Nesophrosyne Kirkaldy 1907 : 160
|
publication ID |
https://doi.org/ 10.5281/zenodo.207804 |
|
DOI |
https://doi.org/10.5281/zenodo.6182588 |
|
persistent identifier |
https://treatment.plazi.org/id/03EB1E0B-FC6F-9867-07A1-CC9D733B6851 |
|
treatment provided by |
Plazi |
|
scientific name |
Nesophrosyne Kirkaldy 1907 : 160 |
| status |
|
Nesophrosyne Kirkaldy 1907: 160 View in CoL
Type species: Euttetix perkinsi Kirkaldy 1907: 160
Nesophrosyne (Nesoreias) Kirkaldy 1910: 573 View in CoL new synonymy
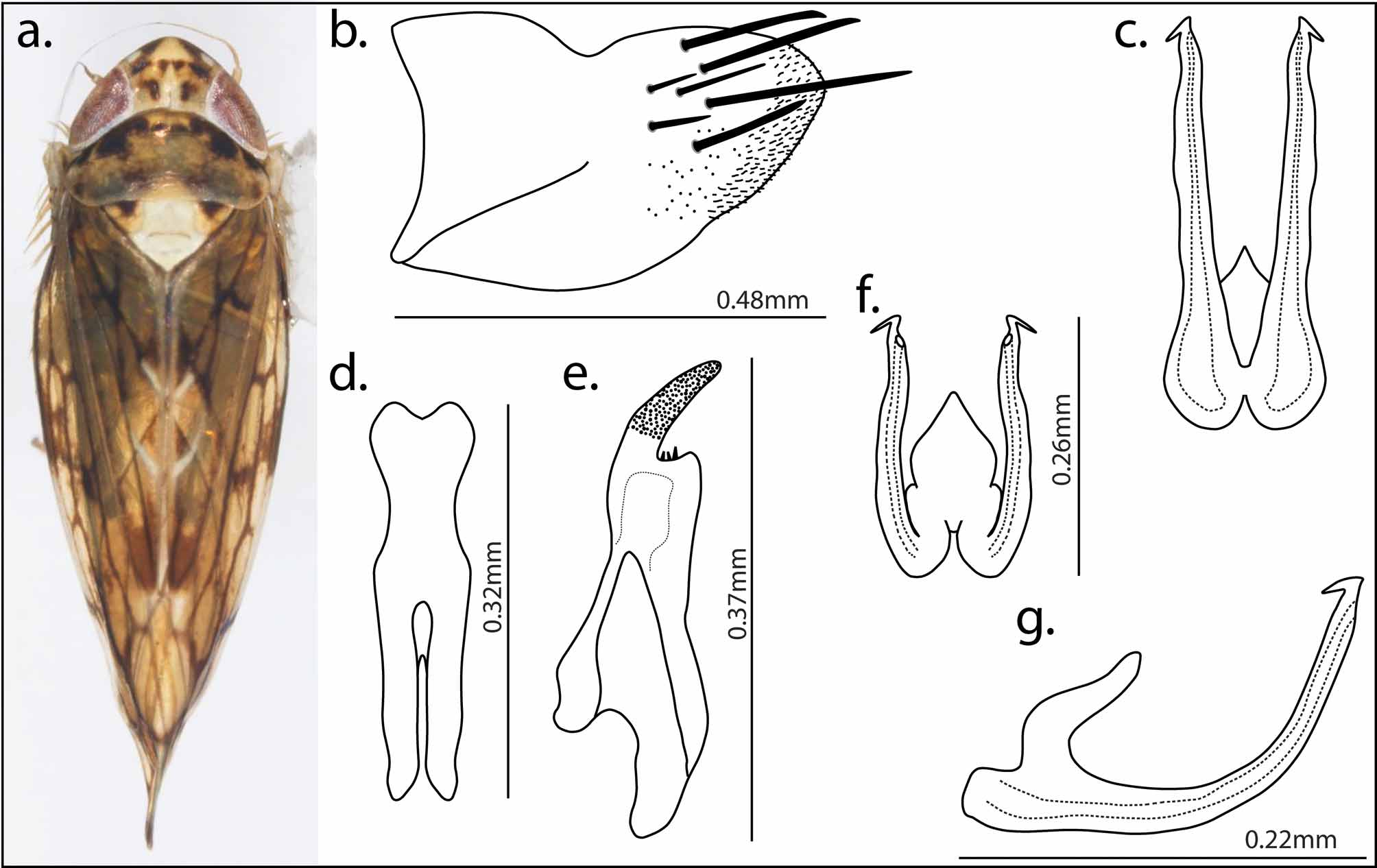
Diagnosis. Nesophrosyne resembles other described genera in the tribe Opsiini , having a bifurcate aedeagus and two gonopores. The genus is most closely allied with Orosius , having two parallel aedeagal arms, curving posterodorsally; gonoducts arising independently from preatria at the base with gonopores usually subapical; styles with hooked posterior processes; the crown sometimes bluntly rounded, although often angularly produced and extended well beyond anterior edge of the eyes (e.g., N. anguilifera ); the outer anteapical cell in the forewing present and sometimes elongate relative to the central anteapical cell; and the forewing flecked in two species ( N. notatula and N. signatula ).
Nesophrosyne differs from Orosius and other opsiine genera in having the aedeagal arms with produced and hooked apical processes, varying from short-straight to long and recurving, but completely absent in some species; the body size larger (3.25mm – 5.5mm), with the smallest species overlapping with Orosius (2.63mm – 3.29mm); the frontoclypeus wide, but variable across species; the forewing often with extensive and distinct pale color patterning sometimes forming a solid saddle-like mark (e.g., N. pluvialis ); the pronotum and forewing without the brown stripes common in Orosius ; the forewing with the outer anteapical cell reduced and sometimes absent, forming a small triangular structure with single R1 vein extending to the outer edge (some species with two); the subgenital plates tapering more gradually and remaining wider at the distal end, without an abrupt medial narrowing ledge and tube-like distal processes ( Orosius ); and the anal tube dorsally sclerotized and with lateral and ventral edges sclerotized.
Description. Dorsum (see Figs. 4–9 View FIGURE 4 View FIGURE 5 View FIGURE 6 View FIGURE 7 View FIGURE 8 View FIGURE 9 ): Small slight (3.25 mm) to large robust (5.5 mm) leafhoppers. Color and coloration patterns varying widely. Crown ranging from bluntly rounded to elongate and extremely produced. Ocelli visible in dorsal view, situated on margin of crown and face. Pronotum as wide as, or in some cases slightly wider than, head; anterior margin rounded, convex; posterior margin straight. Forewing with large central anteapical cell and smaller outer anteapical cell; central anteapical cell narrowed medially; outer anteapical cell variable in size and shape from nearly the length of the central anteapical cell to completely absent (varies within and between species). Clavus of many species with well-formed, conspicuous saddle mark ( Fig. 7 View FIGURE 7 ).
Venter: Frons and clypeus broad, width nearly 3/4 length; lateral margins not sinuate. Clypellus elongate, lateral margins subparallel.
Genitalia: Pygofer with 16 or fewer macrosete on posterior half of apical lobe, extending beyond posterior edge; fine stout microsete covering posterior half of apical lobes ( Figs. 4–6 View FIGURE 4 View FIGURE 5 View FIGURE 6 b & 8–9b). Valve rounded, notched symmetrically on either side of posterior apex. Subgenital plate elongate, curved posterodorsad, tapering towards distal point; lateral edges hirsute from base to apex, with long filamentous and macrosete (usually 5) interspersed (not unique to this genus). Anal tube long, not fully membranous dorsally, with well sclerotized band; laterally and ventrally sclerotized. Aedeagus bifurcate with two aedeagal arms and gonopores; aedeagal arms curving posterodorsally, height of arms variable from sub-anal tube to terminating at the dorsal edge of pygofer; gonopore near apex, usually subapical, opening posteromesially; aedeagal arms with produced apical processes hooked anterolaterad, length from short-straight (~1/10 length of aedeagal arm) to long and recurving (~1/4–1/5 length of aedeagal arms), completely absent in some species; base of gonoducts not connected, forming two preatria; anterior base of aedeagus with apodeme pointed dorsad ( Figs. 4–6 View FIGURE 4 View FIGURE 5 View FIGURE 6 c,f,g & 8–9c,f,g). Styles with well-developed posterior processes, curved posterolaterad; preapical lobe with fine microsete ( Figs. 4–6 View FIGURE 4 View FIGURE 5 View FIGURE 6 e & 8–9e). Connective ‘Y’ shaped; body thinning medially, appearing hourglass shaped; anterior arms variably shaped and splayed ( Figs. 4–6 View FIGURE 4 View FIGURE 5 View FIGURE 6 d & 8–9d).
Material examined. A total of 1085 specimens from at least 101 distinct morphological species were examined. Sampling from individual islands is as follows: Hawai’i Island, males = 232, females = 301, species = 32; Maui, males = 118, females = 93, species = 32; Lana’i, males = 1, females = 3, species = 3; Moloka’i, males = 18, females = 12, species = 4; O’ahu, males = 59, females = 37, species = 15; Kaua’i, males = 105, females = 106, species = 16.
Discussion. Despite intraspecifc variation in Nesophrosyne , color patterning offers useful characters for species identification (we refer readers to Zimmerman’s (1948) well-illustrated review of Nesophrosyne for a photographic menagerie of the genus’ external morphological diversity). However, external and internal genitalia provide a more reliable suite of morphological characters for species determination. These characters, combined with molecular data and species’ geographic ranges, provide congruent lines of evidence for Nesophrosyne species and cryptic species groups. Results presented here provide evidence of cryptic sibling species, which may be common in other lineages associated with widespread host plants. Molecular characters may be necessary for identification of species within these groups.
Nesophrosyne’s biogeographic origin and relationships to other Pacific cicadellid genera are currently unknown. Kirkaldy (1907, 1910) proposed a relationship to Nephotettix , but he did not explain the basis for this hypothesis. Osborn (1935) strongly argued for a Scaphoideus -like ancestor derived from either America or Asia, [colonizing] during the early history of life on the islands.
A plausible origin for the Hawaiian Nesophrosyne lineage is the genus Orosius found along the western Pacific Rim including Japan, Southeast Asia, and the Oceania region. Linnavuori (1960a, 1960b, 1975) first examined this relationship, and based on similarities in the bifurcate aedeagus, blunt crown and flecked forewing of some Nesophrosyne species, presence of an elongate outer anteapical cell, hollowing near the base of the antennae, and overlapping shape and size of the frontoclypeus of some species, he combined the genera with Orosius as a subgenus of Nesophrosyne . Ghauri (1966) later reexamined Linnavuori’s conclusions, determining that morphological differences, including the anal tube structure, subgenital plate shape, divergence of frontoclypeus shape in some species, and hooking distal process found only in Nesophrosyne were sufficient to preserve Nesophrosyne and Orosius as separate genera. Linnavuori (1975) conceded, commenting that a thorough investigation of the biotic and morphological diversity of both genera is necessary to resolve the question. He further reiterated the opinion that Nesophrosyne originated from eastern Oceania. Both Ghauri (1965) and Linnavuori (1975) noted the presence of hooking apical processes on the aedeagal arms in Nesophrosyne , which are absent in Orosius . A more thorough investigation of Nesophrosyne genitalia reveals that the apical processes are absent in some species and the genitalic affinities between the two genera are stronger than previously thought. The relationship needs to be examined phylogenetically.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
Nesophrosyne Kirkaldy 1907 : 160
| Bennett, Gordon M & O’Grady, Patrick M 2011 |
Nesophrosyne (Nesoreias)
| Kirkaldy 1910: 573 |
Nesophrosyne
| Kirkaldy 1907: 160 |