Rioraja agassizii (Müller & Henle, 1841)
|
publication ID |
https://doi.org/ 10.1590/1982-0224-2021-0059 |
|
DOI |
https://doi.org/10.5281/zenodo.11059757 |
|
persistent identifier |
https://treatment.plazi.org/id/AE455563-4E09-543B-4D9B-FD32FAE5FBC2 |
|
treatment provided by |
Felipe |
|
scientific name |
Rioraja agassizii (Müller & Henle, 1841) |
| status |
|
Rioraja agassizii (Müller & Henle, 1841) View in CoL
( Figs. 2–9 View FIGURE 2 View FIGURE 3 View FIGURE 4 View FIGURE 5 View FIGURE 6 View FIGURE 7 View FIGURE 8 View FIGURE 9 ; Tabs. 1–3 View TABLE 1 View TABLE 2 View TABLE 3 )
Uraptera agassizii Müller, Henle, 1841:155–156 (original description; type locality: “ Brazil ”). — Garman, 1913:367–368, figs. 53, 68 (systematic account). — Marini, 1929:452 (listed) View in CoL .
Raja agassizii Günther, 1870:465 (British Museum; catalogue). —Miranda Ribeiro, 1923:34 (Rio de Janeiro, Brazil; catalogue). —Ringuelet, Aramburu, 1960:46 ( Argentina; catalogue). —Gomes et al., 1997:96 (cervicothoracic synarcual cartilage). —Gadig, 1998:50 (listed). —Andreata et al., 2002:1142 (listed).
Raja agassizii Miranda Ribeiro, 1961:4 (Rio de Janeiro; catalogue). —Soares et al., 1999:201 (São Paulo, Brazil; diet). —Estalles et al., 2009:60 ( Argentina; reproductive biology). —Muto et al., 2001:217 (São Paulo, Brazil; diet). —Costa, Chaves, 2006:1 (Paraná and Santa Catarina, Brazil; listed).
Raja (Rioraja) agassizii Menni, 1973:419 , 422, fig. 2 ( Argentina; catalogue). —Roux, 1977:118 ( Brazil and Argentina; listed). —Menni et al., 1984:63 ( Argentina; catalogue). —Andreata, Séret, 1995:581 (Espírito Santo, Brazil; listed). —Deynat, Séret, 1996:81 (squamation). —Barbosa, Gomes, 1998:127 (juvenile external morphology).
Rioraja agassizii Whitley, 1929:254 (original description; type locality: Rio de Janeiro, Brazil). — McEachran , Dunn , 1998:286 (systematic study). — Compagno , 1999:489 (listed). — Mazzoleni , Schwingel , 1999:63 ( Itajaí , Santa Catarina, Brazil; listed). — Menni , Stehmann , 2000:88 ( Argentina, Uruguay and Brazil; listed). — Gomes , Gadig , 2003:29 (São Paulo, Brazil; listed). — McEachran , Aschliman , 2004:101 (systematic study). — Compagno , 2005:533 (listed). — Gomes , Paragó , 2005:55–62, figs. 1, 5, 9 (mucous pores canals). — Oddone et al., 2006:43 (Rio Grande do Sul, Brazil; reproductive biology). — Colonello et al., 2007:277 ( Uruguay and Argentina; reproductive biology). — Cousseau et al., 2007:14 ( Mar del Plata , Argentina; catalogue). — Ebert , Compagno , 2007:116 (listed). — Oddone et al., 2007a:415 (reproductive biology). — Oddone et al., 2007b:594 (reproductive biology). — Oddone , Vooren , 2008:5 (egg capsules). — Gomes et al., 2010:166–167, fig. 263 (Rio de Janeiro, Brazil; catalogue). — Last et al., 2016:944 (listed). — Moreira et al., 2017:1– 12 (clasper morphology). — Gomes et al., 2019:297, fig. 286 (Rio de Janeiro, Brazil; catalogue). — Coelho et al., 2020:1–16 ( Brazil; ecological niche model). — Soares et al., 2020:493–500 (scapulocoracoid morphology).
Raja agassizii meta (in part) Miranda-Ribeiro, 1953:406 (type catalogue, Museu Nacional , Rio de Janeiro, Brazil) .
Raja agassizii picta Miranda Ribeiro, 1953:406 (type catalogue, Museu Nacional, Rio de Janeiro, Brazil) View in CoL .
Raja platana Miranda Ribeiro, 1961:4 (Toku Maru; listed).
Lectotype. MNHN 2430, male, 405 mm TL, 259 mm DW ( Brazil) [designated herein].
Diagnosis. Rioraja agassizii distinguishes from Atlantoraja species by lacking caudal fin (vs. present in Atlantoraja spp. ); posterior margin of anterior nasal flaps straight (vs. rounded); one interdorsal thorn (vs. none); ventral surface of the disc cream in color with few small dark spots (vs. dark ventral surface); dark blotch on the ventral surface of snout (vs. absent); mucous pore canals less numerous and rounded (vs. more numerous and elongated); distance between the first dorsal fin and posterior lobe of pelvic fin somewhat similar to the distance between the first dorsal fin and caudal tip (vs. first dorsal fin closer to the caudal tip in Atlantoraja castelnaui ); clasper with a dorsal terminal 1 cartilage triangular and with a conspicuous proximal shelf (vs. inverted U-shape and shelf absent); shape of the ventral terminal cartilage like an elongated spoon (vs. Y-shape in A. cyclophora , flattened in A. platana and wrench-shaped in A. castelnaui ).
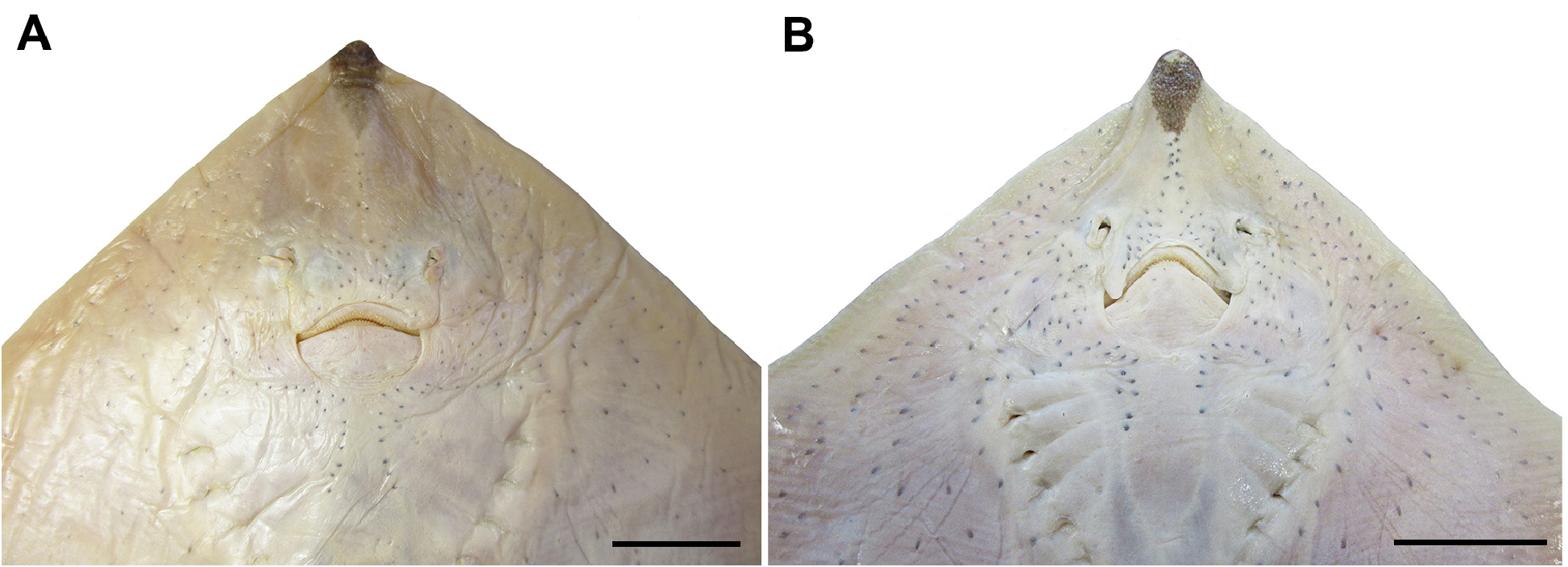
External description. Proportional dimensions of males and females are presented in Tab. 1 View TABLE 1 . Disc rhomboidal with straight or sinuous pectoral anterior margins (in adult males) according to the degree of maturity and posterior pectoral margins slightly convex; 1.2–1.3 times wider than long ( Figs. 2–3 View FIGURE 2 View FIGURE 3 ). Snout long and obtuse, slightly projecting beyond anterior disc margin and 0.2 times the disc length ( Figs. 2–4 View FIGURE 2 View FIGURE 3 View FIGURE 4 ). Posterior lobe of disc covering the anterior portion of the anterior lobe of pelvic fin. Preorbital length 1.2–1.6 times greater than prenasal length and 1.0–1.2 times the preoral length.
Eye and spiracle closely set; spiracles projecting obliquely from midline ( Fig. 2B View FIGURE 2 , 3 View FIGURE 3 ). Eye length greater than spiracle length and goes 1.3–2.5 in times the interorbital distance; adult males with slightly greater eyes than females and juveniles.
Anterior nasal flap broad, not fused medially, about four times the lateral flap and covering the mouth commissure ( Fig. 4 View FIGURE 4 ); lateral margin slightly sigmoid with an anterior notch close to the nasal aperture, posterior margin straight and smooth, and inner margin nearly straight. Lateral flap tube-like, not fringed and outlining the incurrent aperture.
Mouth shape strongly arched and displaying a V-shape in males ( Fig. 4 View FIGURE 4 ). Distance between incurrent apertures 0.9–1.2 times the distance between mouth corners. Teeth with a flat oval base and acute single cusp. Total tooth count in upper jaw 39–51 arranged in four rows and in lower jaw 36–48 arranged in five rows; higher tooth counts in adult females than in males ( Tab. 2 View TABLE 2 ).
Gill openings slitlike and medially situated to the propterygia; first one 1.5–1.9 times greater than fifth and third slightly larger than the first ( Fig. 4 View FIGURE 4 ). Distance between first gill slits 1.7–2.2 times the distance between fifth gill slits ( Tab. 1 View TABLE 1 ); females presenting the last gill slits more widely spaced (6.4–8.3% TL) than males (5.4–6.0% TL).
Pelvic fin deeply concave outwardly with anterior and posterior portions continuous externally but forming distinct lobes ( Fig. 3 View FIGURE 3 ). Anterior lobe similar in size to the posterior one in juveniles and females; 1.4 times posterior lobe in adult males. Anterior margin of the anterior lobe nearly straight close to its origin and convex distally. Origin of anterior lobe slightly anterior and ventral to pectoral fin insertion. Distal tips of radials well-marked and prominent along the posterior margin of anterior lobe. Posterior lobe with strongly convex lateral margin and straight inner margin. Clasper inner length 1.4–2.2 times pelvic posterior lobe length and 1.5–1.6 times clasper outer length.
Dorsal fins rounded, interdorsal space corresponding to 3.0–6.8% of total length, slightly greater in adult males than in adult females ( Tab. 1 View TABLE 1 ). First dorsal fin 1.1–1.2 times higher than the second. Distance from axil of the pelvic fin to first dorsal fin origin 1.2–1.4 times the distance from first dorsal fin origin to caudal tip.
Caudal region slender and elongate, clearly distinct from disc and tapering from pelvic base to its distal tip. Caudal region greater than the disc; distance from cloaca to tail distal tip 1.1–1.2 times the distance from the snout to cloaca. Caudal fin absent.
Squamation. Skin in juveniles smooth on dorsal and ventral surfaces; prickles and thorns more conspicuous in adult specimens. Preorbital, interorbital and midorbital thorns absent in all examined specimens. Mediocaudal and interdorsal thorns more abundant in adult females than in adult males and juveniles of both sexes ( Tab. 2 View TABLE 2 ). Rostral, postorbital, spiracular, interspiracular, nuchal and scapular thorns in adult females, absent in adult males and juveniles. Alar thorns in adult males varying from 72 to 76 arranged in three horizontal rows (8–23).
Coloration. Color pattern light brown dorsally and whitish ventrally with a dark spot on the anterior tip of snout ( Fig. 3 View FIGURE 3 ). Diffuse dark ocellus with a paler center on the medial portion of pectoral fins in most specimens ( Figs. 3A, E, F View FIGURE 3 ). Dorsal surface with light and diffuse and scattered spots mostly in females ( Fig. 3C View FIGURE 3 ). Dark saddles on the caudal region in juveniles, absent in adult specimens. Ventral surface of disc with sensory and mucous pores as black streaks and spots.
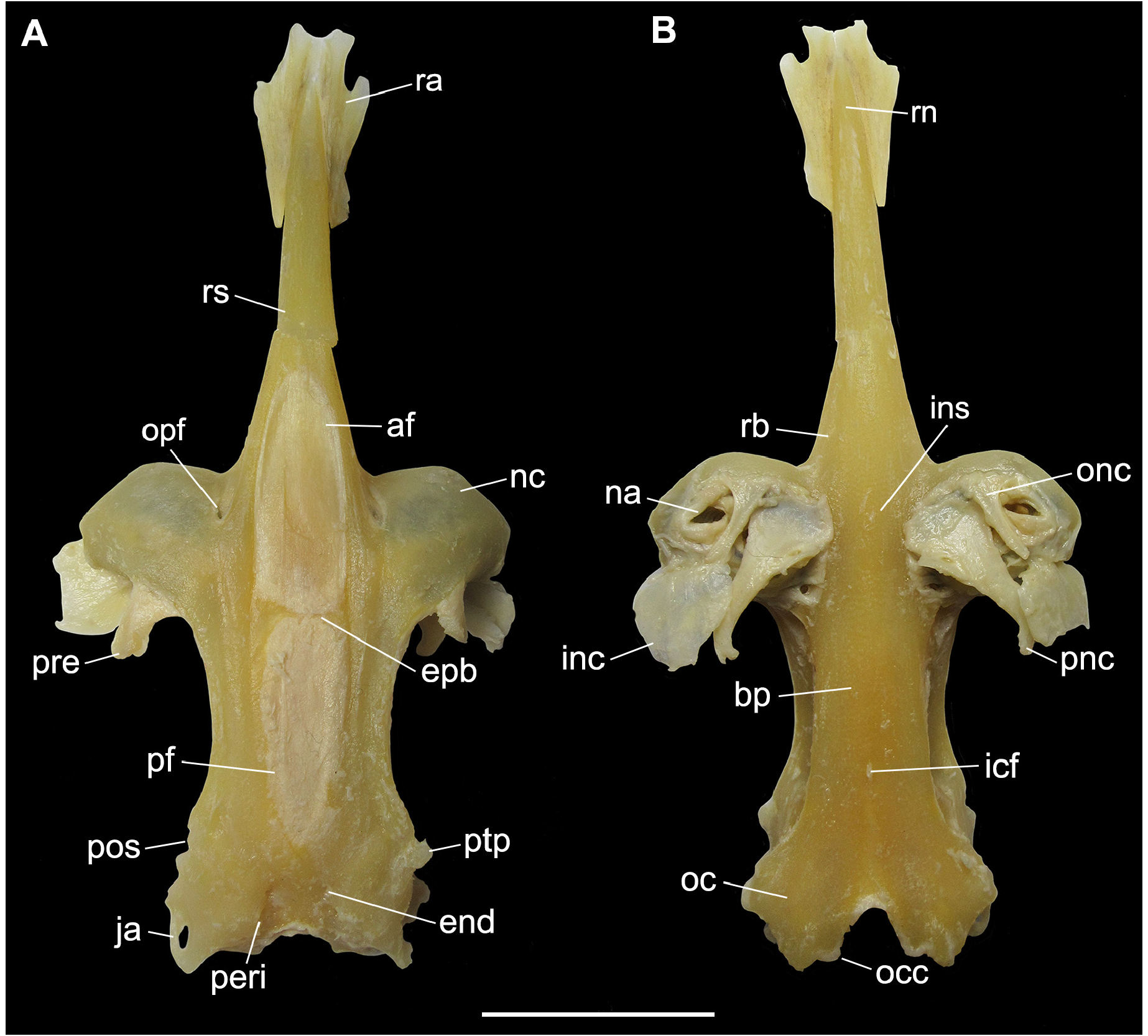
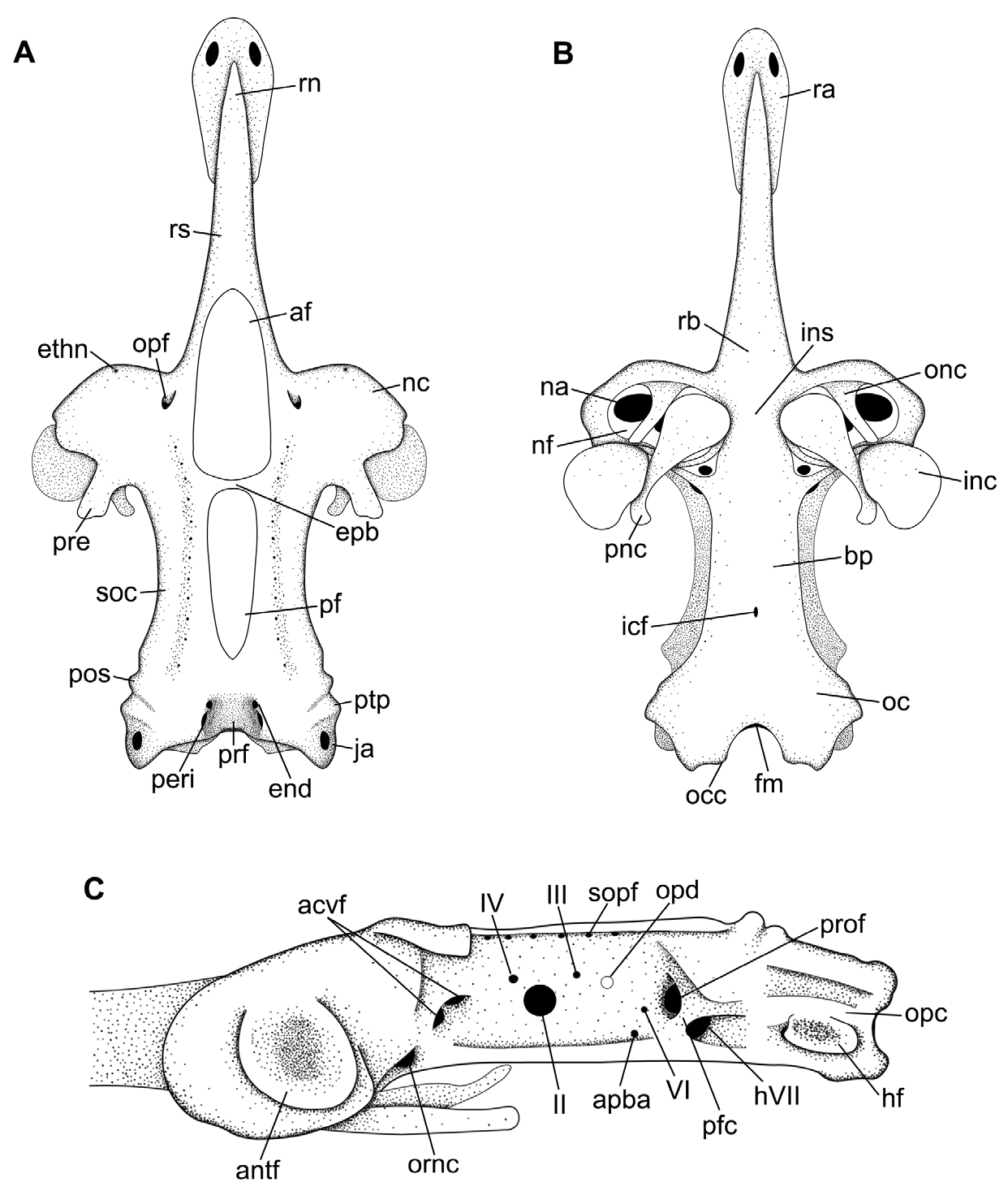
Neurocranium. Largest width at nasal capsules, 1.0–1.1 times in the nasobasal length ( Tab. 3 View TABLE 3 ). Rostrum delimitated posteriorly by the intersection between nasal capsules and rostral cartilages at ophthalmic foramen and extends anteriorly to the tip of rostral appendix ( Figs. 5–6 View FIGURE 5 View FIGURE 6 ). Rostral appendix fused to the rostral cartilage, wider at the rostral node, tapering posteriorly, its length 0.9–1.6 times in rostral cartilage length and 2.4–2.8 times rostral appendix width. Rostral cartilage not flexible, large dorsally from its base to ophthalmic foramina, narrowing towards its anterior tip, its length 1.0–1.4 times in nasobasal length. Furrow present on dorsal surface of rostral cartilage, extending from the anterior region to the anterior margin of anterior fontanelle, extending throughout rostral cartilage in lateral view.
Males (n = 26) Females (n = 36)
Measurements Range Mean Range Mean Nasobasal length 52.0–72.4 55.8 48.3–80.3 56.2 Rostral length 39.1–49.5 45.7 24.6–52.3 45.7 Rostral appendix length 47.5–67.6 56.3 43.6–54.8 49.8 Rostral appendix width 28.2–55.0 45.1 28.6–57.7 43.4 Rostral base width 7.9–17.2 14.3 10.2–20.4 14.9 Width across nasal capsules 46.2–60.4 50.2 44.2–65.6 51.3 Nasal aperture width 10.3–22.4 17.8 15.4–23.0 18.4 Internasal distance 8.0–14.6 10.3 7.9–15.2 11.1 Interorbital distance 13.2–25.3 18.4 16.2–24.6 18.9 Anterior fontanelle length 17.1–31.0 23.5 21.2–34.4 25.5 Anterior fontanelle width 15.6–22.1 17.6 14.3–16.7 15.5 Epiphyseal bridge width 2.1–4.5 2.4 2.8–4.1 2.9 Posterior fontanelle length 21.3–32.8 24.9 19.8–34.4 24.4 Posterior fontanelle width 5.1–10.3 7.1 5.8–11.1 7.6 Basal plate width 10.7–26.0 13.7 10.2–18.5 13.8 Width across otic capsules 22.0–31.0 25.0 17.1–36.1 25.3
Nasal capsule obliquely oriented to the cranial roof and perpendicular to the rostrum, with anterior edge slightly anterior to the rostral cartilage base ( Figs. 5A View FIGURE 5 , 6A View FIGURE 6 ). Foramen of ethmoidal nerve on anterodorsal surface of the nasal capsule, and lateral to the foramen lies the antorbital facet which articulates to the antorbital cartilage. Nasal aperture wide with a rounded anterior margin and nearly straight posterior margin; ophtalmicus foramen anteriorly and oronasal canal more posteriorly. Internasal space 4.0–4.4 times in the greatest cranium width. Outer nasal cartilage triangular, dividing incurrent and excurrent apertures laterally oriented ( Figs. 5B View FIGURE 5 , 6B View FIGURE 6 ). Inner nasal cartilage rounded and laterally expanded, its length two-thirds of its width, it lies on posterior margin of nasal apertures, extending from the internasal septum to the lateral margin of nasal fontanelle. Posterior nasal cartilage broad anteriorly, tapering posteriorly, curved distally, oblique to anteroposterior axis, its posterior tip more laterally situated than the anterior one ( Figs. 5B View FIGURE 5 , 6B View FIGURE 6 ).
Cranial roof extends from the posterior margin of the anterior fontanelle to the parietal fossa, slightly posterior to the posterior fontanelle and limited laterally by the supraorbital crest ( Figs. 5A, C View FIGURE 5 ). Two dorsal fontanelles; anterior fontanelle wider than the posterior, teardrop-shaped beginning at posterior third of rostral cartilage, extending to anterior edge of supraorbital crest ( Figs. 5A View FIGURE 5 , 6A View FIGURE 6 ), 1.4–3 times longer than wide and relatively wider in males than in females. Posterior fontanelle almost the same length of the anterior one, extending from the epiphysial bridge to the parietal fossa. Epiphysial bridge slender with a vertical extension on its median portion; bridge corresponding to 16–21.6 times in nasobasal length. Preorbital process well-developed on the posterior third of nasal capsule and continuous to the supraorbital crest.
Basal plate begins anteriorly at the shallow ethmoidal fossa right after the nasal capsules, runs to the posteroventral end of the optic region and ends at the ventral margin of the foramen magnum ( Figs. 5B View FIGURE 5 , 6B View FIGURE 6 ). A single foramen for the internal carotid artery is situated on the posterior third at the midline. Basal plate narrower than cranial roof, its width 5.4–9.4 times in nasobasal length ( Tab. 3 View TABLE 3 ).
Orbital region delimited anteriorly by the posterior margin of the nasal capsules, posteriorly by the anterior margin of otic capsules, dorsally by the cranial roof, ventrally by the basal plate, and medially by the orbital wall, which is perforated by several foramina to the passage of cranial nerves and blood vessels ( Fig. 6C View FIGURE 6 ). Oronasal canal ventrally close to the basal plate. Foramina for the anterior cerebral vein on the same vertical line between the oronasal canal and the foramina for the ophtalmicus nerve. Large foramen for the optic nerve ( II) posterior to the foramina for the anterior cerebral vein and ventral to a single foramen for the trochlear nerve ( IV). Foramen for the oculomotor nerve ( III) posterior to the optic nerve and anterior and dorsal to optic pedicel. Foramen for the afferent pseudobranchial artery ventral to the optic pedicel and close to the basal plate. Prootic foramen for the passage of the trigeminus and facialis nerves ( V + VII) as the second largest aperture of the orbital wall and slightly posterior to the foramen for the abducens nerve ( VI). Prootic foramen separated posteriorly from the hyomandibular ramus of the facialis nerve by the prefacial commissure, close to the otic region ( Fig. 6C View FIGURE 6 ).
Otic region as the second widest region of the neurocranium ( Figs. 5–6 View FIGURE 5 View FIGURE 6 ), composed of otic capsules, impressions of semicircular canals, parietal fossa and foramina for the endolymphatic (smaller aperture) and perilymphatic canals. Behind the sphenopterotic
crest, there is a well-developed opistotic crest superior that runs dorsally to the otic capsule and ends close to the occipital region. The ventral portion of the otic region is delimited laterally by the opistotic crest inferior and the hyomandibular facet.
Occiptal region on the posterior surface of the chondrocranium, composed by the large foramen magnum and the lateral edges of the hyomandibular facet. On each side of the foramen magnum lies the occipital condyle and lateral to it the foramen for the vagus nerve (X). Lateral to the foramen for the vagus nerve lies the foramen for the glossopharyngeal nerve ( IX).
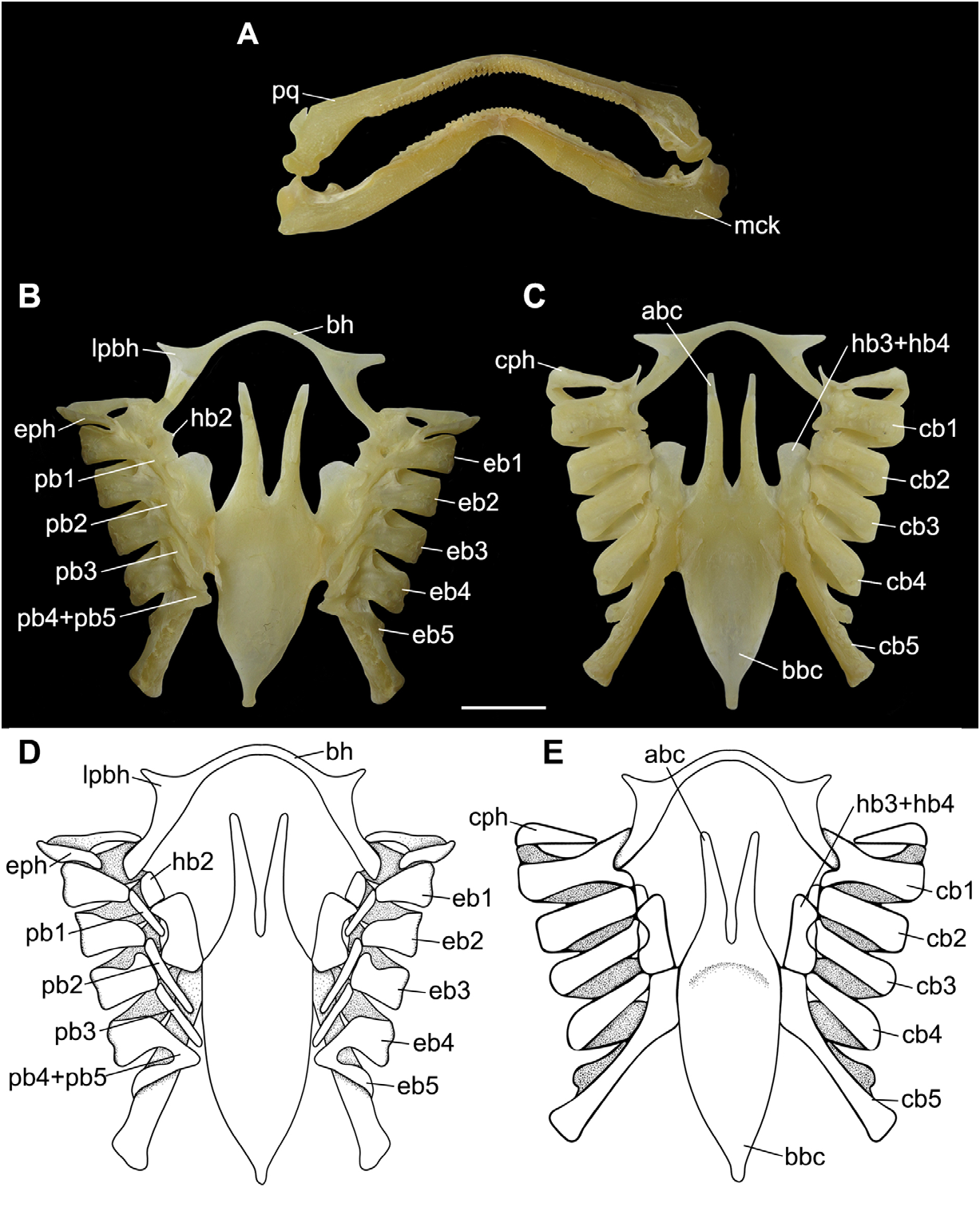
Visceral skeleton. Upper jaw concave medially; lower jaw strongly convex ( Fig. 7A View FIGURE 7 ). Both jaws tapering towards midline. Hyomandibular cartilage tapering anteriorly. Dorsal (epi-) and ventral (cerato-) pseudohyoid rodlike, laterally to the basihyal cartilage ( Figs. 7B–D View FIGURE 7 ). Basihyal cartilage wide, slender, strongly arched, with well-developed anterolateral projections. Hypobranchial 2 one-third the length of posterior hypobranchials, rounded and restricted to the medial portion of ceratobranchials 1 and 2. Hypobranchials 3 and 4 fused, not reaching the anterior portion of basibranchial copula. Ceratobranchials 1–4 overlapping medially; ceratobranchial 5 articulating directly to the basibranchial copula. Epibranchials 1 and 5 longer than medial ones. Pharyngobranchials 2 and 3 longer than the first; gill pickax half-length of the pharyngobranchial 1. Anterior processes of basibranchial copula straight, tapering anteriorly and shorter than the posterior portion of basibranchial copula. Cardiobranchial cartilage elongate; accessory cartilage of the basibranchial copula triangular and corresponding to one-fifth of the cardiobranchial cartilage.
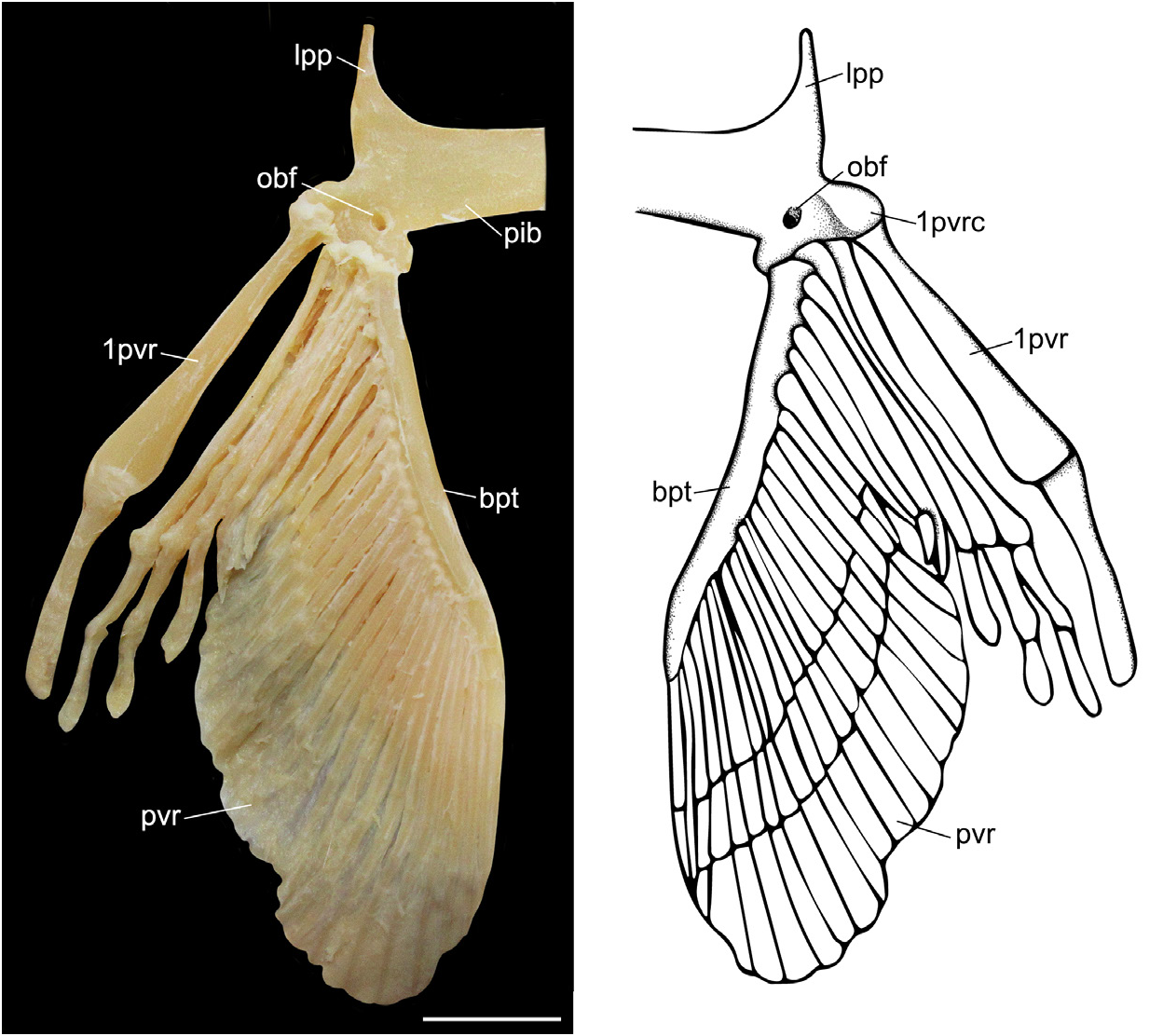
Pelvic girdle and fin. Pelvic bar transverse and nearly straight, with a process extending forward from the outer corner at each end ( Fig. 8 View FIGURE 8 ). Median ischio-pubic region and lateral iliac regions with obturatorial nerve foramina for diazonal nerves. Lateral prepelvic process triangular and directed anteriorly. Condyle for articulation ventrolateral, with enlarged anterior radial; obturator foramina posterior to the puboischiadic condyle. Pelvic anterior lobe with six radial segments; first radial segment stout, elongate, with very concave articular surface for puboischiadic condyle. Posterior lobe with 20 radial segments.
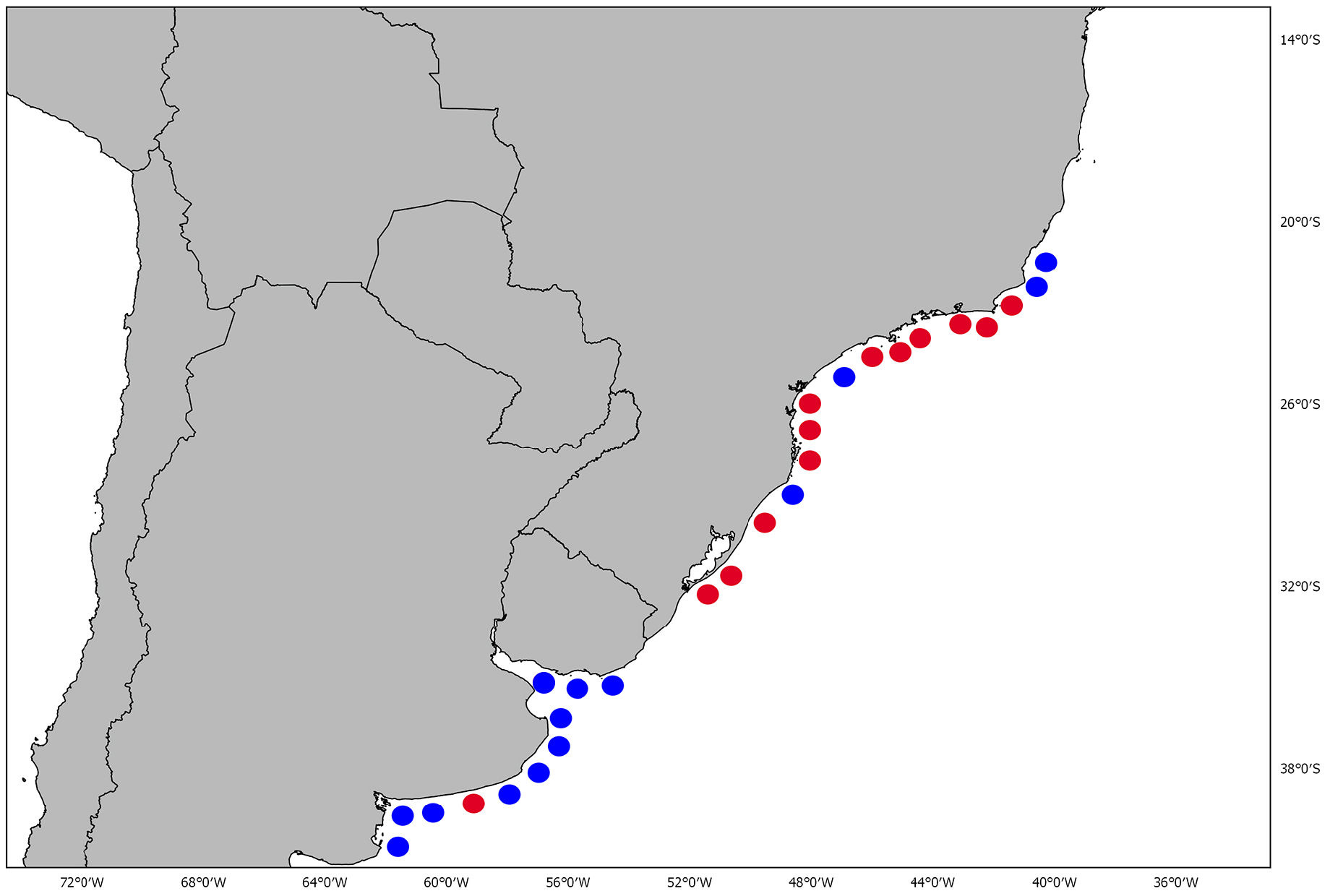
Geographic distribution. This species is endemic to the coastal waters of southwestern Atlantic occurring from Espírito Santo to Rio Grande do Sul, Brazil , Uruguay and Northern Argentina ( Fig. 9 View FIGURE 9 ; see also, Menni, 1973; Menni et al., 1984; McEachran, Miyake, 1990; Andreata, Serét, 1995; Gadig, 1998; Mazzoleni, Schwingel, 1999; Cousseau et al., 2000, 2007; Meneses, Paesch, 2003; Coelho et al., 2020). It has been reported in temperate waters (Menni, 1973; Bellisio et al., 1979; Menni et al., 1984; Menni, Stehmann, 2000) at depths up to 150 m (Andreata, Séret, 1995; Vooren, 1997; Cousseau et al., 2000, 2007) although rare over 120 meters depth (Oddone et al., 2007a).
Ecological notes. Largest female and male specimens recorded in Argentina, 710 mm and 629 mm in TL, respectively (Cousseau et al., 2007; Estalles et al., 2009). Females become sexually mature at 200 mm DW and the first sexual maturation takes with 250 mm DW, while males become mature at 200 mm DW (495–577 mm TL in females and 420–520 mm TL in males, Cousseau et al., 2007; 320 mm TL in Oddone et al., 2007a,b; 570 mm and 504 mm in TL for females and males respectively, Estalles et al., 2009). Ovulation, egg-laying and presence of sperm in the seminal vesicle all year round and at least one peak in the sexual activity (Oddone et al., 2007a,b).
Siqueira (1999) found that in specimens of Rioraja agassizii captured in Santos, São Paulo State, the egg-laying starts by the right uterus. Egg capsules bright and light brown, laterally keeled and symmetrically convex with a smooth surface. Mean total length of the egg capsules 47 mm and mean width 31 mm (Oddone, Amorim, 2006). Distance from anterior apron to apex of anterior horn and flange with less than 50% of the length of egg case with well-defined filaments and strongly adhered to the lateral keel. Lateral edge of 61 to 68 mm (Mabragaña et al., 2009).
Stomach contents have been reported to include crustaceans ( Alpheidae, Amphipoda, Caridea, Copepoda, Cirripedia, Isopoda, Stomatopoda , Ogyrididae , Penaeidae , Solenoceridae , Sicyonidae ), nematodes, polychaetes, and teleost fishes ( Bothidae , Ophidiidae , Scorpedinidae, Serranidae , Bathracoididae, Ophichthidae , Bragmacerotidae) (Muto et al., 2001; Pasquino et al., 2011; Motta et al., 2016).
Common name. Rio skate.
Conservation status. This species is categorized as ‘Vulnerable’ (VU) (Pollom et al., 2020).
Remarks. Müller, Henle (1841) described the species Uraptera agassizii based on four specimens of which one is housed in Berlin (ZMB, not examined) and three in Paris (MNHN) but only one was found in alcohol and examined in this study (MNHN 2430, Fig. 2B View FIGURE 2 ).
Whitley (1929) designated the genus Rioraja in substitution of Uraptera because it was already pre-occupied by Uraptera Billberg, 1820, a genus of Lepidoptera (Insecta). However, subsequent authors used the species name Raja agassizi (e.g., Miranda Ribeiro, 1907, 1923; Bigelow, Schroeder, 1953; Ruschi, 1965; Menni, 1972b, 1973; Sadowsky, 1973; Figueiredo, 1977; Lucena, Lucena, 1981; Menni et al., 1984; Gomes et al., 1997; Barbosa, Gomes, 1998). Later, Menni (1973) synonymized the name Uraptera in Rioraja as proposed by Whitley (1929).
According to Roux (1977: 118), a specimen of Rioraja agassizii was collected in Fernando de Noronha, which represents the northernmost record for the species. The specimen is deposited in the MNHN under the catalog number 1984 -0019 (311 mm TL, 202 mm DW) and its identification was confirmed by the last author (ULG). Nevertheless, its catch locality remains uncertain and may have been mislabeled, since the species is only found in the continental waters of southwestern Atlantic.
Material examined. Argentina. MACN 3825, male, 316 mm TL, 222 mm DW, Puerto Quenquén. Brazil. Paraná: AC.UERJ 1105, male, 26°20’S 47°52’W. AC.UERJ 1106, male, 25°3’S 46°16’W. Rio de Janeiro: AC.UERJ 1360, male, 295 mm TL, Marambaia Restinga. AC. UERJ 1361, male, 310 mm TL, Marambaia Restinga. AC. UERJ 1362, male, 297 mm TL, Marambaia Restinga. AC.UERJ 1363, male, 267 mm TL, Marambaia Restinga. AC.UERJ 1367, male, 315 mm TL, Marambaia Restinga, Angra dos Reis. AC.UERJ 1380, female, 474 mm TL, 320 mm DW, Rio de Janeiro. AC.UERJ 1386, male, 440 mm TL, Rio de Janeiro. MNRJ 578, male, 367 mm TL, 230 mm DW, Farol de Santana. MNRJ 591, 5 females, 270–441 mm TL, 2 males, 311–381 mm TL, Rio de Janeiro. MNRJ 593, female, 450 mm TL, 282 mm DW, Farol de Santana. UERJ 695, female, 385 mm TL, 329 mm DW, Barra de Guaratiba. UERJ 741, female, 177 mm TL, 128 mm DW, Ilha Grande. UERJ 743, female, 270 mm TL, 168 mm DW, Ilha de Santana. UERJ 747, female, 277 mm TL, 190 mm DW, Ilha de Santana. UERJ 782, female, 430 mm TL, 313 mm DW, Ilha Rasa. UERJ 788, male, 306 mm TL, 182 mm DW. UERJ 943, 3, 90–149 mm TL, between Macaé and Niterói. UERJ 1178, 2 males, 360–436 mm TL, Macaé. UERJ 1179, female, 490 mm TL, 350 mm DW, Macaé. UERJ 1428, male, 188 mm TL, 116 mm DW, Ilha Grande. UERJ 1525, female, 294 mm TL, 190 mm DW, Maricá. UERJ 1569, female, 466 mm TL, 332 mm DW, Barra de Guaratiba. UERJ 1580, male, 532 mm TL, 345 mm DW, Barra de Guaratiba. UERJ 1581, female, 466 mm TL, 342 mm DW, Barra de Guaratiba. UERJ 1716, female, 153 mm TL, 95 mm DW, Ilha Grande. USU 119, 2 males, 318–335 mm TL, 2 females, 395–434 mm TL, Rio de Janeiro. USU 598, 4 females, 356–503 mm TL, Guaratiba. USU 2216, male, 495 mm TL, 419 mm DW. Rio Grande do Sul: MCP 7120, male, 260 mm TL, 159 mm DW, Torres. MCP 7122, male, 237 mm TL, 152 mm DW, Torres. MCP 7156, female, 586 mm TL, 381 mm DW, Torres. MCP 7158, female, 277 mm TL, 172 mm DW, Torres. MCP 7444, male, 421 mm TL, 263 mm DW, between Farol da Solidão and Farol da Conceição. UERJ 879, male, 356 mm TL, 239 mm DW, between Ilha do Bom Abrigo, São Paulo, and Paranaguá, Paraná. UERJ 880, male, 323 mm TL, 213 mm DW, between Ilha do Bom Abrigo, São Paulo, and Paranaguá, Paraná. UERJ 892.1, female, 90 mm TL, 60 mm DW, between Southern Ilha Bela, São Paulo, and Ilha Grande, Rio de Janeiro. UERJ 892.2, male, 172 mm TL, 102 mm DW, between Southern Ilha Bela, São Paulo, and Ilha Grande, Rio de Janeiro. Santa Catarina: AC.UERJ 1107, male, 27°20’S 48°2’W. MCP 1523, male, 318 mm TL, 205 mm DW, Florianópolis. MCP 4834, female, 360 mm TL, 262 mm DW, Ponta das Canas, Florianópolis. São Paulo: AC.UERJ 1044, male, 245 mm DW, Santos. AC.UERJ 1047, male, 355 mm TL, 240 mm DW, Santos. AC.UERJ 1048, male, 365 mm TL, 240 mm DW, Santos. AC.UERJ 1050, male, 360 mm TL, 245 mm DW, Santos. AC.UERJ 1051, male, 250 mm DW, Santos. AC.UERJ 1052, male, 390 mm TL, 265 mm DW, Santos. AC.UERJ 1053, male, 380 mm TL, 240 mm DW, Santos. AC.UERJ 1054, male, 230 mm DW, Santos. AC.UERJ 1055, male, 250 mm DW, Santos. AC.UERJ 1056, male, 240 mm DW, Santos. AC.UERJ 1057, male, 220 mm DW, Santos. AC.UERJ 1070, male, 390 mm TL, 255 mm DW, Santos. AC.UERJ 1071, male, 350 mm TL, 240 mm DW, Santos. AC.UERJ 1073, male, 395 mm TL, 250 mm DW, Santos. AC.UERJ 1074, male, 209 mm DW, Santos. AC.UERJ 1075, 231 mm DW, Santos. AC.UERJ 1102, male, Santos. AC.UERJ 1103, male, Santos, 24°11’S 46°29’W. AC.UERJ 1104, male, Santos, 24°39’S 46°17’W. AC.UERJ 1108, male, Santos 24°48’S 47°23’W. AC.UERJ 1109, male, Santos. AC.UERJ 1110, male, Santos. AC.UERJ 1111, male, Santos. AC.UERJ 1112, male, Santos. AC.UERJ 1113, male, Santos. AC.UERJ 1114, male, Santos. AC.UERJ 1115, male, Santos. AC.UERJ 1116, male, Santos. AC.UERJ 1117, male, Santos. AC.UERJ 1381, female, 567 mm TL, 365 mm DW, Santos. MNRJ 588, male, 356 mm TL, 226 mm DW. MNRJ 50512, female, 505 mm TL, 23º11’30”S 41º2’55”W. MZUSP (uncatalogued), male, 495 mm TL, 419 mm DW. MZUSP (uncatalogued), 3, 410– 561 mm TL. MZUSP 117280, female, 543 mm TL. UERJ 830, male, 138 mm TL, 85 mm DW, Santos. UERJ 897, 4, 112–202 mm TL, Ilha do Trigo. UERJ 898, 5, 102–135 mm TL. UERJ 1719, female, 472 mm TL, 323 mm DW, Ubatuba.
| V |
Royal British Columbia Museum - Herbarium |
| VI |
Mykotektet, National Veterinary Institute |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Rioraja agassizii (Müller & Henle, 1841)
| Soares, Karla D. A., Moreira, Renan A. & Gomes, Ulisses L. 2021 |
Uraptera agassizii Müller, Henle, 1841:155–156 (original description; type locality: “ Brazil ”). — Garman , 1913:367–368, figs. 53, 68 (systematic account). — Marini, 1929:452 (listed)
| Muller, Henle 1841: 156 |