Sisyra panama Parfin & Gurney, 1956
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4802.2.10 |
|
publication LSID |
lsid:zoobank.org:pub:3B340088-6420-40CE-8740-9E9239B8B206 |
|
DOI |
https://doi.org/10.5281/zenodo.10564344 |
|
persistent identifier |
https://treatment.plazi.org/id/A62B87AF-FF87-C85B-3BD0-8AEE175B73BE |
|
treatment provided by |
Plazi |
|
scientific name |
Sisyra panama Parfin & Gurney, 1956 |
| status |
|
Sisyra panama Parfin & Gurney, 1956 View in CoL
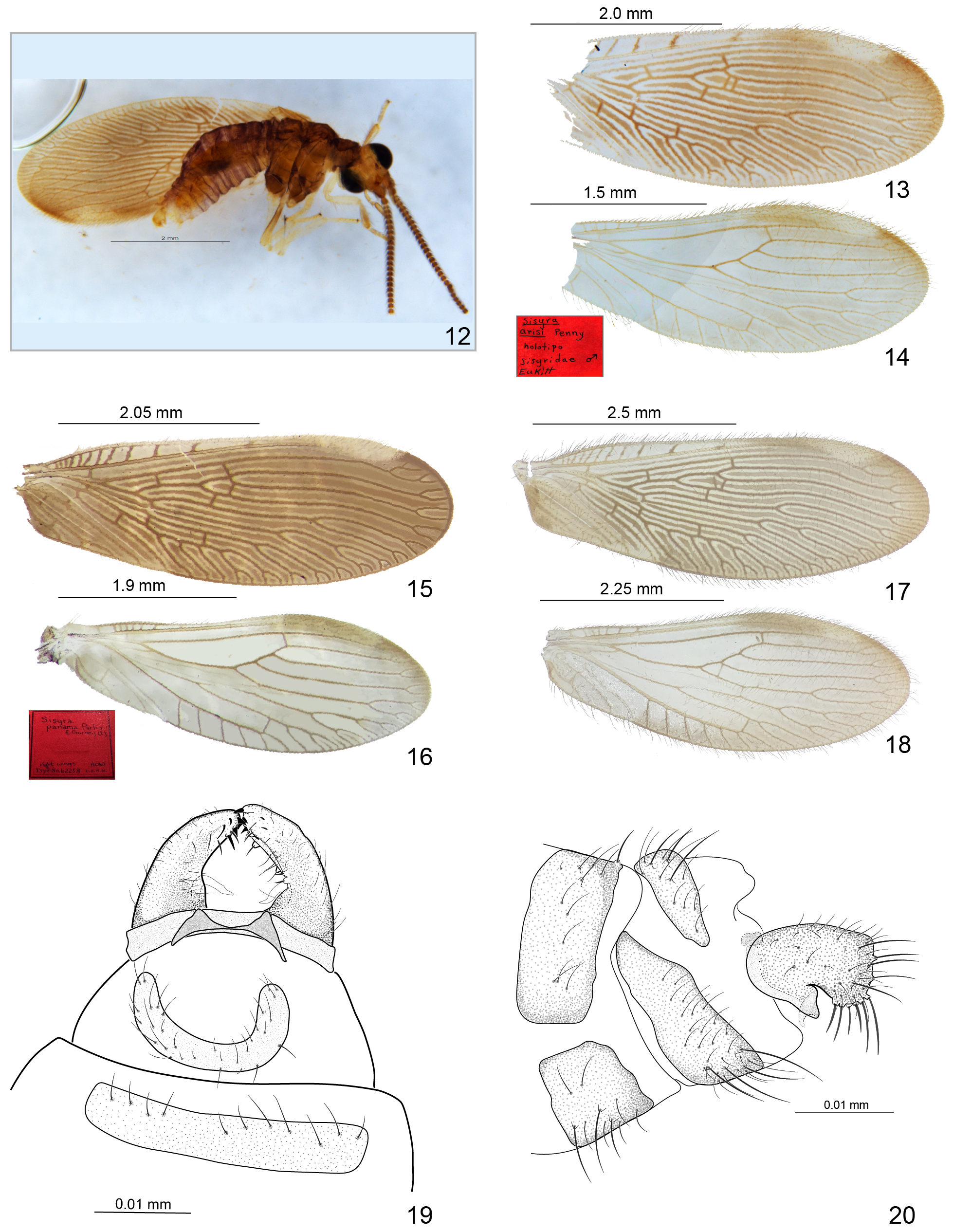
( Figs 1, 3–4, 6 View FIGURES 1–7 , 12–20 View FIGURES 12–20 )
Sisyra panama Parfin & Gurney, 1956:474 View in CoL [description]; Penny 1981:166 [record]; Flint 2006:281 [record]; Hamada et al. 2014:281 [bionomic notes and record]; Bowles 2015:4 [record]; Assmar & Salles 2017:87 [record].
Sisyra ariasi Penny 1981:168 View in CoL [description]. NEW SYNONYMY.
Remarks. Both holotypes of S. ariasi ( Figs 12–14 View FIGURES 12–20 ) and S. panama ( Figs 15–16 View FIGURES 12–20 ) were examined and no significant differences were found between the two in the lateral view. Other views are not possible to analyze because the male holotype of S. ariasi is fixed on slide and the genitalia is not cleared ( Fig. 12 View FIGURES 12–20 ).
Penny (1981) affirmed that S. panama and S. ariasi have two distinctive crossveins between “R3 and R4+5” (RP2 and RP3) ( Fig. 13 View FIGURES 12–20 ). However, the additional crossvein found between these veins in the right forewing of S. ariasi holotype is a modification exclusive to this wing, and this additional crossvein is not present in the left forewing of the same specimen ( Fig. 12 View FIGURES 12–20 ). A male specimen of S. panama , collected in Salvador, Bahia, presented this same modification in the left forewing and in the hind wing ( Figs 17–18 View FIGURES 12–20 ). Besides, S. panama has only one crossvein between RP2 and RP3, according to Parfin & Gurney (1956).
Regarding the male genitalia, both illustrations and descriptions of these two species are not detailed. Parfin & Gurney (1956) described the male gonocoxite 9 of S. panama as “stout”, “with long setae and distal inner teeth” and “caliper-like”. In addition, the authors described the basal portion of the “paramere” (gonostyle 11) as triangular. The illustration shows the postero-dorsal margin elongated in relation to the ventral one ( Parfin & Gurney 1956). In contrast, according to Penny (1981) S. panama male gonocoxite 9 is “short, quadrangular, with several small teeth at the apex”, lacking the “ventral thumb-like lobe”, present in S. ariasi . The last cited characteristic would be the only difference between the two species in the male genitalia ( Penny 1981).
Comparing the male genitalia, there are no significant differences between S. ariasi and S. panama ( Figs 12, 19–20 View FIGURES 12–20 ). Sisyra panama has a ventral “thumb-like lobe”, which is the gonostyle 11, called “parameres” by Parfin & Gurney (1956). In addition, the apex of the male gonocoxite 9 of both species are truncate, although Penny (1981) illustrated the gonocoxite 9 of S. ariasi with the apex rounded compared to slightly rhomboidal in S. panama ( Figs 12, 20 View FIGURES 12–20 ). It is possible that the apparent differences are due to the angle at which the illustrations were made. As can be seen in Penny (1981), the genitalia are drawn in an oblique position relative to S. panama . Due to the absence of distinctive characters between the two species, Sisyra ariasi Penny, 1981 , NEW SYNONYMY, is now attributed to S. panama Parfin & Gurney, 1956 . Description and illustration of the male genitalia of S. panama are provided here ( Figs 19–20 View FIGURES 12–20 ).
This is the first record of Sisyra panama and the family Sisyridae from Caatinga biome in Brazil. The specimens of S. panama were collected in two highland areas, Serra de Ibiapaba, Ubajara (Ceará state) and Caeté-Açú, Palmeiras (Bahia state), where orographic rainfall is common, thus enabling the existence of wet forest islands surrounded by Caatinga vegetation ( Queiroz et al. 2017). Serra de Ibiapaba is characterized as a “Brejo de Altitude”, and it is floristically similar to the Amazonia biome ( Takiya et al. 2016; Queiroz et al. 2017). Caeté-Açú is a district of Palmeiras city, located in the Chapada Diamantina Complex, which has phytophysiognomies similar to the Atlantic rainforest and it contains the only Rupestrian Grassland vegetation of the Caatinga ( Queiroz et al. 2017).
Male genitalia ( Figs 19–20 View FIGURES 12–20 ). Ectoproct and sternite 9 transversal, with numerous long dentigerous-based setae and simple setae. Ectoproct larger dorsally, rounded margins. Sternite 9 long, quadrangular, posterior margin longer than anterior laterally. Gonocoxite 9 stout, quadrangular, truncate apex with inner distal teeth dorsally. Numerous long, large dentigerous-based setae on gonocoxite 9 posterior margin, mostly postero-ventrally, simple setae throughout. Gonostyle 11 triangular in lateral view, resembling a ventral “thumb”, apex acute, curving to midline.
Bionomic notes. Two adults were collected during different periods and observed alive, in order to analyze their behavior. A male and a female were observed, the first was kept alive for three days, and the last for approximately 14 days, being fed with a mix of water and honey, placed on cotton. Both individuals landed on the cotton to feed on the solution.
It was observed that the specimens mostly walked, instead of flying in the enclosure. While walking, they occasionally stopped, vibrated their maxillary palps, made a “back and forth” movement with the body, touched their abdomen on the surface or got really close to it, and then starts walking again (behavior also reported to S. nigra (Retzius, 1783) by Rupprecht (1995)). This vibration and abdominal movements have been reported for some Neuropteran families, including Sisyridae , as a conspecific communication and courtship behavior ( Henry 2005). The specimen emits the vibration that travel through the substrate and is received by the other nearby individuals through the subgenual organs in the legs ( Devetak & Amon 1997; Devetak 1998). In Sisyra , the use of the mandibles and the abdomen to produce vibration have been reported by Rupprecht (1995; 1997) and Henry (2005), respectively. Here, it was observed the maxillary palp was the body part that vibrated.
Related to the flight behavior, both specimens started flight from a vertical landing position by dropping itself from the surface and then flying. When it was in a horizontal position, a spiral flight was performed, until it reached a certain height. In both cases, their flight was without apparent pattern.
Material examined. BRAZIL: Bahia, Palmeiras, Caeté-Açú, Vale do Capão , rio do Batista , 30.vi–03.vii.2018, Pennsylvania white light trap, Calor , Assmar , Gomes , Miranda & Tavares, 1 female ( UFBA, N0033 ); Salvador, 19BC, córrego Cascão, 12°58’16” S 38°30’39” W, 19.viii.2014, white sheet, Gomes, V GoogleMaps .; Ferreira, E; Alves; Lima, 1 female ( UFBA, N0038 ); same data, except Alphaville I, Residencial Itaparica, L. Tavares’s house, 12°56’39” S 38°23’50.7” W, 01.viii.2019, Tavares, L., 1 male ( UFBA, N0044 ); same data, except 02.x.2019, Tavares, L. 1 female ( UFBA, N0048 ) GoogleMaps . Ceará, Ubajara, Serra do Ibiapaba, Parque Nacional de Ubajara , Murimbeca stream, 03°49’18.9” S 40°54’16.6” W, 868 m, 28.x.2011, white light pan trap, Gomes, V GoogleMaps & Duarte, T., 1 male ( UFBA, N0041 ) .
| V |
Royal British Columbia Museum - Herbarium |
| T |
Tavera, Department of Geology and Geophysics |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Sisyra panama Parfin & Gurney, 1956
| Assmar, Alice Carvalho & Calor, Adolfo Ricardo 2020 |
Sisyra ariasi
| Penny 1981: 168 |
Sisyra panama
| Parfin & Gurney 1956: 474 |