Aspidonema kashmirensis, Nazir & Tahseen, 2020
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4780.1.9 |
|
publication LSID |
lsid:zoobank.org:pub:F35ACEFE-02C9-4B3F-AD23-8FD1D299129A |
|
DOI |
https://doi.org/10.5281/zenodo.3853007 |
|
persistent identifier |
https://treatment.plazi.org/id/D51A8797-FFDC-FFFF-FF7D-FCF9FEA29A65 |
|
treatment provided by |
Plazi |
|
scientific name |
Aspidonema kashmirensis |
| status |
sp. nov. |
Aspidonema kashmirensis sp. nov.
( Figs. 1–5 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 View FIGURE 5 )
Measurements. Table 1.
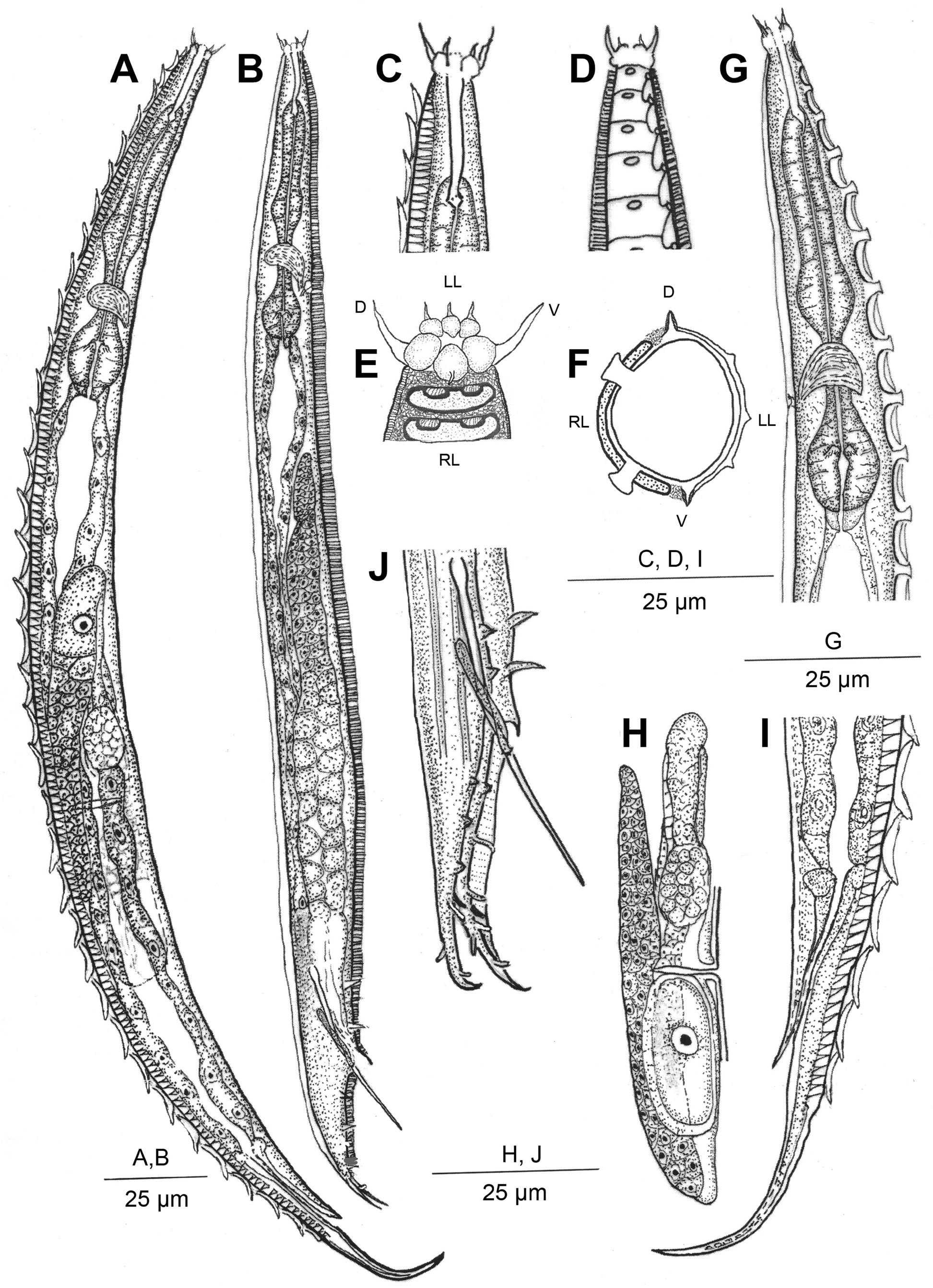
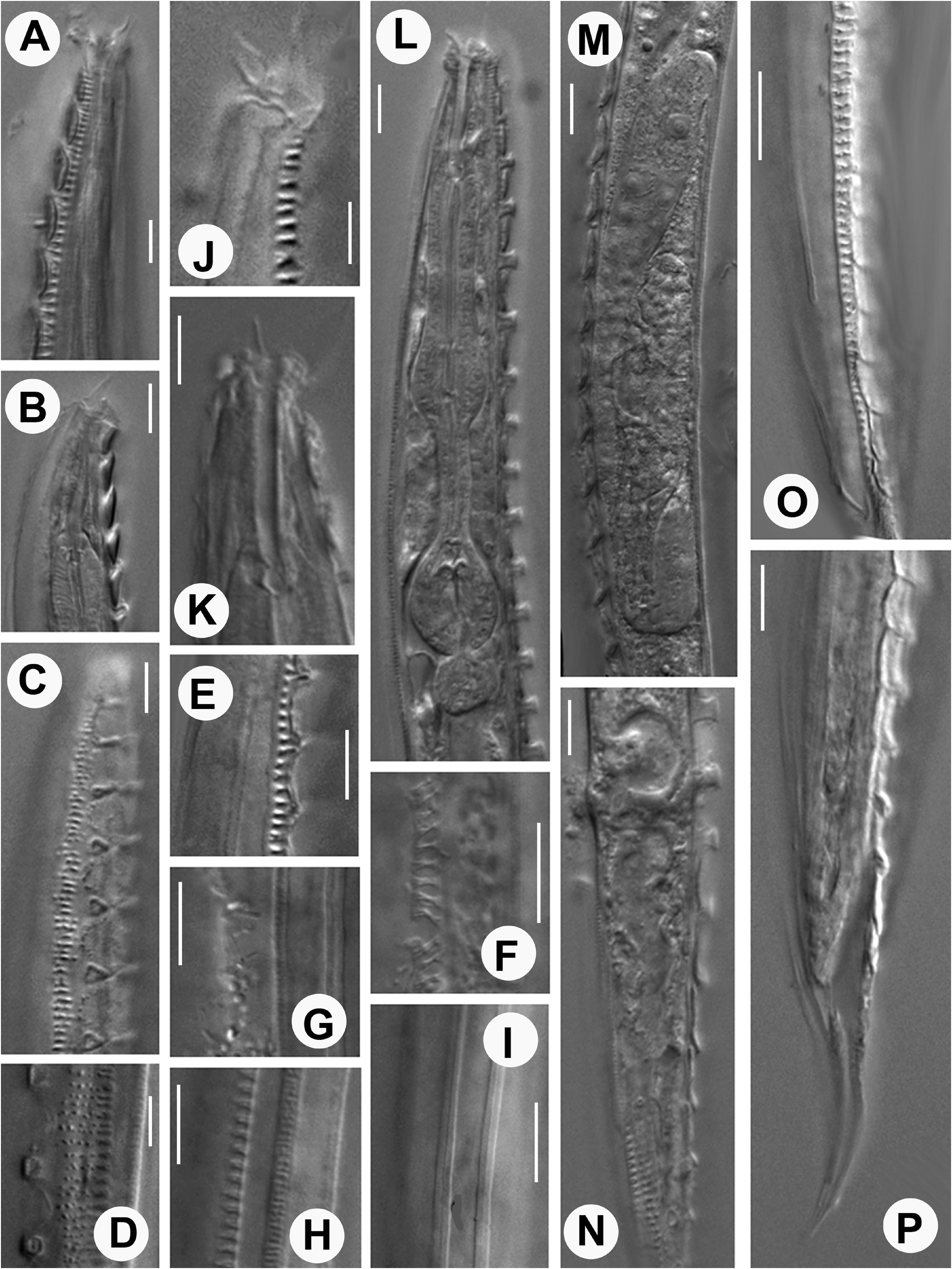
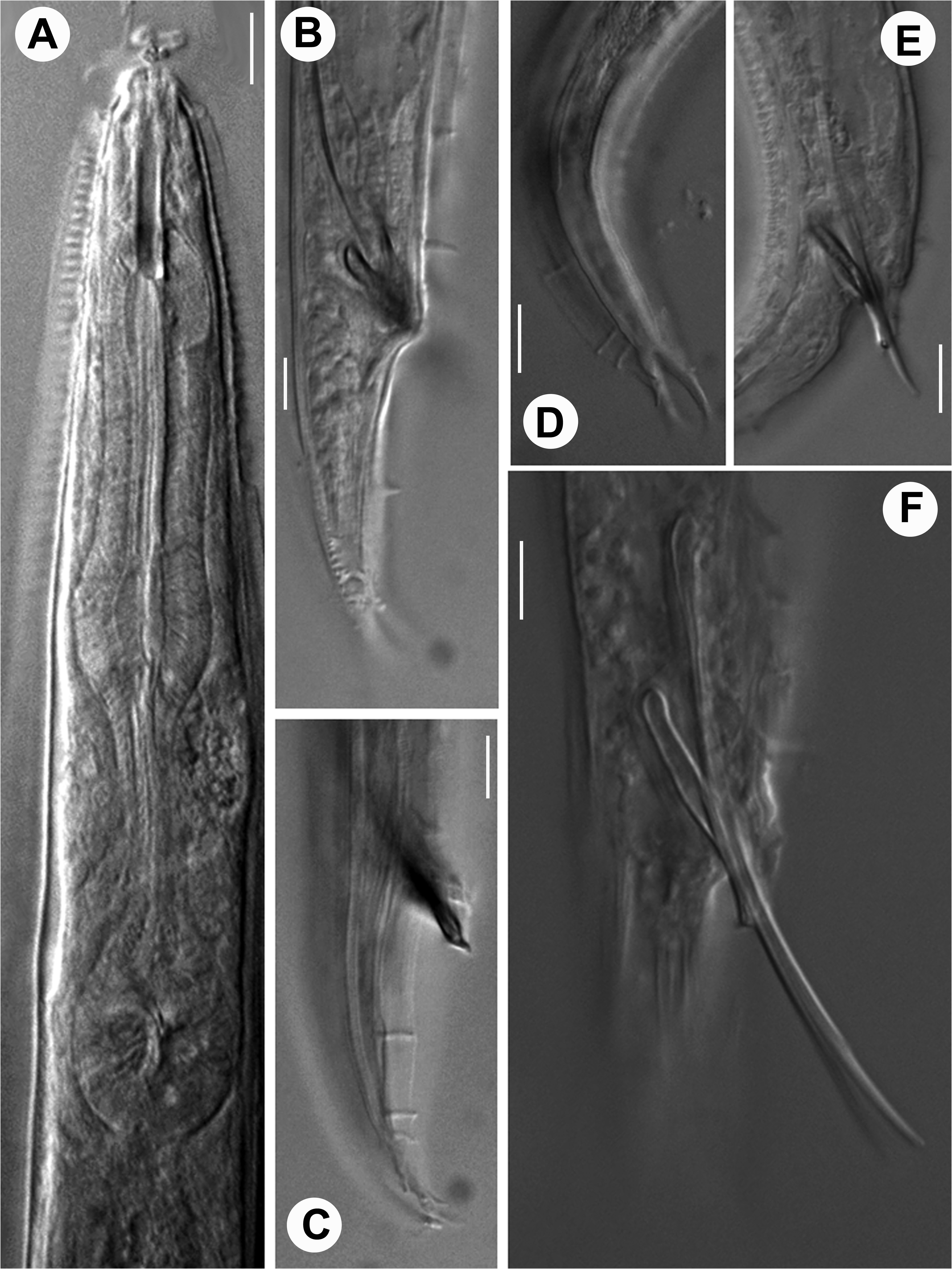
Description. Adult: Small stout nematodes, body slightly curved upon fixation. Body asymmetrical, with right side strongly ornamented and left side provided with longitudinal ridges. Right side provided with transversely elongated-ovoid shields of 14–16 µm x 4–5 µm dimension, interspersed with a reticulate cuticular network. Network provided with small polygonal cavities, bordering the shields along ventro-sublateral and dorso-sublateral sides. Shields porous with numerous fine pores present on surface. Pre-equatorial shields display anterior margins slightly overlapping the preceding shield while those post-equatorial in position show posterior margins overlapping the proceeding one. Each shield with a pair of elliptical grooves/sockets to accommodate cuticular warts emerging from cuticle underneath. Warts in 32–47 pairs, arranged in two rows starting from anterior part to the posterior part of the nematode. Warts 2–3 µm high; placed 3–5 µm apart from each other; a few anterior and posterior warts smaller than those on rest of the body; anteriorly placed warts with a slightly bent anterior edge while the posteriorly placed warts with a slightly bent posterior edge. Cuticular shields porous with strongly sclerotized borders; adhering to 6–7 underlying cuticular annules which show peg-like remnants after shedding of shields; reticulate network surrounding the shields from all sides. Left side of body with fine transverse striations and 5 conspicuous elevated longitudinal ridges. Each ridge in turn formed of two closely placed thinner ridges giving an impression of three lines under LM. Both outer ridges on mid-dorsal and mid-ventral sides appearing relatively strongly radiated or ribbed under LM. Lips offset, asymmetrical. Right lips slightly larger, raised forming a small flap-like structure with large, thick, seta. Two small, thin setiform extensions present on left lips. Labial papillae obscure.Amphids small elliptical pores indiscernible under LM. Stoma long, tubular, 3–4 times lip region diameter in length. Cheilostom weakly cuticularised, gymnostom with parallel walls, slightly curved in a few specimens in posterior region; metastegostom with fine, nearly indiscernible denticles. Pharynx muscular, with prominent, 43–57 µm long, swollen metacorpus, 10–16 µm long isthmus and a rounded to pyriform basal bulb, 12–19 µm long and 8–15 µm wide with a grinder and single-chambered haustrulum. Pharyngeal sleeve weakly developed, covering base of the stegostom. Nerve ring thick, obliquely surrounding the isthmus at ca 45%–70% of the pharyngeal length from anterior end. Secretory-excretory pore inconspicuous. Cardia 5–7 µm long. Intestine granular with wide lumen. Rectum narrow with rectal glands.
Female: Reproductive system amphidelphic. Ovaries reflexed, with flexures nearly reaching vulva; oocytes arranged in one to two rows at distal end and in single row proximally. Spermatheca ovoid to rounded bearing round to amoeboid sperms. Occasionally a single intra-uterine egg measuring 28–32 µm x 12–14 µm present. Vagina about one-third of the corresponding body width in length; vulva a transverse slit, equatorial to post-equatorial in position; vulval lips protruding; obliterating sublatero-ventral or outermost ridge of the left half. Tail conoid with pointed terminus.
Male: Small-sized body; similar to female in morphology except for bifid tail. Testis monorchic, reflexed. Spicules long, extremely slender, thin and free. Capitulum knob-shaped. Gubernaculum one fourth of the total spicule length, with distal end beak-like or flanged. Tail conoid and terminally bifid. Bifid tail showing asymmetry in bursal flaps. Genital papillae arranged in eight pairs comprising 2 pre-cloacal and 6 post-cloacal pairs. Pre-cloacals relatively larger and more strongly developed than post-cloacals. Unlike the other conical genital papillae, GP4, GP5, and GP6 are tubular and open at the edge of the right bursal flap. Bursa on left side rudimentary or nearly absent and genital papillae relatively smaller. Phasmids between GP5 and GP6.
Type habitat and locality. Mint beetles bearing Aspidonema kashmirensis sp. nov. were collected from a hilly area with geographic coordinates 34° 17’ 55.2336’’ N and 74° 28’ 12.5256’’ E in Sopore , Kashmir, India in the month of July, 2018 GoogleMaps .
Type specimens. Holotype female, ten paratype females and three paratype males on the slide “ Aspidonema kashmirensis sp. nov. no. NIK/1–8” deposited in the nematode collection, Department of Zoology , Aligarh Muslim University, Aligarh, UP, India.
Diagnosis and relationship. Aspidonema kashmirensis sp. nov. is characterised by having its body covered on the right side with a row of transversely-ovoid, porous, cuticular shields with strongly cuticularized margins, bordered by a reticulate network and bearing 32–47 pairs of warts in elliptical grooves; the left side of body is provided with five longitudinal ridges each marked by three lines under LM; stoma occasionally curved; pharyngeal collar surrounding the base of stoma; vulva usually post-equatorial; spermatheca ovoid; uterus occasionally with single intra-uterine egg; males with dorsally reflexed testis; long, very slender and free spicules; gubernaculum distally beak-like or flanged, tail bifid with bursa relatively well developed on right half and reduced on left one and eight pairs of genital papillae comprising 2 pre-cloacal, 6 post-cloacal pairs. Pre-cloacals relatively larger and stronger than post-cloacals. Phasmids between GP5 and GP6.
Aspidonema kashmirensis sp. nov. closely resembles A. weingaertnerae in most morphometric values, but differs in morphological details, although comparisons could not be made on the basis of SEM details due to the lack of SEM data for A. weingaertnerae . The new species differs from A. weingaertnerae in having a relatively more slender body (a = 13.1–20.3 vs 11.2–12.9) and lower b (3.2–4.2 vs 4.1–5.9). A. kashmirensis sp. nov. also differs in having relatively smaller number (32–47 vs 40–45) of wart pairs; shields horizontally ovoid with smooth outline (vs squarish to rectangular with angular outline); each shield having anteriorly-located paired elliptical grooves (vs posteriorly-located paired rectangular indentations) to accommodate warts; presence (vs absence) of metastegostomal denticles and males with relatively longer spicules (40–52 µm vs 38–44 µm in A. weingaertnerae ).
A. kashmirensis sp. nov. differs from A. stammeri in having relatively shorter body (L = 224–337 µm vs 289– 429 µm); relatively lower b (3.2–4.2 vs 4.1–6.0) value; horizontally extended ovoid shields nearly covering the body diameter (vs short ovoid shields covering about 1/2–1/3 of body diameter), shields showing sockets or indentations to accommodate paired warts (vs warts independently placed alternate to shields; fewer pairs (32–47 vs 50–59) of warts; and males with relatively longer spicules (40–52 µm vs 39–42 µm in A. stammeri ).
The new species differs from A. ruehmi in having relatively lower b (3.2–4.2 vs 4.4–6.4) and c (7.2–10.7 vs 10.7–12.9) values; horizontally extended ovoid shields nearly covering the body diameter (vs short rounded to ovoid shields covering about 1/3–1/4 of body diameter), shields showing sockets or indentations to accommodate paired warts (vs warts independently placed alternate to shields; more pairs of warts (32–47 vs 30–34); males with longer spicules (40–52 µm vs 30–34 µm and a greater number of genital papillae [16 genital papillae (excluding phasmids), vs 13 genital papillae in A. ruehmi apud Meyl, 1961 ].
A. kashmirensis sp. nov. differs from A. scheucherae in having a relatively smaller b (3.2–4.2 vs 4.3–4.7) value; horizontally extended ovoid shields nearly covering the body diameter (vs oblong to ellipsoidal shields covering about 1/3–1/4 of body diameter), shields showing sockets or indentations to accommodate paired warts (vs warts independently placed alternate to shields; more pairs of warts (32–47 vs 21–26); males with longer spicules (40–52 µm vs 36–40 µm and greater number of genital papillae [16 or eight pairs (excluding phasmids) vs 10 genital papillae or five pairs in A. scheucherae ].
The new species differs from A. formosa in having relatively lower b (3.2–4.2 vs 4.2–4.6) and c (7.2–10.7 vs 12.3–17.9) values; horizontally extended ovoid shields nearly covering the body diameter (vs short ovoid shields covering about 1/3–1/4 of body diameter), shields showing sockets or indentations to accommodate paired warts (vs warts independently placed alternate to shields); metastegostom with minute denticles (vs without denticles) and males present (vs not found in A. formosa ).
Etymology. The species name ‘ kashmirensis ’ is derived from the region, Kashmir, where the host insect was collected.
Remarks. Nematodes associated with insects show complex life history traits. Phoretic nematodes use insect hosts as a means of transport to a new environments for better food opportunities. Bark beetles and scarab beetles have been found to be common insect associates for phoretic rhabditids and diplogastrids ( Sudhaus 2008). Bunonematids, like other bacteriophagous rhabditids, have been reported from habitats rich in organic matter including moss, decaying wood, humus-rich soil, dung and beetle frass.
The new species A. kashmirensis , isolated from the mint leaf beetle ( Chrysolina herbacea ), is the first report of a bunonematid isolated directly from any beetle in India. The eggs of mint beetles ( Fig. 6 View FIGURE 6 A–C) hatch in April to May, with the larvae feeding on and tunnelling into the mint roots. They pupate in late May or early June and the adults emerge during early July. The adults also feed on mint leaves and cause extensive damage. The association of nematodes with beetles is likely not by sheer chance but rather may involve some cues or chemoreception. Bunonematids are bacteriophagous nematodes and were observed in the present study emerging from the excised guts of mint leaf beetles. These beetles have been reported to harbour a special gut microbial flora that helps detoxify the monoterpenes produced by the mint plant ( Pizzolante et al., 2017), and the nematodes are probably attracted to this microbial population. It is also possible, however, that the nematodes may transport those special bacteria. The late emergence of nematodes (i.e., nearly 8 days after the plating of beetles) suggests a state of advanced endophoresy leading to conditions of necromeny. Endophoresy usually involves the presence of dauer forms inside the body of the host insect ( Sudhaus 2008), which were indeed found in large numbers in the present study.
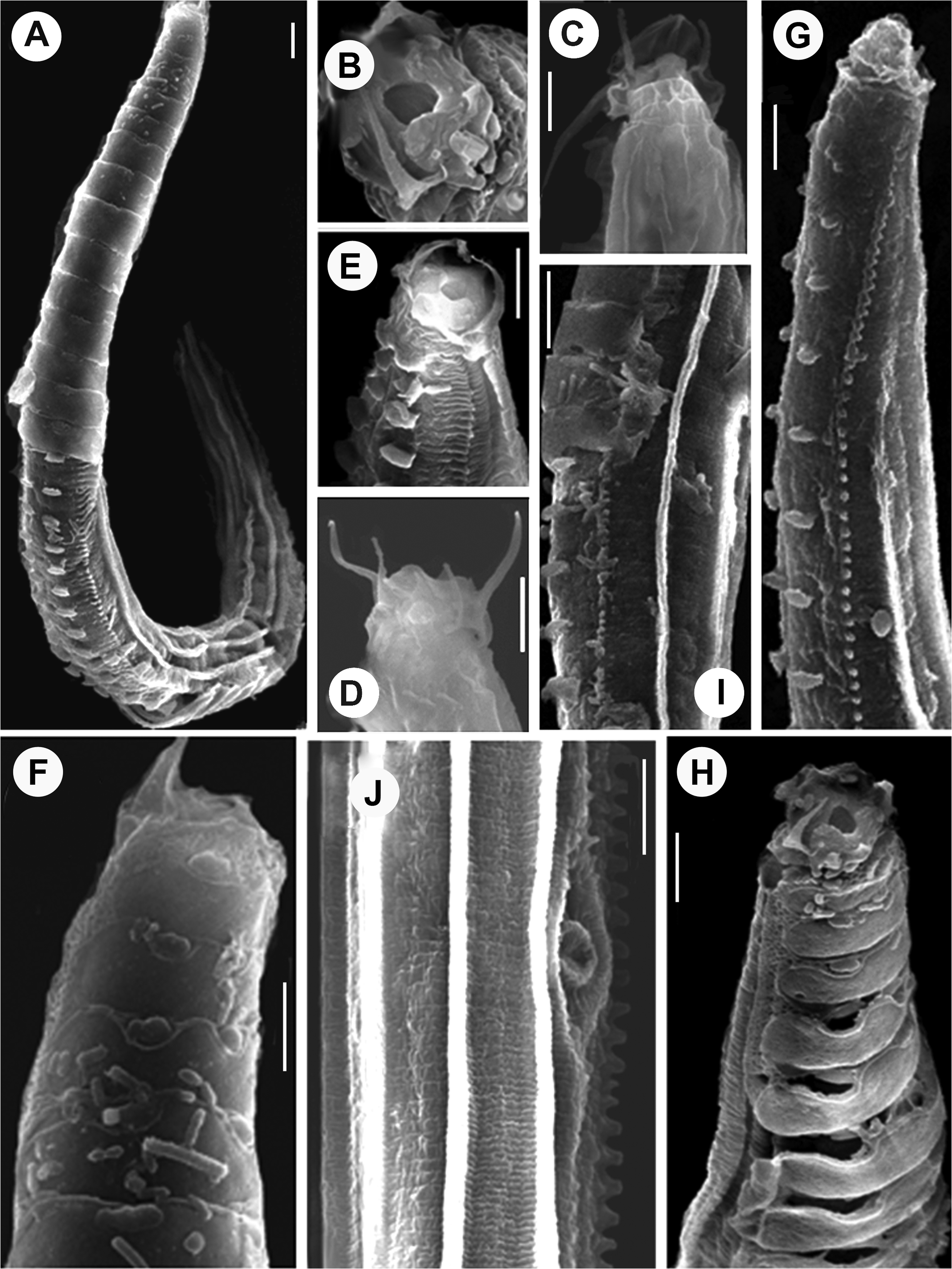
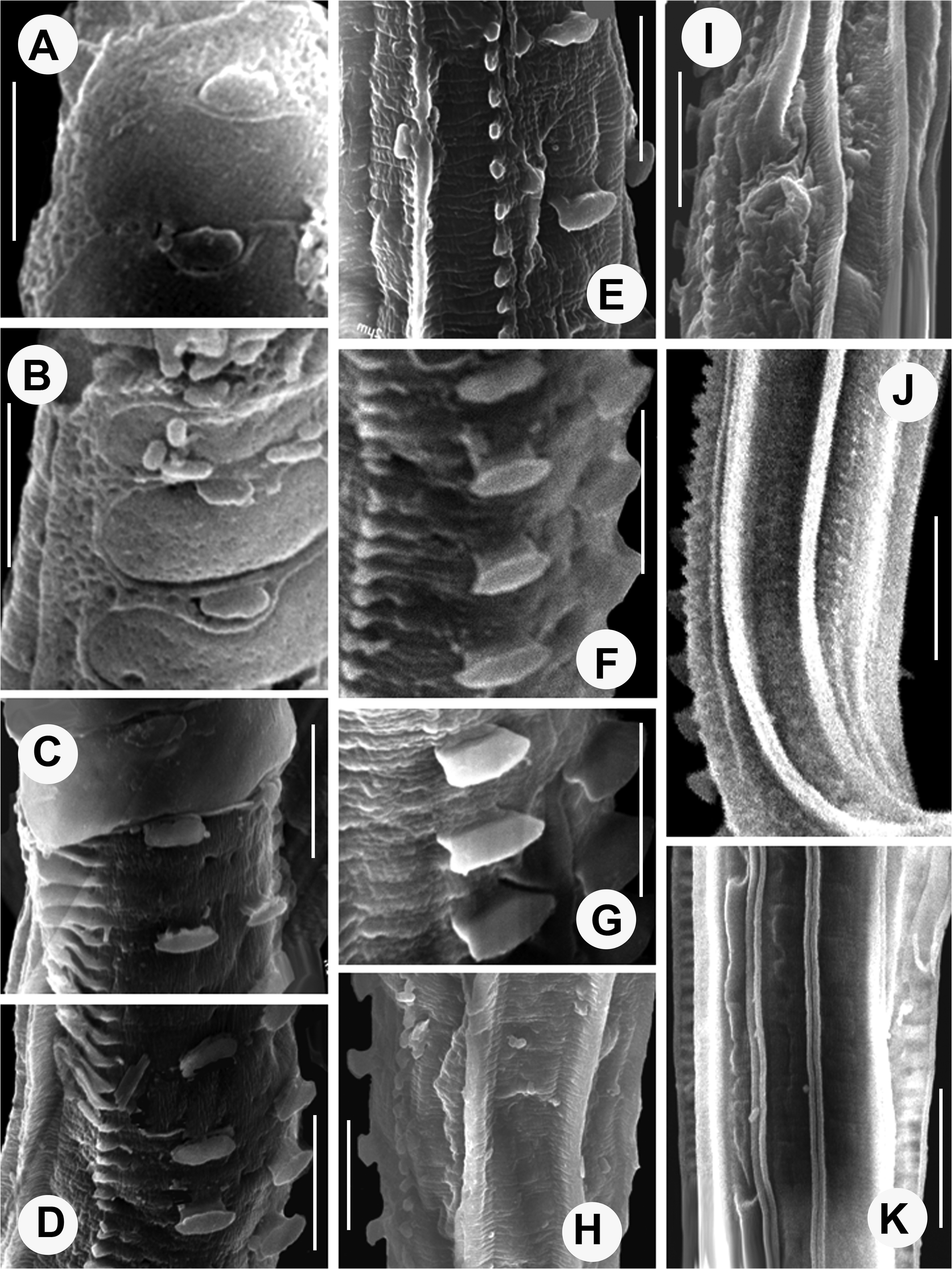
Bunonematids have mostly been reported from unusual locations including the crevices and tree holes, away from the soil environment and show ornamented bodies studded with conspicuous warts, and complex networks. SEM studies of bunonematid body surfaces have revealed intricate details of these ornamentations. The new species described here possesses porous, transversely ovoid cuticularised shields with strongly cuticularized margins surrounded by a reticulate network on the right side of the body ( Fig. 3A, B View FIGURE 3 ) while the left side of the body possesses five longitudinal ridges that appear to be striated ( Fig. 3 View FIGURE 3 H–K). The prominent cuticular striae continuing through the elevated ridges give a fin-like/striated appearance to these ridges ( Fig. 3I, K View FIGURE 3 ). On right side, the cuticularised shields adhere to the underlying transverse striae (6–7 in number) and the small peg-like paired warts present on the ventro-sub lateral side emerge through the elliptical sockets of the cuticularised shields. The origin of the warts and shields seems to differ with the warts originating in the cuticle while the shields and network show an extracuticular nature ( Fig. 3C View FIGURE 3 ). These extracuticular structures appear to offer resistance to abrasive surfaces and provide protection to the underlying cuticle through sloughing off ( Fig. 3C, D View FIGURE 3 ) under friction ( Bernard 1979). However, there is no regrowth/ regeneration once the shield /ornamentation is sloughed-off. Ultrastructural study by Jägerskiöld (1905) revealed that the ridges and tubercles are formed by evagination and thickening of the median zone of the cuticle thus confirming their cuticular origin. As a result, even after removal of the shields as well as the adjoining network, the warts remain intact ( Fig. 3C, F, G View FIGURE 3 ) ( Raski & Jones 1973, Bernard 1979). After detachment of the shields from the underlying cuticular striations, the remnants of the shields adhering to the striae surfaces present a fish-bone appearance and gradually erode away, leaving very small irregular remnants ( Fig. 3 View FIGURE 3 C–G) attached to the body surface. The importance of this unique bunonematid cuticular ornamentation was posited to have locomotory significance ( Steiner 1921, Stauffer 1924, Raski & Jones 1973), but TEM studies have not indicated any musculature associated with these structures ( Van Gansbeke et al. 2004). The effect of the peculiar asymmetry of bunonematids on locomotory abilities of females of Aspidonema kashmirensis sp. nov. was studied through simple observations of culture plates containing 1.2% nutrient agar plated with E. coli strain OP 50 in our laboratory. We observed that nematodes preferred to move on their left sides and avoided turning onto their heavily ornamented right sides. These results support the reports of Steiner (1921), Stauffer (1924), Raski & Jones (1973) and of Van Gansbeke et al. (2004).
Ornamentation in bunonematid nematodes may vary with age. Therefore, a taxonomic study based only on a very few individuals may be misleading as it is possible that adults with intact cuticular ornamentations, when compared with others whose shields and cuticular network have been removed or sloughed off, may be misinterpreted as different species.
| UP |
University of Papua and New Guinea |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |