Bursaphelenchus hildegardae, Braasch, 2006
|
publication ID |
https://doi.org/ 10.11646/zootaxa.5071.1.9 |
|
publication LSID |
lsid:zoobank.org:pub:A5367C32-1687-4785-8955-263EDCD9C92B |
|
DOI |
https://doi.org/10.5281/zenodo.5725536 |
|
persistent identifier |
https://treatment.plazi.org/id/861387C8-B916-785E-FF7D-A595CE4AF8F6 |
|
treatment provided by |
Plazi |
|
scientific name |
Bursaphelenchus hildegardae |
| status |
|
( Figs. 1–6 View FIG View FIG View FIG View FIG View FIG View FIG )
MEASUREMENTS
See Table 2. View TABLE 2
DESCRIPTION
Adults
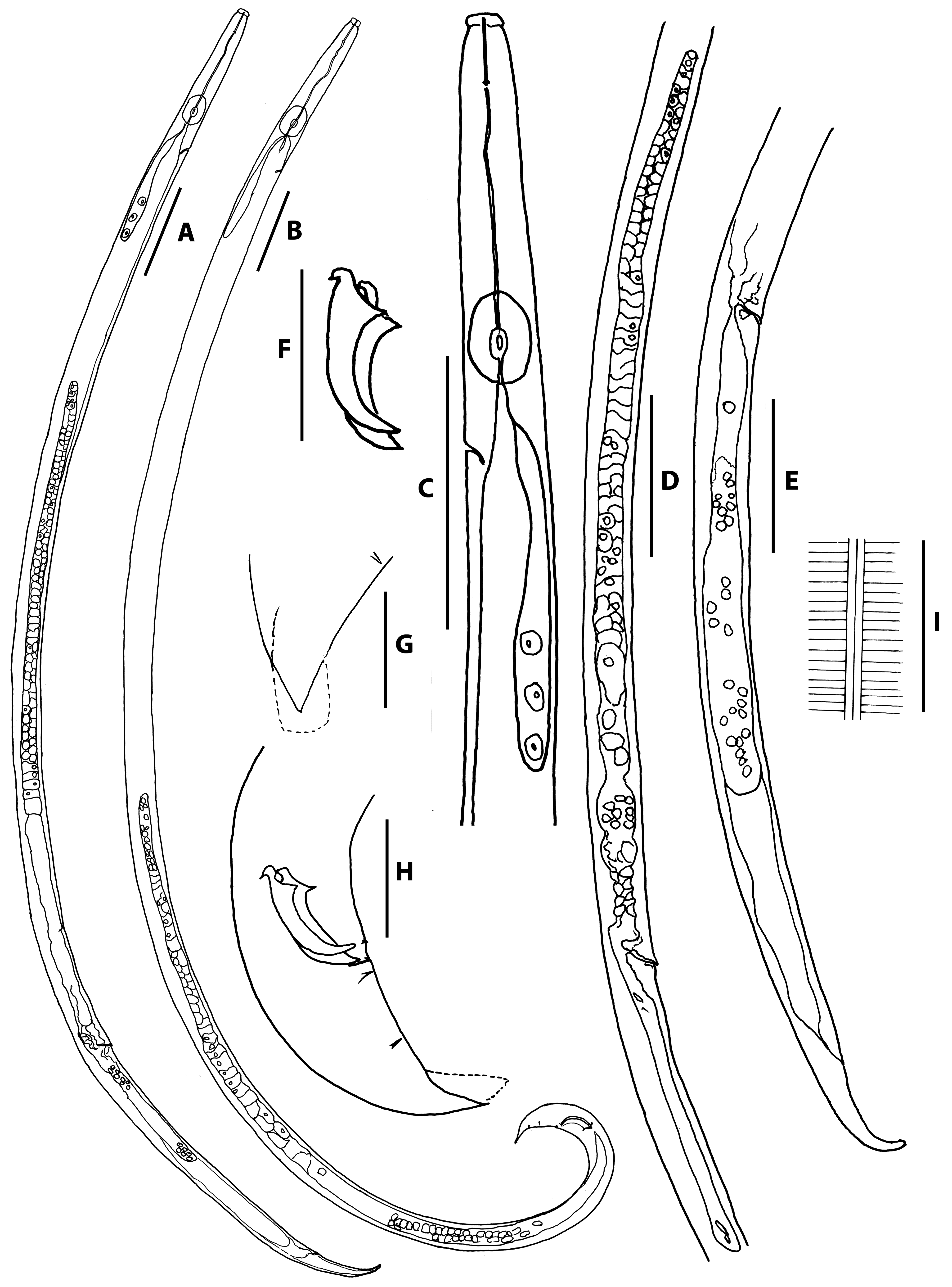
Body length ranging from 839–1163 and 807–1250 μm for male and female, respectively. Body cylindrical, moderate to slender, i.e., a = 44.6–60.1 and 41.0–58.5 for male and female, respectively. Cuticle thin, annulated, lateral field bearing three lines, i.e., two ridges. Head distinctly offset from body, separated by a clear constriction ( Figs. 1A, B & C View FIG ; 2A & E View FIG ). Stylet with narrow lumen comprising a short cone ca 39% – 45% of total stylet length and a shaft with slightly clear basal swelling. Procorpus cylindrical, ending in well-developed median bulb, i.e., 19.6 μm in length x 12.6 μm in width for female and 19.1 μm in length x 12.1 μm in width for male. Metacorpal valve clearly observed, i.e., 5.1–6.0 μm in length x 3.9–4.3 μm in width for female and 4.2–5.9 μm in length x 3.3–4.7 μm in width for male, located at middle of, or slightly posterior to, centre of median bulb. Dorsal esophageal gland orifice opening into lumen of metacorpus mid-way between anterior end of metacorpal valve and anterior end of metacorpus. Pharyngo-intestinal junction ca 3.5–6.0 μm posterior to median bulb. Dorsal pharyngeal glands overlapping intestine dorsally, extending ca 154–203 μm long for female and 142–178 μm long for male, posterior to median bulb. Position of excretory pore posterior to median bulb ca 93–119 μm long for female and 96–115 μm long for male from anterior head end, respectively. Nerve ring surrounding esophageal glands and intestine slightly posterior to pharyngo-intestinal junction. Hemizonid at ca 7.5–10.0 μm posterior to excretory pore for female and 8.0–13.0 μm posterior to excretory pore for male.
Female
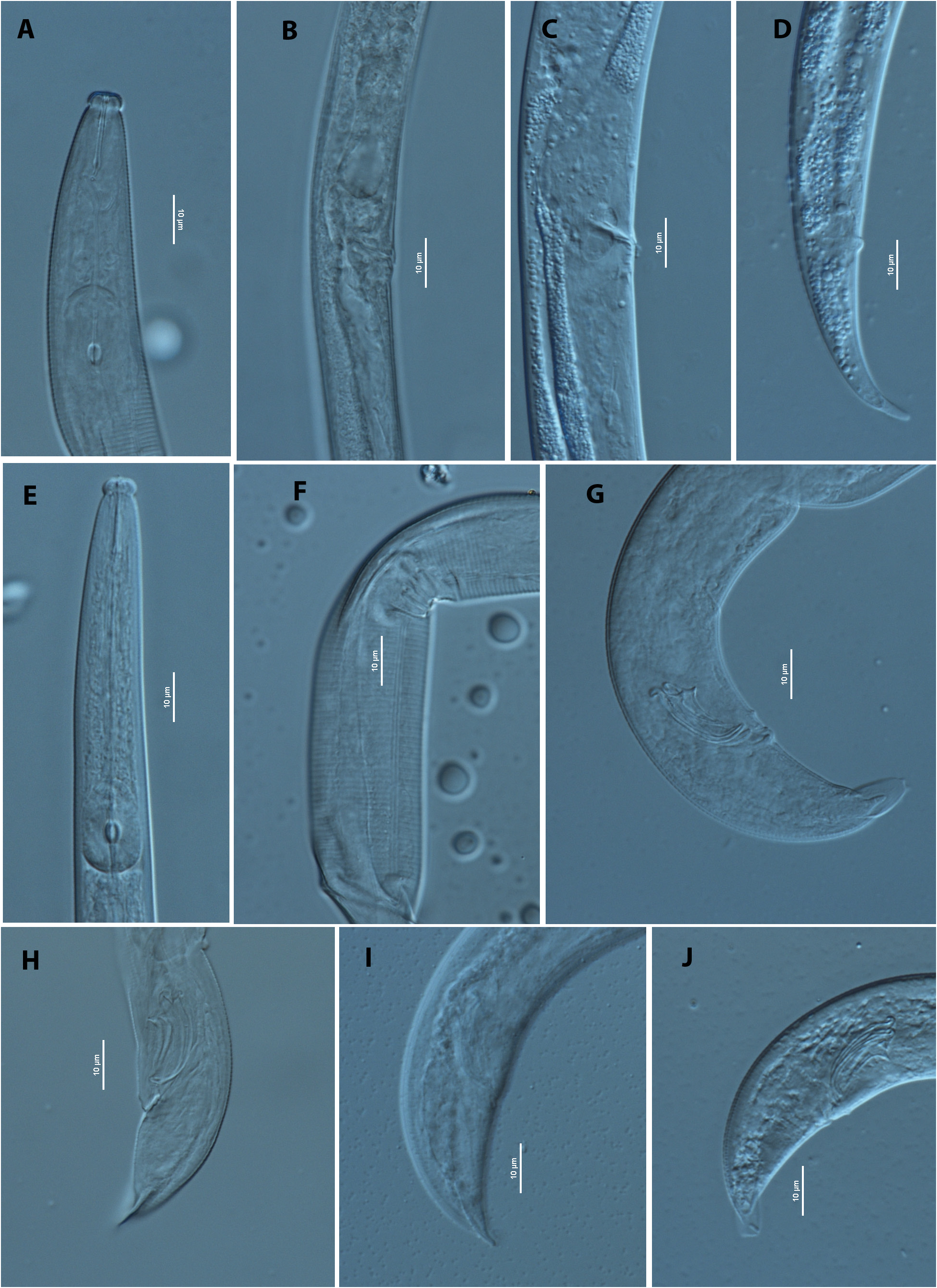
Body smoothly ventrally arcuate when killed by heat ( Fig. 1B View FIG ). Cuticle marked by fine transverse striations, ca. 1.0 μm wide. Lateral field with three lines, ca. 1.8 μm wide in midbody ( Fig. 1I View FIG ). Head set off by a distinct constriction, ca. 3 μm high, 8 μm wide ( Fig. 2A View FIG ). Stylet slender, almost without basal swellings, shaft forming about 2/3 of total stylet length. Procopus cylindrical. Median bulb elongated oval with conspicuous centrally placed valve plates. Oesophageal gland lobe extending dorsally for about five body widths long, down the body. Nerve ring located closely posterior to metacorpus. Ovary anteriorly outstretched. Developing oocytes in multiple rows in ovary. Oviduct tube-like. Spermatheca oval shaped, filled with well-developed sperm. Crustaformeria quadricolumella form, conspicuous. Uterus irregularly rounded. Vagina perpendicular to body surface, slightly inclined anteriorly. Vulval opening with distinctive short vulval flap ( Figs. 1D & E View FIG ; 2B & C View FIG ). Post-uterine sac (PUC) long and conspicuous, i.e., 6–7 vulval body diam. long, often containing sperm. Rectum and anus present, functional. Tail bent ventrally, elongate, conoid shape. Tail tip simple and blunted, no indentation observed before the end.
Male
Anterior body part and cuticle similar to those of female ( Fig. 2E View FIG ). Tail region strongly ventrally arcuate when killed by heat ( Figs. 1B View FIG ; 2G View FIG ). Gonad outstretched, sometimes reflexed backwards, occupying 45–55% of total body length. Spermatocytes and spermatozoa arranged in multiple rows, tightly packed in testis. Tail appearing distinctly claw-like at terminus in lateral view. Lips of cloacal aperture slightly protruding. Spicules paired, relatively straight, rosethorn-shaped. Capitulum of spicule well developed, short condylus with a relatively thin dorsally hooked end (ca. 2 μm long) and a pointed short triangular rostrum (ca. 2 μm long) with pointed tip. Spicule blade ventrally curved consisting of smoothly and clearly ventrally curved and well cuticularised dorsal limb ( Figs. 1F & H View FIG ; 2 G & H View FIG ). Distal tip of spicule thin, truncate, without cucullus. Gubernaculum absent. Bursal flap present, starting from level of posteriormost genital papillae (P4). Seven genital papillae, i.e., one ventral papilla (P1) and three subventral paired papillae (P2, P3, P4) present: precloacal P1, ventral, 2–3 μm anterior to cloacal opening (CO); P2 on subventral body adanal or slightly anterior to CO; P3 on ventro-subventral body, located mid-way between CO and tail tip; P4 on ventro-subventral body, located at tip of bursal flap or slightly posterior. P4 slightly smaller than P1, P2 and P3. Bursal flap roundish-rectangular to oval in shape, surrounding tail terminus, not indented ( Figs. 1G & H View FIG ; 2J View FIG )
Dauer Juvenile
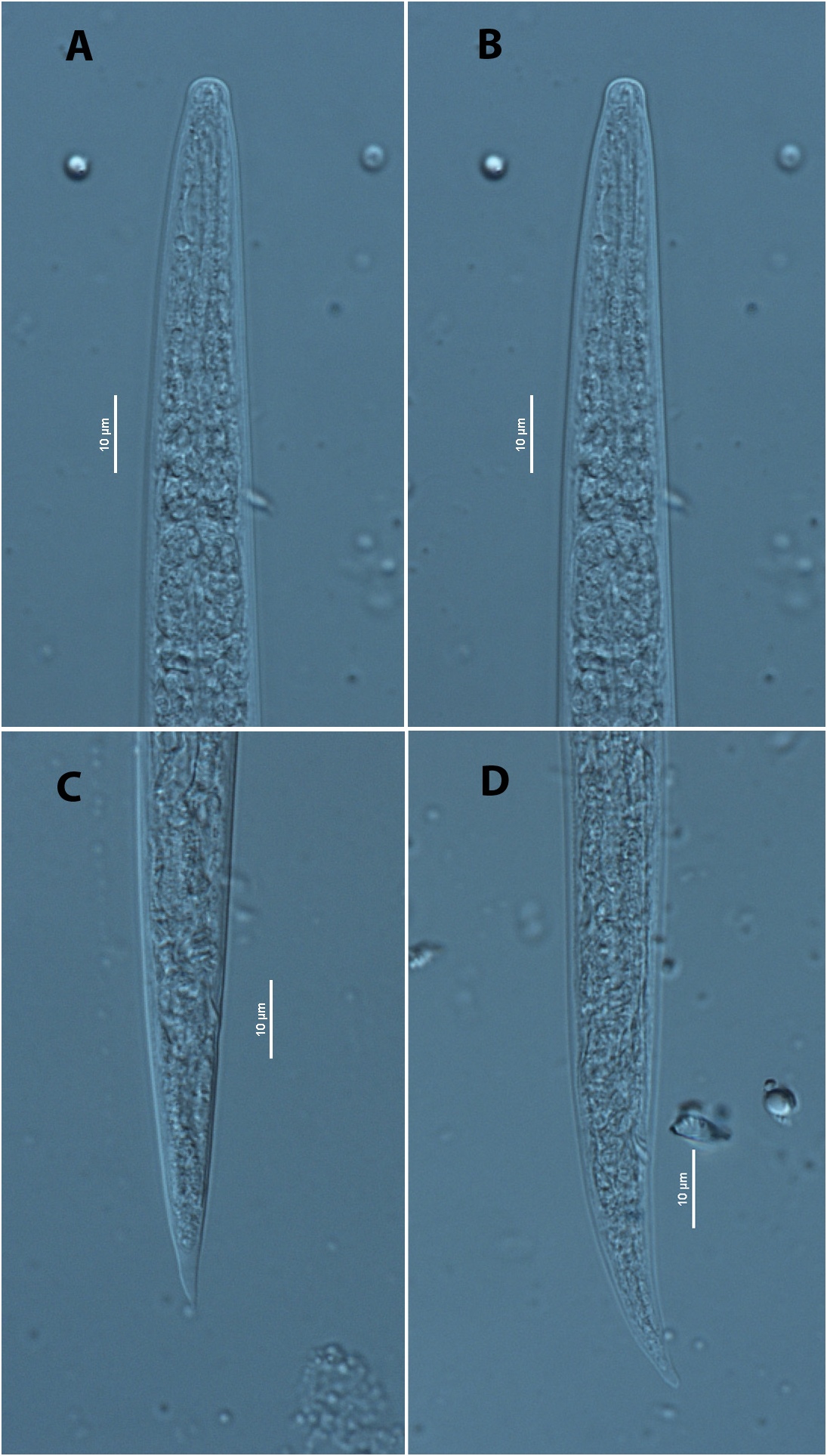
Dauer juvenile found under the elytra of H. ater . Body slender, 569–635 μm long. Cuticle with smooth surface, fine annulation was observed clearly, lateral lines not seen. Median bulb oval, well-developed, 17.0 μm in length x 10.9 μm in width (Figs. A & B). Metacorpal valve clearly observed, 3.8 μm in length x 2.9 μm in width, present at middle of, or slightly posterior to, centre of median bulb. Excretory pore not conspicuous. Hemizonid not observed. Cephalic region dome-shaped (Figs. A & B). Stylet observed with some examined specimens. Pharyngo-intestinal junction not conspicuous. Pharyngeal gland lobe not observed. Anus present, conspicuous ( Fig. 3D View FIG ). Tail conical, bluntly pointed ( Figs. 3C & D View FIG ).
HOST AND LOCALITY
Bursaphelenchus hildegardae , extracted from pine wood ( Pinus radiata ) and bark beetle ( H. ater ) samples collected from Kaingaroa Forest in the central North Island of New Zealand ( NZ) (38° 24’ 36.394”S; 176° 33’ 44.989” E) in March and April 2019; and from Douglas fir ( Pseudotsuga menziesii ) from Waipori, Dunedin in the South Island of NZ (45° 56’ 29.222” S; 170° 5’ 29.27” E) on 11 November 2020.
MATERIAL EXAMINED
Seven females (slide nos NNCNZ 3350–3352), seven males (slide nos NNCNZ 3353–3354) and five dauer juveniles (slide nos NNCNZ 3355) from Kaingaroa Forest; five females (slide nos NNCNZ 3356–3358) and three males (slide nos NNCNZ 3359–3362) from Waipori deposited at the National Nematode Collection, New Zealand (NNCNZ).
MORPHOLOGICAL DIAGNOSIS AND RELATIONSHIPS
The New Zealand isolate of B. hildegardae is morphologically closest to the original description of B. hildegardae ( Braasch et al., 2006) . However, it varies from the original description in the male with bursa shape, spicule size and b index. In the original description, it states that the male has a distinct V-shaped dorso-ventrally visible terminal bursa, but in the New Zealand specimens of B. hildegardae it has not been observed ( Figs. 1G View FIG ; 2G & J View FIG ). The spicule sizes were 23–28 μm vs 18–24 μm; and the b values were 8.6–10.2 vs 10.2–12.7 in the German and New Zealand isolates respectively.
The New Zealand isolate of B. hildegardae is also morphologically similar to B. eggersi , B. elytrus Massey, 1971 , B. glochis Brzeski & Baujard, 1997 , B. newmexicanus Massey, 1974 and B. tusciae Ambrogioni & Palmisano, 1998 . However, it can be differentiated from them by spicule shape, particularly by the thin hook-like condylus ( Table 3 View TABLE 3 ).
MOLECULAR PHYLOGENETIC RELATIONSHIPS
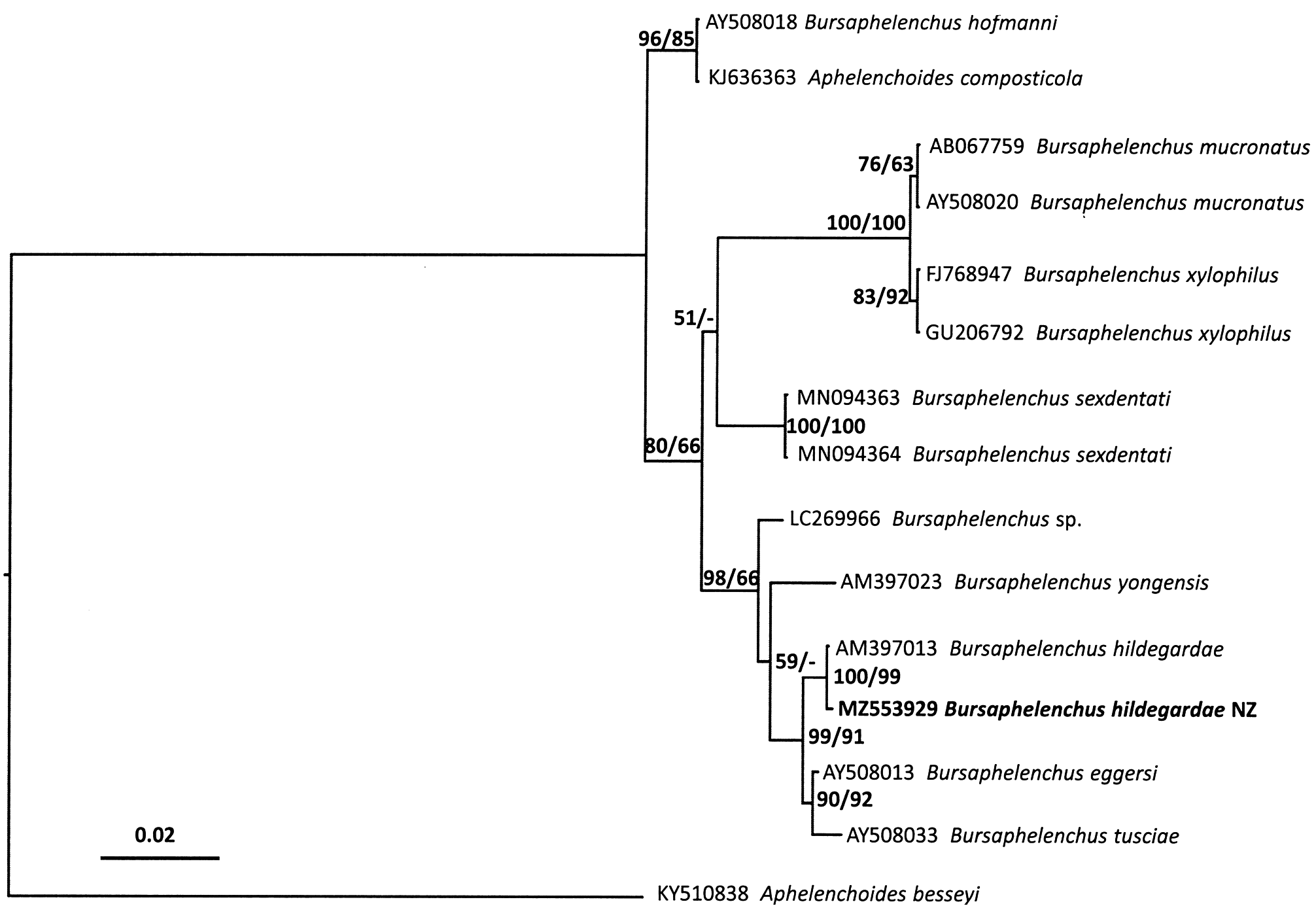
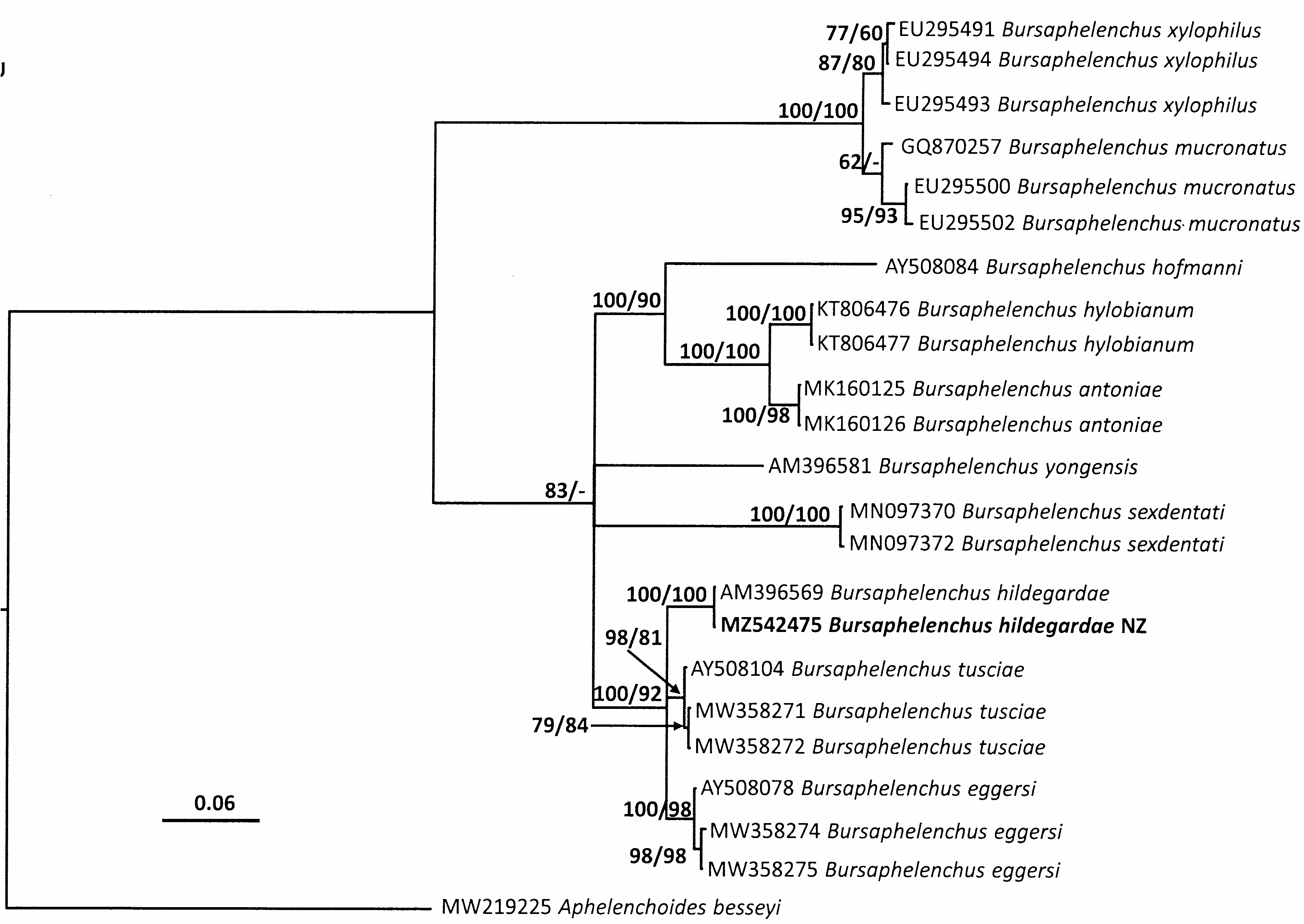
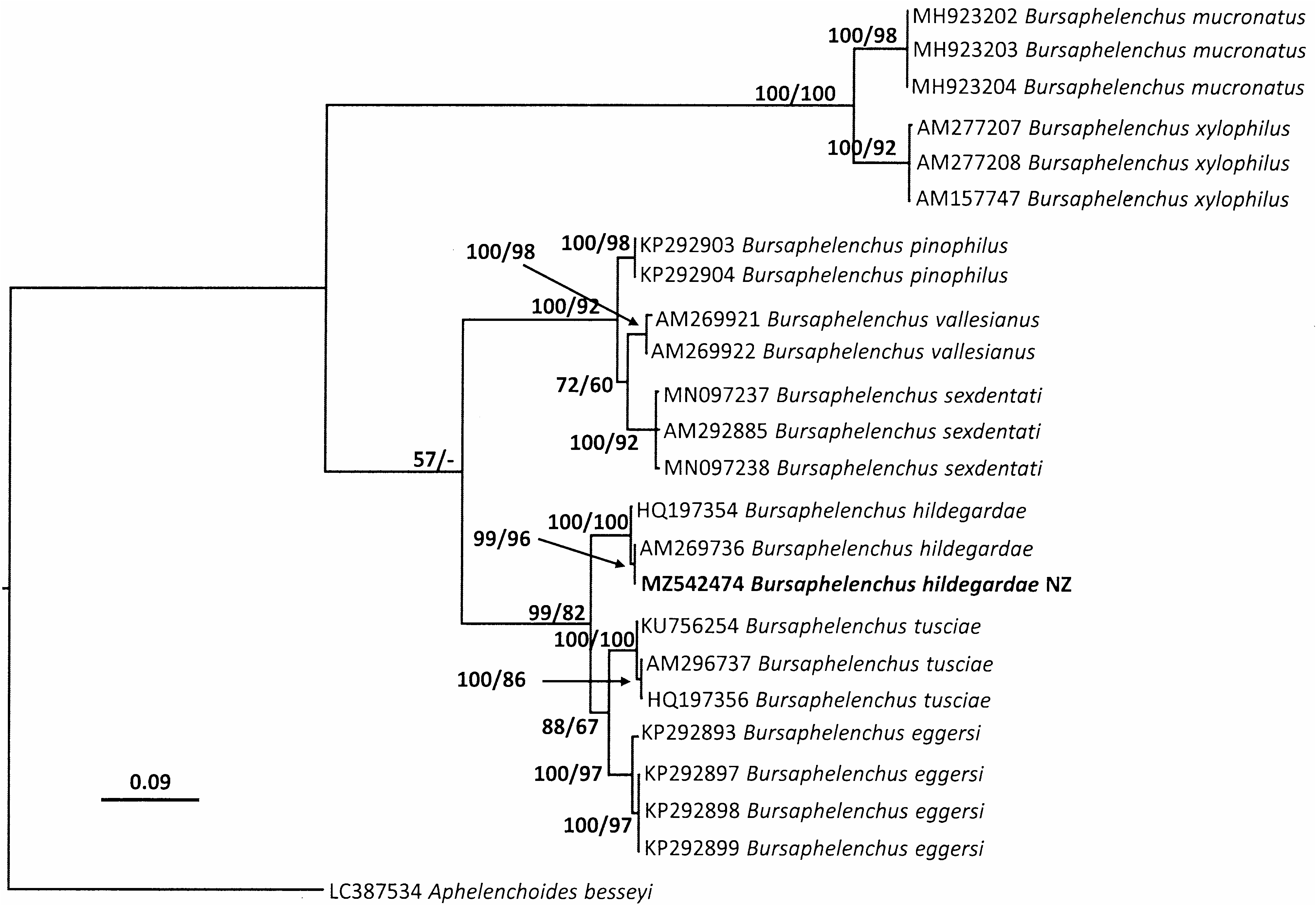
Sequences for partial SSU, D2/D3 and ITS were amplified from a female, male, juvenile and dauer juvenile respectively. These PCR products were subjected to direct sequencing and aligned with those sequences from published B. hildegardae data. Comparisons of trees inferred from Bayesian analyses are shown in Figs 4–6 View FIG View FIG View FIG . Molecular phylogeny of near full length SSU, D2/D3 expansion segments of LSU and ITS region indicate that the specimen collected in New Zealand is phylogenetically close to B. hildegardae ( Figs 4–6 View FIG View FIG View FIG ). Molecular analyses of an individual female, male, juvenile and dauer juvenile of the nematode confirmed they are the same species. All three trees derived from SSU, D2/D3 and ITS sequences, grouped the New Zealand isolate of B. hildegardae together with the isolates of B. hildegardae from Germany.
The consensus tree inferred from SSU ( Fig. 4 View FIG ) shows that B. hildegardae is clustered with the only SSU sequences of B. hildegardae ( AM397013 View Materials ) available in GenBank, with a posterior probability of 100% and a bootstrap of 100% support, respectively. The results of a BLAST search also showed that New Zealand B. hildegardae is nearly identical to the B. hildegardae sequences ( AM397013 View Materials ) having 99.9% with one bp difference, and close to B. eggersi ( AY508013 View Materials ) having 99.4% with nine bp difference and B. tusciae (AY08033) having 98.9% with 16 bp difference for the 18S sequences.
The consensus tree inferred from LSU ( Fig. 5 View FIG ) shows that the New Zealand isolate of B. hildegardae is grouped with the only LSU sequences of B. hildegardae ( AM396569 View Materials ) available in GenBank, with a posterior probability of 100% and a bootstrap of 100% support respectively. From the results of a BLAST search, the sequences of 28S from the New Zealand isolate were almost identical to the only B. hildegardae sequences ( AM396569 View Materials ) having 99.7% with two bp difference. It is also close to two B. eggersi sequences ( AY508078 View Materials and MW258275) having 95.9% and 95.8%, and two B. tusciae sequences ( AY508104 View Materials and MW358271 View Materials ) having 96.4% and 96.5% identity for the 28S sequences, respectively.
The consensus tree inferred from ITS ( Fig. 6 View FIG ) shows that the New Zealand isolate of B. hildegardae is monophyletic with a posterior probability of 100% (a bootstrap of 100%) when two ITS sequences of B. hildegardae ( AM269736 View Materials and HQ197354 View Materials ) available in GenBank are included. The results of a BLAST search showed that the sequences of ITS from the New Zealand isolate were 100% identical to the B. hildegardae sequences ( AM269736 View Materials ) and close to a sequences from Spain ( HQ197354 View Materials ) having 99.48% identity for the ITS sequences, respectively.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |