Cracticotaenia adelaidae, Mariaux & Georgiev, 2018
|
publication ID |
https://doi.org/10.5852/ejt.2018.440 |
|
publication LSID |
lsid:zoobank.org:pub:DB80A42B-5C53-455B-86A4-2003D6F03522 |
|
DOI |
https://doi.org/10.5281/zenodo.3846840 |
|
persistent identifier |
https://treatment.plazi.org/id/5F32D506-1E05-421F-B891-DDB9E89F4195 |
|
taxon LSID |
lsid:zoobank.org:act:5F32D506-1E05-421F-B891-DDB9E89F4195 |
|
treatment provided by |
Valdenar |
|
scientific name |
Cracticotaenia adelaidae |
| status |
sp. nov. |
Cracticotaenia adelaidae sp. nov.
urn:lsid:zoobank.org:act:
Figs 34–38 View Figs 34–38
Etymology
The species name is derived from the name of the City of Adelaide, where the type series was collected.
Material examined
Holotype
AUSTRALIA: South Australia, Adelaide , 34°54′ S, 138°36′ E, 27 Oct. 1983, David Shulz leg. (AHC 36516).
GoogleMapsParatypes
AUSTRALIA: 5 specs, same data as for holotype (AHC 36517–36525).
Additional material from the same host
AUSTRALIA: South Australia 3 specs, South Australia, Ashbourne, 35°18′ S, 138°36′ E, Dec. 1969, Michael Smyth and Lee leg. (AHC 29591); 2 specs, very fragmented, Coromandel Valley, 35°6′ S, 138°36′ E, 4 Apr. 1969, Michael Smyth leg. (AHC 29592); about 30 specs, Coorong, 36°2′ S, 139°33′ E, Oct. 1935, D. Brunswick leg. (AHC 230). – Victoria 2 specs, Torquay, 38°18′ S, 144°18′ E, May 1979, Karl Harrigan leg., from Strepera sp. ( Artamidae , Passeriformes ) (AHC 29596).
Other material
AUSTRALIA: from Strepera versicolor (Latham, 1801) : 6 specs and numerous fragments, no locality, 12 Dec. 1910 (AHC 533). – From Corcorax melanorhamphos (Vieillot, 1817) ( Corcoracidae , Passeriformes ): 4 specs, Queensland, Eidsvold, 25°22′ S, 151°5′ E, Sep. 1918, Thomas L. Bancroft leg. (AHC 531, AHC 29598, co-infection with a Dilepididae , see above); 12 specs, South Australia, Elwomple, 35°17′ S, 139°32′ E, 19 Oct. 1937, collector unknown (AHC 3368).
Other comparison material
Type material of Monopylidium fieldingi ( Maplestone & Southwell, 1923) , from Cracticus destructor [now C. torquatus ] in Queensland, Townsville (The Natural History Museum, London 1977.11.3.34, 2 slides).
Host
Gymnorhina tibicen (Latham, 1801) (syn. Cracticus tibicen ) ( Artamidae , Passeriformes ).
Intensity
Six specimens.
Description
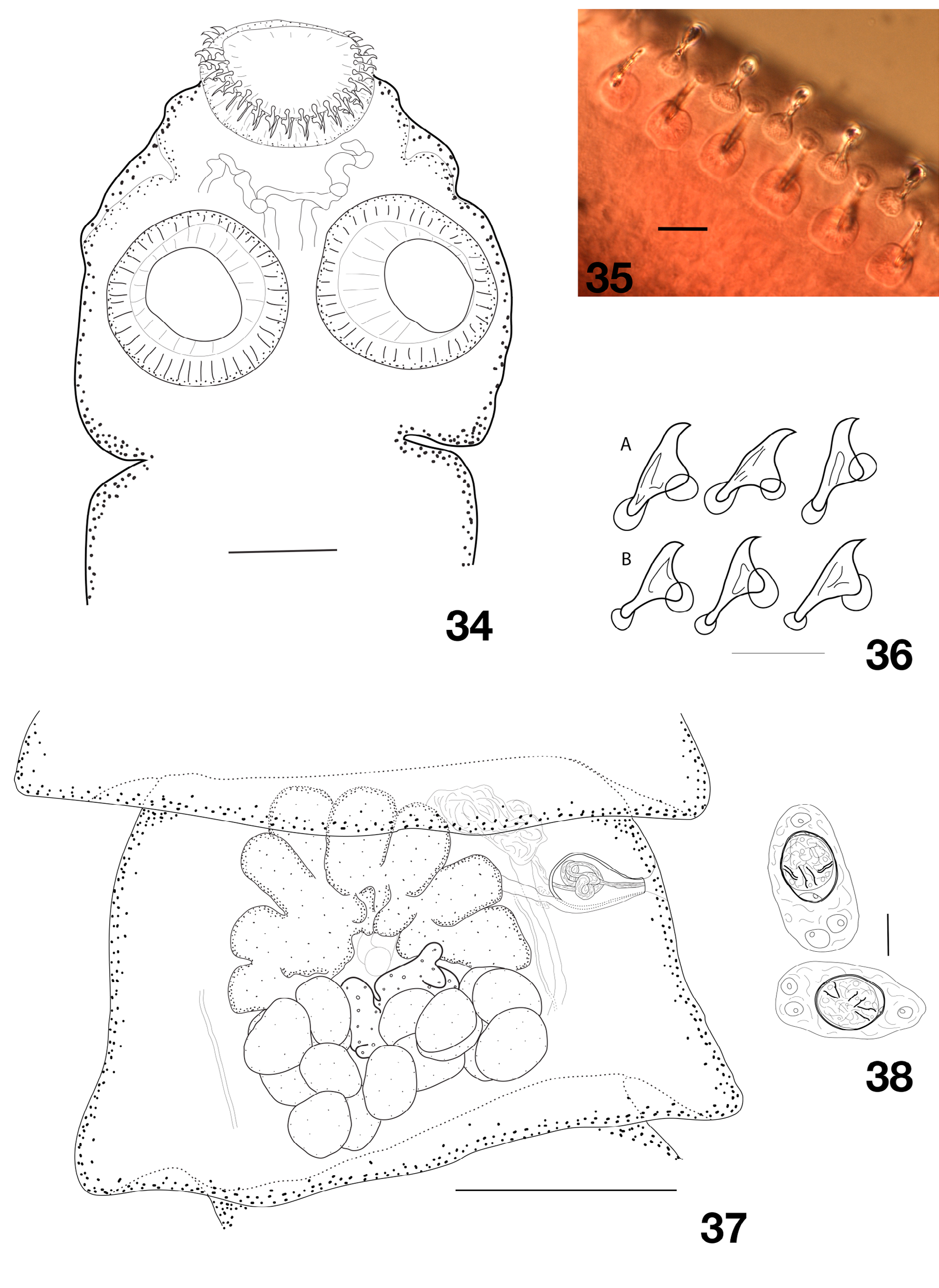
Body of medium size, with maximum length 35.5 mm and maximum width 1.3–2.12 mm (1.7, n = 6) at level of pregravid or early gravid proglottides. Most complete specimen consisting of 240 proglottides. Proglottides craspedote, wider than long, progressively becoming longer, up to slightly longer than wide when gravid. Scolex rounded, not markedly delineated from neck, with diameter 350–475 (407, n = 8), Suckers unarmed, muscular, rounded, 142–198 (169, n = 35) in diameter. Rostellum muscular, cushion-like 128–188 (149, n = 8) in diameter ( Fig. 34 View Figs 34–38 ). Rostellum bearing double crown of regularly arranged hooks, 52–61 (56, n = 8) in number. Hooks triangular, blade terminating with a short, curved tip. Handles and guards bearing well-developed epiphyses, more developed on guard. Epiphyses of hooks in posterior row clearly larger than those of hooks in anterior row ( Figs 35–36 View Figs 34–38 ). Anterior hooks 23.5–25 (24, n = 4), posterior hooks 21–22 (21.5, n = 5) long. Neck 192–257 (216, n = 5) wide; proglottization starting shortly behind neck. Genital pores unilateral (a single side-switch observed), opening at border of first third of proglottis length margin. Ventral osmoregulatory canals up to 60 wide, connected posteriorly by anastomosis in each proglottis. Dorsal osmoregulatory canals up to 12 wide. Genital ducts passing between osmoregulatory canals. Genital atrium small. Internal organs closely packed in short and wide proglottides, their shape and relative position clearly visible only in very relaxed or extended proglottides.
Testes 12–18 (14.5, n = 38) in number, in two or, rarely, three layers, in one continuous posterior field, slightly extending laterally up to level of anterior margin of vitellarium ( Fig. 37 View Figs 34–38 ). External vas deferens discrete, convoluted, in antero-poral part of median field, at distance from cirrus-sac. Cirrus-sac short, with strong walls, most often pear-shaped, sometimes more oval, 90–118 × 43–58 (103 × 51, n = 57), not always reaching poral longitudinal osmoregulatory canals, sometimes slightly overlapping them. Internal vas deferens forming several coils. Cirrus straight with well marked walls, often appearing refractory.
Vitellarium central, compact, often heart-shaped or slightly transversely elongated, variable in shape. Ovary fanlike when not contracted, with deep lobes filling most of anterior part of proglottides. Mehlis’ gland central and anterior to vitellarium, connected to seminal receptacle by wide and often clearly visible canal that may become almost as wide as seminal receptacle. Seminal receptacle oval to fusiform, remaining rather small, reaching up to 150 × 50, dorsal and between ovary wings or over poral wing, closely to proximal extremity of cirrus-sac. Vagina opens dorsally and slightly posteriorly to male pore, mostly straight, with faint sphincter ( Fig. 37 View Figs 34–38 ).
Uterus starts its development in late mature proglottides as a postero-central and dorsal mass, developing into a lateraly-extended multisaccular and reticulate cavity with well marked wall, then becoming saccate. Outer shell of eggs delicate, oval 62–73 × 33–38 (68 × 35, n = 11), becoming more circular and irregular in broken proglottides. Embryophore thin and regular, oval, 30–41 (35, n = 23) in diameter. Oncosphere subspherical to slightly elongate, 30–40 (35.5, n = 18) in diameter. Embryonic hooks: central 13–15.5 long (14.5, n = 14) long, lateral 11–13 (12, n = 17) long ( Fig. 38 View Figs 34–38 ).
Remarks
This material is very similar to Cracticotania fieldingi ( Maplestone & Southwell, 1923) , the only species know in the genus. Cracticotania fieldingi was described from Cracticus destructor (now C. torquatus , Artamidae ) in Queensland and attributed to Monopylidum by Maplestone & Southwell (1923). It was later placed in a newly erected monotypic genus by Spasskii (1966) and transferred to the Metadilepididae by Kornyushin & Georgiev (1994). Our material fits the most recent generic diagnosis of these authors with the notable exception that only a very faint vaginal sphincter is visible. Besides this character, it differs from the single known species in the genus by a smaller number of rostellar hooks (52–61 vs about 80), a slightly fewer testes (12–18 vs 16–21) and the differently-shaped and shorter cirrus-sac (globular vs pipette-like and 90–118 vs 130). It should, however, be noted that we examined the type material of C. fieldingi and the measurements provided by Maplestone & Southwell (1923) do not fully correspond to our observations. Although only two slides, mostly with gravid proglottides, are available, we noted a somewhat longer cirrus-sac (up to 180) and fewer testes (12–15). This, however, does not prevent the unambiguous separation of our material from C. fieldingi and we place it in a new species which we name Cracticotaenia adelaidae sp. nov.
The specimens found in Corcorax Lesson, 1831 fit the above description of Cracticotaenia adelaidae sp. nov. in Gymnorhina tibicen quite well for most metric and meristic characters that can be observed. The main observed differences are in slightly smaller suckers (123–147 vs 155–198) and possibly more rostellar hooks (62–66 vs 52–61), and the faint vaginal sphincter that could not be observed. Keeping in mind that these observations are based on a limited number of incomplete specimens, we consider for now that these small variations (which may be partially exaggerated due to the poor preservation state of our specimens) are likely to be intraspecific.
Although both belonging to the Corvides (see Jønsson et al. 2016), the hosts of C. adelaidae sp. nov. are not closely related and belong to clades that have probably diverged some 20 to 30 Ma ago ( Jønsson et al. 2016; Moyle et al. 2016). The Australian magpie ( G. tibicen ) and the white winged chough ( C. melanorhamphos ) have however similar diets and habitats ( Russell et al. 2017) as well as overlapping ranges (hosts reported in this study were captured a mere 100 km apart). This relative phylogenetic proximity and quite similar ecology probably largely explain the hosting of shared parasites.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |