Saciperere catuaba Huber & Carvalho, 2019
|
publication ID |
https://doi.org/10.11646/zootaxa.4546.1.1 |
|
publication LSID |
lsid:zoobank.org:pub:D2C9F49A-9B76-40AE-9A60-CAE9B99BA547 |
|
DOI |
https://doi.org/10.5281/zenodo.5449751 |
|
persistent identifier |
https://treatment.plazi.org/id/E21587DB-FFA1-FFEF-FF11-FCAC4F49F852 |
|
treatment provided by |
Plazi |
|
scientific name |
Saciperere catuaba Huber & Carvalho |
| status |
sp. nov. |
Saciperere catuaba Huber & Carvalho View in CoL sp. n.
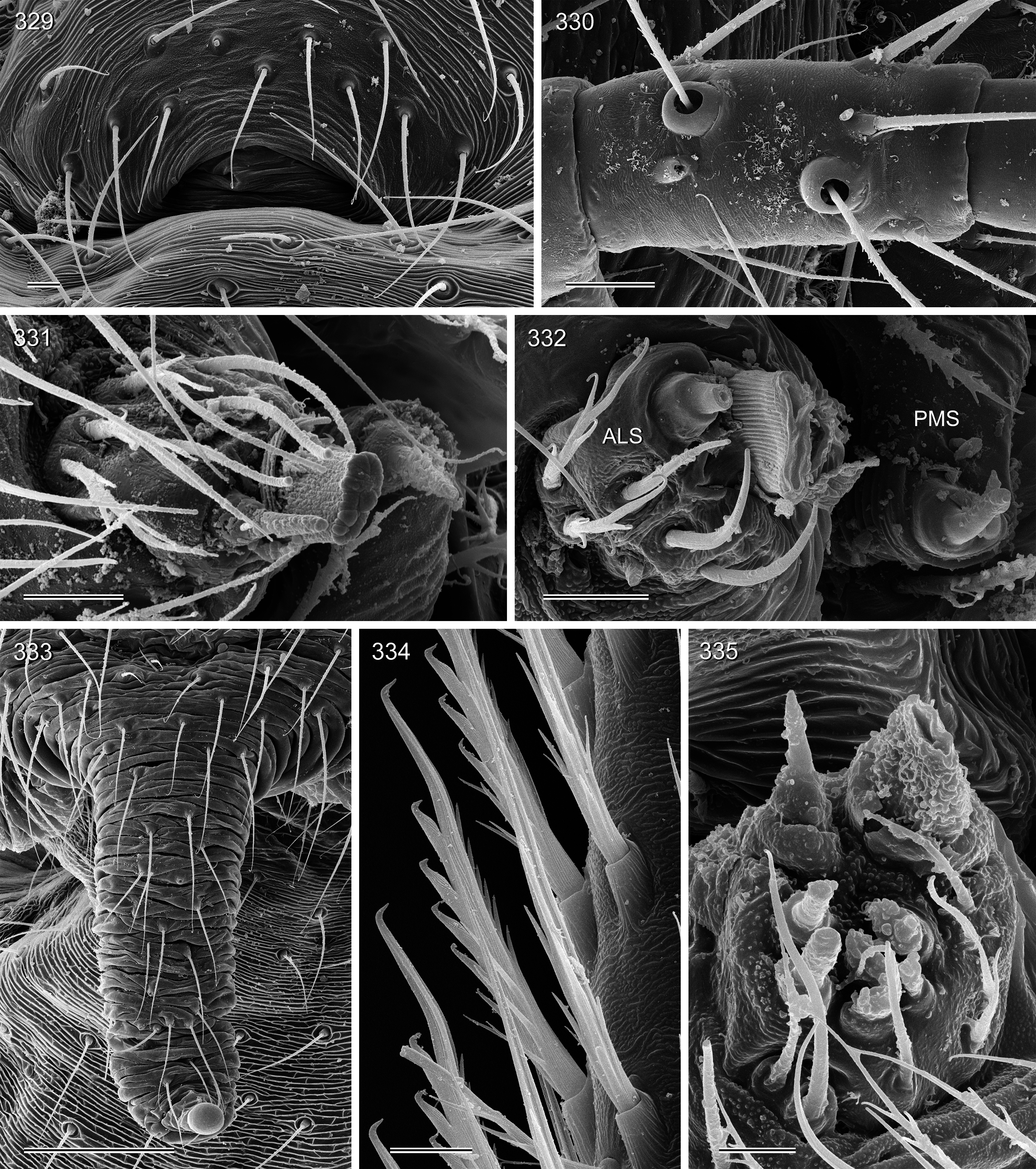
Figs 213–239 View FIGURES 213–214 View FIGURES 215–220 View FIGURES 221–229 View FIGURES 230–239 , 323–332 View FIGURES 322–328 View FIGURES 329–335
Gen.n. Br15-45: Eberle et al. 2018 (molecular data); Huber et al. 2018: fig. 3.
Type material. BRAZIL: ♂ holotype, UFMG (22742) ; 3♀ paratypes, UFMG (22603, 22743); 1♀, CHNUFPI (2554); and 4♂ 19♀ paratypes, ZFMK ( Ar 20633–34), Acre, Senador Guiomard, Fazenda Experimental Catuaba ( 10.08°S, 67.625°W), 225 m a.s.l., 26–27.x.2016 ( B.A. Huber, L.S. Carvalho) GoogleMaps ; 4♂ 16♀ paratypes, ZFMK ( Ar 20635), same locality at 10.078°S, 67.619°W, 200 m a.s.l., 29.x.2016 ( B.A. Huber, L.S. Carvalho) GoogleMaps .
Other material examined. BRAZIL: 3♀ 1 juv., ZFMK ( Br 16-304), and 1♀, CHNUFPI (2551), all in pure ethanol, same data as types . 1♀ 2 juvs, ZFMK (Ar 20636), and 1♀ in pure ethanol, ZFMK ( Br 16-311), Acre, Cruzeiro do Sul, forest near Cruzeiro do Sul Internacional Airport ( 7.596°S, 72.767°W), 200 m a.s.l., 30.x.2016 ( B.A. Huber, L.S. Carvalho, E.O. Machado) GoogleMaps . 1♀ 1 juv., ZFMK ( Ar 20637), and 3 juvs in pure ethanol, ZFMK ( Br 16-317), Amazonas , Guajará , forest at Fazenda Colorado , ‘site 2’ ( 7.528°S, 72.655°W), 210–240 m a.s.l., 31.x.2016 ( B.A. Huber, L.S. Carvalho, E.O. Machado) GoogleMaps . 4♀, CHNUFPI (2537), and 1 juv. in pure ethanol, ZFMK ( Br 16-330), Amazonas, Tabatinga , forest at the Brazilian army base Comando de Fronteira Solimões , 8° Batalhão de Infantaria ( 4.244°S, 69.92– 69.93°W), 90 m a.s.l., 2–4.xi.2016 ( B.A. Huber, L.S. Carvalho) GoogleMaps . 3♀, CHNUFPI (2542, 2546); 2♀, UFMG (22602, 22605); and 1♂ 3♀ 1 juv., ZFMK (Ar 20638), and 1 juv. in pure ethanol, ZFMK ( Br 16-296), Rondônia, Itapoã do Oeste, Floresta Nacional de Jamari , ‘site 1’ ( 9.193°S, 63.107°W), 130 m a.s.l., 23.x.2016 ( B.A. Huber, L.S. Carvalho) GoogleMaps . 3♂ 6♀ 2 juvs, ZFMK ( Ar 20639), and 5 juvs in pure ethanol, ZFMK ( Br 16-300), Rondônia, Itapoã do Oeste , Floresta Nacional de Jamari , ‘sites 2 & 3’ ( 9.22°S, 63.93°W— 9.26°S, 63.92°W), 110 m a.s.l., 24.x.2016 ( B.A. Huber, L.S. Carvalho) GoogleMaps . 2♀, CHNUFPI (2489, 2539); 1♀, UFMG (22608); 2♂, ZFMK (Ar 20640), and 2♀ 1 juv. in pure ethanol, ZFMK ( Br 16-261), Pará, Belterra, Floresta Nacional de Tapajós , km 83 ( 3.049°S, 54.928°W), 95 m a.s.l., 14–18.x.2016 ( B.A. Huber, L.S. Carvalho) GoogleMaps . 1♀ 1 juv., ZFMK (Ar 20641), and 1♀ in pure ethanol, ZFMK ( Br 16-281), Pará, Belterra, Floresta Nacional de Tapajós , km 72, ‘site 2’ ( 2.940°S, 54.933°W), 140 m a.s.l., 17.x.2016 ( B.A. Huber, L.S. Carvalho) GoogleMaps . 2♂ 1♀, CHNUFPI (2495, 2507, 2543); 1♀, UFMG (22607); 2♂ 8♀ 3juvs, ZFMK ( Ar 20642–43); and 4 juvs in pure ethanol, ZFMK ( Br 15-201, 206), Alagoas, near Murici , Estação Ecológica de Murici ( 9.247°S, 35.838°W), 350–400 m a.s.l., 18–19.v.2015 ( B.A. Huber, L.S. Carvalho) GoogleMaps . 1♀, UFMG (22606); and 1♂ in pure ethanol, ZFMK ( Br 15-204), Alagoas, near Murici, Estação Serra do Ouro ( 9.242°S, 35.838°W), 500 m a.s.l., 19.v.2015 ( B.A. Huber, L.S. Carvalho) GoogleMaps . 1♂, ZFMK (Ar 20644), and 1 juv. in pure ethanol ( Br 15-220), Pernambuco, near Bonito, forest near Cachoeira da Gruta ( 8.547°S, 35.712°W), 380 m a.s.l., 24–25.v.2015 ( B.A. Huber, L.S. Carvalho) GoogleMaps . 1♀ 1 juv., ZFMK (Ar 20645), and 1 juv. in pure ethanol, ZFMK ( Br 15-228), Pernambuco, Tamandaré, Reserva Biológica de Saltinho ( 8.727°S, 35.178°W), 50 m a.s.l., 26.v.2015 ( B.A. Huber, L.S. Carvalho) GoogleMaps . 1♀, CHNUFPI (2528), Pernambuco, São Vicente Ferrer , near road PE-089 ( 7.621°S, 35.463°W) GoogleMaps . 1♀, CHNUFPI (2532); 1♀, UFMG (22610); 2♂, ZFMK (Ar 20646); and 1♀ 1 juv. in pure ethanol ( Br 15-240), Paraíba, Areia, Parque Estadual da Mata do Pau Ferro ( 6.965°S, 35.748°W), 600 m a.s.l., 31.v.2015 ( B.A. Huber, L.S. Carvalho) GoogleMaps .
Etymology. The species name is derived from the type locality; noun in apposition.
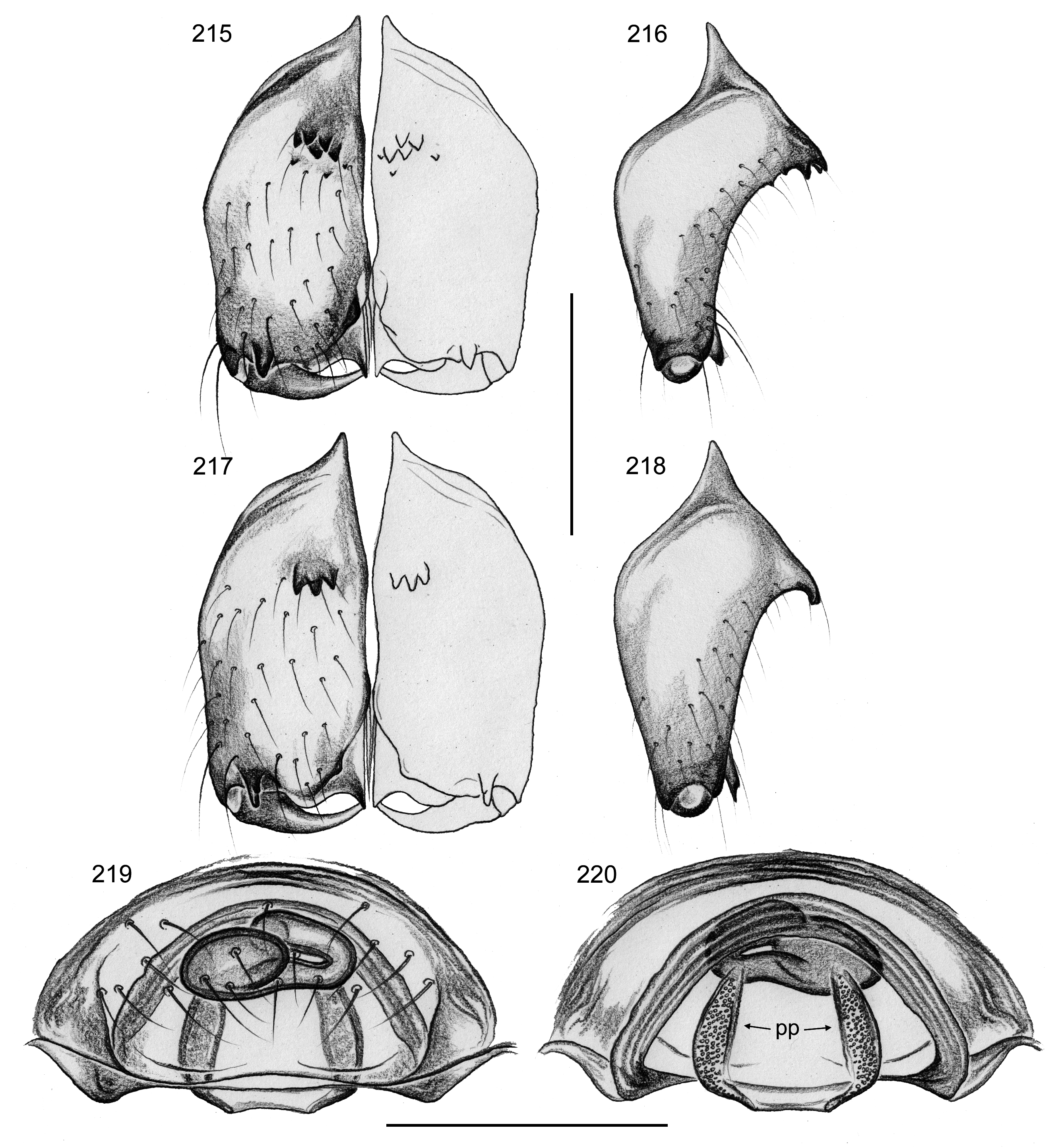
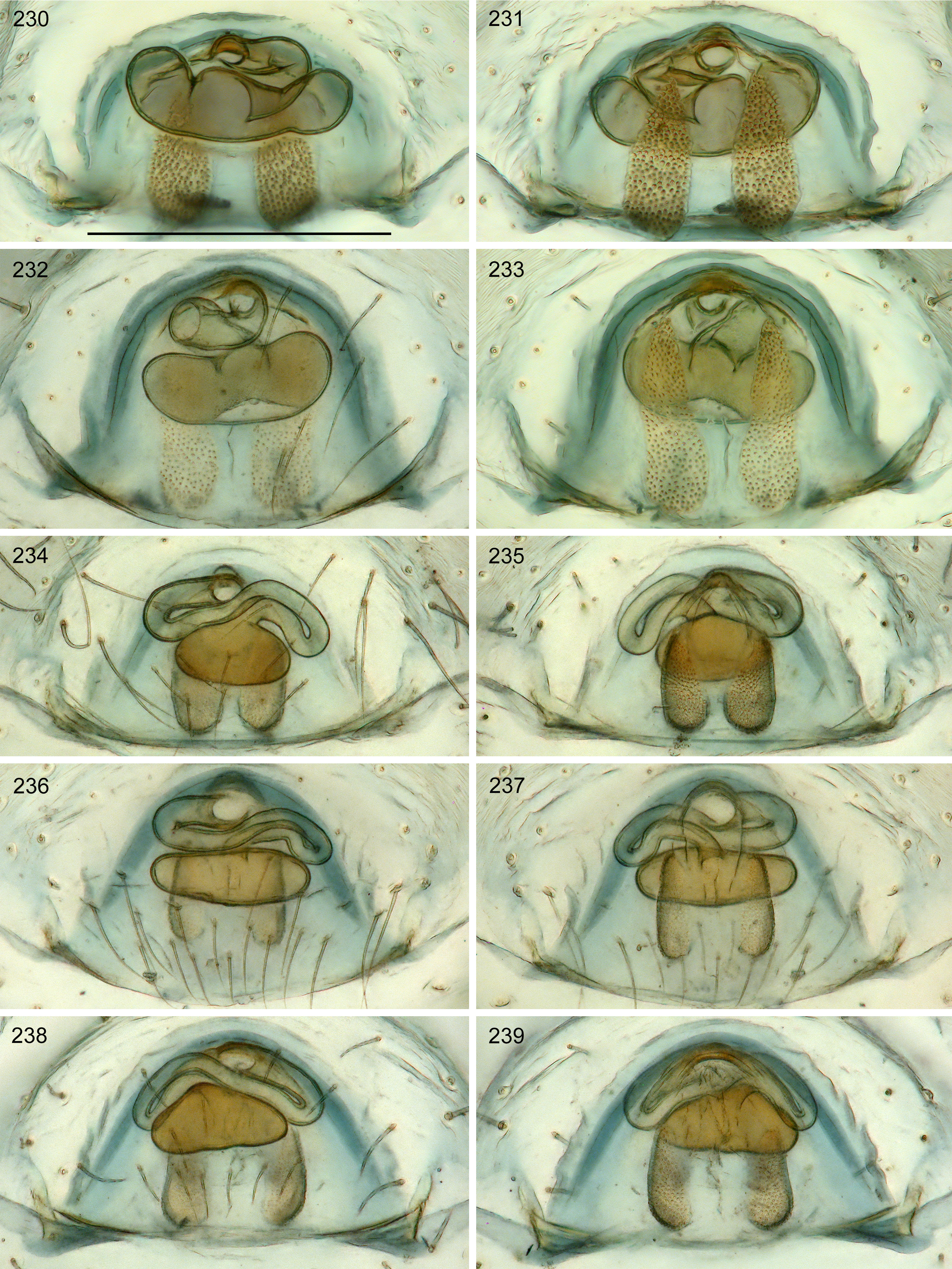
Diagnosis. Males are easily distinguished from all other known pholcids by armature of male chelicerae: proximal frontal processes with 3–5 short conical apophyses, distal apophyses in front of fang joints, and simple sclerotized humps near laminae ( Figs 215–218 View FIGURES 215–220 ); also by slender S-shaped procursus with thin sclerite at tip accompanied by transparent membrane ( Fig. 214 View FIGURES 213–214 ; similar to certain species in Pisaboa ). Females with distinct ‘receptacle’ (visible through cuticle in uncleared specimens) connected to ‘valve’ area by variably long tube ( Figs 219–220 View FIGURES 215–220 , 224–239 View FIGURES 221–229 View FIGURES 230–239 ).
Description. Male ( holotype). MEASUREMENTS. Total length 1.7, carapace width 0.63. Distance PME-PME 130 µm; diameter PME 70 µm; distance PME-ALE 40 µm; distance AME-AME 70 µm, diameter AME 25 µm. Leg 1: 21.6 (5.4 + 0.3 + 5.1 + 9.2 + 1.6), tibia 2: 3.1, tibia 3: 2.1, tibia 4: 2.8; tibia 1 L/d: 93.
COLOR (in ethanol). Carapace pale ochre-yellow, sternum whitish; legs ochre yellow with brown rings on femora (subdistally), tibiae (proximally, subdistally), and metatarsi (proximally). Abdomen monochromous pale greenish gray, white dorsal marks of life specimens lost in ethanol.
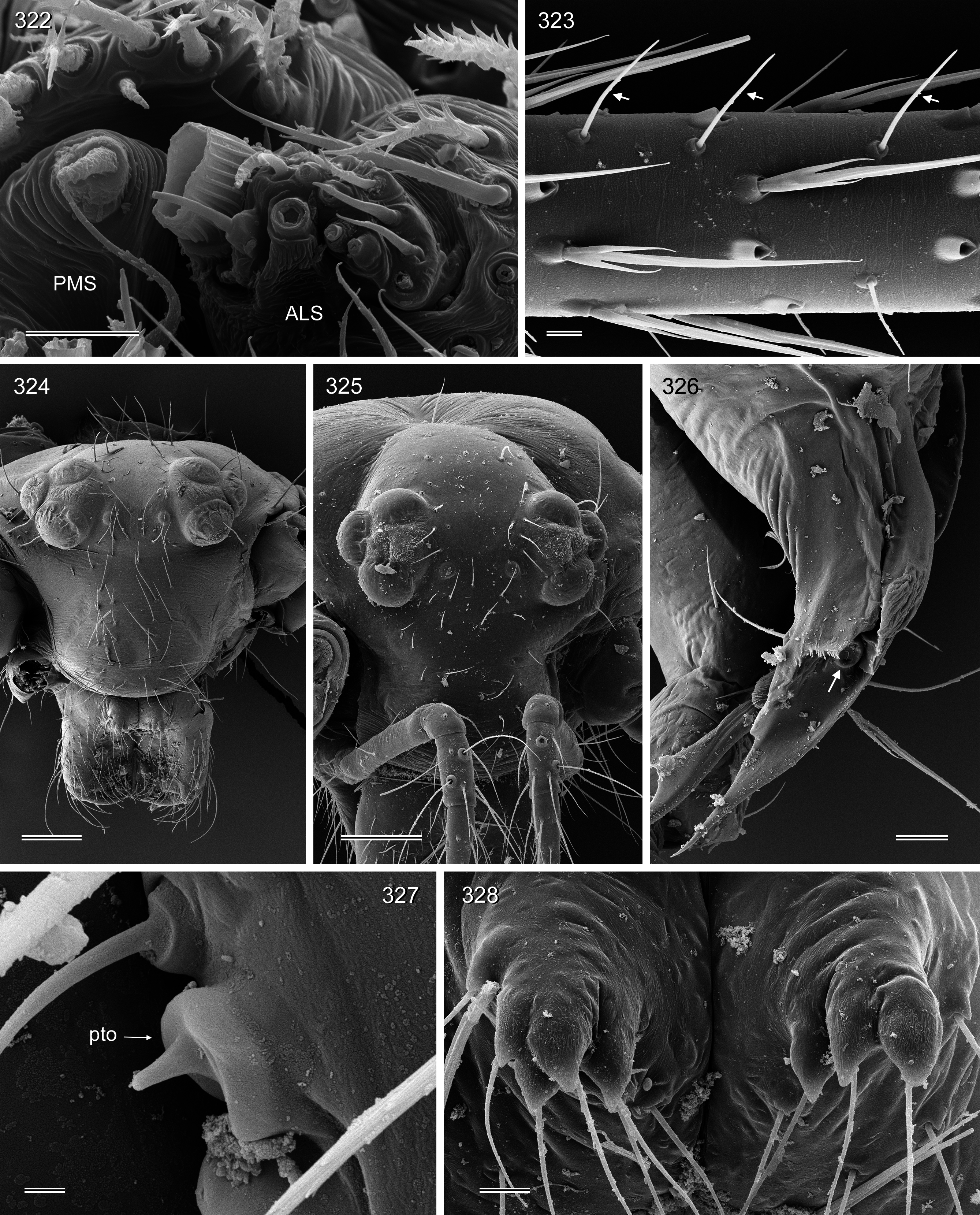
BODY. Habitus as in Fig. 221 View FIGURES 221–229 . Ocular area barely elevated. Thoracic furrow shallow. Clypeus unmodified. Sternum wider than long (0.50/0.34), unmodified. Gonopore without epiandrous spigots ( Fig. 329 View FIGURES 329–335 ). ALS with only two spigots each, one strongly widened and one pointed ( Fig. 331 View FIGURES 329–335 ).
CHELICERAE. As in Figs 215–216 View FIGURES 215–220 , with three pairs of modifications: proximal frontal processes with several (4–5) short conical apophyses ( Fig. 328 View FIGURES 322–328 ); distal apophyses in front of fang joints; and simple sclerotized humps near laminae.
PALPS. As in Figs 213–214 View FIGURES 213–214 ; coxa with retrolateral apophysis, trochanter barely modified, femur with retrolateral process proximally and distinct (but weakly sclerotized) ventral apophysis distally; tarsal organ exposed ( Fig. 327 View FIGURES 322–328 ); procursus long but simple, distal slender process accompanied by transparent lamina (barely visible in dissecting microscope); bulb with large, partly sclerotized conical process with subdistal sperm duct opening ( Figs 213 View FIGURES 213–214 , 326 View FIGURES 322–328 ).
LEGS. Without spines and curved hairs; with many vertical hairs on all femora and tibiae ( Fig. 323 View FIGURES 322–328 ); retrolateral trichobothrium of tibia 1 at 5%; tibia 1 without prolateral trichobothrium (present on other tibiae); tarsus 1 with ~20 pseudosegments.
Male (variation). Tibia 1 in 21 other males: 4.7–5.9 (mean 5.2). Cheliceral armature slightly variable: apophyses on proximal processes in most Amazonian specimens as in type (i.e. in a patch; Fig. 215 View FIGURES 215–220 ), in Atlantic Forest specimens in a transversal row ( Fig. 217 View FIGURES 215–220 ); males from Tapajós resemble males from Atlantic Forest in this respect. Distal apophyses and humps near laminae only slightly variable in size.
Female. In general similar to male ( Figs 222–223 View FIGURES 221–229 ) but with usual low density of vertical hairs on legs. Tibia 1 in 62 females: 3.3–4.6 (mean 4.0). Epigynum very simple externally, weakly sclerotized, internal ‘valve’ and ‘receptacle’ visible through cuticle ( Figs 224–225 View FIGURES 221–229 ). Internal genitalia with pair of pore plates of variable shape ( Figs 226–239 View FIGURES 221–229 View FIGURES 230–239 ) and median duct of varying length connecting to oval or elongated ‘receptacle’; ‘receptacle’ usually in asymmetric position; 39 females from type locality: 18 right-sided, 21 left-sided (i.e., antisymmetric; cf. Huber et al. 2007). ALS spigots as in male ( Fig. 332 View FIGURES 329–335 ).
Notes. All specimens above are tentatively assigned to one species but the observed variation in both males and females suggests that this needs to be re-evaluated, ideally based on much larger samples and the addition of molecular data. ‘Receptacle’ shape varies strongly even within populations (compare Figs 226–229 View FIGURES 221–229 and 236–239 View FIGURES 230–239 ), suggesting that this is not a good species character. Male cheliceral armature and female pore plate shape may indicate morphological species limits but very few males are available (from several localities none), and a proper evaluation of pore plates requires the clearing of many more female abdomens. Thus, at this point a single, easily distinguished but potentially polytypic species seems clearly preferable.
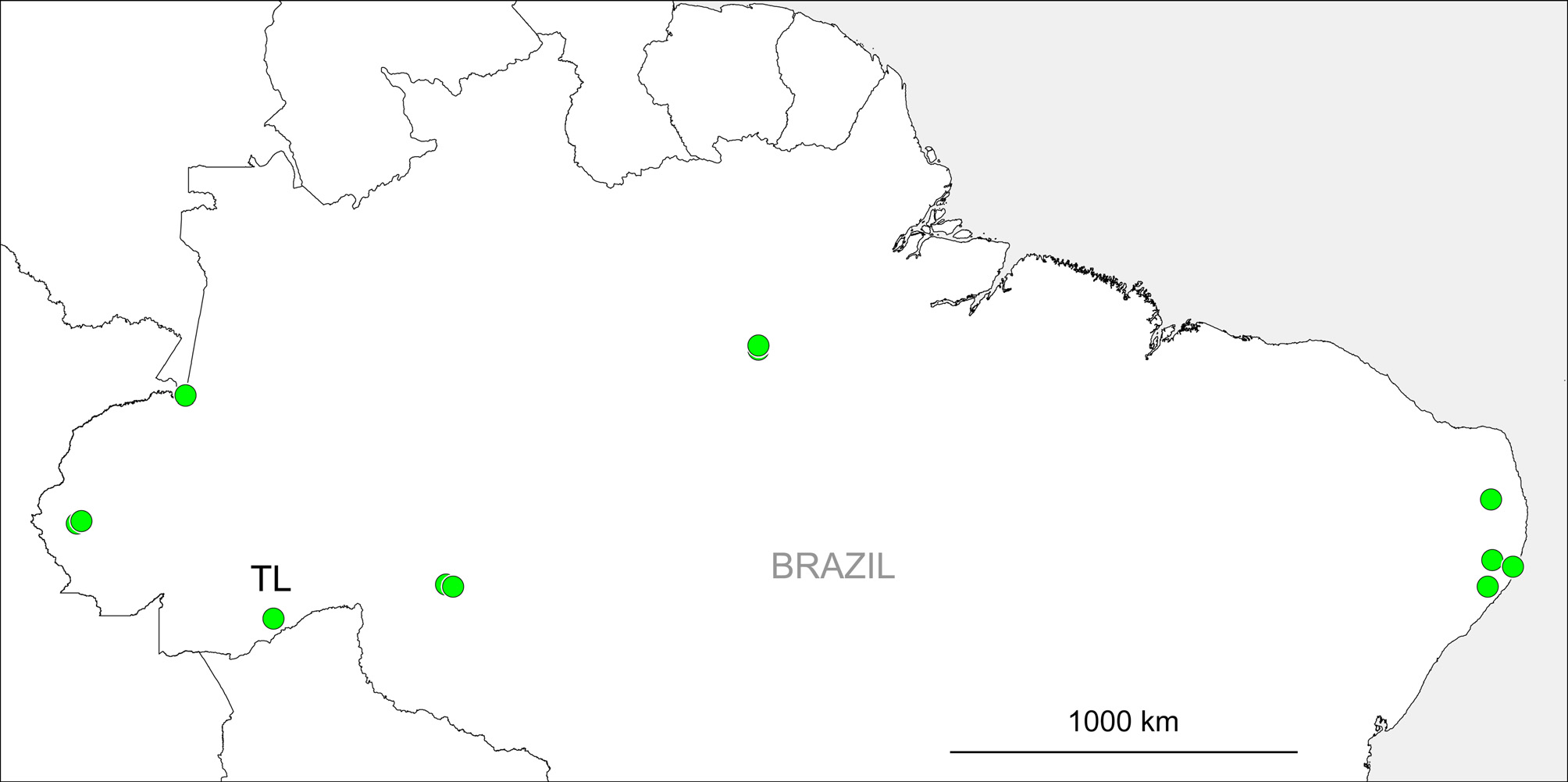
Distribution. Widely distributed in Brazil ( Fig. 347 View FIGURE 347 ), likely to range into neighboring Colombia, Peru, and Bolivia. This is one of only four pholcid species currently known to occur both in the Amazon and in the Atlantic Forest (the other species are Mesabolivar kathrinae Huber, 2015 , and two species where Amazonian and Atlantic Forest populations might in fact be separate species: Metagonia taruma Huber, 2000 and Metagonia beni Huber, 2000 ; L.C. Carvalho & B.A. Huber, unpubl. data). Most of the other ~190 formally described Brazilian Pholcidae are restricted to one of these forest blocks (very few species are described from other regions in Brazil).
Natural history. This species was found on the undersides of green leaves in the vegetation. There seemed to be a strong preference for certain hairy leaves: at the type locality, numerous specimens were collected from one species of plant with hairy leaves but very few from the many other plant species with otherwise suitable leaves (size and elevation from ground) present at the site. The spiders built domed sheet webs that had the apex connected to the underside of the leaf. Males and females were often found together.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
SubFamily |
Modisiminae |
|
Genus |