Pseudoliparis swirei Gerringer & Linley, 2017
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4358.1.7 |
|
publication LSID |
lsid:zoobank.org:pub:84494502-9E85-49DA-8530-092AF8918D88 |
|
DOI |
https://doi.org/10.5281/zenodo.6016523 |
|
persistent identifier |
https://treatment.plazi.org/id/0391DE73-FF98-FF91-98B7-FA6F5F959253 |
|
treatment provided by |
Plazi |
|
scientific name |
Pseudoliparis swirei Gerringer & Linley |
| status |
sp. nov. |
Pseudoliparis swirei Gerringer & Linley View in CoL sp. nov.
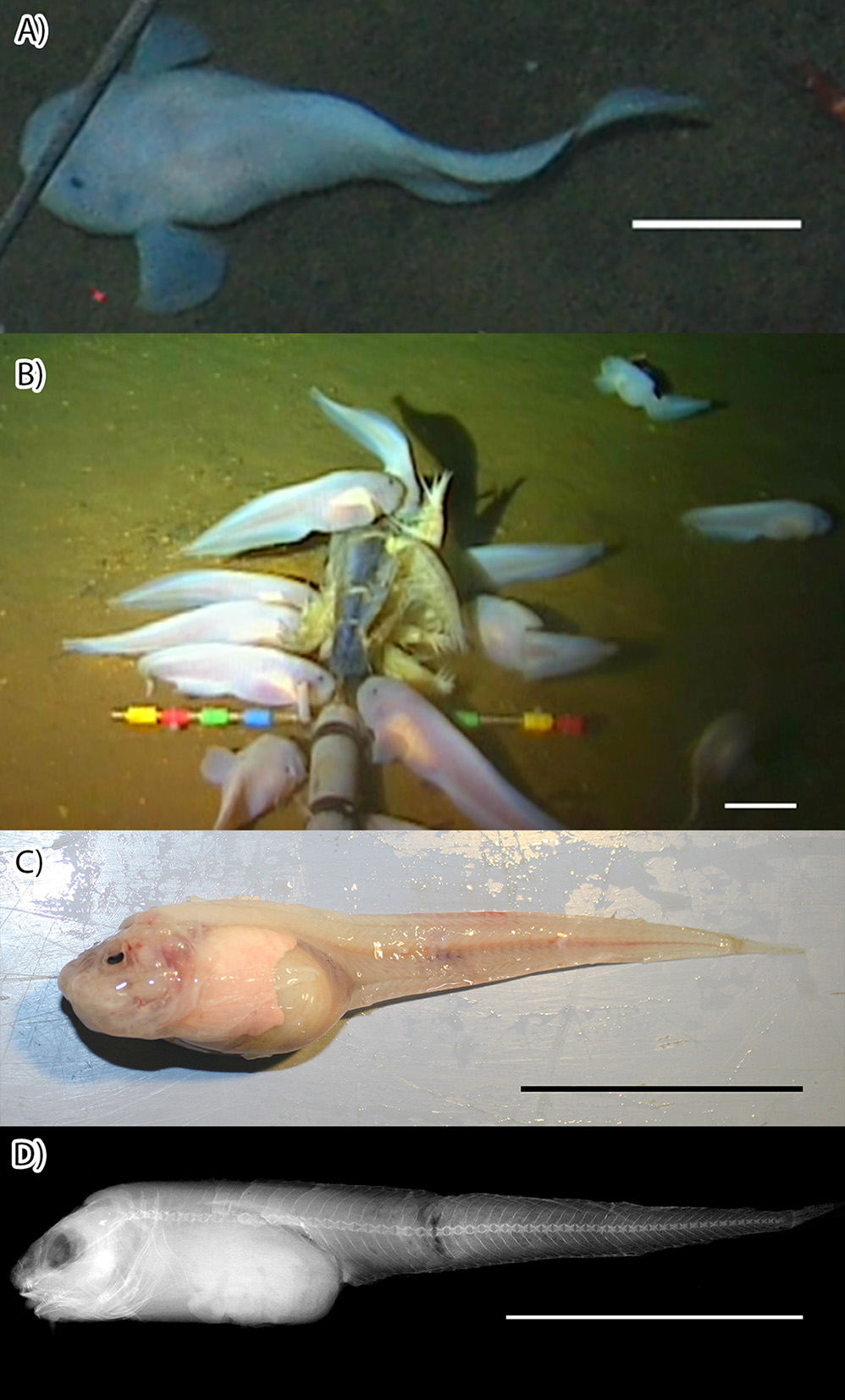
Figures 2 View FIGURE 2 , 3 View FIGURE 3 , 4 View FIGURE 4 , 5 View FIGURE 5 ; Tables 2, 3
Mariana snailfish: Linley et al. 2016 (page 105, Figure 4a View FIGURE 4 )
Mariana snailfish: Linley et al. 2017 (page 42, Figure 6.43)
Mariana snailfish/Mariana liparid: Gerringer et al. 2017a (page 111)
Mariana liparid/ Liparidae View in CoL sp. nov: Gerringer et al. 2017b (page 137)
Diagnosis. Andriashev and Pitruk (1993) define the genus Pseudoliparis as having a well-developed disk and one pair of nostrils and lacking pseudobranchia and pleural ribs, with four radials in the pectoral girdle, which has neither notches nor fenestrae. In this genus, the hypural plate is divided by a distal slit ( Andriashev and Pitruk, 1993). Like the other in this genus, Pseudoliparis swirei sp. nov. ( Figures 2 View FIGURE 2 , 3 View FIGURE 3 ) displays these characters, including a moderately well-developed disk, although this is easily damaged in collection. Pseudoliparis swirei sp. nov. can be distinguished from the two other known Pseudoliparis species with the following characters. Pseudoliparis swirei sp. nov. differs from P. belyaevi in the presence of a distinct lower pectoral-fin lobe, similar to that seen in P. amblystomopsis (Andriashev, 1955) . Pseudoliparis swirei has more dorsal-fin rays 55 (51–58) than P.
amblystomopsis 49 (49–52), more anal-fin rays 48 (43–49) compared to 43 (42–45), and more vertebrae 61 (56– 62), compared to 55–57, although these ranges somewhat overlap. Head length is shorter in P. swirei sp. nov. (17.0–21.7 %SL) than P. amblystomopsis (21.6–24.0 %SL). Comparisons were made according to ranges presented by Andriashev & Pitruk (1993). Pseudoliparis belyaevi is known only from the Japan Trench, P. amblystomopsis from the Japan and Kurile-Kamchatka trenches, P. swirei only from the Mariana Trench.
Description. Vertebrae 61 (56–62), dorsal-fin rays 55 (51–58), anal-fin rays 48 (43–49), caudal-fin rays 13 (11–14), pectoral-fin rays 30 (28–32), pectoral radials 4, pyloric caeca 7 (5–9). Ranges of measurements and counts are presented in Table 2. Ratios are presented in Table 3. All individual measurements and counts are available in Supplementary Table 1. Micro-CT scans and a 3D rotatable model of the holotype are available in the Supplementary Material.
Head small, low, and wide, lateral profile anteriorly rounded and rising slowly to occiput, where the angle increases. Head depth about equal to body depth or lower, depending on abdominal fullness. Snout blunt, nostrils single, nares on horizontal with center of eye. Mouth broad, horizontal, subterminal, moderately large; upper jaw reaching to below middle of orbit, oral cleft reaching to below anterior edge of orbit. Teeth simple, sharp canines, innermost largest, arranged in approximately 9 (6–11 maxilla, 7–13 mandible) oblique, irregular rows of up to 20 (6–17 maxilla, 8–20 mandible) teeth each, forming a moderately wide band (2–4 teeth wide) in each jaw ( Figure 4 View FIGURE 4 ). Larger individuals had more teeth per row and more rows of teeth. Maxilla with prominent symphyseal gap, slight gap present in mandible. Pharyngeal teeth well developed, long, sharp, strongly fixed on globular tooth plates. Eye very small, about 10% head length. Orbit large, its dorsal margin well below that of head. Gill opening small, located completely above pectoral fin, width 5% SL. Opercular flap fleshy, broadly triangular, opercle terminates in two small spines below the flesh. Cephalic pores small, easily damaged; few remaining. Eight widely separated infraorbital and mandibular pores, lacking raised rims. In fresh specimens, the infraorbital and mandibular pores appear sunken and anteriorly orientated within the gelatinous tissue of the head ( Figure 4 View FIGURE 4 ).
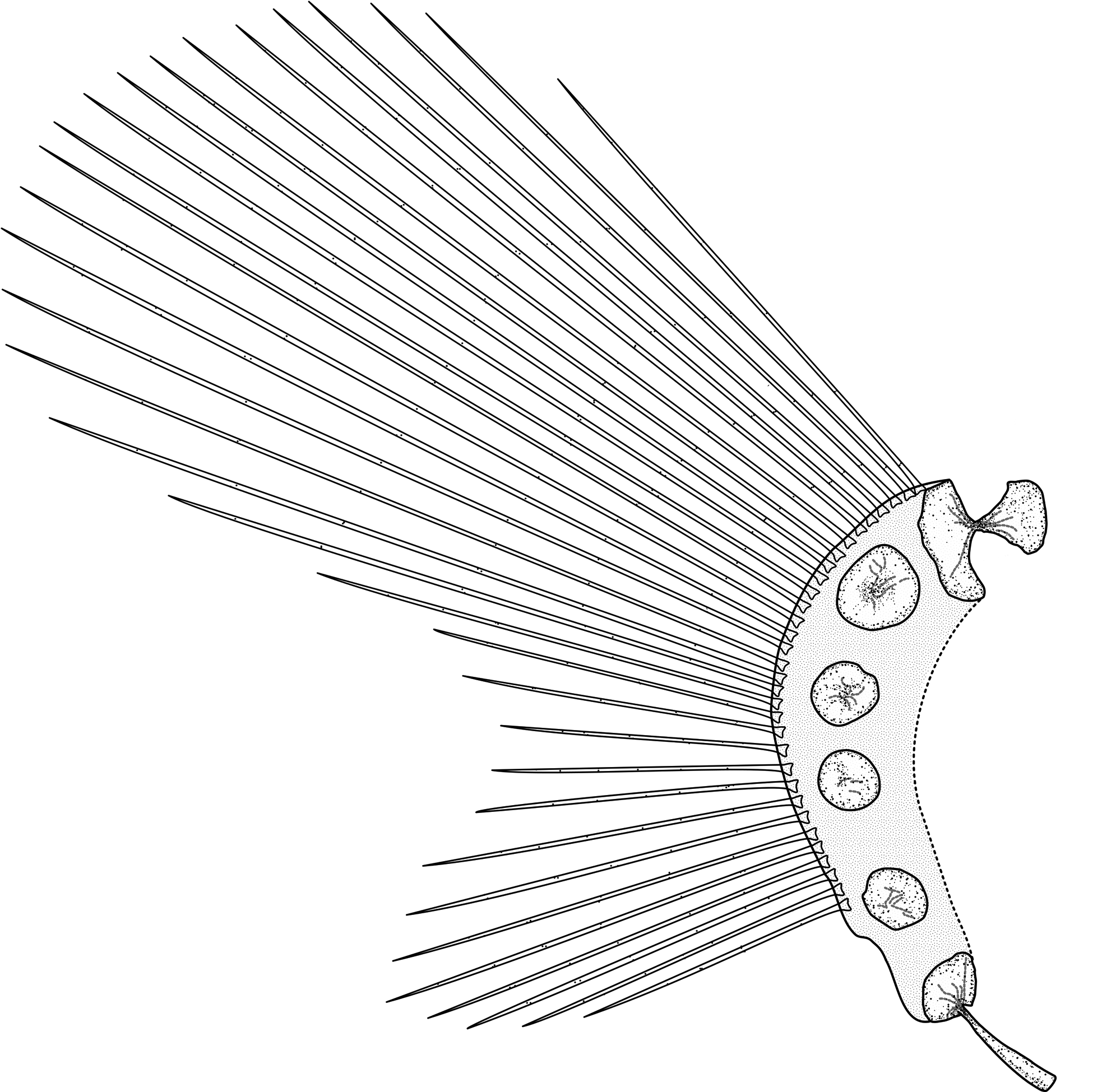
Pectoral fin divided into lobes by a moderately deep notch, rudimentary rays absent. Notch rays 5 (3–6), clearly more widely spaced than those of upper and lower lobes, more so in larger individuals. Upper and lower lobe rays closely spaced. Dorsal-most pectoral-fin ray on horizontal between level of upper jaw and lower margin of orbit. Symphysis of pectoral fins and anteriormost ray below rear of orbit. Upper lobe about 15.2% SL (12.6– 19.8), lower about 7.8% SL (6.3–12.1). Upper lobe almost extending to anal-fin origin, lower lobe distinct, reaching well behind disk to below middle of upper lobe base. Pectoral radials four, fenestra absent; of four specimens examined (USNM 438996/HADES 200024, USNM 438994/HADES 200027, USNM 438997/HADES 200033, USNM 438989/HADES 200096); one (USNM 438996HADES 200024) had (1+1+1+1), and three (USNM 438994/HADES 200027, USNM 438997/HADES 200033, USNM 438989/HADES 200096) had (3+1) radials, generally round, notches and foramina absent ( Figure 5 View FIGURE 5 ). Radials gradually and irregularly decreasing in size from R1 (largest) to R4 (smallest). Distal radials present under the base of all rays, with the exception of the two most ventral rays (Supplementary Figure 1 View FIGURE 1 ). Scapula double-headed, posterior head larger and broader than anterior head, coracoid with broad head and long slender helve.
%SL %HL
Holotype Holotype and Paratypes n Holotype Holotype and Paratypes n Fresh
Head Length 20.6 14.5–21.9 (18.7±1.8) 33
Snout Length 7.2 4.3–9.3 (6.4±1.0) 33 35.0 26.3–45.5 (33.8±5.3) 37 Eye Width 2.1 0.9–2.9 (1.8±0.5) 33 10.0 5.3–15.4 (9.9±2.4) 37 Body Depth 18.6 18.6–31.2 (24.5±3.1) 18 90.0 90.0–156.8 (128.7±17.9) 18 Preanal Fin Length 37.1 36.5–49.5 (42.0±3.5) 33 180.0 180.0–295.8 (228.0±26.1) 37 Disk present, oval, longer than wide, below cheek and gill cavity between pectoral fin notches; well behind pectoral symphysis. Bones fully developed but weakly calcified; all elements present. Disk and pectoral girdle supported by a pair of clearly visible and strongly developed muscles extending anteriorly to pectoral symphysis, probably infracarinalis anterior (D.L. Stein, personal communication). Disk structure supporting a thin layer of tissue, often damaged or missing entirely; disk margin only slightly thicker than more central tissue. In cross section, disk rays clearly flattened as if to support disk margin.
Body depth dependent on reproductive state and fullness of stomach, usually much deeper than head depth, but shallow above vertebral column. Abdominal cavity long; peritoneum and body cavity extending to about 40% standard length. Total vertebrae 61 (56–62); anterior 13 (11–14), caudal 46 (44–49). In the 19 individuals in good enough condition to be radiographed, a double ray is present at or near anal fin first ray, usually between the second and third haemal spines of the caudal vertebrae. Pre-dorsal length about 27% SL (18.6–32.6), dorsal fin origin between fourth and fifth vertebrae (origin after vertebrae 3–5). Pre-anal-fin length about 42% SL (36.3–49.5)*. Anus far posterior to disk, roughly 2/3 of distance from disk to anal fin origin. Pyloric caeca usually 7 (5–9), located left ventrally in body cavity; thick, digitate, usually separated into two distinct size classes, most commonly 4 short and 3 long, longest about 8.7% SL (5.9–11.7), shortest 3.7% SL (2.4–5.3). Longer caeca generally grouped together. Hypural with obvious suture; caudal fin most commonly of 13 (11–14) rays, ventral one or two often rudimentary. Skin thin, transparent; subdermal extracellular matrix (SECM; Eastman et al. 1994) thick below skin and between muscle bands. Total and standard lengths were approximately 10% shorter after preservation. The subdermal extracellular matrix is also lost after capture and preservation, resulting in changes to shape and proportion (Gerringer et al. unpublished data). With increased use of visual in situ techniques, reporting of both fresh and preserved specimen features will become increasingly useful.
The 37 individuals used for description varied in size from 89–235 mm SL, apparently covering a wide developmental range for the species. Some characters correlated significantly with ontogeny, explaining much of the variation in ratios. Both the upper and lower pectoral-fin lobe lengths as a percentage of SL decreased significantly with increasing SL (upper: F1,25=11.88, p<0.01, R2=0.322; lower F1,23=5.05, p<0.05, R2=0.180). Proportional orbit width decreased with increasing standard length (F1,18=26.25, p<0.01, R2=0.593).
In life, body pinkish-white, skin and peritoneum transparent; internal organs (liver, stomach, pyloric caeca) and muscles of trunk clearly visible through skin and thin abdominal wall. Anterior bundles of epaxial muscle thick, becoming less densely packed posteriorly. Some larger specimens with dusky skin on head. Pyloric caeca orange; most individuals entirely lacked both internal and external pigmentation. In alcohol, except for those with dusky heads, specimens uniformly pale.
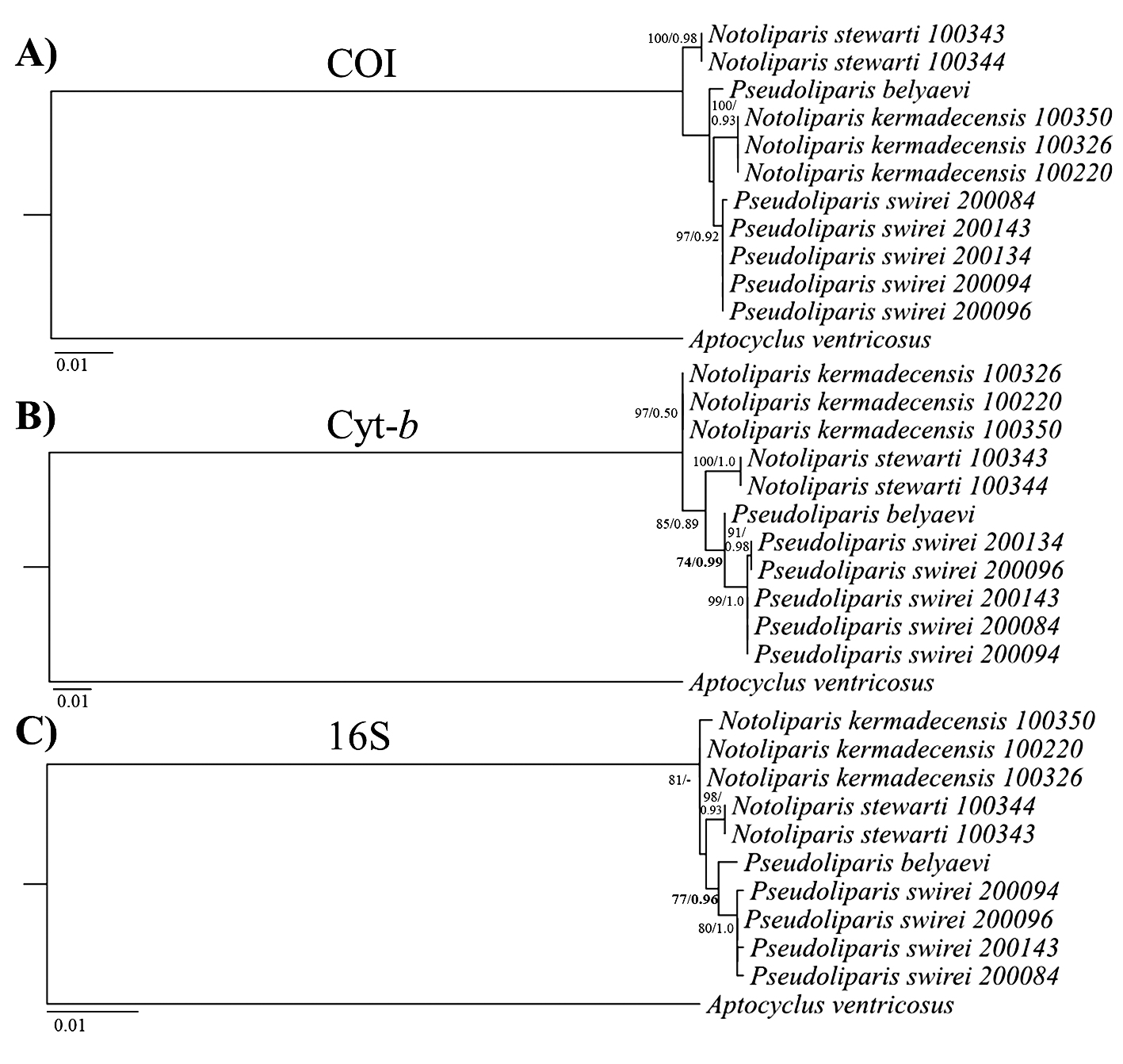
Phylogenetic inference supports placement within the genus Pseudoliparis , with P. swirei sp. nov. more closely related to P. belyaevi of the Japan Trench than to the Kermadec Trench liparids ( Notoliparis kermadecensis ; N. stewarti ). Phylogenetic relationships of P. swirei sp. nov. and closely-related species based on 16S, COI, and Cyt- b are presented in Figure 6 View FIGURE 6 , with estimates of evolutionary divergence among species reported in Table 4. Both the 16S and Cyt- b trees support placement of P. swirei sp. nov. as most closely related to P. belyaevi (>74% ML bootstrap support,>0.96 Bayesian posterior probability), with highly concordant topology. Genetic distances between the two species of Pseudoliparis are 0.6% at Cyt- b and 0.7% at 16S rRNA ( Table 4), with distances of 1– 1.4% (Cyt- b) and 0.8–1% (16S rRNA) to the Notoliparis species. COI lacked sufficient polymorphism to resolve the relevant nodes, with low bootstrap support observed for the placement of P. swirei sp. nov. relative to P. belyaevi and N. kermadecensis ( Figure 6 View FIGURE 6 ). Genetic distances among species also were the lowest at COI, at <1% or three nucleotide substitutions observed between the sequences of Pseudoliparis . Phylogenetic analyses conducted using the more closely-related Careproctus outgroup taxa, but with a shorter alignment (696-bp 16S rRNA, 744-bp Cyt- b, 645-bp COI) found congruent results to the trees presented in Figure 6 View FIGURE 6 ( Aptocyclus outgroup), but with lower bootstrap support for most nodes (Supplementary Figure 2 View FIGURE 2 ). The 16S rRNA phylogeny supported placement of the new species into Pseudoliparis (67% ML bootstrap support/0.75 Bayesian posterior probability), and moderate support was observed at Cyt- b (0.82 Bayesian posterior probability, ML unresolved). There was still no significant resolution of generic placement in the COI phylogeny with the Careproctus outgroup species. Sequences from Pseudoliparis swirei sp. nov., Notoliparis kermadecensis , Notoliparis stewarti (this study) are available under GenBank accession numbers KY659176 View Materials – KY659204 View Materials (Supplementary Table 2).
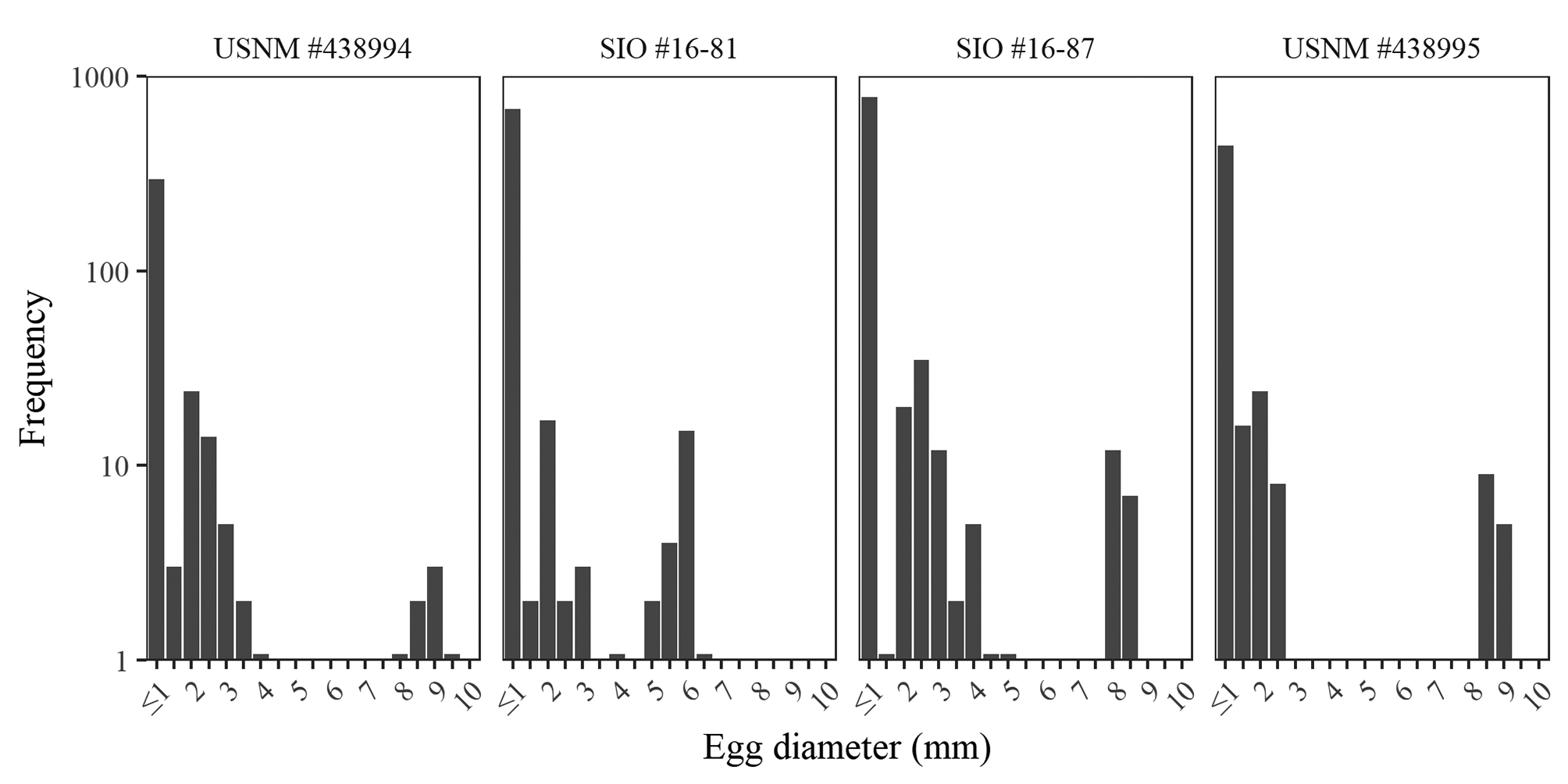
Reproduction. Holotype is immature. Ripe females had eggs up to 9.4 mm diameter, among the largest teleost eggs recorded ( Tyler and Sumpter, 1996), 0.4 mm smaller than the largest record ( Matallanas et al., 1990). The eggs were unsorted within gonad, with the largest eggs free and interspersed within a matrix of smaller eggs. No developmental structures were visible within even the largest eggs. Two distinct size classes of eggs present with up to 23 large eggs (> 5 mm) and up to 851 small eggs of less than half the diameter of the larger size class. There were rarely intermediate stages ( Figure 7 View FIGURE 7 ). Individuals with only small eggs had maximum egg sizes ranging from 0.7 to 1.4 mm. Genital papilla visible in freshly collected males, oriented anteriorly.
Distribution. Known only from the Mariana Trench at capture depths from 6,898–7,966 m, individuals likely this species were recognized in video at depths 6,198–8,098 m ( Linley et al. 2016; Jamieson & Linley, unpublished data).
Etymology. The Mariana Trench famously houses the ocean’s deepest point, at Challenger Deep, named for the HMS Challenger expedition which discovered the trench in 1875. Their deepest sounding of 8,184 m, then the greatest known ocean depth, was christened Swire Deep after Herbert Swire, the ship’s First Navigating Sublieutenant ( Corfield 2003). We name this fish in his honor, in acknowledgment and gratitude of the crew members that have supported oceanographic research throughout history.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.