Givotia rottleriformis, Griff.
|
publication ID |
https://doi.org/ 10.1016/j.phytochem.2016.03.002 |
|
DOI |
https://doi.org/10.5281/zenodo.10530380 |
|
persistent identifier |
https://treatment.plazi.org/id/03ABE15A-FF83-FFC9-6346-AF4674E8FD01 |
|
treatment provided by |
Felipe |
|
scientific name |
Givotia rottleriformis |
| status |
|
2.1. Identification of benzoylsalicylic acid in seed coats of G. rottleriformis View in CoL
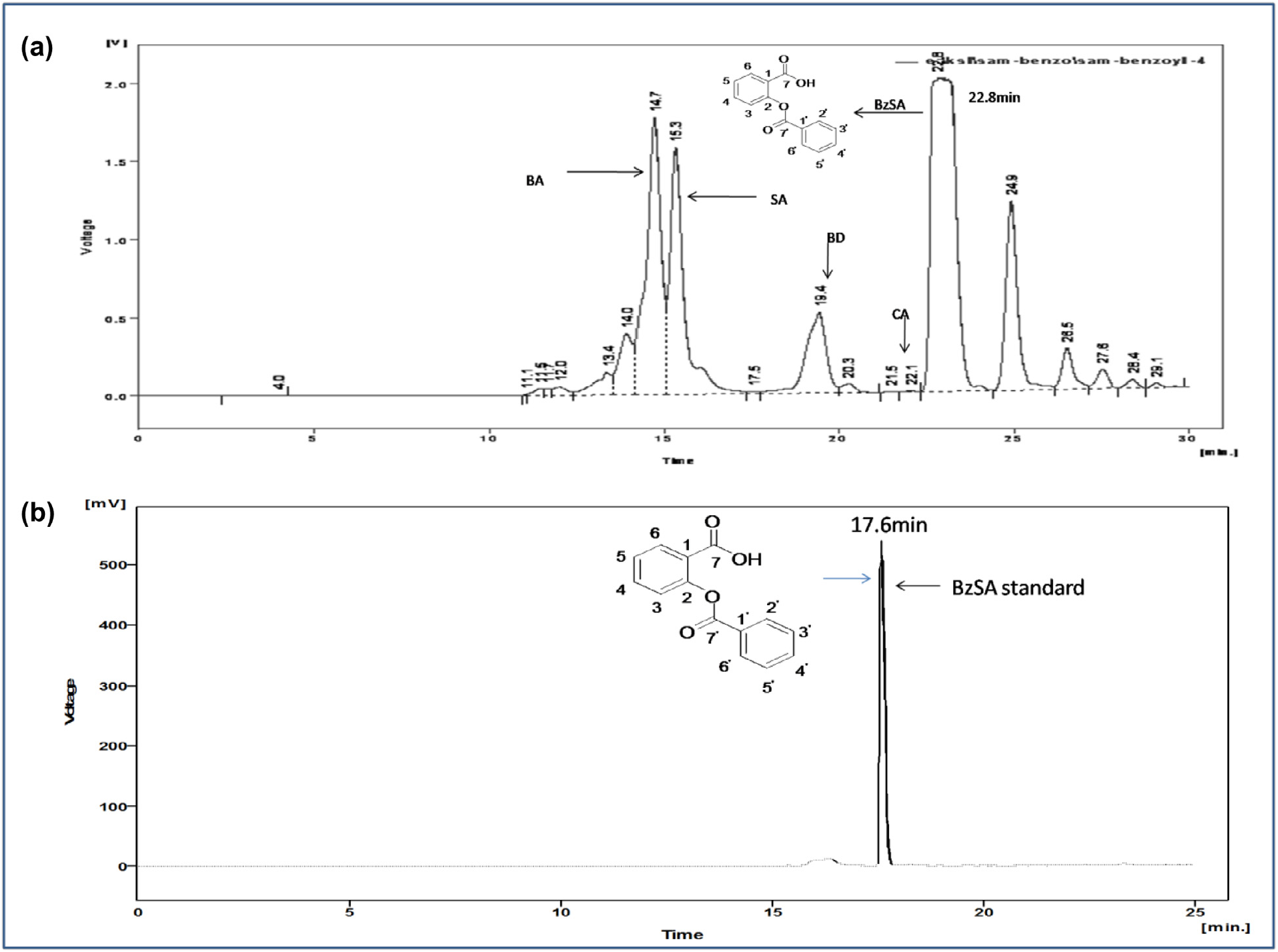
We extracted the total seed coat compounds in methanol (MeOH) and the methanolic crude seed coat extract was fractionated by open silica column chromatography (see Experimental procedure, Section 4.3.1) and the eluted fractions (1–7) were tested for their SAR inducing bioactivities against TMV in tobacco (Supplementary Table S2 View Table 2 ). Among all the fractions, a fraction number 3 was effective in reducing the development of TMV-induced lesion number and diameter. This active fraction showed a group of peaks with different retention times (RT) when resolved by RP-HPLC ( Fig. 1a View Fig ). The major compound of the peak from fraction 3 that eluted at RT 22.8 min ( Fig. 1a View Fig ) was purified and tested for its purity by analytical HPLC with RT at 17.6 min ( Fig. 1b View Fig ). The purified compound was found to be active against TMV and was characterized as benzoylsalicylic acid (CCDC with accession number 90056) using single crystal X-ray diffraction analysis (Supplementary Fig. S2a and b View Fig ). Further structural analysis of purified BzSA was carried out using IR and NMR (Supplementary Fig. S3a–c View Fig ). The mass of the purified BzSA was determined by GC–MS/MS as 242 Da (Supplementary Fig. S4 View Fig ).
Although the seed coats of Givotia are a rich source of BzSA (0.5 mg /gm DW), the other parts like leaves (0.1 mg /gm DW) and bark (0.15 mg /gm DW) also contained this compound (Supplementary Fig. S5a and b View Fig ). The higher accumulation of BzSA, a phenolic compound, in seed coats of G. rottleriformis could be due to its importance in seed germination, seedling growth and interaction with soil microbes. It is well known from the literature that phenolics function as signals in plant–microbe interactions ( Raskin, 1992). Emerging evidence implicates the role of SA in seed germination, flowering, thermogenesis, plant growth and development, and tolerance to abiotic stresses such as drought, chilling, heavy metal toxicity, heat and osmotic stress in plants ( Khan et al., 2015; Rivas-San Vicente and Plasencia, 2011).
The chemical structure of BzSA along with SA and ASA are depicted in Fig. 2a–c View Fig . The possibility of BzSA biosynthesis depends on the availability of free SA and benzoyl-CoA ( Fig. 2d View Fig ). Literature suggests that SA and benzoyl-CoA are present in plants and the biosynthesis of SA in plants takes place via CoA-dependent or independent way, and during this process benzoyl-CoA is formed as intermediate by the oxidation of cinnamic acid ( CA) to benzoic acid ( BA) ( Ribnicky et al., 1998). Previously, it has been reported that the majority of the endogenously synthesized SA are rapidly converted and stored as biologically inactive derivatives via glucosylation and methylation since accumulation of SA has adverse physiological consequences ( Dempsey et al., 2011; Park et al., 2007; Vlot et al., 2009). Recent studies have shown 2,3-dihydroxybenzoic acid (2,3 DHBA) as a hydroxy derivative of SA ( Zhang et al., 2013). However, the benzoylation of SA has not been reported so far. In this study, we report benzoylation of SA for the first time in plants.
| NMR |
Natuurhistorisch Museum |
| SA |
Museum national d'Histoire Naturelle, Laboratiore de Paleontologie |
| CA |
Chicago Academy of Sciences |
| BA |
Museo Argentino de Ciencias Naturales Bernardino Rivadavia |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.