Scapsipedus icipe Hugel & Tanga, 2018
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4486.3.9 |
|
publication LSID |
lsid:zoobank.org:pub:E96EC360-E058-4ED6-96CE-65D1A08382BD |
|
DOI |
https://doi.org/10.5281/zenodo.5962087 |
|
persistent identifier |
https://treatment.plazi.org/id/B53A836A-2E75-FFA2-FF51-FB46FEBAFDF3 |
|
treatment provided by |
Plazi |
|
scientific name |
Scapsipedus icipe Hugel & Tanga |
| status |
sp. nov. |
Scapsipedus icipe Hugel & Tanga View in CoL , n. sp.
Figures 1–15 View FIGURES 1–6 View FIGURES 7–9 View FIGURES 10–12 View FIGURE 13 View FIGURE 14 View FIGURE 15 , Table 1–2.
Scapsipedus icipe Hugel & Tanga nov. sp., here described.
Distribution. Kenya ( Figure 15 View FIGURE 15 ).
Holotype. Male. icipe Duduville campus, Kasarani , Nairobi, Kenya, Lat. 01° 13' 14.6'' S, Long. 036° 53' 44.5'' E, Alt. 1612 m, 0 4th March 2014, Tanga Mbi leg, NMK GoogleMaps .
Paratypes. Males. same as holotype, 2♂ NMK, 3♂ MNHN, 3♂ coll. SH. Females: same as holotype, 2♀ NMK, 3♀ MNHN, 3♀ coll. SH.
The species was also found in other localities (not included in the seria typica), as illustrated in Figure 15 View FIGURE 15 .
Diagnosis. Size large for the genus (body length> 21 mm); head moderately but distinctly widened below the eyes (frontal view, Fig. 1 View FIGURES 1–6 ; unlike S. latus Gorochov, 1988 where it is strongly widened; unlike S. mjagkovi Gorochov, 1988 and S. thesigeri Gorochov, 1993 where it is weakly widened), head black with a distinct yellow band between the eyes, no continuous lighter line around upper and posterior part of the eye (unlike S. latus , S. steinbergi Gorochov, 1988 , S. amplus Gorochov, 1988 ), genae with a yellow ventral line from the posterior part of the eye to mandibles and posterior margin of head (side view, Fig. 3 View FIGURES 1–6 ; similar to S. latus , somewhat similar to S. thesigeri ; unlike S. mjagkovi , S nigriceps Chopard, 1954 , S. flavomarginatus ( Chopard, 1934) , S. obscuripes ( Chopard, 1962) which have no yellow pattern on genae, occiput with a fine yellow band from the top of the eye to the posterior margin of the head (side view, Fig. 3 View FIGURES 1–6 ; unlike S. nigripes and S. flavomarginatus which have uniformly black heads, unlike S. mjakovi and S. amplus which have discontinuous bands, and unlike other species that have different patterns). Male genitalia similar to S. mjagkovi , distal projection of pseudepiphallus narrowing regularly at the basis (not strongly narrowing unlike S. mjakovi and S amplus , in dorsal view, Fig. 12 View FIGURES 10–12 ), terminal part blunt-Vshaped (in dorsal view, Fig. 12 View FIGURES 10–12 ; unlike all other species) widened ventral lobe of pseudepiphallus (the part with setae) rounded apically (side view, Fig. 11 View FIGURES 10–12 ; as in most species but unlike S. mjagkovi where it is obliquely truncated). Pseudepiphallic parameres with a particularly wide lateral anterior branch, and with a particularly short median anterior branch that hardly reaches the mid length of the lateral branch (unlike all other species, including S. amplus , the latter of which shares with S. icipe a paramere with a very wide lateral anterior branch; ventral view, Fig. 10 View FIGURES 10–12 ).
Description. Size large for the genus ( Table 1). Head moderately but distinctly widened below the eyes (frontal view, Fig. 1 View FIGURES 1–6 ); black with a distinct yellow band between the eyes, genae with a yellow ventral line from posterior part of the eye to mandibles and posterior margin of head (side view, Fig. 3 View FIGURES 1–6 ), occiput with a fine yellow band from the top of the eye to the head posterior margin, with two faint sub-median lines (dorsal view, Fig. 2 View FIGURES 1–6 ); mandibles brown, palpi yellow with black apex; clypeus dark with a yellow sagittal line and ventral margin; labrum black; eyes of average size. Pronotum slightly larger anteriorly; anterior margin inconspicuously convex, posterior margin inconspicuously concave; disk of pronotum black with inconspicuous transverse light patterns on the middle, with well distinct lateral band on the distal two thirds, these bands are spotted with black (dorsal and side views, Fig. 2, 3 View FIGURES 1–6 ); lateral lobes with a yellow pattern on the ventral margin (side view, Fig. 3 View FIGURES 1–6 ). Legs. Fore and midlegs of light color, femora often with black spots on proximal and distal ends, tibiae often infuscate near knees; hind femur brown with dark knee, hind tibia black. Wings. Forewings not reaching the end of abdomen; dark, with a lighter area around harp in males ( Fig. 4, 5 View FIGURES 1–6 ), near the fold in females ( Fig. 7 View FIGURES 7–9 ). Male file with ca. 170–190 teeth; teeth density increasing distally ( Fig. 6 View FIGURES 1–6 ); area with numerous setae distally to the file (ventral view, Fig. 6 View FIGURES 1–6 ). Hindwings often present. Abdomen black dorsally with yellow spots, yellow ventrally. Male genitalia. Pseudepiphallus with a relatively narrow distal projection, distal projection narrowing regularly at the basis, terminal part blunt-V-shaped (in dorsal view, Fig. 12 View FIGURES 10–12 ), ventral lobe of pseudepiphallus with setae, relatively acute, rounded apically (side view, Fig. 11 View FIGURES 10–12 ). Pseudepiphallic parameres without posterior branch, with particularly wide lateral anterior branch, with particularly short median anterior branch hardly reaching the mid length of the lateral branch (ventral view, Fig. 10 View FIGURES 10–12 ). Ovipositor much longer than hind femur, moderately curved ( Fig. 7–9 View FIGURES 7–9 ).
All values are in mm. F: femur; FW: forewing; IOW: inter ocular width; L: length; Ovp: ovipositor; T: thorax; W: width.
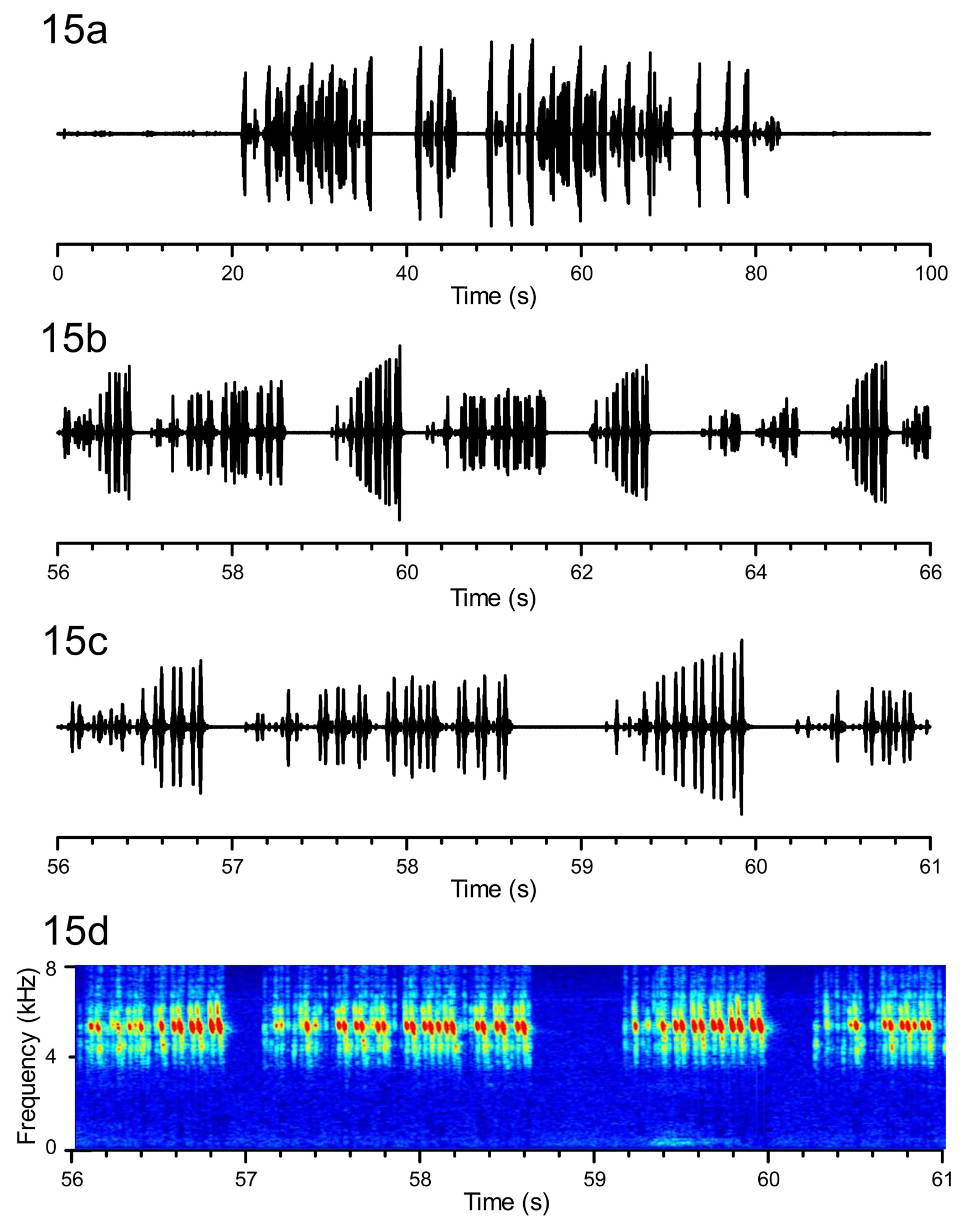
Acoustic behavior ( Figures 13–14 View FIGURE 13 View FIGURE 14 ). The call of S. icipe n. sp. ( Fig. 13 View FIGURE 13 ) is made up of more or less regularly repeated sentences containing five syllables, the first syllable having a lower amplitude than the four others. At 28±1°C, 50 ± 8% RH and photoperiod of L12: D12, sentences are repeated every 2.70 ± 0.51 s (min: 2.04; max: 4.19 s; 4 specimens), these last 356 ± 116 ms (min: 228 ms; max: 704 ms; 4 specimens), the frequency peaks at 5.77 ± 0.04 kHz (min: 5.69; max: 5.84; 4 specimens). The courtship song of S. icipe n. sp. ( Fig. 14 View FIGURE 14 ) consists of irregularly repeated sequences, usually containing repeated pairs of syllables; the frequency peaks at similar frequency as the call.
Molecular data. No sequences of Scapsipedus spp were available in databases. The percentage identities obtained from GenBank for the samples to the closest linking relative was 90% to Gryllus bimaculatus isolate 0 88 (MF046161.1) with query coverage of Ž 99%. Scapsipedus icipe sequences have been deposited in Genbank (Accession number MH923437 View Materials - MH923446 View Materials ).
Etymology. This new cricket is named after the type locality, International Centre of Insect Physiology and Ecology ( icipe ), Duduville Campus, Nairobi, Kenya.
Biology. This species is very widespread and inhabits various zones from Lowlands (19 m a. s. l) to highlands (2672 m a. s. l). Adults occur throughout the year, commonly in and close to human settlements where they hide under and between stones, waste places, logs, dry grasses or leaves and in cracks (crevices) in the earth during daytime especially in open degraded forest landscapes. It is also found in grassland habitats. Although, preliminary studies have shown that the species is easy to breed in captivity, the biology of this species is largely unknown. Males are territorial and will fight off other males but allow any number of females to coexist in the same shelter. Females lay their eggs into humid soil and the pinhead crickets hatch in 13–14 days at 28±1°C, 60±5% RH and a photoperiod of L12: D12.
Remark. This species has been reared for three years in the research facility at the International Centre of Insect Physiology and Ecology ( icipe ), Duduville Campus, Nairobi, Kenya. It has been demonstrated through several research activities that it is a very promising species for mass rearing for food and feed. The best rearing conditions have been extensively tested and will be published in different journals elsewhere (Magara et al submitted).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |