Reliquantha, Roháček, 2013
|
publication ID |
https://doi.org/ 10.5281/zenodo.5740784 |
|
publication LSID |
lsid:zoobank.org:pub:17F1D510-AA62-4279-B592-2768D9B5D24E |
|
persistent identifier |
https://treatment.plazi.org/id/AB613866-4E1F-FFA2-4450-FBC8E82DFEFB |
|
treatment provided by |
Marcus |
|
scientific name |
Reliquantha |
| status |
gen. nov. |
Reliquantha View in CoL gen. nov.
Type species. Reliquantha variipes View in CoL sp. nov., hereby designated.
Diagnosis. (1) Head distinctly higher than long. (2) Eye large, broadly oval, with longest diameter subvertical. (3) Frons moderately broad; frontal triangle long, largely (including ocellar triangle) microtomentose. (4) Ocellar triangle elevated and ocelli relatively large. (5) Frontal lunule small but distinct. (6) Occiput dorsally distinctly concave. (7) Antenna geniculate between pedicel and 1st flagellomere, the latter strongly compressed laterally. (8) Arista very short-ciliate. (9) Palpus whitish, small and slightly clavate, with only 1 (subapical) seta longer. Cephalic chaetotaxy: (10) pvt relatively short, convergent; (11) vte and oc long, vti somewhat shorter; (12) oc normal, not erect; (13) 3 ors, anterior distinctly shorter than others; 1 microsetula in front of anterior ors; (14) several medial microsetulae in front of frontal triangle; (15) postocular setulae short, in single row; (16) vi relatively long, subvibrissa well developed; (17) peristomal setulae small and sparse.
(18) Thorax very slightly narrower than head, more or less shining, despite some microtomentum. (19) Scutellum strongly convex, postscutellum well developed. Thoracic chaetotaxy: (20) 1 hu, 2 npl (anterior distinctly longer); (21) 1 moderately long sa, 1 longer pa; (22) 1 distinct moderately long prs; (23) 2 postsutural dc, both relatively long; (24) ac microsetae not very numerous but in 4 rows, ending slightly beyond level of anterior dc; (25) 2 sc (apical longest of thoracic setae and strong, laterobasal short); (26) 1 minute upcurved ppl; (27) 2 relatively long stpl (anterior slightly shorter) plus 1 microseta in dorsal half of sternopleuron; only 2 setae on its ventral corner. (28) Femora distinctly variegated (yellow and brown); (29) tibiae darkened in the middle, yellow on apices; (30) f 1 without ctenidial spine; (31) t 2 with distinct but short ventroapical seta and 3 adjoining thickened setulae; (32) male f 3 with posteroventral row of short and thick setae, 4–5 distal of which shortened and thickened. (33) Wing long, moderately narrow; (34) wing membrane unicolourous. (35) C with inconspicuous thicker setulae among fine hairs on Cs 2; (36) R 2+3 long, bent parallel to C, ending slightly farther from apex of R 4+5 than M; (37) R 4+5 very slightly bent; (38) cell dm moderately long and narrow; cross-vein r-m situated in the middle of dm cell. (39) CuA 1 ending near, A 1 far from wing margin. (40) Anal lobe and alula well developed, relatively broad.
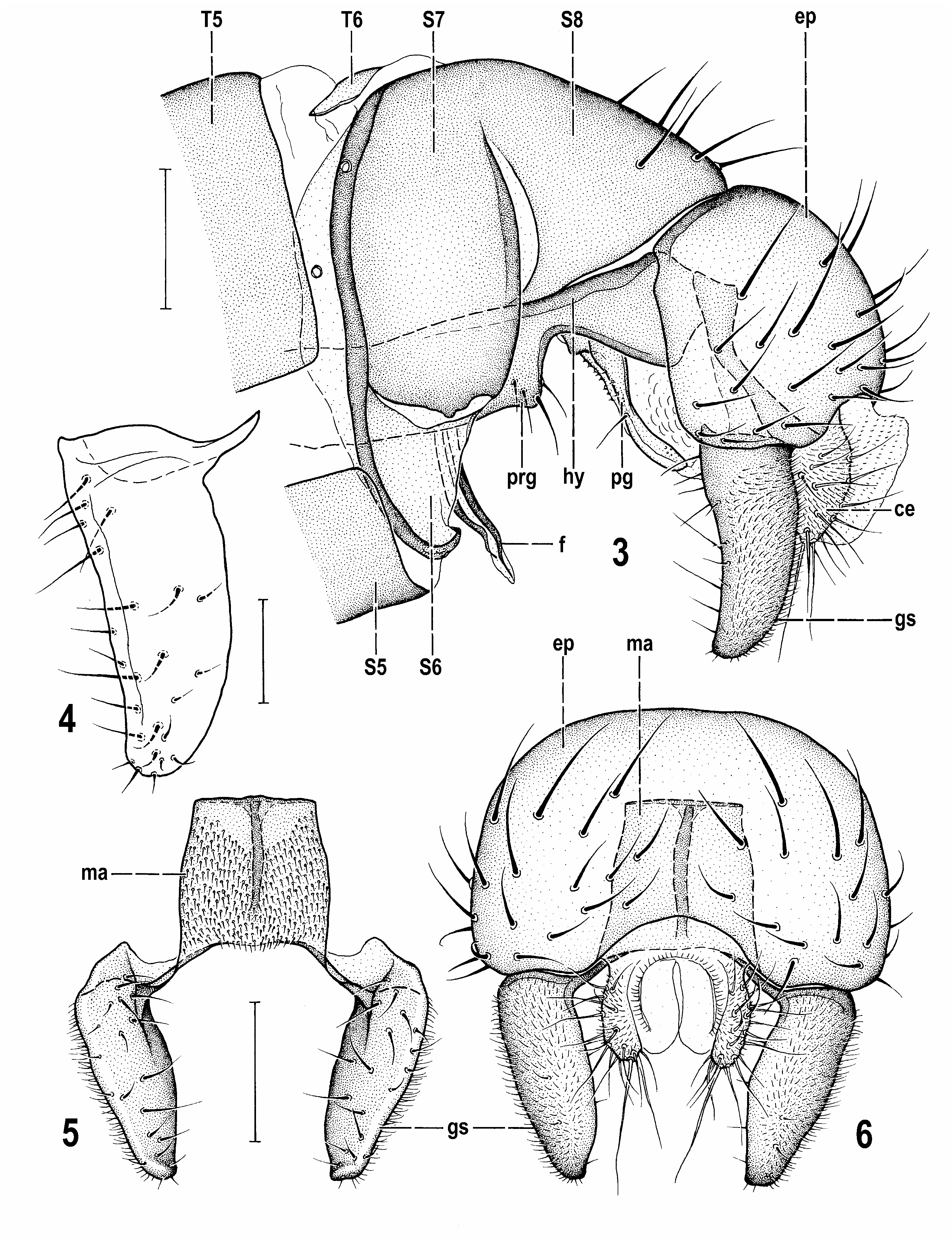
Male abdomen. (41) T1 separate from T2, at least dorsally; (42) T2–T5 large and broad. (43) S2–S5 becoming wider posteriorly, all brown. Male postabdomen: (44) T6 short, transverse, bare and well sclerotized although paler-pigmented than S6–S8. (45) S6, S7 and S8 partly coalesced together but their borders distinct. (46) S6 short, strongly asymmetrical, band-like tapered on both sides, with longest middle part separated horizontally from S7. (47) S6 and S7 entirely bare, both with usual dark marginal ledge. (48) S8 relatively long, less asymmetrical, more setose and situated dorsally.
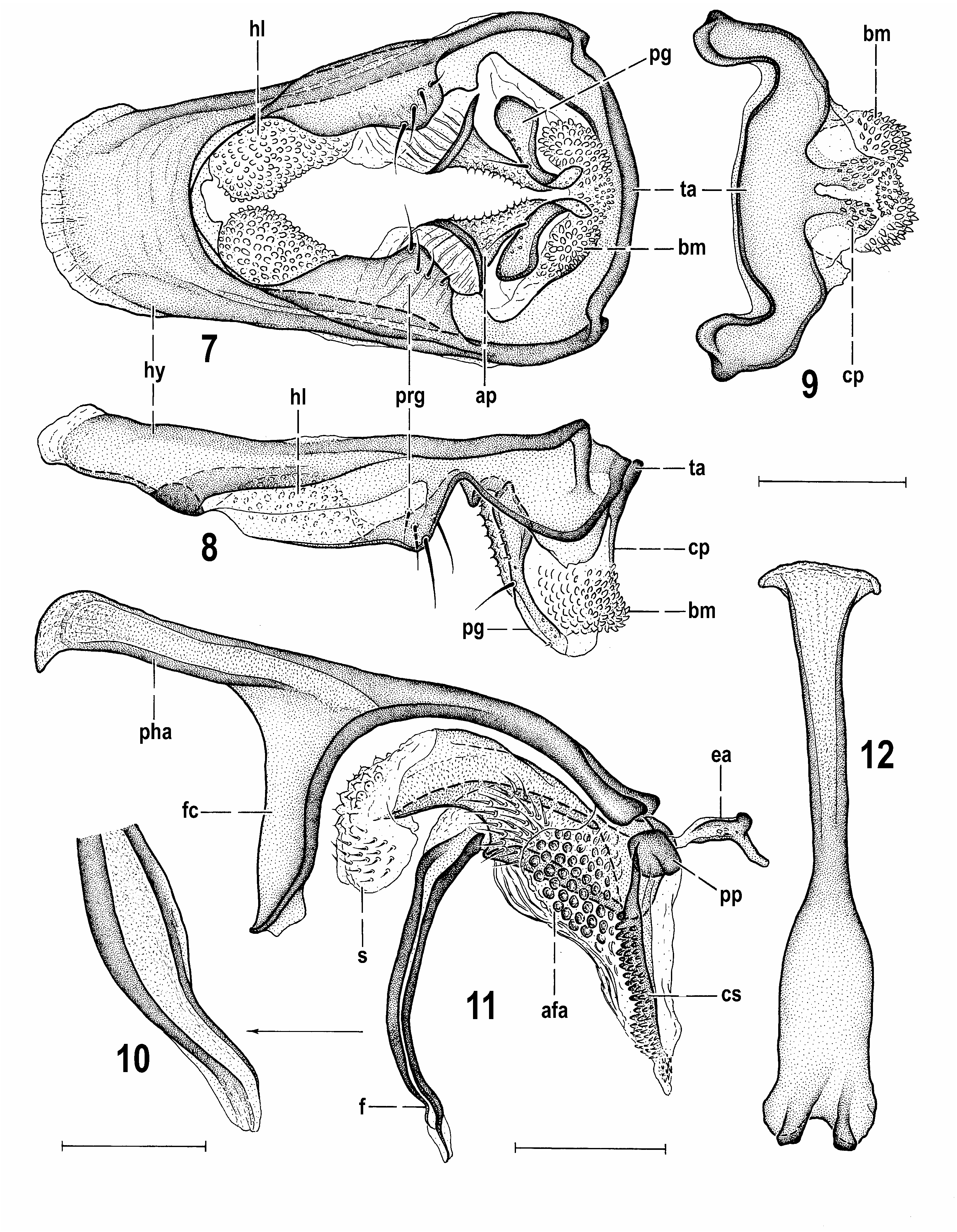
Male genitalia. (49) Epandrium of moderate size, setose, without particularly long setae. (50) Anal fissure very reduced, low. (51) Medandrium high, almost oblong, its anterior surface covered with dense short setulae. (52) Cercus pale-pigmented, laterally somewhat compressed. (53) Gonostylus darker than epandrium, simply elongate, with rounded apex and external surface largely covered by micropubescence. (54) Hypandrium with anterior flat lobes not projecting dorsally, weakly sclerotized but distinctively tuberculate; (55) transandrium robust, with short, flat and forked caudal process. (56) Pregonite relatively large, anteriorly flat and fused to hypandrium, posteriorly angular and separated by deep ventral notch and with only one (posterior) group of setae. (57) Postgonite slender, long, slightly bent, with some microsetulae in addition to usual anterior seta. (58) Anterior to postgonite there is an additional sclerotized projection ( Fig. 7 View Figs 7–12 , ap) covered with spinulose tubercles. (59) Phallapodeme with laterally dilated and flattened and shortly forked basal part. (60) Phallophore short, rather simple, including ventral part; (61) distiphallus composed of distally membranous saccus and slender sclerotized filum. (62) Saccus with relatively small membranous part, distinct basal and internal sclerites, basally with spine-like setae on left side, distally with tubercles and short setae in membrane; (63) filum formed by 2 long, dark, band-like sclerites terminating in narrowed membranous apex. (64) Aedeagal part of folding apparatus attached to base of aedeagus and phallapodeme and provided with dense pale rounded tubercles combined with dark striae. (65) Connecting sclerite strongly sclerotized, dark, and armed with dense blunt spines. (66) Basal membrane below caudal process with dense tuberculiform or short spines. (67) Ejacapodeme of moderate size, with subterminal digitiform process, on very short duct.
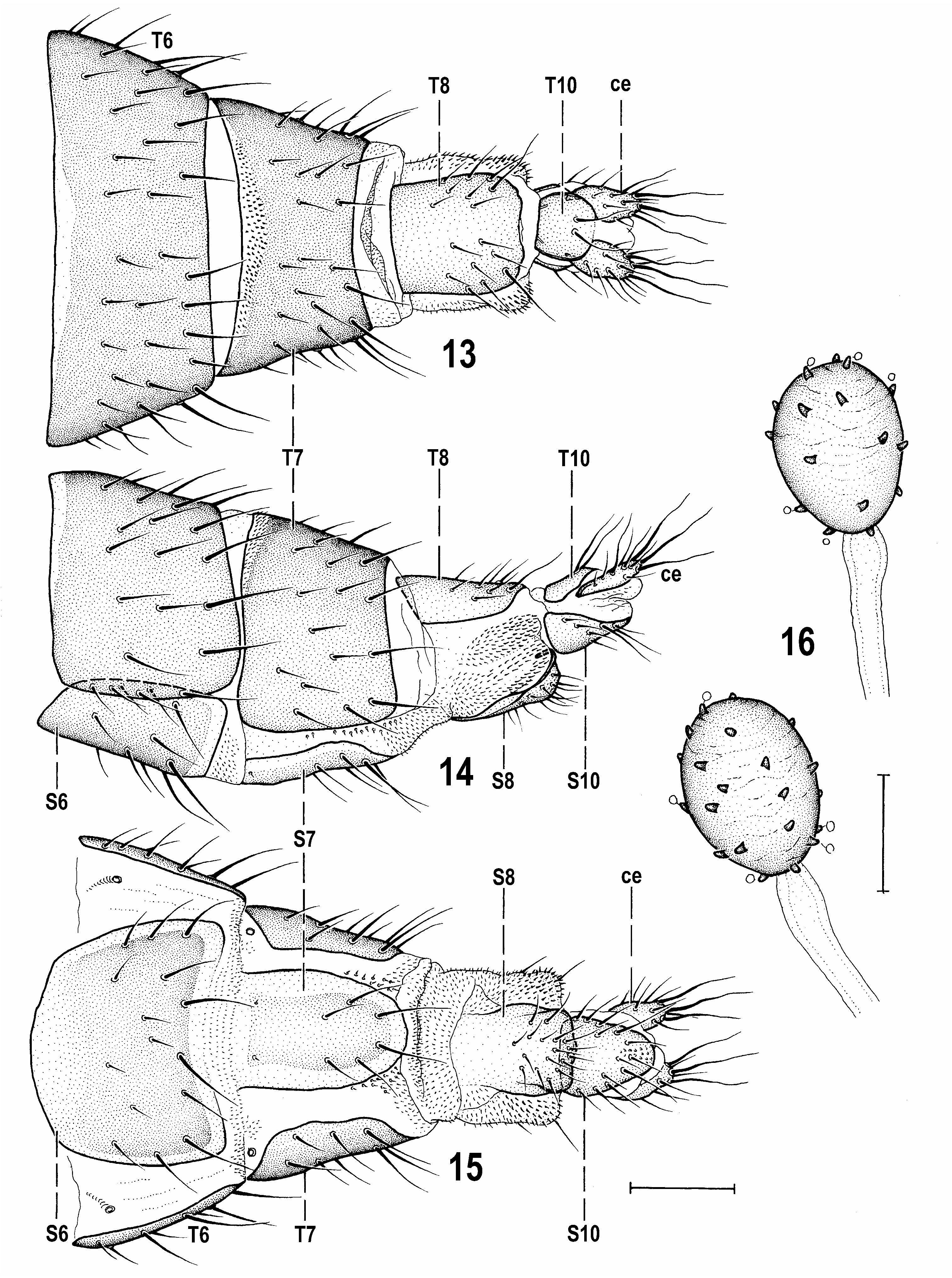
(68) Female abdomen relatively shining, with broader terga (T3–T5) and slightly narrower sterna (S2–S5). (69) Postabdomen relatively long, caudally gradually tapered, telescopically retractable from 7th segment, with less sclerotized and paler S7, T8, S8, T10 and S10. (70) T6 large, broad, also S6 relatively large (wider than S5). (71) T7 and S7 disparate. (72) S7 of elongate ligulate shape, pale, lacking micropubescence and with reduced setosity. (73) Membrane between T7 and S7 narrow and provided posteriorly with distinctive microsetae. (74) T8 simple, somewhat convex and without micropubescence; (75) S8 narrow, elongate, undivided, devoid of micropubescence and its posterior part dorsally curved and somewhat invaginated into 8th segment. (76) Internal structures of female genital chamber (uterus) reduced, with paired plates absent and with only (77) one rather small, somewhat transverse pale-pigmented annular sclerite. (78) Ventral receptacle short, on broad short duct, unpigmented, with plain surface and distally attenuating as a slender projection. (79) Accessory glands small, on slightly dilated plain ducts. (80) Spermathecae (1+1) small, simply ovoid, with scattered short and blunt spines; ducts very short, without distal collar. (81) T10 small, somewhat rounded, bare except for 1 pair of dorsal setae; (82) S10 elongately triangular, with long marginal setae but without micropubescence. (83) Cercus rather short, dorsoventrally flattened, without micropubescence, with a number of fine setae, 3 on apex markedly longer.
Discussion. The new genus clearly belongs to the subfamily Anthomyzinae (for its concept and diagnosis see ROHÁČEK 1998, 2006) and is characterized by a unique combination of plesiomorphic and derived, i.e. (supposedly) apomorphic characters. This fact proved to be the main stumbling block in uncovering the relationships of Reliquantha gen. nov., particularly when a number of ‘apomorphies’ recognised were found to be shared with various, apparently distantly related, genera. In addition, a more detailed comparison with the fossil genera Grimalantha Roháček, 1998 and Lacrimyza Roháček, 2013 unfortunately cannot be made because internal structures of the postabdomen and genitalia are largely unknown in these extinct groups of the subfamily.
Reliquantha gen. nov. is best diagnosed by the following combination of characters (those apomorphic are marked as ‘A’ and those unique as ‘U’ in parentheses, numbering of characters is the same as in the above diagnosis): (4) ocellar triangle elevated and ocelli relatively large; (11) 3 ors; (27) only 2 setae on ventral corner of sternopleuron (A); (28) femora variegated with brown and yellow (A); (30) f 1 without ctenidial spine (A); (32) male f 3 with posteroventral row of short and thick setae (A); (47) S6 and S7 entirely bare (A); (50) anal fissure of epandrium very reduced, low (A); (51) medandrium with anterior surface covered with dense short setulae (U); (54) hypandrium with distinctly tuberculate anterior lobes (A); (56) pregonite separated by posterior ventral notch and with only one (posterior) group of setae (U); (58) internal genitalia with an additional sclerotized projection anterior to postgonite (U); (59) phallapodeme with laterally dilated and flattened basal part (A); (62) saccus basally with spine-like setae on left side (A); (63) filum formed by 2 long, dark, band-like sclerites; (65) connecting sclerite strongly sclerotized and tuberculate (A); (72) female S7 elongate, lacking micropubescence (A); (75) S8 undivided, without micropubescence, with posterior part invaginated into 8th segment (?A); (77) only annular sclerite present in the female genital chamber (A); (78) ventral receptacle short, unpigmented, with plain surface and slender terminal projection (?A); (80) spermathecae on very short ducts (A); (83) female cercus without micropubescence and with 3 long setae on apex (U).
The unique (U) characters (= hitherto unknown in other representatives of Anthomyzidae ) are the most diagnostic. However, at least some of them may not necessarily be apomorphic. For example, the dense fine setosity of the medandrium (51, Fig. 5 View Figs 3–6 ) may well be an ancestral (hence plesiomorphic) character that was lost in other recent lineages of Anthomyzidae , or it could reappear as an atavism only in Reliquantha gen. nov. and, if so, it should be treated as apomorphic. On the other hand, the posteriorly notched pregonite (56, Fig. 8 View Figs 7–12 ), the additional paired sclerite in the male internal genitalia (58, Fig. 7 View Figs 7–12 ) and the female cercus lacking micropubescence and terminating in 3 long setae (83, Fig. 13 View Figs 13–16 ) can be considered apomorphic, the latter being unknown also in all fossil genera of Anthomyzidae (cf. ROHÁČEK 1998, 2013).
Noteworthy are some of the above derived (apomorphic, A) characters which are, however, shared with some extant or ancient genera. The reduced number of ventral setae on the sternopleuron (27) is only shared with one species of the fossil genus Lacrimyza ( L. lacrimosa Roháček, 2013 ); both known species of Lacrimyza also possess the greatly reduced anal fissure of the epandrium (50); these two characters could thus indicate a closer relationship of the latter genus with Reliquantha gen. nov. Moreover, Lacrimyza has the femora and tibiae dark with only the apices lightened (thus resembling the variegated legs of Reliquantha but more similar to those of Fungomyza Roháček, 1999 ) and f 1 lacking the ctenidial spine (as in Reliquantha and a number of other recent and fossil genera, including Grimalantha and some Protanthomyza species ). The ctenidial spine on f 1 (a ground plan character of Anthomyzidae ) is considered to be lost many times in various clades of Anthomyzidae (cf. ROHÁČEK 2009, 2013) and, consequently, its loss (30, Fig. 20 View Figs 17–22 ) cannot directly demonstrate the relationships of these taxa. The same is true for the male femora armed with short and thickened setae (32, Fig. 22 View Figs 17–22 ), an apomorphic structure which evolved independently as a consequence of behaviour during copulation in the majority of lineages including the fossil Lacrimyza (cf. ROHÁČEK 2013: 467).
The completely bare male S6 and S7 (47, Fig. 3 View Figs 3–6 ) is a rather unusual feature; normally both these sclerites bear 1–3 setae, sometimes one of them (more often S6) can be bare (e.g. in Quametopia Roháček & Barber, 2011 , see ROHÁČEK & BARBER 2011) and the setosity of these sclerites can be variably developed even within one genus ( ROHÁČEK & BARBER 2005). The tuberculate anterior lobes of the hypandrium (54, Fig. 7 View Figs 7–12 , hl) also are not a unique apomorphy of Reliquantha because similarly structured lobes also occur in Paranthomyza Czerny, 1902 although they are differently shaped and dorsally projecting in the latter genus ( ROHÁČEK 2006: Fig. 485). The setose left basal side of the saccus (52, Fig. 11 View Figs 7–12 ) also seems to be a distinct apomorphy, but a similar armature is known in Carexomyza Roháček, 2009 (see ROHÁČEK 2006: Fig. 502), Amygdalops Lamb, 1914 (cf. ROHÁČEK 2004) and, much finer setosity can also be seen in Paranthomyza (cf. ROHÁČEK 2006: Fig. 483).
The heavily sclerotized and tuberculate connecting sclerite (65, Fig. 11 View Figs 7–12 , cs) is another distinctive apomorphic feature of Reliquantha because in the majority of (extant) Anthomyzidae this structure is weakly developed and pale pigmented (although often spinulose). However, a well-sclerotized and spinose connecting sclerite occurs also in some Anthomyza species (e.g. A. dissors Collin, 1944 , see ROHÁČEK 2006: Fig. 119) or in both Epischnomyia species , but in the latter case it is markedly shortened ( ROHÁČEK 2009: Fig. 113). Very diagnostic of Reliquantha seem to be the female postabdominal sclerites (S7, T8, S8, T10, S10) that are devoid of micropubescence (72, 75, Figs 13–15 View Figs 13–16 ). Such extensive reduction of micropubescence is surely apomorphic (in other known genera only some of these sclerites, usually T10 and/or S10, are not pubescent). The micropubescence is even absent on the cerci which should be considered a unique apomorphy of the new genus (see above). Also the female S8 of Reliquantha is unusual (75, Fig. 15 View Figs 13–16 ), of (plesiomorphic) undivided shape, but with the posterior part bent dorsally, recurved and partly invaginated ( Fig. 17 View Figs 17–22 ) into the 8th segments (as in some taxa of the Anthomyza clade where, however, S8 is longitudinally divided). The polarity of this character is difficult to determine because there are several possibilities for how it could have evolved. The same holds for the short, unsclerotized and terminally projecting ventral receptacle (78, Fig. 18 View Figs 17–22 ) being hardly comparable to that of other known (extant) genera of Anthomyzidae – only the unrelated Amygdalops species have a short and membranous ventral receptacle (see ROHÁČEK 2004, 2008) somewhat resembling that of Reliquantha . The reduction of internal sclerotization of the female genital chamber with paired sclerites absent and only the annular sclerite preserved (77, Fig. 19 View Figs 17–22 ) seems to be also apomorphic but a similar situation occurs as a homoplasy in some other genera, e.g. the unrelated Anagnota Becker, 1902 and Santhomyza Roháček, 1984 (the annular sclerite is also absent in the latter genus, cf. ROHÁČEK 2006). Spermathecae of Reliquantha are worthy of discussion. Their simple ovoid shape is undoubtedly plesiomorphic but the short spermathecal ducts (80, Fig. 17 View Figs 17–22 ) are considered apomorphic and, apart from Reliquantha , were found also in species of Fungomyza, Arganthomyza Roháček, 2009 (see ROHÁČEK & BARBER 2013) and in Receptrixa Roháček, 2006 . The small spermathecae of the latter genus (cf. ROHÁČEK 2006: Fig. 518) are somewhat similar to those of Reliquantha but otherwise these groups are very dissimilar. It should be noted that somewhat less shortened spermathecal ducts occur in Carexomyza and other genera, too.
Generally the plesiomorphic features distinctly predominate in the new genus, viz. complete chaetotaxy of head (including 3 ors and subvibrissa) and thorax (with all macrosetae present), large eye with subvertical diameter, rather long frontal triangle, unicolourous wing with venation unmodified, C with spinulae small but present, pregenital sterna relatively large, gonostylus simply shaped, filum formed by 2 ribbon-shaped sclerites ( Fig. 11 View Figs 7–12 ), female S7 disparate, simple ovoid spermathecae etc. Most of these characters (if known) are shared with the fossil Lacrimyza , including the distinctly elevated frontal triangle and relatively large ocelli (4). Besides Lacrimyza there are only a few genera of Anthomyzidae where these plesiomorphies (and, particularly, 3 ors) are present, as discussed under the latter genus by ROHÁČEK (2013): the fossil Grimalantha from Dominican amber, and extant Fungomyza , Santhomyza and Zealantha Roháček, 2007 . However, Grimalantha , Santhomyza and Zealantha differ significantly from Reliquantha in general habitus in addition to strong dissimilarities in structures of the male and female genitalia. Fungomyza , on the contrary, closely resembles the new genus in general appearance, including the dark and relatively shining body, structures of the frons, and the variegated femora (cf. ROHÁČEK & BARBER 2004, ROHÁČEK 2009). In spite of all this, R. variipes cannot be affiliated with Fungomyza because of the absence of most of the synapomorphies of this genus (e.g. subvibrissa absent, male S8 prolonged, medandrium narrowed dorsally, phallophore with ventral process, saccus with adpressed surface spinulae, S8 longitudinally divided and invaginated into 8th segment, female annular (= looped) sclerite reduced, spermathecae with terminal invagination, etc., see ROHÁČEK 2009). On the other hand, the new genus surprisingly shares with Fungomyza some apomorphies, which probably evolved independently as homoplasies: the variegated femora (mentioned above), the caudal process of the transandrium forked and the spermathecal ducts markedly shortened.
Based on the above comparison it is suggested that the closest relative of Reliquantha gen. nov. is possibly the Eocene genus Lacrimyza . The apomorphic features shared by these two genera (discussed above) are few in number but others may occur in the internal structures of the male and female genitalia which cannot be studied in detail in amber fossils. However, the genus Lacrimyza is diagnosed by several apomorphies lacking in Reliquantha , viz. oc arising close to each other and peculiarly erect (see ROHÁČEK 2013: Figs 14A, 14E View Figs 13–16 ); ac microsetae very sparse and with two medial rows situated very close to each other; also 2 stpl arising unusually close to each other; male femora (f 3 in particular) thickened; epandrium with 1 pair of strong setae; male cercus reduced, small; aedeagal part of folding apparatus densely pubescent; female T7 dorsomedially pale-pigmented; female S7 only marginally dark. In addition, Lacrimyza differs in having the wings somewhat shortened and narrowed with rm situated more proximally, very reduced peristomal setulae, prolonged male S8, very short and transverse female S8, with the latter sclerite, S10 and cerci distinctly micropubescent, etc. All these characters clearly prevent inclusion of R. variipes within the genus Lacrimyza .
The genus Reliquantha can be separated from other extant Palaearctic genera of Anthomyzidae using the key in ROHÁČEK (2009) where it runs to couplet 5 (see ROHÁČEK 2009: 13) which is to be modified and supplemented as follows:
5(4) Arista short-ciliate; head higher than long; 3 strong ors. ......................................... 5a
– Arista pectinate or long-plumose; head longer than high; only 2 strong ors. ........... 6
5a(5) Thorax unicolourous dark brown; legs with femora brown and yellow variegated; 2 dc; male f 3 with a row of short thick posteroventral setae ( Fig. 22 View Figs 17–22 ); cross-vein r-m situated in the middle of dm cell ( Fig. 1 View Figs 1–2 ); anal fissure of epandrium very reduced, low ( Fig. 6 View Figs 3–6 ); pregonite simple, without projections ( Fig. 8 View Figs 7–12 ). Female S10 and cerci without micropubescence ( Fig. 15 View Figs 13–16 ); cerci short and robust ( Fig. 13 View Figs 13–16 ). ....................... ................................................................................................. Reliquantha gen. nov.
– Thorax yellow to pale brown, with dark longitudinal bands on mesonotum and/or pleuron; legs with femora unicolourous yellow; 3 dc (anterior short); male f 3 with uniform fine setosity; r-m situated markedly distal to the middle of dm cell ( ROHÁČEK 2006: Figs 535, 536); anal fissure of epandrium well developed, high and narrow ( ROHÁČEK 2006: Figs 524, 538); pregonite with slender rod-like projection and a shorter process ( ROHÁČEK 2006: Figs 527, 539). Female S10 and cerci distinctly micropubescent ( ROHÁČEK 2006: Fig. 531); cerci long and slender ( ROHÁČEK 2006: Fig. 543). ............ ......................................................................................... Santhomyza Roháček, 1984
Etymology. The name of the genus is constructed from the abbreviated conjunction of reliqu [us] + anth [omyz] a because the taxon is probably a relic of an otherwise extinct clade of Anthomyzidae .
Species included. Reliquantha variipes sp. nov. (described below) from Great Britain.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.