Triberta De Prins
|
publication ID |
https://doi.org/ 10.11646/zootaxa.3741.2.1 |
|
publication LSID |
lsid:zoobank.org:pub:E37C82A2-27DA-42DE-A298-838578F6B179 |
|
DOI |
https://doi.org/10.5281/zenodo.6157653 |
|
persistent identifier |
https://treatment.plazi.org/id/EA5C9E21-9D30-0470-D8FC-677579E8BF2A |
|
treatment provided by |
Plazi |
|
scientific name |
Triberta De Prins |
| status |
gen. nov. |
Triberta De Prins View in CoL gen. nov.
Type species: Lithocolletis cistifoliella Groschke, 1944 , by present designation.
Triberta is assigned to the subfamily Lithocolletinae on the basis of eleven nuclear genes and the following putative morphological synapomorphies: hindwing vein Rs parallel to vein M at the basal half of wing; adults rest parallel to surface, adult head with occipital tuft; internal feeding of all larval instars within the mine.
Diagnosis. According to the phenotypic, biological and ecological characters Triberta resembles other genera of Lithocolletinae , but according to the wing venation it is the closest to the monotypical Nearctic genus Protolithocolletis Braun, 1929 . We define Triberta as a genus-group taxon of the clade Triberta + Macrosaccus + ( Leucanthiza + Chrysaster ) (Fig. 01). The ecological trait defining Triberta is that pupation occurs outside the mine (a trait shared with Chrysaster and Leucanthiza ) in a transparent oval-shaped and slightly raised cocoon ( Fig. 59 View FIGURES 56 – 59 ). In contrast the pupation of Cameraria occurs within an upperside blotch mine under a flat circular cocoon while in Protolithocolletis “the pupa and its cocoon are invariably found within the mine” (Braun 1929: 39). The last instar larva of Chrysaster spins an ellipsoid silken cocoon at the edge of a contracted leaf (Kumata 1961) and the larva of Leucanthiza weaves its cocoon on some substance on the surface of the ground (Clemens 1859). The shape and structure of this cocoon spinning clearly separates the new genus Triberta from the latter two genera Chrysaster and Leucanthiza . In addition to the ecological and molecular characters, Triberta may be distinguished from all remaining lithocolletine genera by the combination of the following morphological diagnostic traits: the forewing pattern with three white fasciae or chevrons strongly angulated at midline, or rows edged basally and apically (a trait easily differentiating Triberta from Cameraria and Cremastobombycia , where fasciae typically are black margined distally); occipital tuft developed like in most Lithocolletinae genera but different from Chrysaster and Leucanthiza where vertex is smooth ( Figs 60, 61 View FIGURES 60 – 61 ). Wing venation is similar in that of Protolithocolletis in retaining 7 apical veins in forewing: R2, R3, R4, R5, M1, M2+3, Cu1 (R1 rudimentary), the cell between R5 and M1 open, but different from Cameraria , Phyllonorycter and Porphyrosela which have only five or four apical veins. The hindwing venation is similar to Cremastobombycia , Hyloconis , Porphyrosela and Protolithocolletis in the presence of M2+3; this vein is lost in Cameraria , Chrysaster , Leucanthiza , Macrosaccus , Neolithocolletis and Phyllonorycter (Fig. 03). Sternum VIII in males forms a characteristic flap, laying under the valvae like in many Lithocolletinae genera except Chrysaster , Leucanthiza , Macrosaccus and Protolithocolletis , a trait easily differentiating Triberta from the latter genera. In Cameraria sternum VIII often bifid caudally, while in Triberta it is shaped like a broad triangle (Figs 0 4, 0 6, 21). Uncus fused with tegumen to form a single sclerite (Kuznetzov 1981) (Fig. 08). The apical part of tegumen in the male genitalia with two apical setae like in Cameraria (Figs 0 5, 09). Apex of tegumen is setose in remaining nine lithocolletine genera except Phyllonorycter . Ventral surface of valvae in Triberta covered with long hair-like setae (Fig. 06), anellus strongly developed, tubular in many species of Cameraria , however differently from Cameraria , Macrosaccus , Leucanthiza and Protolithocolletis saccus in being simple, not prominent; aedoeagus is rather short, ca. 2/3 of valval length, bifurcate at coecum, a trait easily differentiating Triberta from all remaining lithocolletine genera; transtilla in Triberta incomplete like in the great majority of Cameraria species and Hyloconis (Figs 06–09, 16–19). In the female genitalia ( Figs 22–25 View FIGURES 22 – 25 ), papillae anales are semi-circular, not flattened like as in Cameraria and Porphyrosela , with the clearly visible separation of papillae caudally; two pairs of apophyses present, the trait evidently separating the new genus from Porphyrosela . The latter genus has only one pair of apophyses. Ostium bursae opens at subanterior area of segment VIII, the trait differentiating the new genus from Cameraria , Cremastobombycia , Phyllonorycter and Porphyrosela ; in most species of the latter genera the ostium bursae opens at the posterior margin of segment VII ( Cameraria , Cremastobombycia , Porphyrosela ) or in different locations within segment VII ( Phyllonorycter ); ductus bursae long, narrow, corpus bursae spherical, signa confined to 1–2 moderately sclerotized oval areas—these traits are shared with many lithocolletine genera but not with Porphyrosela , which has no signum, and not with Macrosaccus and Neolithocolletis , where the signum area consists of numerous microscopic spicules scattered on the wall of corpus bursae or arranged in linear series on subcaudal part of corpus bursae. Larva constructs an abaxial mine ( Figs 56–58 View FIGURES 56 – 59 ). Larva of Triberta is of ‘cylindrical’- type like that in Phyllonorycter and clearly different from Cameraria , the genus whose larva belongs to the ‘flat’- type group (Chambers 1878; Chapman 1902; Braun 1908). As was noted by Stehr (1987) in lepidopteran leaf miners the mandibles and hypopharynx provide diagnostic structures that are useful for generic diagnoses. Davis et al. (2013) indicated that in tissue feeding gracillariid larvae the anterior margin and dorsal surface of hypopharynx are highly diagnostic: the hypopharynx of larval Cremastobombycia bears a transverse series of six, relatively large, spinose, digitate lobes (Davis et al. 2013: Figs 13–15 View FIGURES 10 – 15 ). The hypopharynx of tissue-feeding Phyllonorycter larvae possesses a series of over 20 much smaller, curved, smooth spines (Davis & Deschka 2001: Figs 56–58 View FIGURES 56 – 59 ). The hypopharynx of the two non-feeding larval instars in Cameraria is mostly smooth and without anterior spines (De Prins et al. 2003: Fig. 10 View FIGURES 10 – 15 ; Davis et al. 2013). The ventral surface of the last instar larva of Triberta is covered with hypopharyngeal striations, which differentiates the latter genus from Cameraria , Cremastobombycia and Phyllonorycter . Pupa without cremaster in Triberta and remaining eight lithocolletine genera, except in Phyllonorycter and Porphyrosela . The cremaster is well-defined in Phyllonorycter and reduced, consisting of four little hooks, in Porphyrosela (Bentancourt & Scatoni 2007) . A thickened sclerotized ridge present along the lateral sides of pupal A1–8, in Triberta , which differentiates the new genus from Phyllonorycter and Cameraria . In the latter genera this lateral cuticle sclerotization is absent. Pupation occurs outside the mine, an ecological trait shared only with Chrysaster and Leucanthiza ; and in a transparent oval-shaped and slightly raised cocoon. Members of the new genus Triberta are known to feed exclusively on Cistaceae . Triberta also differs from Phyllonorycter and Cameraria in a concatenated dataset of eleven nuclear gene sequences.
To facilitate the assignment of lithocolletine moths to Triberta , a summary of the foregoing diagnoses is provided: Adult: 1) wing venation: presence of 7 terminal veins in forewing, presence M1 and M 2 in hind wing like in Protolithocolletis ; 2) male genitalia: sternum VIII developed, saccus undeveloped, a pair of setae on apex of tegumen, aedoeagus bifurcate at coecum; 3) female genitalia: ostium bursae opens in subanterior area of segment VIII; compact signum on corpus bursae present. Larva: 4) feeds on Cistaceae , differing from all other genera of Lithocolletinae ; hypopharynx of last instar larva covered with striations. Pupa: 5) pupation outside the mine; pupa with lateral cuticle sclerotization and without cremaster. Molecular: 6) Triberta is not nested within two wellsupported clades Phyllonorycter + Cremastobombycia and Cameraria .
Description. Adult (Figs 0 2, 06–25). Small moths, forewing length ca. 2.5–4.5 mm.
Head. Vertex plate joined with frontoclypeus, head tufted with erect long piliform scales, whitish posteriorly, ochreous anteriorly; frons covered with appressed, smooth, shiny white scales with ochreous apices; eyes large, ocular index ca. 0.6–0.7, interocular index ca. 1.0. Antenna ca. 20% shorter than forewing, smooth scaled, filiform; scape short, thickened, bearing pecten of different lengths, flagellomere 1 ca. 1.6× shorter than scape, flagellomere 2 ca. 10% longer than flagellomere 1. Proboscis developed, naked, of medium length, ca. 1.7× length of labial palpus. Maxillary palpus small, rudimentary, two-segmented, apical maxillary palpomere almost globular. Labial palpus moderate, porrect, filiform, drooping, straight, with ratio of labial palpomeres from base 1.0:1.8:2.0(Fig. 02).
Thorax: Forewing length 2.5–4.5 mm; forewing ground colour beige-ochreous with intermixture of dark brown and white fasciae or chevrons which are highly variable in angulation and width; white markings are edged on both sides basally and apically; fringe very long, particularly at dorsal margin, reaching width of wing in forewing and ca. 3× width of wing in hindwing; fringe of hindwing very long, exceeding 4× width of hindwing. Descaled forewing lanceolate, slender, and with strongly acute apex. Venation with 10 veins, distal margin with 7 veins R2, R3, R4, R5, M1, M2+3, Cu1; R1 is rudimentary, M is forked to form M1 and M2+3, Cu1 separate, CuP indistinct (fold) over entire length, A1 strong, separate; discal cell is open with absence of crossvein between R5 and M1 and with weak connection between R2 and R3. Hindwing lanceolate, maximum width/hindwing length is 0.15, venation 5 veined; Sc very short, terminating near base of costa; Rs very long, extending almost to apex of hindwing; M1 and M2 stalked; basal 2/3 of M1+2 indistinct, parallel to Rs; distal part of M1 terminates at 0.7 the distance to dorsal margin; M2 terminates at 0.6 of dorsal margin, Cu1 strong, ending at 0.4 of dorsal margin; A1 vestigial (Fig. 03). Frenulum in male is a single stout bristle; frenula in female are two tightly appressed bristles. Legs slender, with darker rings; epiphysis on foreleg absent, mid-tibia bearing a pair of tibial spurs; hind tibia thickened, with long fine loose hairs, long medial and short apical spurs; hind tarsus smooth, slender, ca. 1.5× as long as tibia.
FIGURE 0 1. Maximum Likelihood tree of Lithocolletiinae. The numbers above nodes denote the bootstrap nodal supports. Only the bootstrap values> 70% are shown.
FIGURES 02–03. Adult morphology of Triberta helianthemella , Italy, Verona, 25/ 30.vi.2010, leg. P. Triberti. 0 2, descaled head prep. DP 3797♀. Scale bar 100 µm. 0 3, wing morphology, descaled wing prep. DP 3809♀. Scale bar 1 mm.
FIGURES 04–09. Male genitalia of Cameraria and Triberta . 0 4, Male genitalia of Cameraria ohridella , Belgium, Tervuren, 11.v. 2012, leg. E. De Coninck. Scale bar as indicated. 0 5, same preparation, tegumen with a pair of apical setae. Scale bar as indicated. 0 6, male genitalia of Triberta helianthemella , Austria, North Tirol, 07.xi.1964; prep. DP 3826♂; arrows indicate the modified sternum VIII. 0 7, same preparation, aedoeagus. 0 8, same preparation, tegumen. 0 9, same preparation, apex of tegumen; arrows indicate a pair of apical setae. Scale bar as indicated.
Abdomen. Anterior margins of abdomen with narrow opening and sclerotized, becoming slightly broader towards S2; the sclerotized anterior margin of the abdomen well connected on T2 and unconnected on S2 or weakly joined by V-shaped narrow sclerotized connection; S2 apodemes of median length, ending well beyond the abdominal opening, slender, with barbed bases, very slender distally; a pair of sclerotized T2 apodemes initiate at the lateral connection corner of abdominal opening, with broad sclerotized bases, slightly shorter than S2 apodemes. A pair of tiny sublateral-medial spicules present on each abdominal sternum. Sternum VIII in adult males developed, flap-like, extended, more or less triangular caudally (Figs 0 6, 16, 19, 21).
Male genitalia ( Figs 16–19 View FIGURES 16 – 21 ). Tegumen rather long, with a pair of subapical setae. Tuba analis sometimes significantly protruded. Valvae symmetrical, setose. Transtilla incomplete. Vinculum developed, crescent or trapezium shaped, without prominent saccus. Anellus developed, strongly sclerotized, tubular, broader at base; juxta present. Aedoeagus ca. ¼ shorter than valva, relatively complex, with bifurcated coecal end and gradually tapering towards slender apex; cornuti absent.
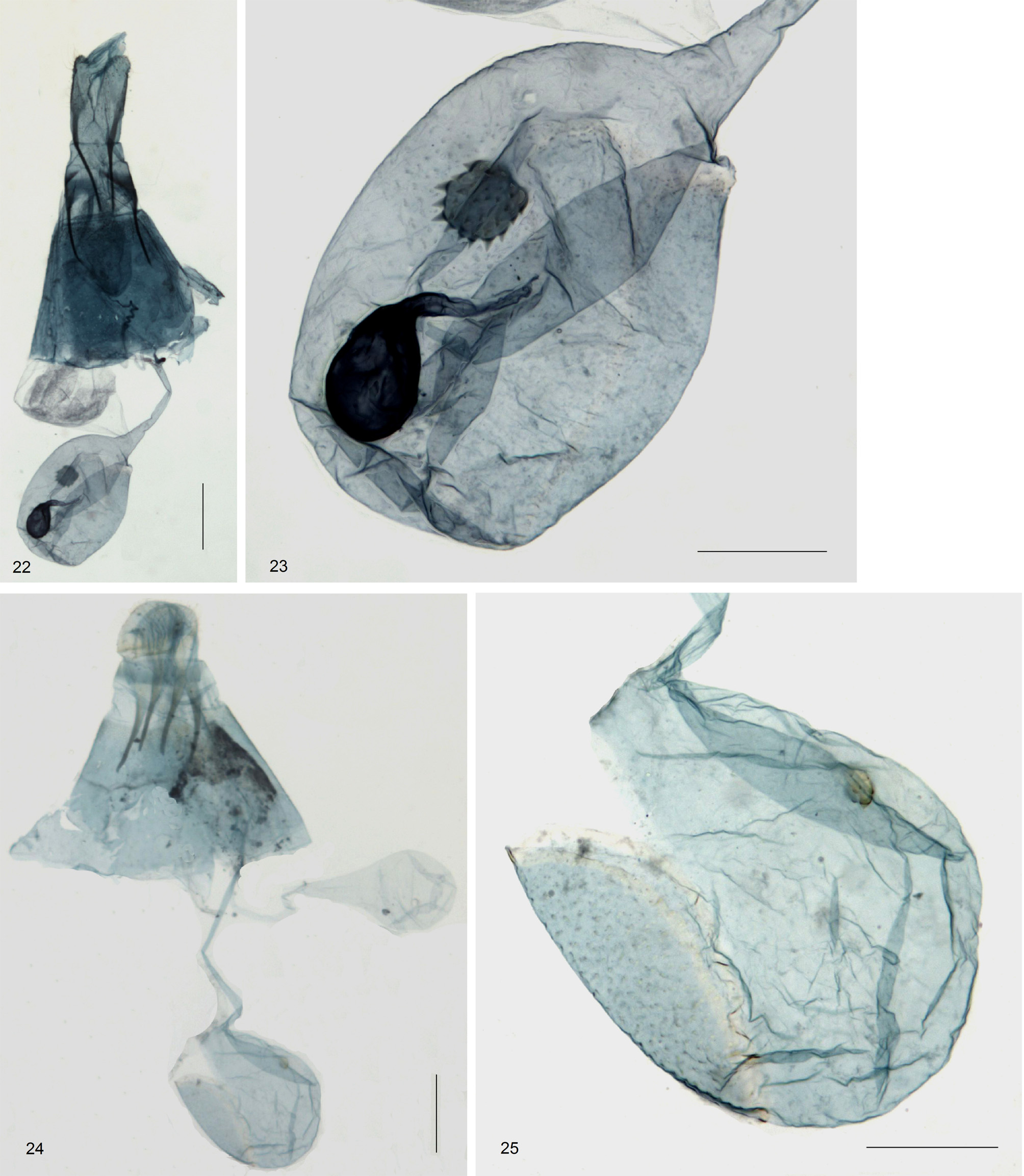
Female genitalia ( Figs 22–25 View FIGURES 22 – 25 ). Papillae anales rounded caudally, not connected laterally. Segment VIII short, well connected to segment VII. Posterior apophyses without basal plate, but thickened basally and gradually acute apically; anterior apophyses originating from sclerotized basal plate of segment VIII, similar in form to posterior apophyses. Ostium bursae opens at the subanterior region of segment VIII. Ductus bursae very narrow, long with sclerotized antrum; ductus seminalis arises ca. midway along ductus bursae ( Figs 22, 24 View FIGURES 22 – 25 ). Corpus bursae distinct from ductus bursae, globular, with two signa regions: a relatively large speculate-sculptured disc along lateral wall of corpus bursae and a strongly sclerotized stellate signum covered with small sharp barbs about midway along the opposite wall of corpus bursae ( Figs 23, 25 View FIGURES 22 – 25 ).
Larva. Last instar larva ca. 3.6 mm long. Head of late instars approximately round with full complement of mouthparts ( Fig. 26, 29–31 View FIGURES 26 – 31 ); 3 pairs of stemmata arranged per pair: stemmata 1 and 2 in a horizontal row distant from each other; stemmata 3 and 4 close to antenna in a vertical row at short distance close to each other; stemmata 5 and 6 in a vertical row touching each other ( Figs 27, 28 View FIGURES 26 – 31 ). Nine setae surround the stemmata of which S3 is very short; antenna 3-segmented with second segment moderately long; sensilla as shown in Fig. 31 View FIGURES 26 – 31 ; labrum strongly bilobed with raised median part (Fig. 33); M1 and M4 present; inner perimeter of labrum without spines; the dorsal surface of the hypopharynx of the last tissue feeding larval instar is covered with hypopharyngeal striations ( Figs 29–31 View FIGURES 26 – 31 ); ventral surface of head covered with numerous short rough spines; maxillary palpus as shown in Figs 29– 31 View FIGURES 26 – 31 ; median lobe of the maxilla with numerous sensory setae of different lengths. Labial palpus bears two sensory setae: one short seta, ca. 2× longer than apical palpomere and one long seta ca. 4x length of apical palpomere. Spinneret a relatively long striate tube ( Fig. 29 View FIGURES 26 – 31 ). Thorax with setae XD1 and XD2, SD1 long, immediately ventral to XD2; SD2 absent on T1; L group of setae long, L2 longer than L1; dorsal surface of T1 covered with both longer and shorter protrubances (Fig. 34); T2 and T3 with long D1 and shorter D2; dorsal surface of thoracic segments covered with tiny spines (Fig. 35). Legs fully developed, coxae with 4 coxal setae; femur and tibia with very long setae; tarsal claw with a sharp spine (Figs 36, 37). Abdomen with bisetose D group on A1–8, unisetose on A9 ( Fig. 42 View FIGURES 38 – 43 ). Prolegs present on A3–5, 10; crochets consisting of 16 larger hooks arranged in biordinal semicircle and four small spines arranged in a row opposite the larger hooks ( Figs 38–40 View FIGURES 38 – 43 ); anal prolegs with crochets consisting of ca. 10–12 rather large hooks arranged in a uniordinal semicircle ( Fig. 43 View FIGURES 38 – 43 ). Anal plate with three pairs of setae ( Fig. 41 View FIGURES 38 – 43 ).
FIGURES 32–37. Late instar larva of Triberta helianthemella , Italy, Verona, 25/ 30.vi.2010, leg. P. Triberti. 32, head and 1st thoracic segment of last instar larva, dorsal view. 33, same preparation, proximal part of head. 34, dorsal surface of T1. 35, dorsal surface of T3. 36, legs of late instar larva, latero-ventral view. 37, median thoracic leg, lateral view. Scale bar as indicated.
Pupa. Maximum length ca. 2.5 mm, width ca. 0.8 mm; body elongate, compact, relatively flat, narrower over last 4 segments ( Figs 44–45 View FIGURES 44 – 49 ); color a bright shade of brown. A thickened sclerotized ridge present along the lateral sides of A1–8. Head smooth, with two pairs of sockets bearing sensillae ( Fig. 46, 47 View FIGURES 44 – 49 ). Vertex furnished with a frontal process (cocoon cutter), which is relatively short, broadly triangular, with slightly wrinkled median surface ( Fig. 46 View FIGURES 44 – 49 ). Forewings long, extending to the posterior margin of abdominal segment A6, and contiguous at their distal ends ( Figs 44 View FIGURES 44 – 49 , 54 View FIGURES 50 – 55. P ). The appendages of antennae longer than forewings and extend up to caudal end of pupa. Labium rather long, ca. 1.7× shorter than proboscis. The hind leg sheaths are slightly shorter than antennal appendages ( Fig. 54 View FIGURES 50 – 55. P ). The distance between the apices of the mesothoracic and metathoracic legs is approximately 1.46× the distance between the apices of the prothoracic and mesothoracic legs. One pair of dorsal setae is present on segments TII–TIII. Abdominal segments A1–5 of more or less equal size, A6–8 strongly decreasing in size. Abdominal segments A3–8 covered with dense, minute spines dorsally ( Fig. 51 View FIGURES 50 – 55. P ); the posterior margin of A1 with a wrinkled band ( Fig. 50 View FIGURES 50 – 55. P ); dorsal surface of each abdominal segment carries a pair of setae, except segment A9 which lacks setae ( Fig. 52 View FIGURES 50 – 55. P ). Terminal segment A10 fully covered with small spinaculae ( Figs 53, 54 View FIGURES 50 – 55. P ); caudal surface of A10 without cremaster but with eight tiny protuberances each carrying a tiny nodule ( Fig. 55 View FIGURES 50 – 55. P ) (Gregor & Patočka 2001: 34; Patočka & Turčáni 2005).
Biology. Larva solitary, feeding exclusively on Cistaceae . Abaxial mine begins as a small corridor that closely follows a thick vein of the plant. It continues into a contracted blotch mine that may occupy the entire leaf; mine becomes transparent when it is fully grown. The tissue-feeding instars remove mesophyll layers of the leaf, and though the mine is formed under the abaxial epidermial layer, from adaxial leaf surface it is visible as a large, contiguous brownish blotch ( Figs 57, 58 View FIGURES 56 – 59 ). However, in some cases the mine can be formed on adaxial surface (see the original description of Lithocolletis cistifoliella here below). Frass distributed either in a row while larva crosses the vein or loose in small accumulations. Pupation occurs outside the mine in a transparent cocoon on a clean background without excrements ( Fig. 59 View FIGURES 56 – 59 ). Biology bivoltine with hibernation as adults is reported.
Host plants: Cistaceae : Cistus creticus L. (= C. villosus L.) (Klimesch 1956: 215); C. incanus L. (Klimesch 1956: 215), C. monspeliensis L. (Walsingham 1908: 976), C. salviifolius L. (Groschke 1944: 122). Helianthemum chamaecistus Mill. (Hartig 1964: 177) , H. nummularium (Cav.) Losa & Rivas Goday (Gregor 1986: 229) , H. sp. (Eckstein 1933: 163; Weber 1945: 389; Gregor & Povolný 1950: 135; Hartig 1964: 177; Szöcs 1981: 216), H. vulgare Gaertn. (Herrich-Schaffer 1861: 20) , Tuberaria guttata (L.) Fourr. (Roüast 1884: 138).
Natural enemies. Eulophidae : Chrysocharis phryne (Walker, 1839) (Noyes 2012) , Cirrospilus staryi Bouček, 1958 (Bouček 1959: 180) , Hemiptarsenus ornatus (Nees, 1834) (Noyes 2012) , Sympiesis gregori Bouček, 1958 (Bouček 1959: 131) .
Distribution. Triberta is a genus of xerothermic habitats in central and southern Europe, the Mediterranean region, and the Canary Islands.
Austria (Knitschke 1927: 97); Canary Islands: La Palma (Hering 1927), Tenerife (Walsingham 1908: 976); Croatia (Wocke 1871: 329); Czech Republic (Nickerl 1894: 29); France (Jourdheuille 1870: 133, 254); Germany (Herrich-Schaffer 1861: 20); Greece (Klimesch 1968: 176); Hungary (Szöcs 1981: 216); Italy ( Mann 1867: 841), Sicily (Amsel 1951: 422), Macedonia (Klimesch 1942: 387); Moldova (Bouček 1961: 21); Serbia (Amsel 1951: 422); Slovakia (Gregor 1986: 229); Spain (Vives Moreno 1994: 54, 557); Switzerland (Weber 1945: 389); Turkey (Stainton 1867: 35).
Generic relationships and species diversity. Based on taxa included in our phylogenetic analysis Triberta is the sister lineage to the clade Macrosaccus +( Leucanthiza + Chrysaster ). The ecological / morphological traits shared by these latter genera are as follows: Triberta , Leucanthiza and Chrysaster pupate outside the mine, the mine of Triberta and Macrosaccus is blotch abaxial, while Leucanthiza and Chrysaster construct adaxial mines; pupa in Triberta without cremaster like in other members of the clade; in the male genitalia, apex of tegumen carries one pair of setae, valvae symmetrical, densely setose; sternum VIII in Triberta is caudally extended, the character that separates Triberta from the other genera of the clade Chrysaster , Leucanthiza and Macrosaccus ; in the female genitalia, ductus bursae very slender and long extending to segment V, corpus bursae in most cases distinct from ductus bursae and bears a compact signum (signa).
In addition to the morphological characters mentioned above, a molecular phylogeny based on eleven nuclear genes places Triberta apart from Phyllonorycter , Cremastobombycia , and Cameraria (Fig. 01).
Two species, both Palaearctic, are currently recognized in the new genus Triberta . The circumscription, delimitation and delineation of Triberta species will be presented in a separate study.
Etymology. The name Triberta is used as a noun in the nominative singular and feminine gender. This name is a patronym in honour of Paolo Triberti, a lepidopterist from the Museo Civico di Storia Naturale, Verona, Italy, who revised and studied many groups of Gracillariidae , the lepidopteran family to which the new genus belongs.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |