Comatella
|
publication ID |
https://doi.org/10.11646/zootaxa.4268.2.1 |
|
publication LSID |
lsid:zoobank.org:pub:0742D287-B82C-4014-A6AC-C357F259D5D7 |
|
DOI |
https://doi.org/10.5281/zenodo.6009076 |
|
persistent identifier |
https://treatment.plazi.org/id/039EDF70-FFB8-A168-FF66-DBA61194F8D9 |
|
treatment provided by |
Plazi (2017-05-16 07:33:13, last updated 2024-11-29 15:24:32) |
|
scientific name |
Comatella |
| status |
|
Comatella AH Clark, 1908
Table 1, Fig. 1 View FIGURE 1 E–L, Fig. 2 View FIGURE 2
Type species. Actinometra nigra Carpenter, 1888 .
Other included taxa (1). Actinometra stelligera Carpenter, 1888 .
Description. Mouth excentric in fully developed individuals; centrodorsal well-developed, circular, and with cirri ( Fig. 1 View FIGURE 1 F, K); IBr2 series united by synarthry; first syzygy at 3+4 on arms arising from IBr; IIBr and beyond of two ossicles; arms arising from IIBr and beyond with first syzygy at 1+2 and/or 3+4 ( Fig. 1 View FIGURE 1 F, K); distal intersyzygial interval 4; distalmost pinnule comb on P6; comb teeth triangular, confluent with interior side of pinnule, reduced in size distally, and tapering to blunt tip ( Fig. 1 View FIGURE 1 L).
Distribution. Tropical Indo-western Pacific from Abrolhos Is, WA, to Lady Elliott I., QLD, Australia, east to Tonga and Samoa, west to Maldives, and north to Ogasawara (Bonin) Is. and Okinawa, Japan, including Chuuk Atoll and Guam ( Chen et al. 1988, AH Clark 1931, Kirkendale & Messing 2003; Kogo 1998, Messing 1998b, Rowe & Gates 1995). Depth range: low intertidal to 32 (perhaps 73) m. Rowe & Gates (1995) list a maximum depth of 160 m for C. nigra , but this may be an error for the 106 m maximum given by AH Clark (1931). Depths greater than ~ 100 m attributed to Comatella by AH Clark (1931) are for species no longer included in the genus: Palaeocomatella decora and “ Comissia ” brachycirra (Messing 2001).
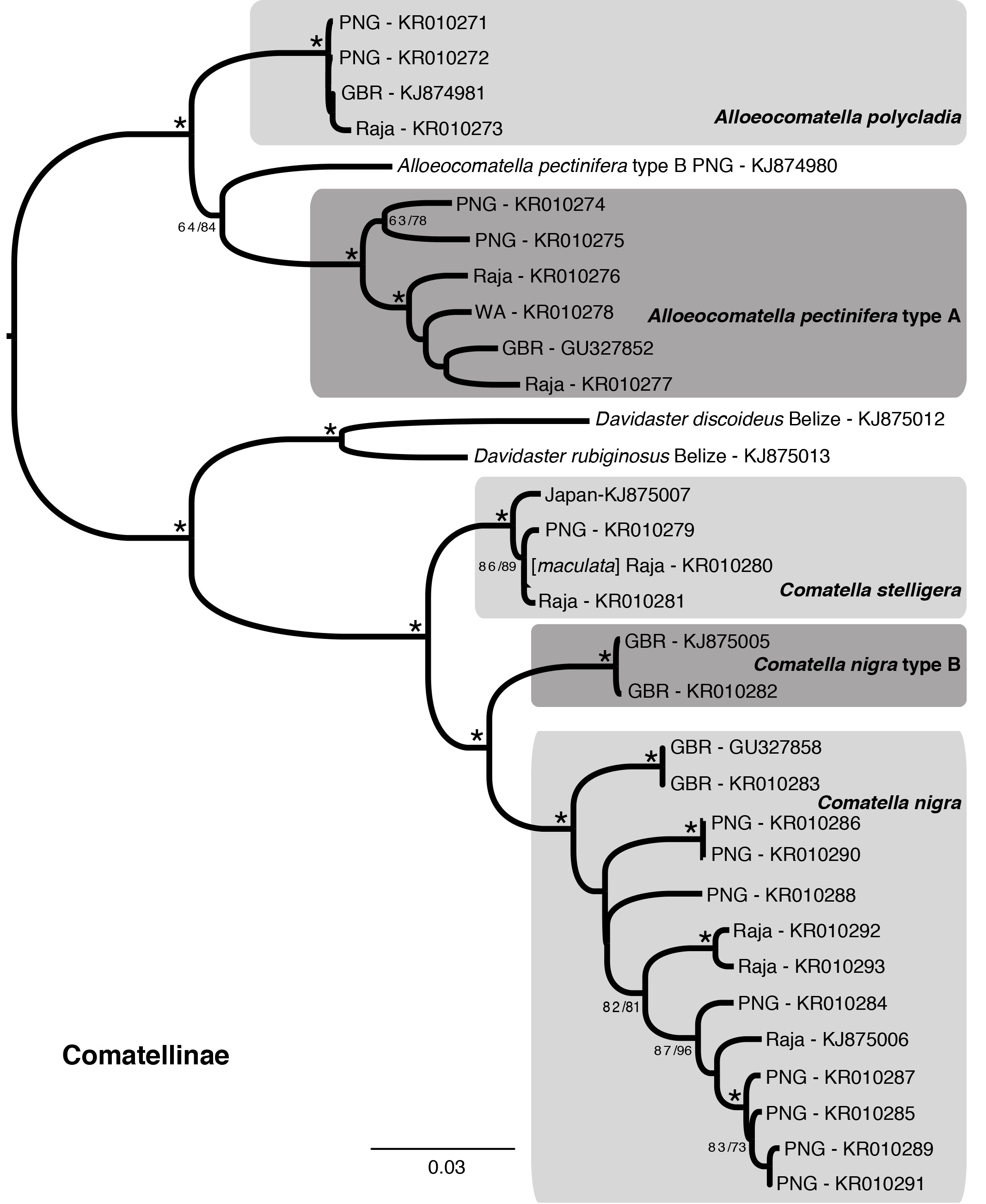
Molecular results. Specimens identified as Comatella nigra and C. stelligera formed reciprocally monophyletic groups in both the parsimony and likelihood analyses ( Fig. 2 View FIGURE 2 ), with a minimum of 5.3% uncorrected COI distance between the taxa. Notably , specimens identified as Comatella nigra from Lizard Island , Australia, showed an even greater intra specific divergence, with up to 6.3% uncorrected COI distance (n=13); this taxon may thus represent at least two different species (= Comatella nigra and Comatella nigra B in Fig. 2 View FIGURE 2 ). Specimens of Comatella nigra type B are minimally 4.9% divergent from remaining specimens of C. nigra with an intra type divergence of less than 0.20%. The remaining C. nigra terminals were up to 4.8% divergent from one another, and may also represent multiple species. The type locality for C. nigra is Bohol , Philippines (syntype: RAME R114); no specimens were sequenced from this region, and we have so far found no clear way to split the group into separate species on either molecular or morphological evidence. The type locality of C. maculata ( NHM 88-11-9 - 103 ) (see below) is Prince of Wales Channel , between Queensland , Australia, and Papua New Guinea; that of C. stelligera ( NHM 88-11-9 -104 ) is Fiji .
Remarks. Comatella differs from all other Pacific comatulids in having comb teeth along the edge of the pinnule closest to the arm (a homoplastic trait shared with western Atlantic Nemaster ) ( Fig. 1 View FIGURE 1 L). Comatella previously included three species that appeared to represent an ontogenetic series based on size. Messing (1998b) synonymized the smallest, C. maculata , with the next largest, C. stelligera . This synonymy is supported by sequencing of specimens herein. Specimens morphologically assigned to C. maculata and C. stelligera were less than 0.08% divergent in COI (uncorrected distance). Molecular results also differentiate C. stelligera from the largest species, C. nigra . In addition to size and molecular data, C. nigra differs from C. stelligera in having asymmetric axils on IIBr and following brachitaxes that produce twisted rays, visible in both juvenile and adults. In C. stelligera , all arms radiate in a single plane.
During the day, Comatella species on reefs are cryptic and curled up beneath coral slabs, rubble or ledges, or deep within branching coral colonies ( Fig. 1 View FIGURE 1 G–J). Arms, especially of large specimens in deeper water (> 15 m), may extend from crevices or under corals with pinnules arranged in a multidirectional, or tetrad, posture (i.e., successive pinnules offset so that they form four rows along an arm) ( Meyer & Macurda 1980). At night, individuals may be completely exposed on coral heads or among dead or living coral branches, or only the arms may be visible. However, on the Great Barrier Reef on expanses of sediment in 12–17 m, Messing et al. (2006:165) found C. nigra “exposed, clinging to the downcurrent side of algae (e.g., Halimeda sp.) with some arms lying at least partly against the sediment. Under unidirectional flow, it forms a multilayered arcuate fan with successive pinnules slightly offset. Under reduced flow, the arms form an irregular meridional array.”
For specimens herein referred to as C. stelligera , we observed two discrepancies relative to previous descriptions: IIIBr sometimes united by syzygy rather than by synarthry only, and 1+2 or 3+4 on undivided arms, rather than 1+2 and 3+4.
Carpenter, P. H. (1888) Report on the Crinoidea collected during the Voyage of H. M. S. Challenger, during the years 1873 - 1876. Part II-The Comatulae. In: Thomson, C. W. & Murray, J. (Eds.), Report of the Scientific Results of the Voyage of H. M. S. Challenger during the years 1873 - 1876, Zoology-Vol. XXVI. Neill & Company, Edinburgh, pp. 1 - 400.
Chen, J. C., Chang, K. H. & Chen, C. P. (1988) Shallow-water crinoids of Kenting National Park, Taiwan. Bulletin of the Institute of Zoology Academia Sinica, 27, 73 - 90.
Clark, A. H. (1931) A monograph of the existing crinoids 1 (3). Bulletin of the United States National Museum, 82, 1 - 916.
Kirkendale, L. & Messing, C. G. (2003) An annotated checklist and key to the Crinoidea of Guam and Commonwealth of the Northern Marianas Islands. Micronesica, 35 - 36, 523 - 546.
Kogo, I. (1998) Crinoids from Japan and its adjacent waters. Special Publication of the Osaka Museum of National History, 30, 1 - 148.
Messing, C. G. (1998 b) An initial re-assessment of the distribution and diversity of the East Indian shallow-water crinoid fauna. Pp. 187 - 192. In: Mooi, R. & Telford, M. (eds.) Echinoderms: San Francisco. Balkema, Rotterdam.
Messing, C. G., Meyer, D. L., Siebeck, U. E., Jermiin, L. S., Vaney, D. I. & Rouse, G. W. (2006) A modern soft-bottom, shallowwater crinoid fauna (Echinodermata) from the Great Barrier Reef, Australia. Coral Reefs, 25, 164 - 168. https: // doi. org / 10.1007 / s 00338 - 005 - 0076 - 3
Meyer, D. L. & Macurda, Jr., D. B. (1980) Ecology and distribution of the shallow-water crinoids of Palau and Guam. Micronesica, 16, 59 - 99.
Rowe, F. W. E. & Gates, J. (1995) Echinodermata. Zoological Catalogue of Australia. CSIRO, Melbourne, 510 pp.
FIGURE 1. Comatellinae. (A – C) Alloeocomatella pectinifera— (A) live, centrodorsal concealed with ten arms exposed from coral, photographed at night; (B) well-developed rounded pentagonal centrodorsal with one row of cirri; (C) combs with> 20 tall, narrow, triangular teeth confluent with the outer edge of pinnule, non-tapering. (D) Alloeocomatella polycladia exposed on coral following removal from under loose coral rubble, with diagnostic red color and> 10 arms, curling. (E – F) Comatella stelligera, preserved — (E) specimen 2, entire animal showing> 20 arms and numerous cirri; (F) specimen 1, well-developed circular centrodorsal with two rows of cirri; arrows indicate syzygies. (G – L) Comatella nigra— (G – J) live, semicryptic on coral reef, showing various color morphs; (K) well-developed circular centrodorsal with one row of cirri; arrows indicate syzygies; (L) combs with triangular teeth, confluent with interior side of pinnule, diminishing distally to a blunt tip. Raja Ampat, Indonesia: A. not vouchered; C. SIO-BIC E 6166. Palau: B. CRECH 236; K. SIO-BIC E 5875; L. SIO-BIC E 6265. Madang, Papua New Guinea: D. MNHN-IE- 2013 - 8123. G. MNHN-IE- 2013 - 8179; I. MNHN-IE- 2013 - 8103; MNHN-IE- 2013 - 8064. Challenger Station 174, near Kandavu, Fiji: E – F. NHM 88.11. 9.104, type specimens 1 and 2. Timor: H. AM J. 25420.
FIGURE 2. Maximum likelihood tree of Comatellinae inferred from COI. Symbols near nodes refer to bootstrap (BS) and jackknife (JK) support scores, for maximum likelihood (ML) and maximum parsimony (MP) analyses, respectively. An asterisk indicates nodes with> 90 % bootstrap and jackknife support. Other scores are represented BS / JK. A hyphen is given for nodes not recovered in MP analyses. Previous morphological identifications in brackets. Taxa labeled as: location - Genbank accession number. Location abbreviations — Great Barrier Reef, Australia (GBR); Madang, Papua New Guinea (PNG); Raja Ampat, Indonesia (Raja); Rowley Shoals, Western Australia (WA). New COI sequences: Genbank KR 010271 - KR 010293.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
1 (by plazi, 2017-05-16 07:37:34)
2 (by ImsDioSync, 2017-05-16 07:38:48)
3 (by ImsDioSync, 2019-03-29 21:34:08)
4 (by ExternalLinkService, 2019-09-26 03:43:52)
5 (by ExternalLinkService, 2022-01-29 17:45:21)
6 (by ExternalLinkService, 2022-02-08 11:52:18)
7 (by plazi, 2023-10-27 16:48:47)