Atrophotergum, Mesibov, Robert, 2004
|
publication ID |
https://doi.org/10.5281/zenodo.157291 |
|
DOI |
https://doi.org/10.5281/zenodo.6269246 |
|
persistent identifier |
https://treatment.plazi.org/id/F92787AB-875B-FFBE-8626-FBAEFC84AFB5 |
|
treatment provided by |
Plazi |
|
scientific name |
Atrophotergum |
| status |
gen. nov. |
Atrophotergum View in CoL n. gen.
Type species: Atrophotergum silvaticum n. sp., by present designation.
Other assigned species: A. bonhami n. sp., A. montanum n. sp., A. pastorale n. sp., A, sodalis n. sp., A. wurrawurraense n. sp.
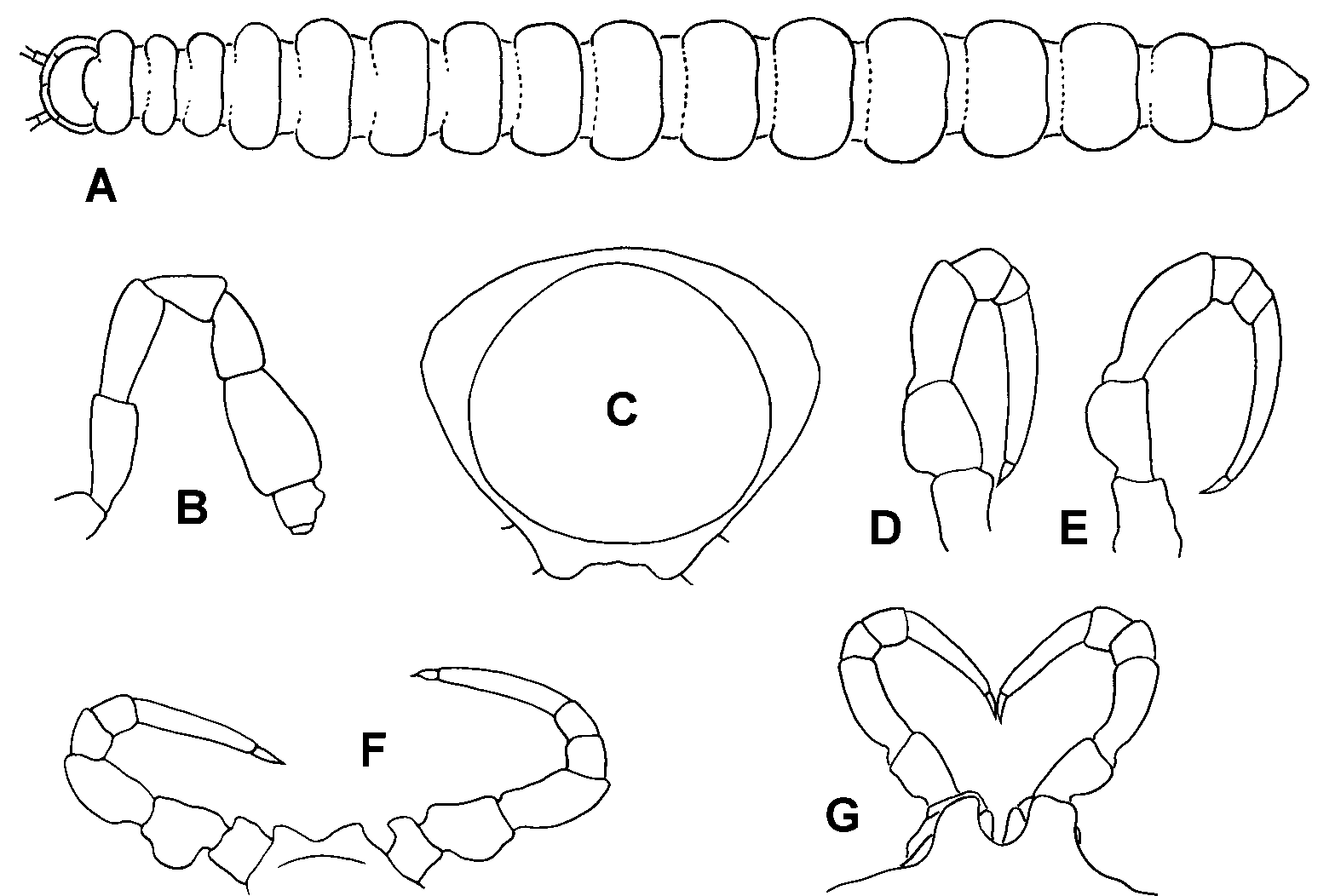
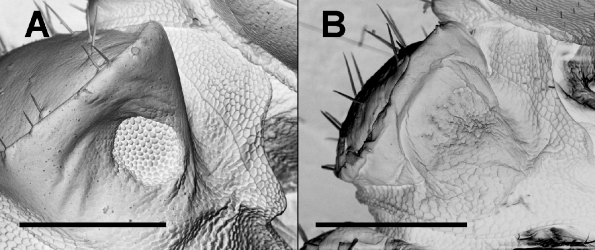
Diagnosis: Small dalodesmids with head + 19 segments and prominent paranota with rounded corners. Males with numerous sphaerotrichomes on anterior legs; a round pit on the underside of the somite 2 paranotum; low, blunt projections on the sternites of the last somites, close to the legs; and somite 3 and 4 metatergites particularly small relative to more posterior metatergites. Clearly distinguished from similar and cooccurring dalodesmids by the solenomerite being carried on a large posterior projection arising midway along the telopodite.
Description: Males 6–14 mm long, 0.5–1.0 mm in maximum vertical diameter. Head moderately setose, antennal sockets slightly impressed, antennae separated by ca. 1.5–1.75 times a base diameter; antennomere 6 the longest and widest ( Fig. 1 View FIGURE 1 B). Collum lightly setose, not as wide as head, anterior margin straight, posterior margin emarginate, corners rounded. Somite 2 metatergite about as wide as head; lateral margin of the paranotum slightly lower than the corner of the collum, anterior margin slightly produced forward; a round pit with a welldefined rim on the underside of the paranotum ( Fig. 3 View FIGURE 3 A). Somite 3 and 4 metatergites distinctly narrower and shorter than all other metatergites ( Fig. 1 View FIGURE 1 A); more posterior metatergites gradually increasing in width and length to somite 13, then decreasing slightly. Waist welldefined on all diplosegments, posterior metatergites with a faintly defined, very shallow transverse depression and three transverse rows of short setae (two anterior to the depression, one posterior); metatergite surfaces slightly roughened. All paranota somewhat inflated ( Fig. 1 View FIGURE 1 C), margins slightly thickened, a few marginal setae on the first few somites. Ozopores on somites 5, 7, 9, 10, 12, 13, 15–17, opening dorsally in the posterior half of the paranotum close to the margin. Legs ( Figs. 1 View FIGURE 1 D, E) incrassate, anteriorly more so; anterior legs ventrally with dense, short setae on coxa, prefemur and femur, and numerous sphaerotrichomes on tarsus, tibia, postfemur and distal end of femur. Tarsi long, ranging from ca. 1.3 to ca. 1.7 times the length of the next longest podomere, the femur. Genital opening on leg 2 coxa a short, mesal projection in the form of a truncated cone. Legpairs 4 and 5 with coxae separated by setose areas on sternum. Legpair 7 widely spaced; legpair 6 closer together, separated by a pair of setose sternal projections; flexed gonopods reaching to legpair 6. Short, blunt projections present on the sternites of the last somites ( 13–17 in the type species), close to the leg bases ( Fig. 4). Spiracles small; on diplosegments, the anterior spiracle opening just dorsal to the anterior leg base, the posterior spiracle opening about midway between leg bases. Preanal ring moderately setose; hypoproct trapezoidal; epiproct gradually tapering to rounded point and projecting slightly past anal valves.
Gonopod aperture subcircular, about half the width of the somite 7 prozonite, the rim
projecting ventrally on each side posterolaterally. Gonopod coxae entirely contained
within aperture; coxae pyriform, tapering posteriorly, very lightly joined posteriorly on
mesal surfaces. Cannula prominent, inserting on mesal side of telopodite base; prostatic
groove running on mesal side of telopodite into the solenomerite flange (see below);
telopodite bare apart from short, dense setae at base and sparse longer setae near base on
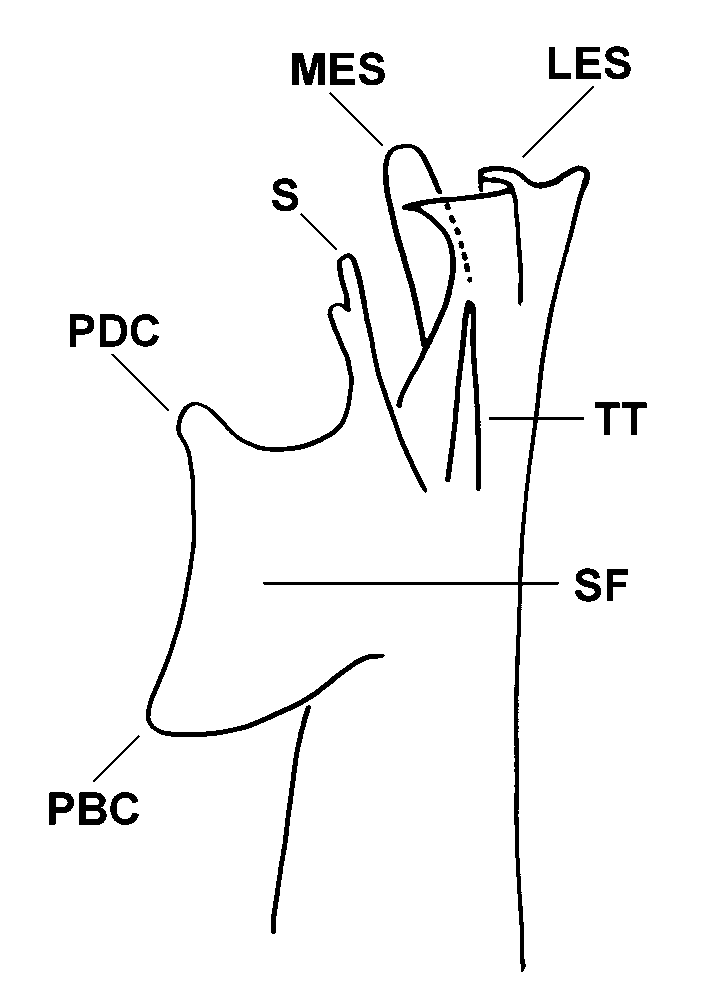
posterior surface. Telopodite with four elements recognizable across all Atrophotergum
spp. and shown diagramatically in Fig. 5 View FIGURE 5 :
(1) Solenomerite flange (SF). A mesolaterally flattened projection arising about midway along the telopodite from the mesal side of the posterior surface. The solenomerite (S) extends from the anterodistal corner of the flange. The posterodistal corner of the flange (PDC) is variously extended and its form is speciesspecific. The posterobasal corner (PBC) is also extended and generally bent laterally.
(2) Tibiotarsus (TT). A rodlike, bluntly pointed process arising just lateral and distal to the solenomerite flange. The implied homology with the tibiotarsus on the gonopod of other Polydesmida is a tentative one based on shape and position.
(3) Mesal end structure (MES). One or two processes arising from the mesal side of the anterior surface of the telopodite, near its tip.
(4) Lateral end structure (LES). The distal end of the telopodite is a complex, flattened structure with its posterior corner extended as a sharp point, distal to the end of the solenomerite, and with the anterior corner variously shaped and produced. Between the two corners, the LES is deeply and narrowly folded (concave laterally).
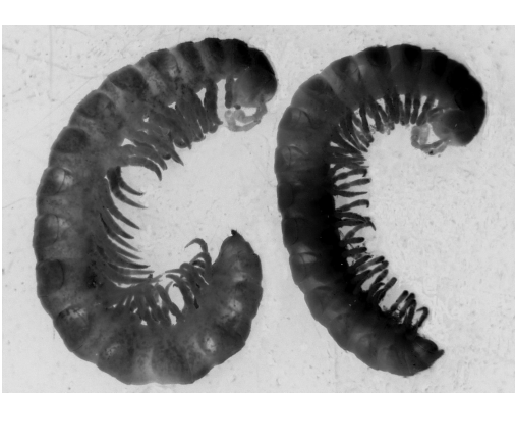
Females of A. montanum n. sp. and A. silvaticum n. sp. slightly smaller and less stout than males, with the posterior somites much less enlarged ( Fig. 2 View FIGURE 2 ); somite 2 pit and lastleg sternal projections lacking; epigynum with posterior rim raised at either end ( Fig. 1 View FIGURE 1 G). Females of other Atrophotergum spp. have not been positively identified, as they cooccur with very similar dalodesmids in other genera.
Etymology: Latin atrophus, atrophy, + tergum, back; neuter gender. So named because of the relatively small size of the somite 3 and 4 metatergites in males.
Remarks: Atrophotergum spp. are highly unusual among Tasmanian Polydesmida in having males larger than females ( Fig. 2 View FIGURE 2 ).
Besides the six Atrophotergum described below, single males have been found which may represent two additional species; these will be described when more material becomes available. One specimen (QVM 23:25669) is from rainforest along the Savage River Pipeline Road in northwest Tasmania, and the other (QVM 23:25670) is from a cave in dolerite pseudokarst on Mt Wellington, near Hobart in the southeast.
Biogeography: The six named Atrophotergum species have a mosaic distribution on the main island of Tasmania, with only minor overlaps of species ranges ( Fig. 12 View FIGURE 12 ). To date, no two species have been found at the same site. Several of the range boundaries appear to coincide with previously recognised faunal divides (‘faunal breaks’) in northern Tasmania. A. bonhami and A. wurrawurraense have a parapatric boundary south of Devonport along the Mersey Break ( Mesibov 1999), and the Mersey Break appears to bound the A. silvaticum distribution on the east. Near Weavers Creek, 20 km east of Launceston, A. pastorale and A. sodalis appear to meet in parapatry along the East Tamar Break ( Mesibov 1997), and the northwest portion of the A. pastorale range appears to end some 60 km further west at the Mersey Break. (In Mesibov (1999), species ‘A1’ and ‘A4’ were said to be in parapatry along the Mersey Break. ‘A1’ is here described as A. silvaticum and ‘A4’ is resolved into A. pastorale , A. sodalis and A. wurrawurraense .) A particularly interesting area for future field work is centered on the town of Latrobe, about 10 km south of Devonport, where the ranges of A. bonhami , A. silvaticum and A. wurrawurraense may meet in threeway parapatry. Unfortunately, much of the native vegetation in north central Tasmania has been cleared for farms ( Mesibov 1999), and native millipedes are very hard to find in remnant patches of native vegetation. These patches are typically overburned, often overgrown with weeds and sometimes infested with the introduced julid millipedes Ophyiulus pilosus (Newport, 1843) and Cylindroiulus spp.
Mosaic distributions of closely related taxa seem to be common among millipedes ( Shelley 1990; Mesibov 2003a). The Atrophotergum mosaic is the third to be documented among Tasmania Polydesmida ( Mesibov 2003b, c).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |