Mesene jimena, Callaghan, Curtis John & Llorente-Bousquets, Jorge, 2011
|
publication ID |
https://doi.org/10.5281/zenodo.277698 |
|
DOI |
https://doi.org/10.5281/zenodo.5679627 |
|
persistent identifier |
https://treatment.plazi.org/id/03B087D0-B32A-2519-FF07-6CF3FA1EF95E |
|
treatment provided by |
Plazi |
|
scientific name |
Mesene jimena |
| status |
sp. nov. |
Mesene jimena sp.nov. Callaghan, Llorente & Luis
Figures.1–4 View FIGURES 1 – 15. 1, 2 , 16–18 View FIGURES 16 – 21 , 22-23 View FIGURES 22 – 26
Description. MALE. ( Figs.1, 2 View FIGURES 1 – 15. 1, 2 , 16-18 View FIGURES 16 – 21 ) Forewing length of HT 12.2 mm, range of material measured 9.74–14.18 mm. n=31, average 13 mm.
Wing shape: Forewing elongated, costa curving slightly before the apex, anal margin slightly curved to anal angle, distal margin slightly convex to M1 before the apex, where it is slightly indented; hindwing costa elongated, slightly curved to a rounded apex, anal margin slightly curved to tornus, where it connects abruptly to the distal margin, distal margin slightly indented at tornus, then curved to costa.
Wing color, dorsal surface: Ground color of both wings uniform light orange, hindwing slightly lighter at the anal margin and costa, fringe of both wings black with scattered lighter scaling between the veins; Wing color, ventral surface: Ground color of both wings lighter than dorsal surface; forewing lighter towards inner margin; apex and costal margin with variable, discontinuous black scaling, distal margin with variable black, slightly elongated triangular markings at the end of the veins, diminishing in size from the apex, fringe black with lighter scaling between the veins. Hindwing margin with slightly elongated black triangular markings along the veins.
Head: Eyes naked; marginal scaling, frons and labial palpi white, short, antennal length about 60% of forewing length, segments black with some white scaling near clubs; clubs elongated, tips paler.
Body: Dorsal color of thorax and abdomen uniform light orange, collar black; ventral surface white, thorax with black scaling where femur folds against it, forelegs white, midlegs femur white, trochanter, tarsal segments black, and hindlegs white with some black on tarsal segments; abdominal tergites with elongated transverse androconial scale pads on the margins of segments 4–7. Abdomen ventrally white with small black marks between the segments.
Genitalia ( Figs. 16, 17, 18 View FIGURES 16 – 21 ) Uncus in lateral view slightly pointed, lobes of uncus slightly squared, broadly separated; ventral lobe of tegumen rounded; scaphus rounded when viewed ventrally, not projecting caudad when viewed laterally, lightly sclerotized; falces long, top bent caudad; vinculum fused to top center of tegumen, uniformly narrow, straight to the level of the valvae, then curved in pronounced “s” shape to saccus and bulging laterally; saccus anteroventral, continuation of vinculum, flat, broad and squared; valvae in lateral view narrow, ending in sharp tips, pointing caudad; in ventral view, valvae bulge slightly to base of pedicel, tips pointed caudad; inner portion of valve fused dorsally by a simple transtilla to opposing valve; aedeagus short, wide, vesica prominent with three groups of long spines pointing caudad when retracted ( Fig.17 View FIGURES 16 – 21 ), one dorsal and two lateral; pedicel broad, short, protruding only slightly beyond valvae at base.
FEMALE ( Figs.3, 4 View FIGURES 1 – 15. 1, 2 , 22, 23 View FIGURES 22 – 26 ): Forewing length average of material measured: 14.32 mm, n=7.
Wing shape: Forewing longer than male with distal margin more curved.
Wing color, dorsal surface: Ground color of both wings uniform light orange, paler than male, costa with marginal black scaling before apex, fringe on both wings black with lighter areas between veins.
Wing color, ventral surface: Ground color of both wings paler orange than dorsally, paler towards anal margin, forewing costa with black scaling, distal margin with small, triangular marginal black marks along the veins, decreasing in size from apex to tornus, distal fringe black at ends of veins, lighter between; hindwing ground color light orange, distal margin with more or less uniform elongated black triangular spots at the ends of the veins, fringe black at ends of veins, lighter between them.
Head: As in male.
Body: Coloration as in male.
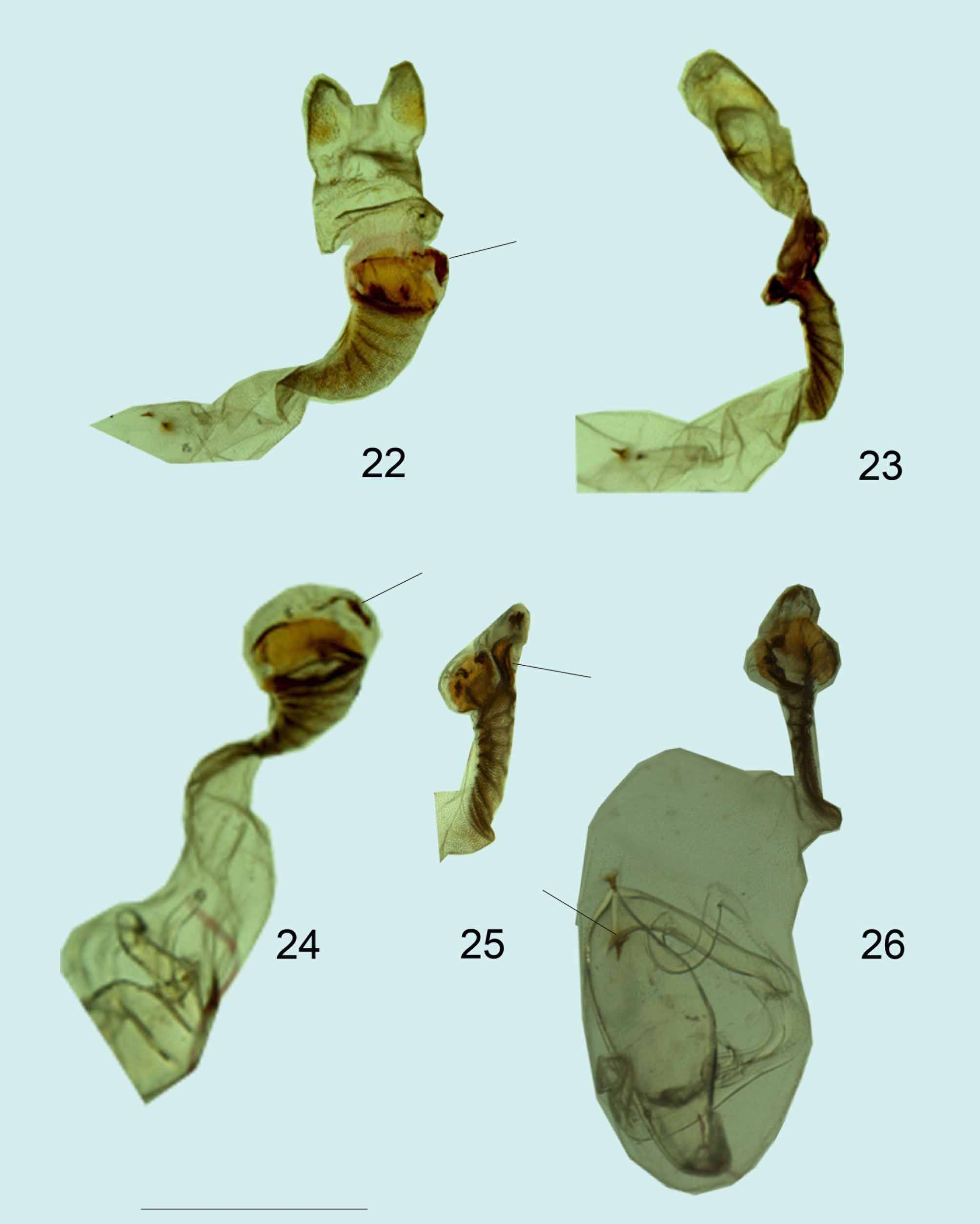
Genitalia ( Figs. 22, 23 View FIGURES 22 – 26 ) Corpus bursae slightly elongated, with two small, invaginate, pointed signa, one longer; ductus bursae short, funnel shaped, bends strongly to left when viewed ventrally, sclerotized with transverse striations, and flat when viewed laterally; ductus seminalis is attached to the right side of the ostium bursae, viewed ventrally; ostium bursae consists of two plates when viewed laterally, the ventral plate larger than the dorsal, slightly flared, round, and bent ventrally; on the right side of the rim when viewed ventrally, is a strongly sclerotized horseshoe-shaped structure; the dorsal plate with amorphous pieces of sclerotized scaly material around the rim in some specimens.
Variation. There is some individual variation in the length of the black marks between the veins on the ventral surface, sometimes absent, vestigial or slightly longer. Also, some specimens lack completely the vestigial black scaling on the ventral abdomen between the segments ( Fig.4 View FIGURES 1 – 15. 1, 2 ).
Types. 57 specimens, all from Mexico; 1 holotype, 1 allotype and 55 paratypes. Holotype male. MEXICO: OAXACA El Azulillo, Candelaria, Loxicha, 29/11/ 2007, 400m MZFC, J. Llorente-A. Luis leg. and a red type label. Allotype female. Río Santiago, Atoyac de Álvarez, Gro, 8/viii/1985 G. Rodríguez; second label ‘Bosque Tropical subcaducifolio–cafetales 680 msnm’, third label ‘Museo de Zoología Lepidoptera 44534, and a red type label. Paratypes: GUERRERO: 36 specimens, 293, Río Santiago Atoyac de Álvarez, 680 m, MZFC, with the following dates: 1 specimen 27/07/1984 (44536), 2 specimens 28/07/1984 (44530, 44541), 3 specimens 16/09/ 1984 (44521, 44523, 44529), 6 specimens 17/09/1984 (44512, 44513, 44514, 44515, 44516, 44517), 6 specimens 18/09/1984 (3136, 44507, 44508, 44509, 44510, 44511), 1 specimen 07/12/1984 (44537), 1 specimen 08/12/1984 (44532), 2 specimens 06/08/1985 (44518, 44533), 1 specimen 07/08/1985 (44519), 1 specimen 08/08/1985 (44524), 1 specimen 09/09/1985 (44535), 2 specimens 22/10/1985 (44526, 44528), 1 specimen 27/10/1985 (44539), 1 specimen 24/11/1985 (44531); 4ƤƤ with the following dates: 1 specimen 28/07/1984 (44540), 2 specimens 08/08/1985 (44520, 44538), 1 specimen 24/11/1985 (44522), MZFC; 23 Puente de Los Lugardo, Atoyac de Álvarez 900m with followings dates: 1 specimen 20/07/1984 (44527), 1 specimen 29/x/1983 (44525) MZFC. Acahuizotla, Chilpancingo de los Bravos, 17/12/1971, Adolfo Ibarra Vázquez leg., 1 specimen, 17/12/1971 CNIN (LEP 25841). OAXACA: 19 specimens, 9 from Azulillo-Río Candelaria, Candelaria Loxicha, 400m, MZFC, J. Llorente-A. Luis leg. 3 specimens 1Ƥ y 13 24/09/2007, 1Ƥ 22/09/2007; 33 El Azulillo, Candelaria Loxicha, 400 m MZFC, J. Llorente-A. Luis leg. 3 specimens with the following dates: 29/11/2007, 27/08/2008, 20/10/2008. Candelaria Loxicha, Adolfo Ibarra Vázquez leg. 2 specimens 1/5/1977 CNIN (LEP 25840, LEP 25844), 1 specimen 2/12/1982 CNIN (LEP 25843); Jesús Saldaña Martínez leg. 1 specimen 27/1/1977 CNIN (LEP 25842); 6 specimens, 23 4ƤƤ Candelaria Loxicha, 500 m, 15/11/1980, (CJC).
The holotype and allotype are deposited in the MZFC. The deposition of the paratypes is indicated by the acronyms or collector.
Etymology. This lovely species is named in honor of Ms Jimena Llorente, wife of the second author.
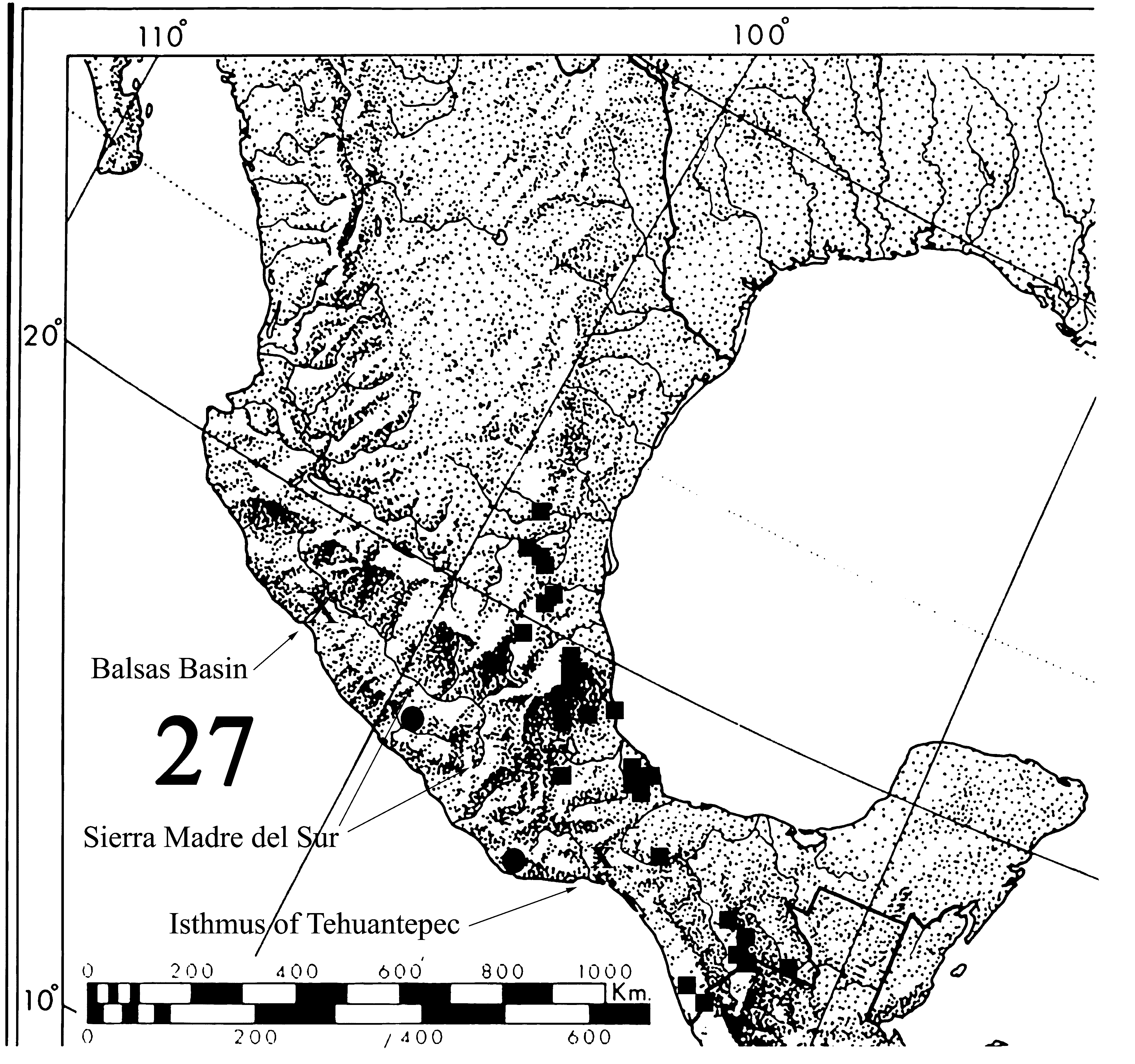
Distribution. ( Fig. 27 View FIGURE 27 ) The species is found in the coastal areas of Guerrero and Oaxaca west of the Sierra Madre del Sur on the Pacific slope.
Habits and habitat. Va rga s et al. (1991, 1994) described the allotype locality, Río Santiago in Sierra de Atoyac, Guerrero: An area at the base of the mountain between 660 and 700 m, somewhat disturbed by coffee plantations which originally was an evergreen tropical forest with riparian vegetation along the rivers Santiago and Las Delicias. The climate is Aw2 (w), very humid with annual precipitation more than 1000 mm. The combination of forest and coffee plantations creates partially shaded microhabitats. At 900 m and joining this locality is the Puente de los Lugardo.
The locality of Azulillo (Candelaria Loxicha, Oaxaca) belongs to a region known as evergreen and semi-evergreen tropical forest, which also contains disturbed cultivated areas, with riparian vegetation along the Río Candelaria. The altitude is lower, between 370 and 500 m, but with the same climate.
M. jimena is on the wing early in the morning between 0800 to 1000 h, and again after 1400 to 1630 h, often in mist or drizzle. They rest on the undersides of large leaves with wings outspread and antennae together. They fly with a mimic unidentified species of Heterocera. The flight is weak and difficult to see in the shade, generally above 3 meters. They fly for periods of 10 seconds before alighting.
Biology. Unknown.
Diagnosis. Hall and Lamas (2007) include the southern Mexican populations of Mesene jimena sp. nov., as conspecific with a polymorphic Mesene margaretta . Maza (1987), Vargas et al. (1991, 1994) and Luis et al. (2004) also were confused by this species ( v. gr. Chimastrum margaretta , Mesene oriens ssp., Mesene margaretta ssp.).We contend, however, that Mesene jimena sp. nov. is a valid species, despite morphological similarities in the genitalia and the existence in both of 4 dorsal abdominal androconial scale pads. We recognize four classes of criteria that can elucidate the question of the species status of Mesene jimena sp. nov. as well as allopatric populations generally. These are morphology, temporal/spatial barriers, biogeographical factors and ecological differentiation. Each of these will be examined in turn with respect to the species status of Mesene jimena sp. nov.
Morphology. Mesene jimena sp. nov. can be easily separated from M. margaretta by the following: The dorsal wing surface of both sexes is uniform light orange-yellow instead of red and with no elongated pointed black lines extending from the apex along the veins, separated (or not) by white scaling1. The red flush in the discal area of the hind wing ( Figs 5, 9, 11 View FIGURES 1 – 15. 1, 2 ) typical of M. margaretta males is lacking. Although both sexes of Mesene jimena sp. nov. have black lines between the veins on the ventral surface, they are much shorter than the same markings in M. margaretta . In Mesene jimena sp. nov., the inner margin of the male ventral forewing is lighter ground color, lacking the black bar ( Figs.6, 10, 12 View FIGURES 1 – 15. 1, 2 ) typical of M. margaretta . The black markings between the segments of the ventral abdomen, a prominent character of M. margaretta , are quite variable in Mesene jimena sp. nov., ranging from total absence ( Fig. 4 View FIGURES 1 – 15. 1, 2 ), to vestigial ( Fig.2 View FIGURES 1 – 15. 1, 2 ). The genitalia of the two phenotypes are practically identical, but, discounting individual variation, there are small but consistent differences among the material examined ( 4 males and 4 females of each species). In Mesene jimena sp. nov. the anteroventrad lobes of the tegument are more rounded, not elongated as in M. margaretta ( Figs. 16, 19, 20 View FIGURES 16 – 21 ). In a lateral view, the vinculum of Mesene jimena sp. nov. is more strongly “S” shaped and bowed more sharply closer to the saccus. ( Fig. 16 View FIGURES 16 – 21 ) In M margaretta , it is straighter or more uniformly curved ( Figs. 19, 20 View FIGURES 16 – 21 ). In the female genitalia, the signa of M. margaretta are more uniform in size and larger ( Fig. 26 View FIGURES 22 – 26 ); the dorsal plate in M. margaretta is larger, with a more pronounced cleavage between the plates ( Fig. 25 View FIGURES 22 – 26 ), and the horseshoe shaped structure less sclerotized and more separated from the ventral plate ( Fig. 24 View FIGURES 22 – 26 ).
Space and time. Spatial and temporal barriers relating to perching behavior have been discussed for Neotropical riodinid butterflies ( Callaghan, 1983; Hall 1999; Hall and Harvey, 2002b). In this case it is shown that closely related congeneric riodinid butterflies often perch at different times and microhabitats ( Callaghan, 1983). If two populations are characterized by perching in different times and localities, presumably this would be an effective potential isolating mechanism, thus reducing their interbreeding potential. This is true in the case of Mesene jimena sp. nov. and M. margaretta . Mesene jimena sp. nov. is active during the early morning and late afternoon, and under inclement weather conditions, whereas M. margaretta males perch in treefalls and along trails in the early afternoon in sunflecks. DeVries (1993) reports activity between 0900 and 1100 h in Costa Rica. In Colombia one of us (CJC) has observed males perching between 1100 and 1400 h. In Mexico, they fly from 900 to 1700 h, peaking around 1200 – 1430 h (Llorente and Luis-Martinez, pers. obs.).
Biogeography. Another indication of species status would be a distribution range corresponding to an area of high endemism among butterflies, as well as other organisms. It has been suggested that there is a high correlation between riodinid species and recognized regions of endemism, particularly in the Amazon ( Hall and Harvey, 2002a). The Pacific region of Mexico shows the greatest amount of endemism in the country (paleo and neo-endemism). The northern limits of the distribution of the Neotropical fauna confronted with strong barriers on the Mexican Pacific slope favor speciation. In the diverse tropical and mesophylic mountain forests of this region endemic
1. The variation on the extent of the black and white apical markings on M. margaretta appears to be within populations and not between them. In the northern coastal range of Venezuela (Cordillera de la Costa) in the Provinces of Aragua and Carabobo, both phenotypes shown in Figs. 9–14 View FIGURES 1 – 15. 1, 2 fly together in the same habitat, from 200 to 1550 m. (Juan C de Sousa, pers. com.). Similar differences are found in populations at Cali, Valle del Cauca and Villavicencio, Meta, in Colombia.(CJC, pers. obs.). In El Salvador, the M. margaretta population shows a considerable reduction of the black forewing markings; however there are also individuals with normal markings. In Mexico and Guatemala, the M. margaretta populations ( Figs.5 – 8 View FIGURES 1 – 15. 1, 2 ) show no significant variation.
subfamilies, genera, species and subspecies are found that characterize the distinct provinces which drain into the Pacific. These provinces are South Sonora, Mexican Pacific Coast, South Sierra Madre Occidental, the West of Transmexican Volcanic Belt, Balsas Basin, and Sierra Madre del Sur (Morrone et al., 1999, 2002; Espinosa et al. 2000, 2008). Morrone (2001: 40 – 42) includes the area where Mesene jimena sp. nov. is found in the biotic province called “Provincia de la Costa Pacífica Mexicana”, and he mentions numerous endemics found there. The 40 characteristic taxa that Morrone enumerates for the Provincia de la Costa Pacífica Mexicana include taxa from the south of Sinaloa y Michoacán, as well as from dry areas like the “islas húmedas”, and two from humid areas where Mesene jimena sp. nov. is found.
Previously, Llorente (1984) had delimited an area of endemism as a sub-region of this province, considering only Oaxaca and Guerrero on the Mexican Pacific Coast between the isthmus of Tehuantepec, a very dry region serving as a barrier between Oaxaca and Chiapas, and the dry Balsas basin on the coast to the northwest. ( Fig. 27 View FIGURE 27 ). To the north, the area is separated by the heights of the Sierra Madre del Sur of over 2000m. This area of endemism is closely related with the Province of the Sierra Madre del Sur, but the limits between both are forests between 1000 and 2000 m that include holm oak forests and cloud forests of mid elevation, as characterized in Llorente and Escalante (1992).
These two barriers also limit many other species of Papilionoidea. For Riodinidae , this region supports endemism on the level of genus and species. Examples are Lamphiotes velazquezi, ( Beutelspacher, 1976) , Callaghan, 1982; Calephelis acapulcoensis, McAlpine, 1971 , Exoplisia azuleja, Callaghan, Llorente & Luis, 2007 , and Theope villai . Beutelspacher, 1981. ( Beutelspacher 1976, Llorente-Bousquets 1988, Llorente-Bousquets & Escalante 1992, Llorente-Bousquets & Luis-Martínez 1992, Llorente-Bousquets et al. 1992, 1993, 2006, Vargas et al. 1996, Luis-Martínez et al. 1996, 2003, Austin et al. 2007, Callaghan et al. 2007).
In contrast, Mesene margaretta ranges from eastern Ecuador ( Hall and Lamas, 2007); Colombia on both slopes of the Andes, the northern Andes of Venezuela, then north through Central America to Mexico (CJC). The two species, M. margaretta and Mesene jimena sp. nov. are allopatric in Mexico, and specimens examined show no signs of interbreeding, nor has a blend zone been discovered despite intensive collecting. Mesene margaretta is found from Chiapas to San Luis Potosí on the Atlantic slope. ( Fig. 27 View FIGURE 27 ) Records from Mexico are shown below from a number of museum collections. Data with the locality of Candelaria, Oaxaca is in error. The material was gathered from local collectors by Edward Welling, a butterfly dealer, and probably represents other localities. Extensive collecting in the Pacific slope in Oaxaca by the junior authors over the last 30 years has failed to record Mesene margaretta from this area.
Mexican specimens of M margaretta examined. A total of 240 specimens. MEXICO. SAN LUIS POTOSÍ: La Mera Ceiba, Tamazunchale 23 Jan. ( MZFC); HIDALGO: Pilcuatla, Tlanchinol 1Ƥ May ( MZFC); PUEBLA: Barranca de Patla 23 Feb., 23 July, 13, 1Ƥ Aug. 133, 3ƤƤ Sept., 13 March, La Ceiba, Francisco Z. Mena 23 Jan. ( MZFC); 33 July, 13, 1Ƥ Aug., 1Ƥ Sept. ( CNIN); Tequesquitla 23 May, 43 June, 23 July, 43, 1Ƥ Aug., 23, 2ƤƤ Sept., 23 Oct., 23 Dec. ( MZFC); 63, 2ƤƤ July, 13 Aug., 273, 12ƤƤ Sept., 93, 1Ƥ Oct., 33 Nov. ( CNIN); VERACRUZ: Barranca de Cayoapa, Tejería, Teocelo 23 Feb., 13, 1Ƥ Aug. 53, 1 Ƥ Sept. ( MZFC); Camino a Santa Rosa, Teocelo 43 Sept., 23 Dec. ( MZFC); Catemaco 2 specimens April, 4 specimens Aug. ( MGCL); 13 Sept. ( CNIN); 13 Nov. ( MZFC); Coatepec 2 specimens ( USNM); Córdoba 2 specimens Jan. ( USNM); Cuetzalapan 1Ƥ July, 13 Aug. ( CNIN); Dos Amates, Catemaco 13 Dec. ( MZFC); El Puente San Marcos 73 Aug., 13 Sept., 13 Oct. ( MZFC); El Puente Texolo, Teocelo-San Marcos 23 July, 63, 1Ƥ Aug., 73 Sept., 13 Oct., 23 Nov. ( MZFC); Fortín de las Flores, Jardín Moctezuma 2 3 17 -IX-49, 21 -XII-68; 43, 2ƤƤ Jan., 13 Sept., 13 Dec. ( CNIN); 23 Dec. ( MZFC); Las Juntas, Llano Grande 13, 1Ƥ Jan. ( MZFC); Presidio, Ixhuatlán del Café 23, 1Ƥ VIII (40, 47, 50), 13, 1Ƥ (42, 45), 13 VII-41 ( MGCL); Catemaco 1Ƥ IV-57, 13, 1Ƥ VIII-57 ( MGCL); 2 specimens March. 6 specimens April, 2 specimens May, 2 specimens July, 2 specimens Aug., 6 specimens Sept. ( AMNH); Santa Rosa 1 specimen May ( USNM) Tapalapan, Santiago Tuxtla 13, 1Ƥ Aug. ( MZFC); Teocelo 13, 1Ƥ Aug. ( MZFC); Zapoapan de Cabañas 1Ƥ Feb. ( CNIN); “Veracruz”, 13 26- IX-1962 ( MGCL). OAXACA: Metates, Santiago Comaltepec 23 May ( MZFC); La Esperanza, Santiago Comaltepec 1Ƥ Mar., Soyolapan El Bajo, Santiago Comaltepec 13 Jan. ( CNIN); CHIAPAS: Escuintla 1 specimen July ( MGCL); 4 Mi. N Cacahoatán on rd. to Unión Juárez nr. Faja de Oro, 1Ƥ 26-IX-1962 ( MGCL); Cacahoatán 1 specimen June ( AMNH); Finca "Guatimoc" 1 specimen July ( AMNH); San Antonio Buenavista (Santa Rosa), Independencia 13 Oct. ( MZFC); Santa Rosa 1 specimen May ( USNM); Unión Juárez 1Ƥ April ( MZFC); Puente Shjubal, 1Ƥ 26-VIII-80 ( MGCL).
Other material examined. GUATEMALA: 23 San Felipe 2000’ Volcan Santa María ( MGCL); 1Ƥ Finca San Rafael, Olimpo Sud 3-VII-65; 13, 1Ƥ Alta Verapaz Tamalhu 1100m 30-VI-63 ( MGCL). EL SALVADOR: 23 La Libertad, 103 22-XI-69 ( MGCL); 63, 2ƤƤ Santa Tecla 7-VI-69 ( MGCL); 1Ƥ San Salvador 2-XI-71 ( MGCL). COSTA RICA: Turrialba, Cartago 16-VII-65 ( MGCL); PANAMA: 13 Cerro Campana, (CJC); C OLOMBIA: 13 1Ƥ VIII Boyacá 400m S. Luis de Garmo 26-II-06; 13 Finca Guayabetal Cundinamarca, 14-II- 92 (CJC); 33 Cunday, Tolima, 22-VIII-93; 13, 1Ƥ, Villavicencio, Meta 5-I-94 (CJC); Santa Cecilia, Risalda vii- 91 (CJC); 13 Río Negro Huila, (CJC); 23 Ciudad Jardín, Cali, Valle, (CJC). VENEZUELA: 13 Palmachal 760m 9-IV-05 (JS); 13 Río Carragua, Barinas 9-II-02 (JS); 13 Barinitas, Barinas, 1I-85 (JS); 1Ƥ Cuyagua, Aragua 23-II-83 (JS); 1 3 Rancho Grande Aragua 1100 28-VI-54. (JS); 13 Pozo del Diablo, Maracay 500m 6-II-86 (JS); Las Trincheras, Carabobo 27-VI-52 (JS); 3ƤƤ Rancho Grande Aragua 1100m VIII-58, VI-65, IX-52 (JS); 1Ƥ El Limón 600m 9-IX-79 (JS); 1Ƥ El Piñal, El Limón VIII-76 (JS); 43 Palmichal, Carabobo 760m 4-V-04 (JS); 13 Parque Nacional Lara Yacambu, El Blanquito 1300m 20-XII-92 (JS); 33 Palmichal 800m Carababo 20-IX-03, 27-VI-06 (JS); 33 El Limón Aragua I-76, I-59, IX-53, (JS); 1Ƥ Palmichal Carabobo 76m IV-03, (JS); 13 Choroni Aragua, 200m 1-V-04 (JS).
Ecology. Mesene jimena sp. nov. is confined to evergreen and semi-evergreen humid forest of Guerrero and Oaxaca Pacific slope below 700 m. It appears this species is associated only with this vegetation and high humidity. Conversely, M. margaretta in Mexico inhabits dry seasonal forest and cloud forest from 400 to 1350 m. In accordance with Llorente (1984, 1996) and Llorente and Escalante (1992) the differentiation of species and subspecies levels in the Mexican Pacific depends on their range of habitat. When they are closely tied to a limited altitudinal range and habitat, their isolation is more complete and the possibilities of divergence to the species level are greater. Conversely, if their distribution is wide and unlimited by climatic and altitudinal barriers, their differentiation between populations is less, as in the case of Pereute charops sspp., Dismorphia amphiona sspp., Rhetus arcius sspp. and M. margaretta , all of which have greater altitudinal and climatic distribution than Mesene jimena sp. nov.
The following table summarizes the differences between the two taxa.
The extent to which the aforementioned criteria will apply to other groups must be tested in practice, with supportive evidence. However, in the case of Mesene margaretta , and Mesene jimena sp. nov., they appear applicable.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |