus; E, fifth thoracopod with detail of the dactylus; F, sixth thoracopod with detail of the dactylus; G, seventh thoracopod with detail of the dactylus; H, eigth thoracopod with detail of the dactylus. Scale bars: A–H, 5 mm; dactyl 1, 3–8, 2.5 mm; dactyl 2, 3.33 mm.
FIGURE 4. Neognathophausia ingens (Dohrn, 1870). A, pleural plates 1–2; B, epimera of sixth pleonal somite, ventral view; C, pleopod 1; D, pleopod 2; E, pleopod 3; F, pleopod 4; G, pleopod 5; H, tail fan; I, apex of telson; J, sympodite of uropod, ventral view; K, sympodite of uropod, dorsal view. Scale bars: A–H, 10.00 mm; J and K, 5.00 mm; I, 2.50 mm.
| EJ |
Ein Yabrud collection catalogue entries at The Hebrew University |
| VI |
Mykotektet, National Veterinary Institute |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Neognathophausia ingens ( Dohrn, 1870 )
| Huber, Augusto Frederico, Ribeiro, Felipe Bezerra & Araujo, Paula Beatriz De 2019 |
Neognathophausia ingens
| Serejo, C. S. & Young, P. S. & Cardoso, I. C. & Tavares, C. & Rodrigues, C. & Almeida, T. C. 2007: 142 |
| Petryashov, V. V. 2005: 959 |
| Wittmann, K. J. & Hernanez, F. & Durr, J. & Tejera, E. & Gonzalez, J. A. & Jimenez, S. 2004: 1261 |
| Haroun, R. & Garrido, M. J. 2003: 68 |
| Durr, J. & Gonzalez, J. A. 2002: 367 |
| Wittmann, K. J. & Wirtz, P. 1998: 512 |
| Petryashov, V. V. 1992: 47 |
Gnathophausia doryphora
| Illig G. 1906: 227 |
Gnathophausia calcarata
| Ortmann, A. E. 1906: 30 |
| Sars, G. O. 1883: 5 |
Gnathophausia
| Gunalan, B. & Kumar, C. P. 2014: 123 |
| Meland, K. & Aas, P. O. 2013: 203 |
| Price, W. W. & Heard, R. W. & Aas, P. & Meland, K. 2009: 923 |
| Casanova, J. - P. & Joung, L. D. & Faure, E. 1998: 60 |
| Casanova, J. - P. 1996: 129 |
| Kathman, R. D. & Austin, W. C. & Saltman, J. C. & Fulton, J. D. 1986: 160 |
| Pequegnat, L. H. 1965: 402 |
| Tattersall, O. S. 1955: 31 |
| Tattersall, W. M. 1951: 25 |
| Fage, L. 1941: 15 |
| Tattersall, W. M. 1939: 224 |
| Illig G. 1930: 407 |
| Sars, G. O. 1883: 4 |
Lophogaster ingens
| Dohrn, A. 1870: 610 |
1 (by plazi, 2019-04-05 08:24:52)
2 (by ImsDioSync, 2019-04-05 09:15:36)
3 (by ExternalLinkService, 2019-09-25 22:56:55)
4 (by ExternalLinkService, 2019-10-09 10:49:23)
5 (by ExternalLinkService, 2019-10-18 15:46:26)
6 (by ExternalLinkService, 2019-10-19 17:36:16)
7 (by ExternalLinkService, 2022-01-29 13:24:19)
8 (by GgImagineBatch, 2022-04-30 04:52:20)
9 (by plazi, 2023-10-30 13:06:39)