Mniophila STEPHENS , 1831
|
publication ID |
https://doi.org/ 10.21248/contrib.entomol.59.1.103-131 |
|
DOI |
https://doi.org/10.5281/zenodo.4794608 |
|
persistent identifier |
https://treatment.plazi.org/id/E101B54F-FFF4-9251-FEB7-4A89FC36FD01 |
|
treatment provided by |
Carolina |
|
scientific name |
Mniophila STEPHENS , 1831 |
| status |
|
Genus Mniophila STEPHENS, 1831
Mniophila STEPHENS, 1831: 330 . – HEIKERTINGER 1930: 1346 (catalogue). – HEIKERTINGER & CSIKI 1940: 520–522 (world catalogue). – KASZAB 1962: 349 (key, description). – MOHR 1966: 206, 261, fig 70: 1 (key). – GRUEV & TOMOV 1986: 312–314, figs 358, 359a, b (key, description). – DOGUET 1994: 549–552, figs 208, 209 (key, description). – KONSTANTINOV & VANDENBERG 1996: 271, 307, fig. 48 (key, description). – COX 1997: 277–279, figs 1–6 (egg, larva). – GRUEV & DÖBERL 1997: 240–241 (distribution, bibliography). – WARCHAŁOWSKI 2000: 25–28, figs 35–40 (key, description, distribution).
Type species: Haltica muscorum KOCH, 1803 , by monotypy.
Distribution:
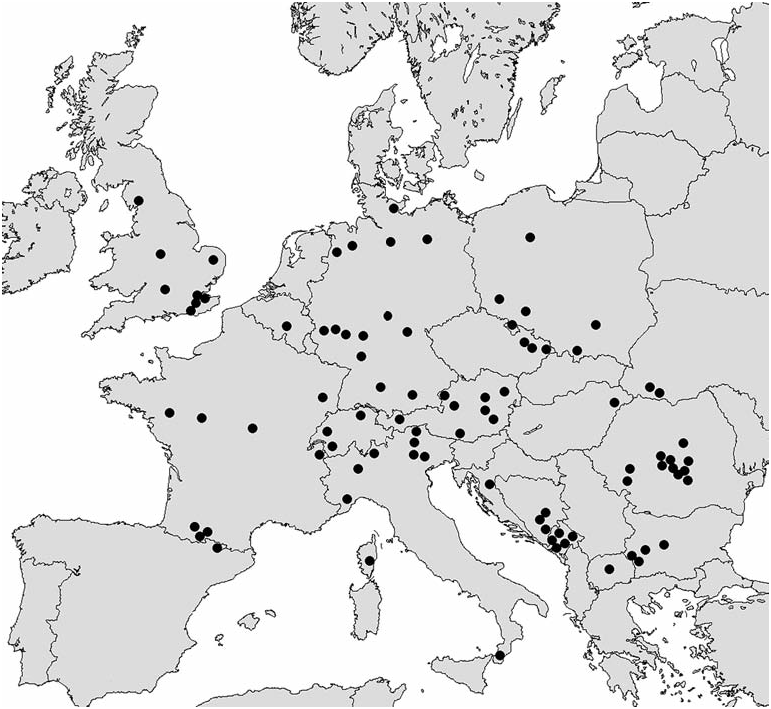
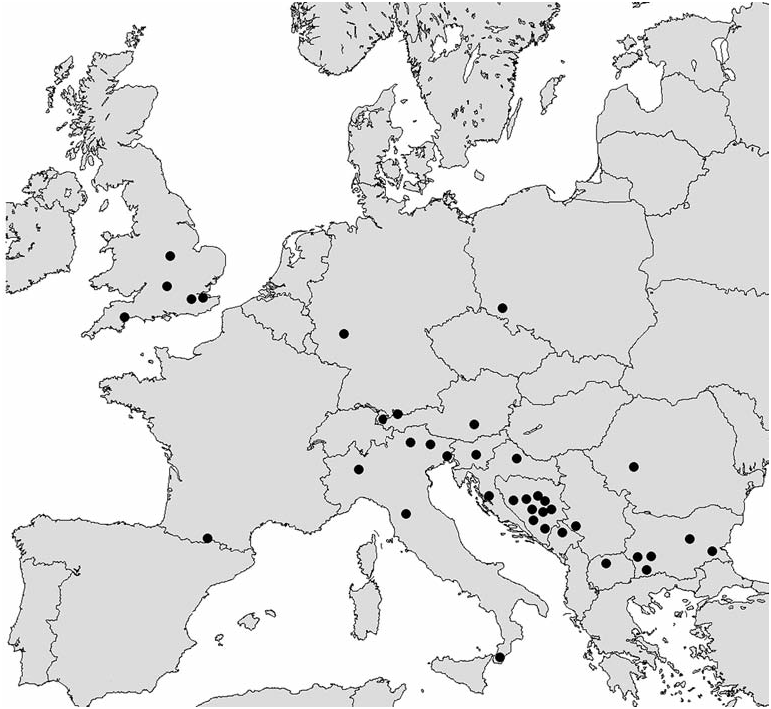
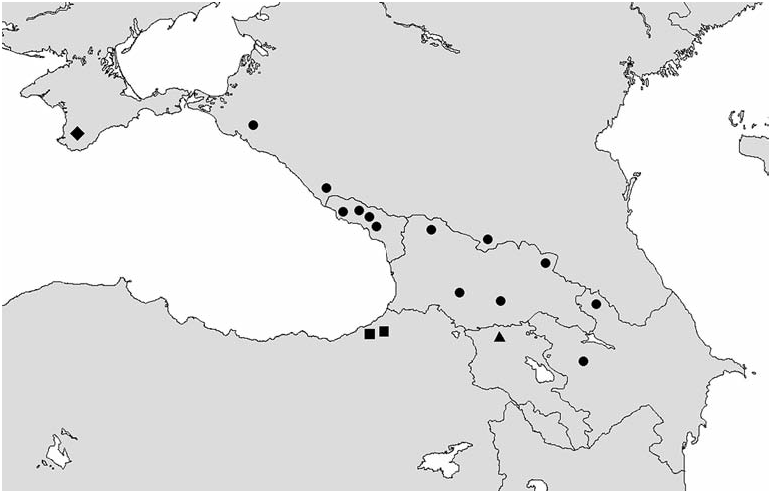
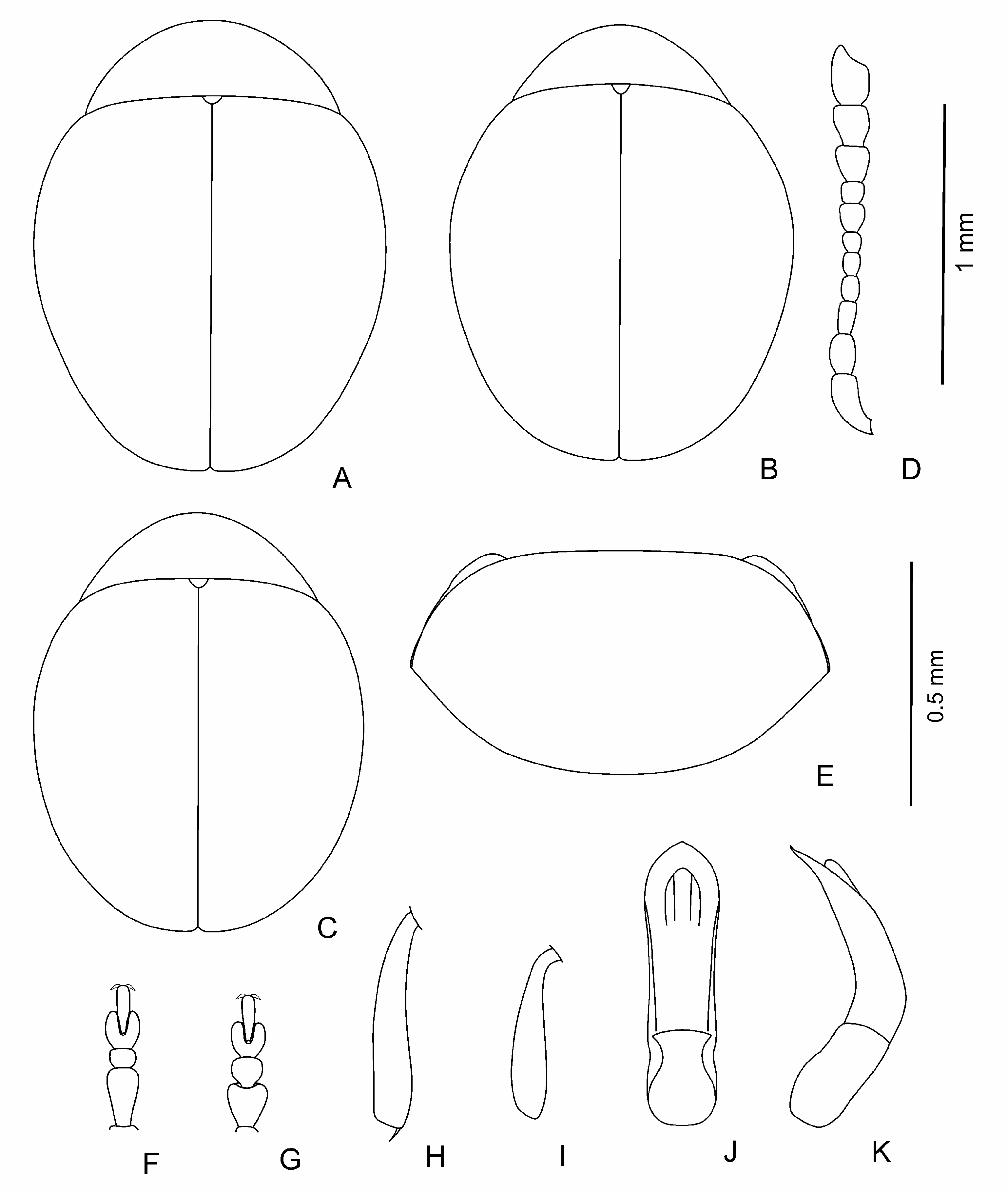
Range of distribution of the genus Mniophila embraces Western and Central Europe, Balkans, Crimea, Caucasus, and Turkey ( Figs 8-10 View Fig View Fig View Fig ). Mniophila muscorum has the widest range ( Fig. 8 View Fig ), from Pyrenees ( Spain) to Carpathians ( Ukraine). The northern boundary of this species is in Dania, Norway, and Sweden. The rest of the species have ranges smaller than Mniophila muscorum . It is worthy to mention the high level of endemism among the species of Mniophila . The only ranges of M. muscorum and M. bosnica , and probably, M. caucasica sp. n. and M. transcaucasica sp. n. are partly overlapping ( Figs 8-10 View Fig View Fig View Fig ).
Redescription:
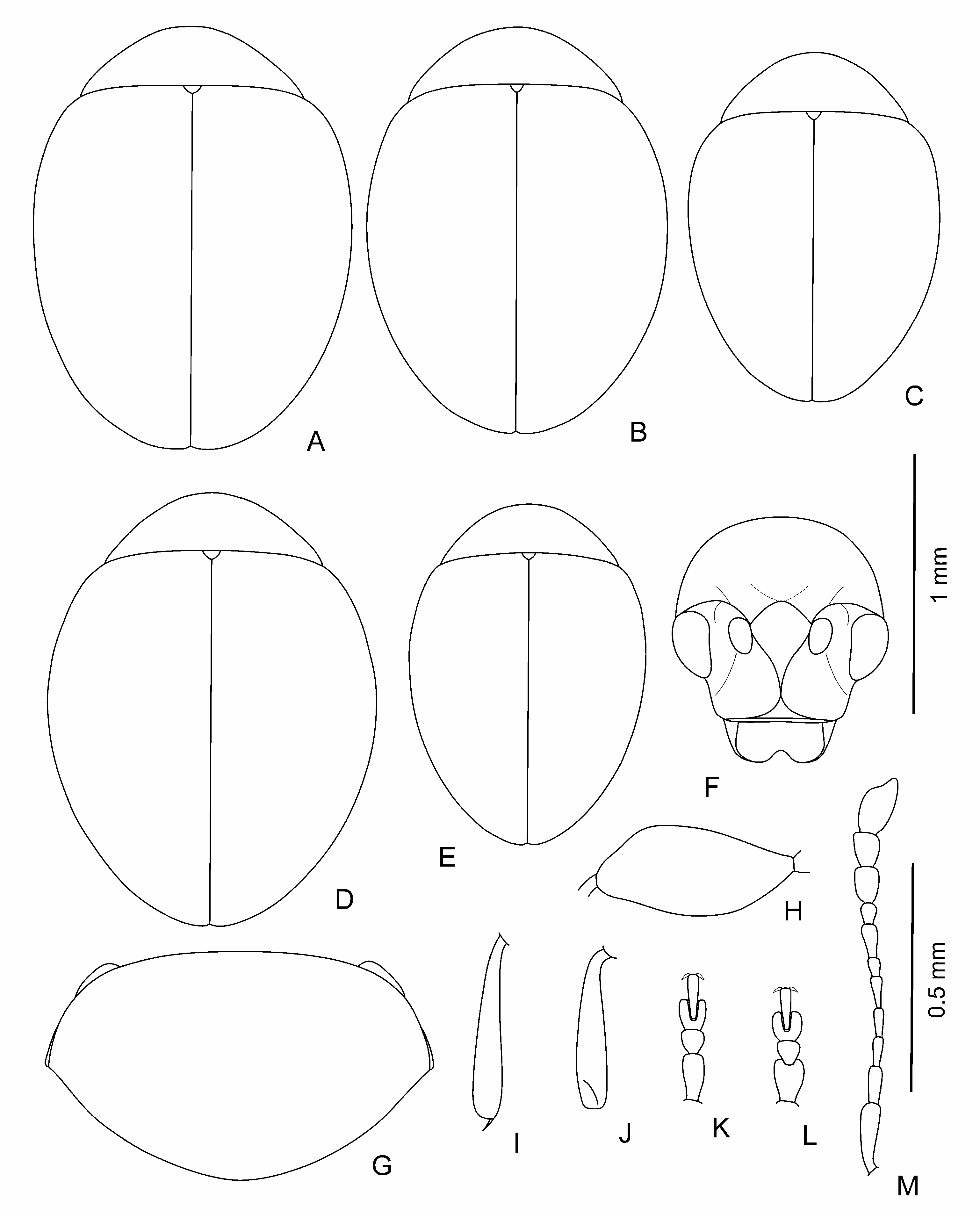
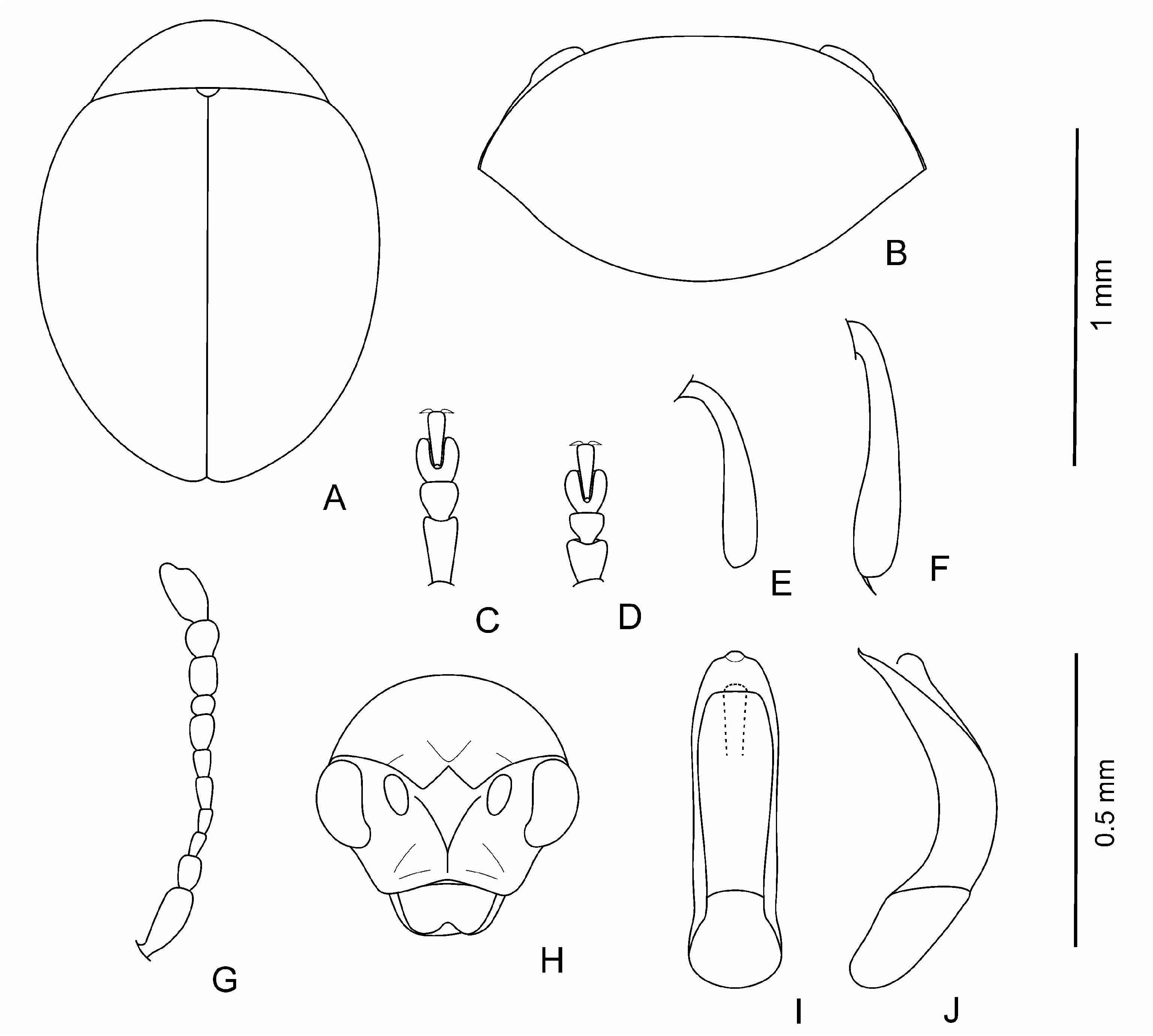
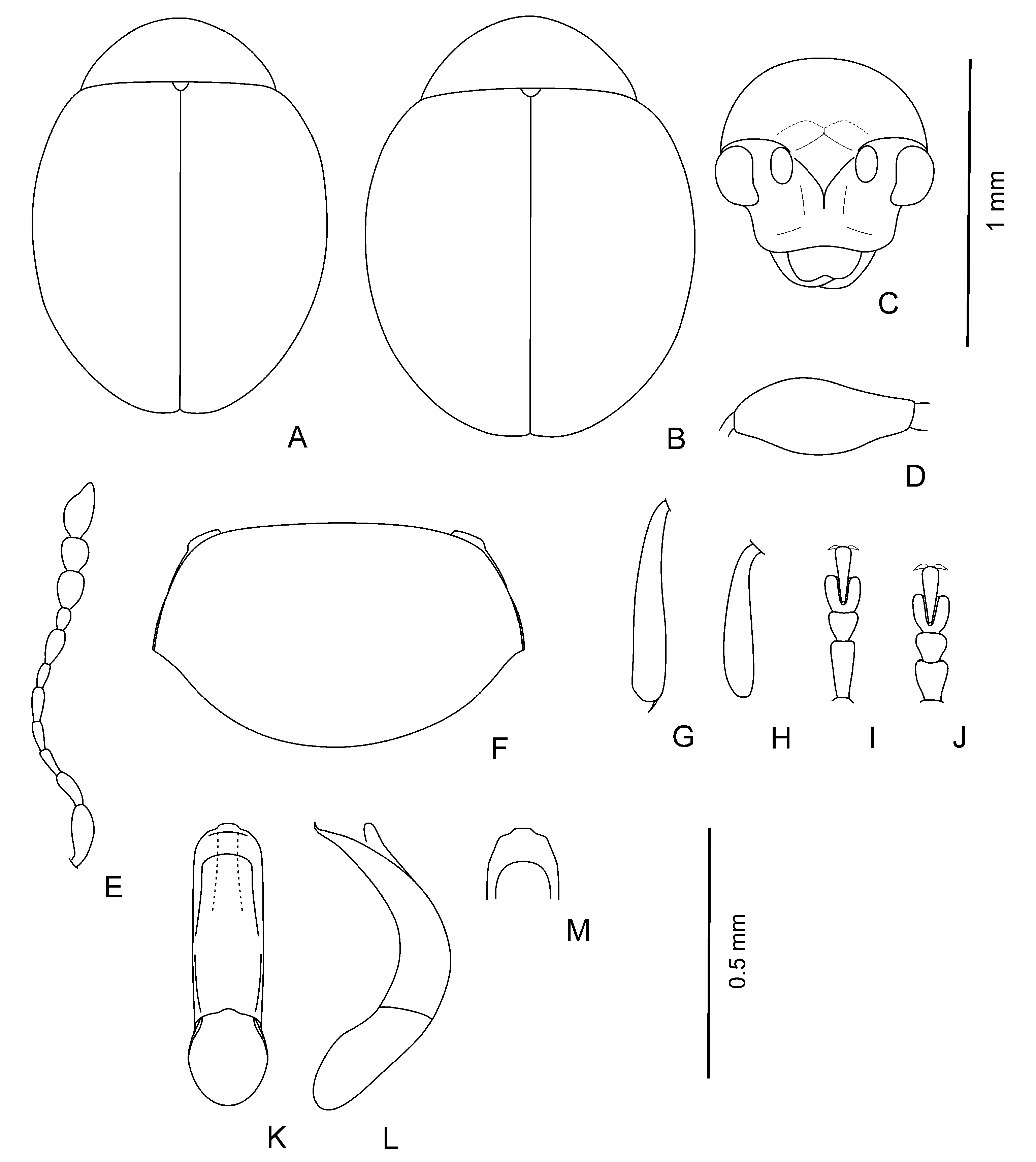
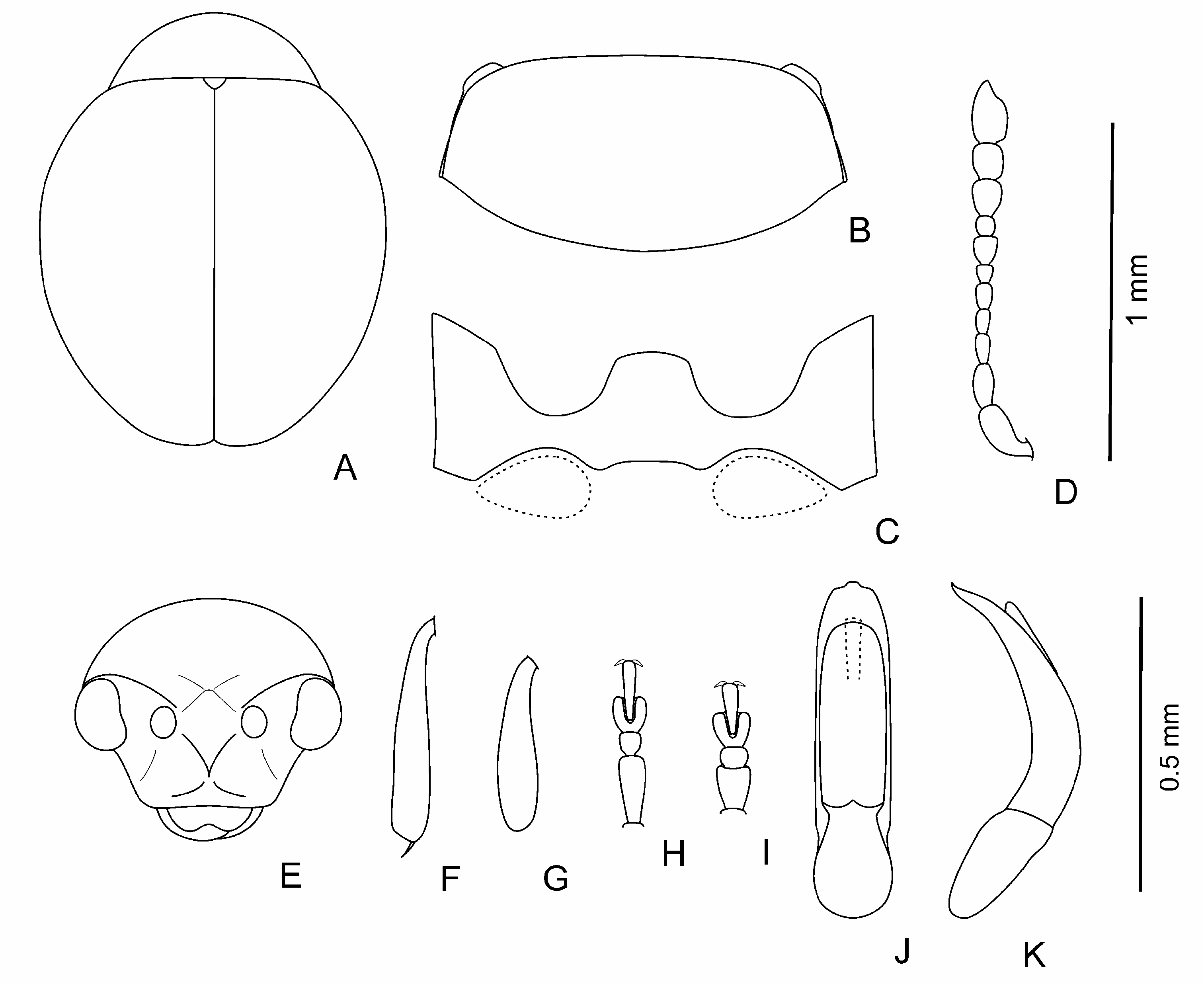
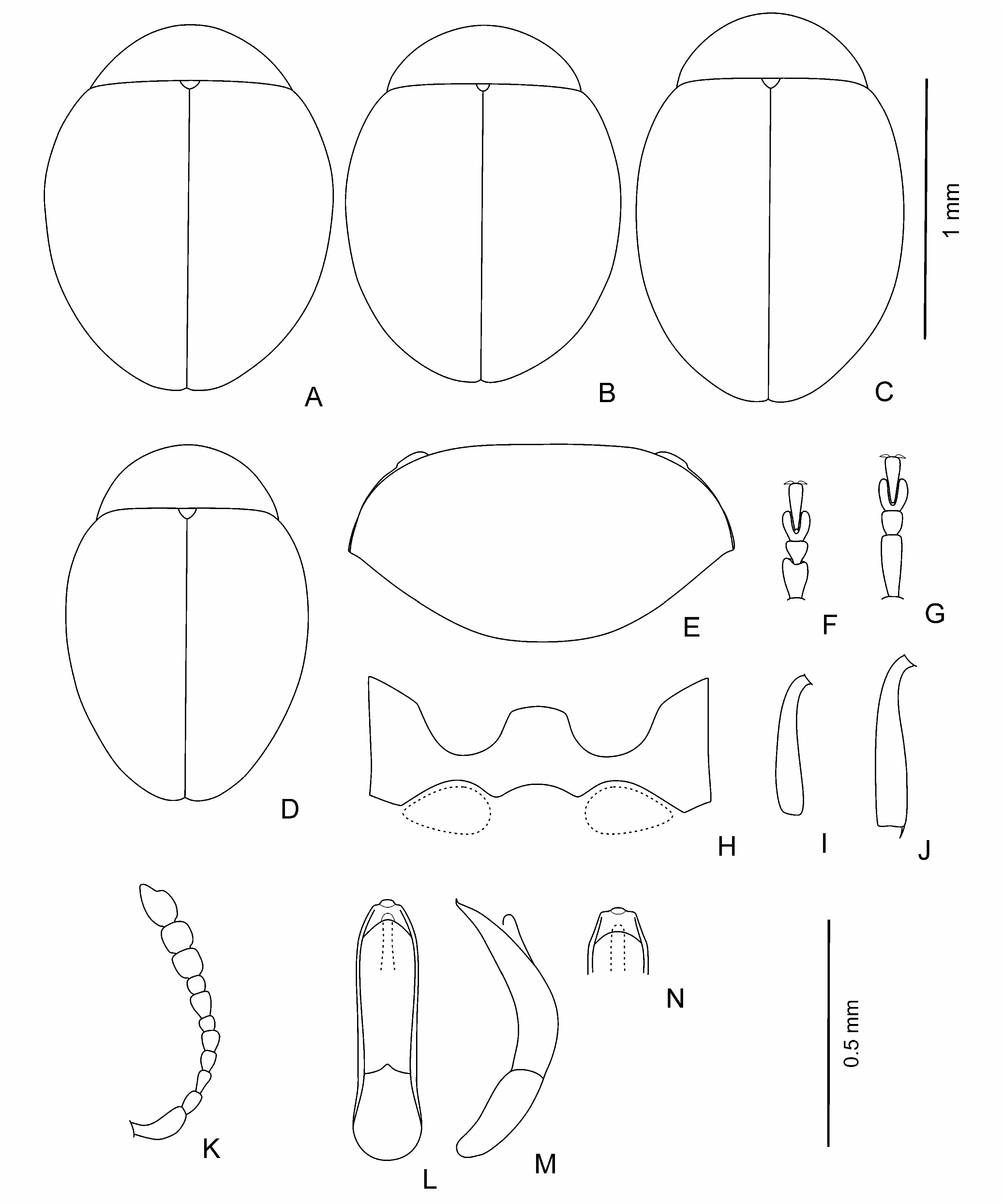
Body dark, brown to black, shining, with greenish or bronzy metallic luster or without one; sometimes in brown individuals pronotum and elytra with light margin; sometimes partly coloured individuals occur; legs and antennae light to dark brown. Body elliptical and moderately convex to clearly rounded and rather convex ( Figs 1 View Fig A-E; 3A-C; 4A; 5A, B; 6A-D; 7A); head not or barely visible from above. Head ( Figs 1F View Fig ; 4H View Fig ; 5C View Fig ; 7E View Fig ) large; vertex large, wide, its surface covered with weakly developed smooth shagrination coarse, well developed, grainy shagrination. Ocular sulci not developed. Frontal calli triangular or rhomboidal, small, its surface smoother than that of the vertex and frons; separated from vertex by thin supracallinal sulci. Frontal ridge distinct, convex, triangular and flattened basally. Anterofrontal ridge weakly convex, straight. Antennal grooves large and deep, covered with large and coarse shagrination. Frontal part of head moderately short to long. Antennal sockets situated closely to each other, separated from eye margin by impressed interval in length is almost equal to socket. Eyes small, elliptical, flattened to convex, widely spaced. Antennae ( Figs 1M View Fig , 3D View Fig ; 4G View Fig ; 5E View Fig ; 6K View Fig ; 7D View Fig ) short; three apical segments distinctly widened; remaining segments thinner; eighth antennomere thinner and smaller than seventh; first and second antennomeres clearly longer and thicker than following five segments. Labrum small, with deep frontal notch situated medially.
Pronotum ( Figs 1G View Fig ; 3E View Fig , 4B; F View Fig ; 6E View Fig ; 7B View Fig ) large, widely transverse. Apical and basal edges with rather thin margins; lateral margin wider. Anterofrontal callosity large, thickened, not forming acute denticle; posterolateral callosity smaller, poorly developed. Basal edge of pronotum curved in sides, distinctly rounded medially or widely rounded. Pronotal surface covered with visible, large shagrination. Punctation usually present, not coarse, sparse, and shallow, poorly visible on pronotal microsculpture, rarely impunctate.
Elytra slightly elongate with elongate elytral apices to clearly rounded, without elongate apices. Punctation variable: impunctate to punctured densely; usually punctures arranged in regular or partly confused striae, depth and frequency, and size of punctures variable; secondary punctation
from undeveloped to well developed, often as large as punctures in striae and dense. Elytral interstices smooth. Hind wings completely reduced.
Pro-, meso-, and metathorax strongly shortened; mesothorax shorter than pro- and metathorax. Procoxal cavities open.
Legs (1I-L; 3F-I; 4C-F; 5G-J; 6F, G, I, J; 7F-I) not long, usually thick, rarely thinner; first protarsomere of male larger and thicker than in female; hind tibiae and often that of middle and foreleg curved, sometimes fore and middle tibiae straight; hind femora usually wide ( Fig. 1H View Fig ), rarely thin ( Fig. 5D View Fig ).
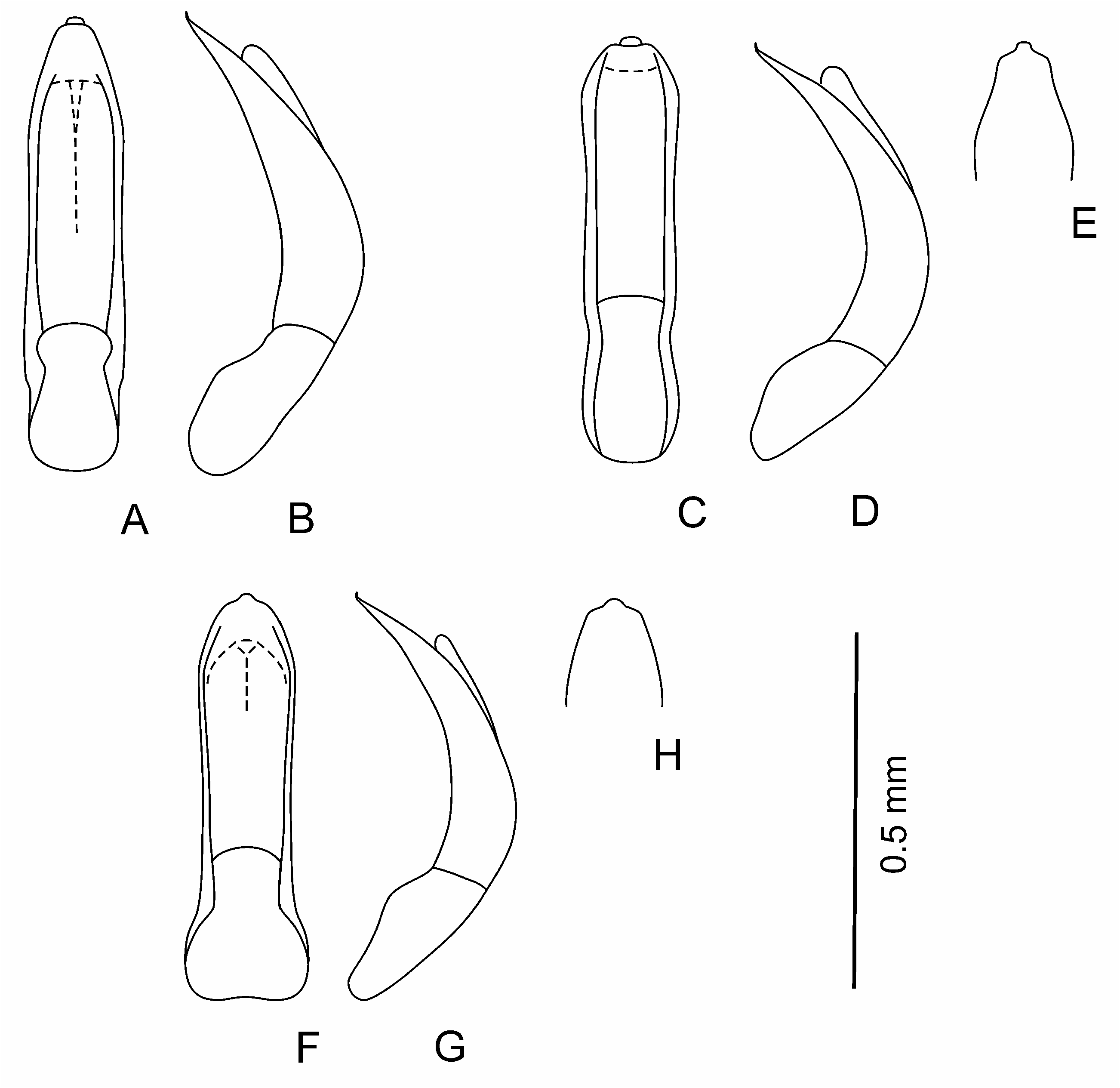
Aedeagus ( Figs 2 View Fig , 3J, K View Fig ; 4I, J View Fig ; 5 View Fig K-M; 6L-N; 7J, K) rather large, curved basally, apical 2/3 almost straight to distinctly curved; apex shape variable: rounded, widened, elongated and narrowing, with or without apical denticle. Spermatheca small, collo large, thick, straight, nodulus comparatively narrow, not very long, duct short, moderately curved, not forming loops, ramus globose, globular.
Body length: 1.1-1.6 mm, width – 0.8-1.2 mm.
Preimaginal stages. Egg and first instar larva have been described by COX (1997).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
Mniophila STEPHENS , 1831
| Nadein, Konstantin S. 2009 |
Mniophila
| WARCHALOWSKI, A. 2000: 25 |
| COX, M. L. 1997: 277 |
| GRUEV, B. & DOBERL, M. 1997: 240 |
| KONSTANTINOV, A. S. & VANDENBERG N. J. 1996: 271 |
| DOGUET, S. 1994: 549 |
| GRUEV, B. & TOMOV, V. 1986: 312 |
| MOHR, K. - H. 1966: 206 |
| KASZAB, Z. 1962: 349 |
| HEIKERTINGER, F. & CSIKI, E. 1940: 520 |
| HEIKERTINGER, F. 1930: 1346 |
| STEPHENS, J. F. 1831: 330 |