Macropsidius kajkanus Mityaev, 1990
|
publication ID |
https://doi.org/ 10.11646/zootaxa.5165.3.5 |
|
publication LSID |
lsid:zoobank.org:pub:C23CFB96-D56F-4C2F-AD98-3C0FD973DD9A |
|
DOI |
https://doi.org/10.5281/zenodo.6838434 |
|
persistent identifier |
https://treatment.plazi.org/id/03BF87A7-C406-FF9C-C984-6EB9CA9CB49B |
|
treatment provided by |
Plazi |
|
scientific name |
Macropsidius kajkanus Mityaev, 1990 |
| status |
|
Macropsidius kajkanus Mityaev, 1990 View in CoL
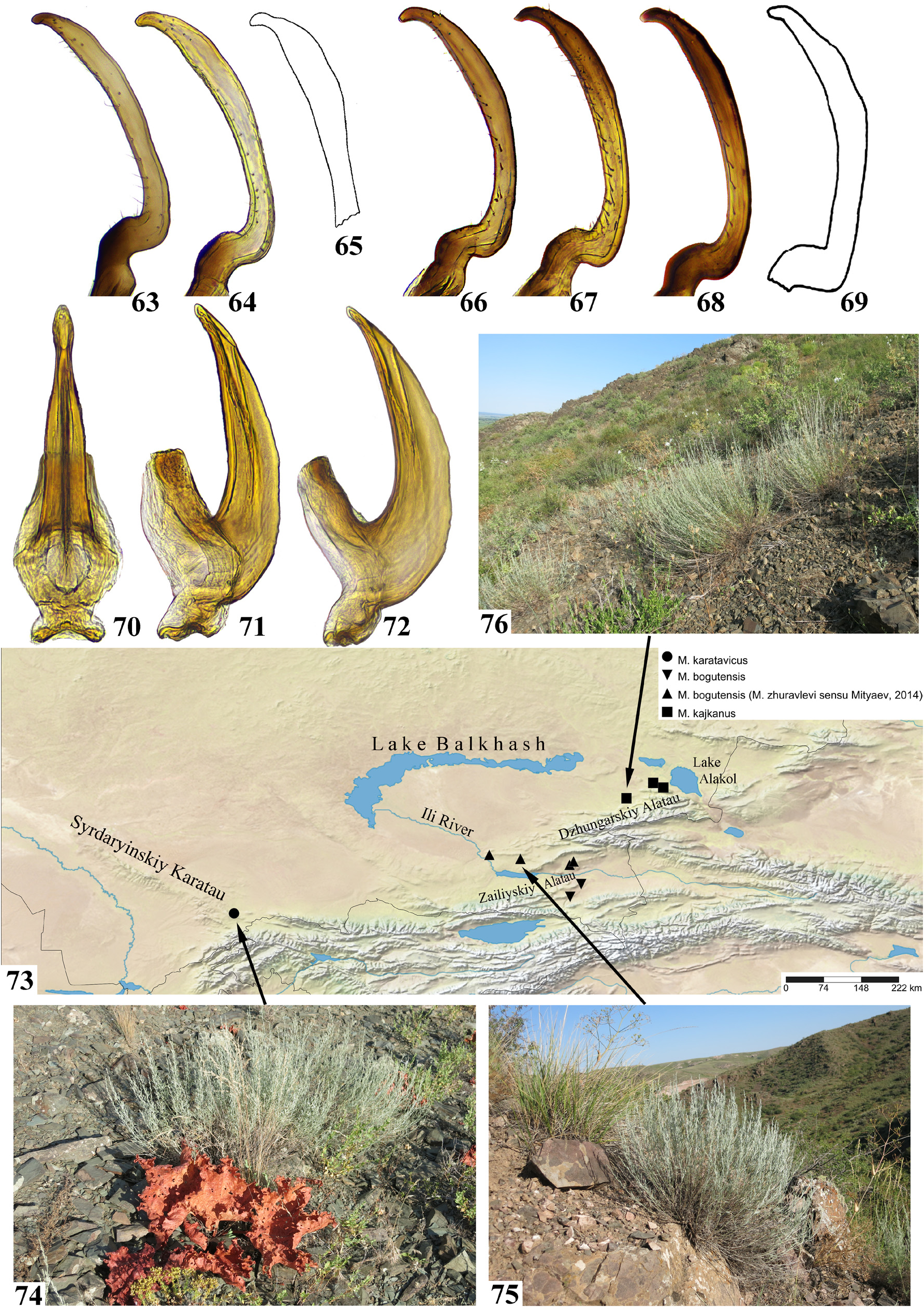
Figs. 29–33 View FIGURES 19–33 , 70–72 View FIGURES 63–76
Description. In appearance and coloration, indistinguishable from most other species of Macropsidius .
Aedeagus in posterior view with narrow lateral carinae slightly not reaching gonopore ( Fig. 70 View FIGURES 63–76 ). Ventral carina with evenly curved margin, runs all along shaft from base to gonopore ( Figs. 71–72 View FIGURES 63–76 ). Aedeagal apex somewhat expanded, with only 1–3 if any blunt denticles on each side. Shape of pygofer appendages and styles same as in most other members of genus.
Hosts. Originally collected from undetermined species of Artemisia ( Mityaev, 1990) ; later A. (Seriphidium) juncea was recorded as a host plant for specimens from the type series ( Mityaev, 2002). Specimens for this study were collected from A. (Seriphidium) juncea growing on stony slopes ( Fig. 76 View FIGURES 63–76 ).
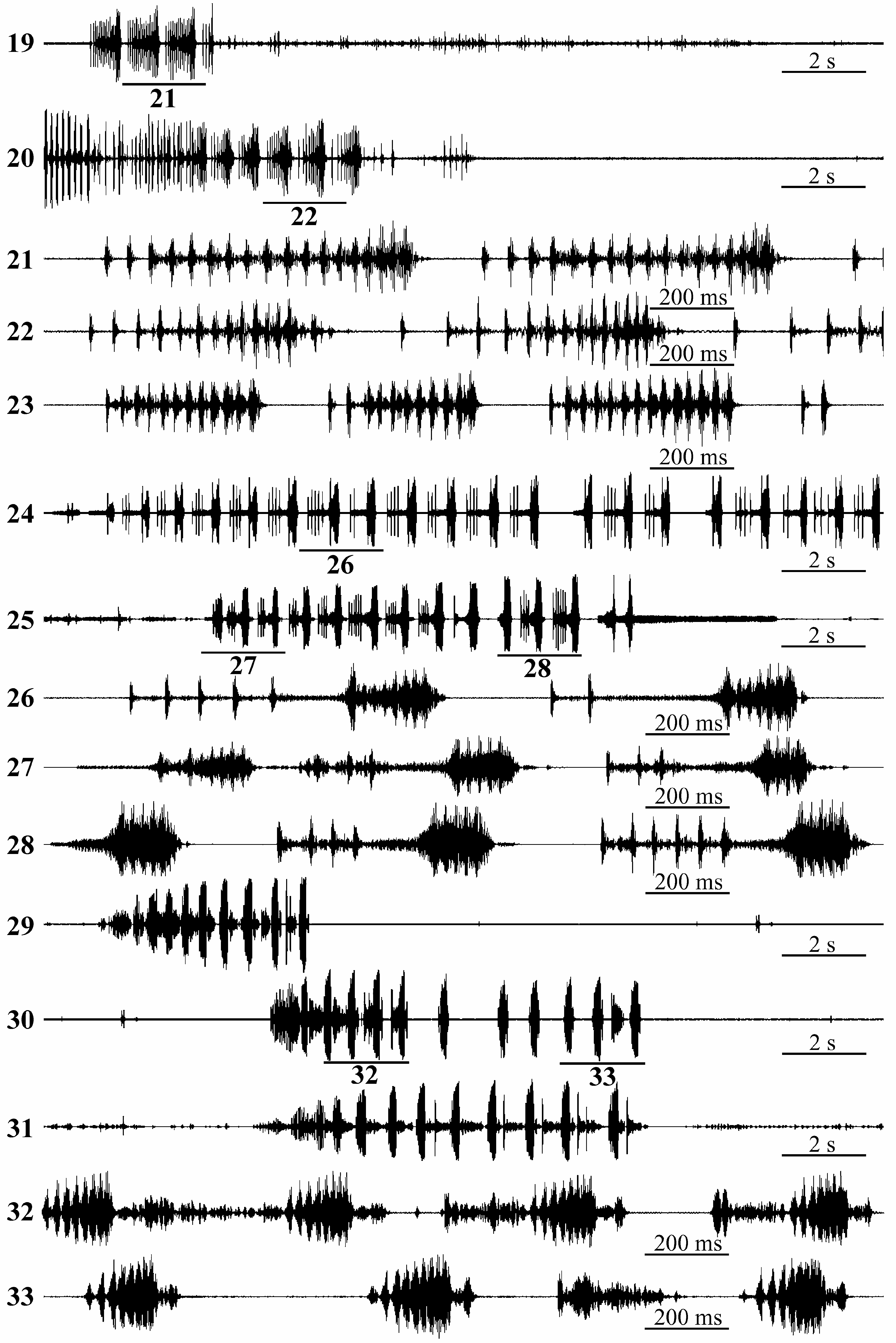
Calling signal. Signals of one male from Southeastern Kazakhstan (foothills of Dzhungarskiy Alatau near the gorge of the Lepsy River at its exit from the mountains to the plain, 13 km south of Kolbay Village, 45.700 N, 80.297 E, from A. (Seriphidium) juncea , 19. VI. 2017) were recorded at 29 o C GoogleMaps .
Male calling signal is a phrase lasting for 5–10 s in the recordings and consisting of syllables following each other with a period from 0.3– 0.4 s at the beginning to 0.8– 0.9 s (occasionally, up to 1 s and more) at the end ( Figs. 29–31 View FIGURES 19–33 ). Each syllable includes the low-amplitude rather variable initial part and the high-amplitude component consisting of 6–10 partially merged pulses ( Figs. 32–33 View FIGURES 19–33 ). Often, low-amplitude parts of syllables gradually reduce towards the end of a phrase, so that the end syllables in a phrase sometimes consist of only high-amplitude component ( Fig. 30 View FIGURES 19–33 ).
On the whole, in temporal pattern, the calling signal of M. kajkanus is similar to that of M. bogutensis . Syllable repetition period ranges in these species considerably overlap, and syllables share similar final component consisting of several partially merged pulses ( Figs. 26–28 and 32–33 View FIGURES 19–33 ). Since the initial low-amplitude part of a syllable in both species is variable, occasionally, some syllables in phrases of two species are almost indistinguishable (two last syllables on Fig. 27 View FIGURES 19–33 and on Fig. 32 View FIGURES 19–33 ).
Distribution. All known records are restricted to the arid foothills of the eastern part of Dzhungarskiy Alatau, Southeastern Kazakhstan ( Fig. 73 View FIGURES 63–76 ).
Remark. In appearance and genitalia shape, M. kajkanus is very similar to M. valiturus and M. araxes ( Tishechkin, 2014) . However, these two species are known only from Caucasus, Transcaucasia, and the adjacent regions of Iran, whereas M. kajkanus was never found outside the eastern part of Dzhungarskiy Alatau. For this reason, their synonymization without comprehensive investigation of Caucasian species would be premature.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |