Huayqueriana
|
publication ID |
https://doi.org/10.1206/0003-0090-404.1.1 |
|
persistent identifier |
https://treatment.plazi.org/id/C03A87C6-FFA5-FF87-FCAE-AA2DFBE2FBFB |
|
treatment provided by |
Carolina |
|
scientific name |
Huayqueriana |
| status |
|
PALEOBIOLOGY OF HUAYQUERIANA
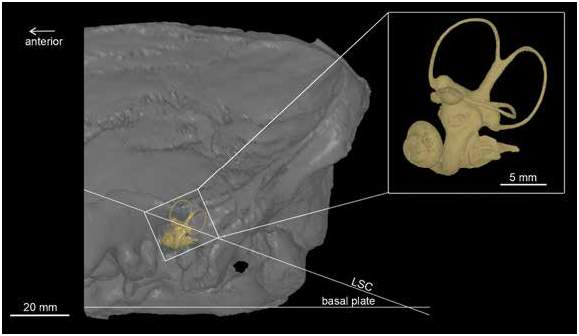
HEAD POSTURE: Following De Beer (1947), the possible life position of the head of Huayqueriana IANIGLA-PV 29 was explored by digitally projecting the 3D model of the inner ear onto the skull, with the plane of the LSC parallel to the horizon ( fig. 27 View FIG ). This orientation is biologically meaningful because the LSC is usually horizontal to the substrate when the animal’s head is in the position of normal visual alertness ( De Beer, 1947; Ekdale, 2013). At rest or during dynamic activities (i.e., locomotion, grooming, feeding) the LSC is displaced relative to the horizon ( Hullar, 2006). When the LSC is horizontal, the ASC and PSC are vertical and would experience the least stimulation when the head is rotated. This disposition allows the organ of balance to function at maximum efficiency ( De Beer 1947).
In IANIGLA-PV 29, the angle subtended by the plane of the LSC and the plane of the base of the skull is about 26°, which is a value similar to that of the horse ( De Beer, 1947) ( fig. 28 View FIG ). Such a head posture in Huayqueriana might have enabled partial binocular vision, as occurs in horses and certain other ungulates (Miller, 1975; Marcus and Sarmiento, 1996). Additionally, among xenarthrans, a strongly inclined LSC was found in armadillos, and regarded as consistent with their “nose down” head position (G. Billet., personal commun., 2016).
BODY SIZE: Our efforts to predict the body mass of IANIGLA-PV 29 using several different approaches are summarized in table 5. Mean body mass predicted by the equations published by Janis (1990) imply that the mass of Huayqueriana cf. H. cristata approached 400 kg. This is a significantly larger body mass than known empirical ranges for allometrically comparable panperissodactyls (Welker et al., 2015) such as extant Tapirus ( T. indicus , 250–375 kg; T. terrestris , 77–300 kg) and wild Equus caballus ( 200– 300 kg) (Nowak and Paradiso, 1983; Silva and Downing, 1995; Clauss et al., 2003). It is also noteworthy that the range of Janis’ estimators is considerable ( 154 to 721 kg), with a percent prediction error above 25%.
A second approach, utilizing algorithm 4.1 of Mendoza et al. (2006), produces a considerably lower estimate of ~ 230 kg for IANIGLA-PV 29 (table 5). The percent prediction error was also lower, at 16%. A third approach, using the equations published by Cassini et al. (2012b), yielded the lowest percent prediction error of all (~6%). Their CR4 statistic utilizes the centroid size of a configuration of 36 3D cranial landmarks to predict body mass, which in this case produced a value of 250 kg.
Interpretation of body mass estimates based on different body parts is often difficult because they produce inherently incompatible results. Thus in the case of Janis’ (1990) equations, considerable disparity exists between results based on dental vs. cranial material of IANIGLA-PV 29 (table 5), with dentally based estimators yielding substantially larger body sizes. As more comprehensive studies show ( Fortelius, 1990; Millien and Bovy, 2010), body mass estimations based on cranial and postcranial measurements are preferable to ones based on dental measurements.
To assess apparent overestimation in a quantitative manner, Cassini et al. (2012b) developed a quantile regression statistic (Qsup) that provides a theoretical maximum body mass for a given centroid size (CS) based on (in this instance) extant ungulates. In the case of IANI- GLA-PV 29, there is no extant ungulate with a CS similar to that of the fossil whose mean body mass surpasses 419 kg (CS maximum, for Bos taurus ), and in light of this we reject results produced by all three of Janis’ dental equations (SUMA, SUMW, SUML). Fariña et al. (1998) reached a similar conclusion when employing Janis’ dental regression equations to predict the body size of Macrauchenia patachonica (e.g., SUML = 1500 kg vs. their mean estimate of 988 kg based on 66 cranial and postcranial equations). This suggests that a body mass of 400 kg for Promacrauchenia , as estimated by Vizcaíno et al. (2012), probably also requires reassessment.
By contrast, the mean for Janis’ nondental equations is ~ 265 kg, a value more consistent with results obtained using algorithm 4.1 of Mendoza et al. (2006) and the CR4 equation of Cassini et al. (2012b). In particular, a value of 250 kg for the body mass of Huayqueriana IANI- GLA-PV 29, using the CR4 statistic of Cassini et al. (2012b), seems more realistic than much larger estimates, and is more in accord with body sizes seen in distantly related taxa such as extant Tapirus and Equus . These results suggest that a rough doubling of body size occurred in macraucheniines between the time of Santacrucian Theosodon (~ 130 kg; Cassini et al., 2012a, 2012b) and Huayquerian Huayqueriana . Further insights into macraucheniine body size will require discovery of associated elements of Huayqueriana , particularly long bones.
LONGEVITY: Given the extremely advanced dental wear of IANIGLA-PV 29, it can be argued that the specimen was approaching its maximum longevity at the time of death. Counting cementum rest lines, which in principle are deposited
TABLE 5
Body mass estimation of Huayqueriana cf. H. cristata
2 Equation from Christiansen and Harris (2005) for multiple estimates ( n). The final body mass (M) average is:
1 1
WM =∑(( Mi /% PEi)* ∑ %PE)* n -2
Mi Mi
where M is the body mass computed by the regression equations and n is the sample size.
3 Equation of Mendoza et al. (2006) after Cassini et al. (2012a): Ln Algorithm 4.1= 0.736*Ln(SUML) + 0.606*Ln(SUMW) + 0.530*Ln(MZW) + 0.621*Ln(PAW) + 0.741*Ln(SC) – 0.157*Ln(SD) + 0.603; in which MZW, muzzle width; PAW, palatal width; SC= PSL, posterior skull length; SD, depth of the face under the orbit; SUML, second upper molar length; SUMW, second upper molar width. Measurements in table 2.
4 Centroid size follows Cassini et al. (2012b): CR4: Log10 (BM)= 3.165*Log10 (CS) – 6.701; Qsup: Log10 (BM)= 3.321*Log10 (CS) – 6.926.
annually, is often considered the most reliable method of calculating longevity, especially if the animal lives in a temperate climate ( Klevezal, 1996) in which there is a distinct low-growth season, usually winter (e.g., Chritz et al., 2009). Age assessment is less precise for mammals living in tropical areas, which lack extreme seasonality. Lines may be indistinct, or two lines may be deposited during a single year ( Klevezal, 1996). But given the fact that the Huayquerias fossil site is in a temperate region and that seasonality in the area was probably already established by the Late Miocene, we consider that these lines represent annual depositions. Nine cementum lines can be counted on the sectioned M2 of IANIGLA-PV 29. However, converting that to a plausible age requires correction. Because the last growth layer (external to the last rest line, where the periodontal ligament attached the tooth to the maxilla) is almost as thick as each preceding layer ( fig. 25 View FIG 25 ), it may be concluded that the animal died at the end of a year. Based on the data of Klevezal (1996) for the M1 and M2 of ungulates, a correction of 0 to 1 year must be added to the cementum count. Therefore, we suggest that IANIGLA-PV 29 died at an age of almost 10 or 11 years.
Longevity is known to be positively correlated with body size in mammals (Schmidt-Nielsen, 1984). We compared the estimated age of IANI- GLA-PV 29 to the longevity of wild extant ungulates approximating a body mass of 250 kg (longevity data from AnAge database, Tacutu et al., 2013; body mass ranges from Nowak and Paradisio, 1983). Longevity is notably greater in the latter. Examples are: ~15 years for the topi, Damaliscus lunatus ; 24 years for the muskox, Ovibos moschatus ; and 22 years, or possibly more, for the elk, Cervus canadensis , or moose, Alces alces . By contrast, ungulates with a longevity in the wild of roughly 10–12 years are conspicuously smaller, with a mean body mass below 100 kg (e.g., the east Caucasian tur, Capra cylindricornis , 50 kg; the markhor, Capra falconeri , 41 kg; the goitered gazelle, Gazella subgutturosa , 49 kg; the Nile lechwe, Kobus megaceros , 90 kg; the gerenuk, Litocranius walleri , 44 kg; the common rhebok, Pelea capreolus , 25 kg; the blue duiker, Philantomba monticola , 6 kg; the mountain reedbuck, Redunca fulvorufula , 30 kg; the brocket, Mazama , 17–33 kg; and the pygmy hog, Sus salvanius, 8 kg). The clear conclusion is that, compared to the longevity of modern wild ungulates of a size similar to Huayqueriana , the latter had a much shorter lifes- pan. Note that the magnitude of this conclusion would only increase if larger size estimates were used for Huayqueriana .
To provide additional context we also sectioned the M1 of an adult Theosodon garrettorum (PIMUZ A/V 4662). Its wear stage is very advanced (styles are completely worn on M1, but on M2–M3 the external outlines are still evident). The specimen exhibited 6 to 7 cementum lines and a thin last growth layer, yielding a likely age of 6–8 years, which is much lower than expected for its body size (ca. 140 kg; Cassini et al., 2012a). Indeed, artiodactyls of similar size live up to ~20 years in the wild (e.g., fallow deer, Dama dama , 25 years; Pere David’s deer, Elaphurus davidianus ,18 years). Our finding opens the question of the influence of environment and food items on longevity reduction in mesodont herbivores during the late Neogene. Unlike notoungulates and many other clades of South American placentals, litopterns failed to develop euhypsodont dentitions as an outcome of adaptation to ecosystem change (Madden, 2015).
DIGESTIVE PHYSIOLOGY: Herbivores utilize different strategies to efficiently digest celluloserich vegetation. The broadest partition is between taxa that ferment plant material in the foregut (stomach and/or specialized portions of the small intestine; e.g., ruminant artiodactyls, hippos, colobine monkeys, muroid rodents, sloths, kangaroos, koalas) as opposed to those that utilize the hindgut (caecum and/or colon; e.g., perissodactyls, proboscideans, hyraxes, ateline monkeys, caviomorph rodents, rabbits, wombats) (Clauss et al., 2003; Fletcher et al., 2010). In general, extant hindgut fermenters have a greater capacity for food intake than foregut fermenters of similar size and diet, and also chew their food more on initial ingestion (Clauss et al., 2003; Fletcher et al., 2010). The most specialized foregut fermenters, among which are ruminant artiodactyls, rely on regurgitation, rechewing, and remixing of digestive fluids to ensure maximum extraction of nutrient value from food. In contrast, after initial intake and chewing, hindgut fermenters rely exclusively on chemical reac- tions in the caecum or colon for nutrient extraction—an ultimately less efficient process.
Although there are marked differences between foregut and hindgut fermenters in digestive physiology, these differences are not reflected osteologically in any obvious way, which makes predictions difficult or impossible for extinct taxa. In principle, differentiation might be expected in the relative occlusal surface area of the cheek teeth For example, occlusal area seems to be larger in hindgut fermenters of similar size and diet, who also chew food more vigorously in the first stages of digestion than foregut fermenters do (Vizcaíno et al., 2006; Cassini et al., 2012a). Differences in jaw mechanics may also be indicative, at least in theory, as jaw size and musculature should be greater in hindgut fermenters of similar body size and diet than foregut fermenter (e.g., Fletcher et al., 2010).
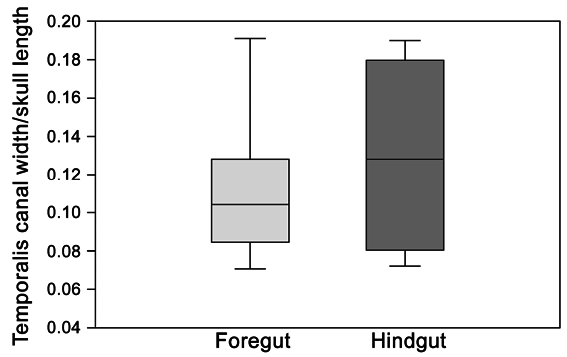
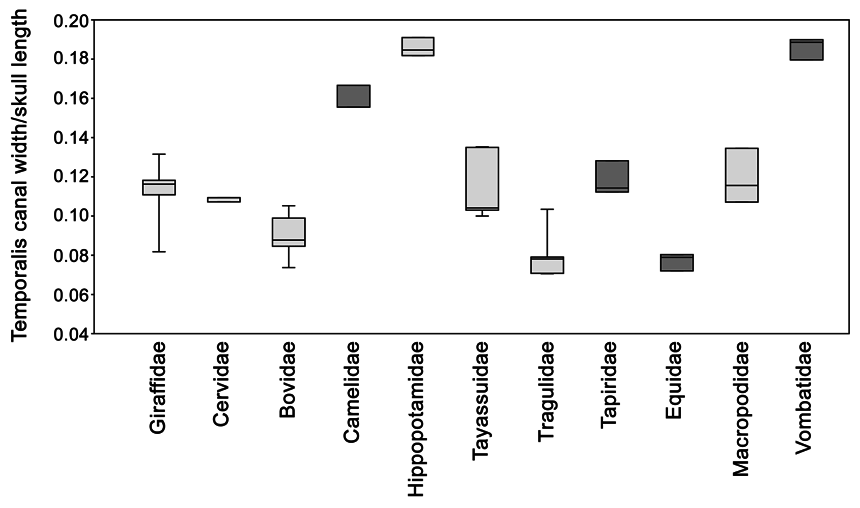
Based on a study of the occlusal surface areas of cheek teeth, Cassini et al. (2012a) demonstrated that all Early Miocene litopterns may have had a gut physiology comparable to hindgut fermenters. In contrast, Madden (2015) concluded that macraucheniids appear to have possessed an inferred masticatory muscle mass more like that of foregut fermenting ruminants than hindgut digesters. He assessed masticatory muscle mass in 24 specimens (representing 11 taxa of various placental groups) on the basis of two osteological measurements, skull length (SL) and width of temporalis canal or temporal fossa (TS). On his analysis the two fermentation styles were found to form two distinct groups (Madden, 2015).
In order to test Madden’s hypothesis statistically, we measured SL and TS in 43 foregut and hindgut fermenters (distributed across 11 taxa; appendix 3). In our analysis, foregut and hindgut fermenters did not form distinct groups ( fig. 29 View FIG ). As the data do not follow a normal distribution as judged by the Shapiro-Wilk and Anderson- Darling tests (p-value <0.01 for the foregut fermenter group), either a nonparametric test or a data transformation is needed to compare the two groups. Using the nonparametric Mann– Whitney U test, we found that there is no signifi- cant difference between the two groups (p-value>0.14). Similarly, after log-transforming the data and checking for both normality of the distribution (Shapiro-Wilk and Anderson-Darling tests, p-value>0.4) and homoscedasticity (Levene’s test, p-value>0.10), we found once again that the two groups were not significantly different (Student’s t-test, p-value>0.09). We also pooled the data by family in order to compare our results to Madden’s analysis ( fig. 30 View FIG ), but with the same result. In sum, our results indicate that extant foregut and hindgut fermenters cannot be separated from one another only using skull length and width of the temporal fossa as proxies for masticatory muscle mass.
The hindgut fermentation strategy seems to be the plesiomorphic condition for ungulates (Prothero and Foss, 2007). In view of the fact that there are no foregut fermenters in crown Perissodactyla , the extant group to which litopterns, notoungulates, and possibly other SANUs are most closely related (Welker et al., 2015; Buckley, 2015), phylogenetic bracketing predicts that litopterns were not either. Further insights may be provided by other predictive models not assessed here (e.g., Cassini et al., 2012a).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.