Phoebe ornator ( Tippmann, 1960 )
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4250.4.4 |
|
publication LSID |
lsid:zoobank.org:pub:DBE44C83-90D5-44E5-BAAC-3512F0392E59 |
|
DOI |
https://doi.org/10.5281/zenodo.6028277 |
|
persistent identifier |
https://treatment.plazi.org/id/96588D12-9762-F04C-E3CB-4953FC35E119 |
|
treatment provided by |
Plazi |
|
scientific name |
Phoebe ornator ( Tippmann, 1960 ) |
| status |
|
Phoebe ornator ( Tippmann, 1960) View in CoL
( Figs. 1 View FIGURES 1 – 3 –6)
Adesmus ornator Tippmann, 1960: 204 View in CoL .
Phoebe ornator Martins & Galileo 1993: 113 View in CoL (comb. nov.); Monné, 2016: 773.
Description. Male. Head from brown to dark brown, except for reddish-brown ventral side. Mouthparts yellowish. Scape, pedicel and antennomeres III–IV from orange-brown to reddish brown; antennomeres V–XI dark brown. Prothorax usually dark brown, but sometimes lighter, mainly anteriorly on pronotum and ventrally. Mesosternum dark brown. Mesepimera and mesepisterna from light brown to dark brown. Metepisterna and metasternum from reddish brown to brown. Elytra with dorsal side dark brown, usually darker toward apex, but could be brown at base, gradually darker toward apex; sides mostly from reddish brown to brown. Legs yellowish, with part of tarsal claws brownish. Abdominal ventrites I–II usually brown or dark brown; ventrites III–V usually reddish brown (sometimes yellowish brown).
Head. Frons transverse, slightly oblique, and slightly convex with large horns at sides of lower eye lobes near antennal socket, distinctly concave from horns to just after upper-eye lobes. Frons, vertex, area behind eyes and genae (except for glabrous, narrow, apical region) with dense, white pubescence obscuring integument, except for three maculae of yellowish-brown (sometimes brown) pubescence (occasionally not visible based on position of head, or fused), placed close to prothoracic margin, not obscuring integument: one transverse on each side behind upper-eye lobes; another centrally, elliptical, with long, erect, sparse setae on frons. Cephalic horns narrowed toward acute apex, obliquely directed upward and not divergent toward sides from base to apex with white, dense pubescence on base and gradually becoming less dense toward apex; length variable, from slightly smaller than width of lower-eye lobe to about 1.5 times width of lower-eye lobe. Ventral side of head mostly glabrous, except for short, moderately sparse setae on submentum. Outer side of mandibles with dense, white pubescence interspersed with long, erect, sparse setae. Distance between upper-eye lobes 0.35 times length of scape; distance between lower-eye lobes in frontal view 0.75 times length of scape. Antennae (based on a single male) 2.8 times elytral length, reaching elytral apex at quarter of antennomere VI; antennal formula (ratio) based on antennomere III: scape = 0.59; pedicel = 0.10; IV = 0.76; V = 0.63; VI = 0.65; VII = 0.61; VIII = 0.59; IX = 0.58; X = 0.57; and XI = 0.61.
Thorax. Prothorax cylindrical and slightly wider than long. Pronotum with dense, white pubescence obscuring integument, except for five subrounded maculae with yellowish-brown pubescence not obscuring integument: one at each side of anterior half; one centrally at basal half; on each side of basal half (almost on lateral side of prothorax). Sides of prothorax with dense, white pubescence obscuring integument except for subcircular macula with yellowish-brown pubescence, not obscuring integument, placed close to anterior margin. Prosternum, prosternal process, most mesosternum and mesosternal process possesses yellowish-brown pubescence that does not obscure integument. Mesepimera, mesepisterna, metepisterna and sides of metasternum with dense, white pubescence obscuring integument; remaining surface of metasternum with yellowish pubescence that does not obscure integument.
Elytra. Dorsal side with dense, white pubescence obscuring integument except for following areas that featured yellowish pubescence (sometimes more brownish) that does not obscure integument (frequently these areas almost disappear because of white pubescence): one subcircular macula close to scutellum; one subcircular macula at basal sixth between suture and lateral carina; one subelliptical maculae near beginning of second quarter, close to suture (occasionally circular but not reaching suture); one subelliptical, usually oblique, macula between suture and lateral carina (sometimes more circular) placed slightly before middle; one elongate, close to suture at middle; one subcircular between suture and lateral carina placed proximal to beginning of distal third, frequently fused with transverse macula that reaches carina and, commonly, almost lateral margin; one transverse from suture to lateral margin near apex. Elytral carina with yellowish pubescence that does not obscure integument (sometimes almost glabrous). Vertical sides of elytra mostly with white, dense pubescence obscuring integument at least partially; narrow area close to margin with yellowish pubescence distinctly not obscuring integument from base to roughly distal quarter (this area wider on basal third). Elytral apex truncate, with short spine at outer angle.
Abdomen. Sides of ventrites I–IV featured white, dense pubescence obscuring integument (sometimes present only on ventrites I–II or ventrites III–IV on distal half or close to apex); remaining surface with yellowish pubescence distinctly not obscuring integument with long, erect, sparse setae.
Dimensions (mm), male. Total length (from apex of cephalic horn to elytral apex), 8.50–10.75; prothoracic length, 1.40–1.95; anterior prothoracic width, 1.60–2.05; basal prothoracic width, 1.50–1.95; humeral width, 2.10– 2.70; elytral length, 5.85–7.20.
Intraspecific variability. Regarding females, the pubescence on the head and vertical sides of the elytra was completely brownish black (Figs 4, 5) or, at most, had white pubescence only on the vertex, behind the eyes and on a small area at the distal quarter of the elytra (Fig. 6) (male frons and vertical sides of the elytra always had white setae; Figs 1–3 View FIGURES 1 – 3 ). The colour variability did not demonstrate any geographical pattern, and though there were no females with white pubescence on their head known from Brazil, females with a completely black head were also found in Bolivia (Fig. 5). In terms of males, extension of the elytral maculation varied by a factor of two, and the apical macula was well separated from the elytral borders ( Figs. 1 View FIGURES 1 – 3 a, 3a) or reached the outer elytral border ( Fig. 2 View FIGURES 1 – 3 a).
Chromatic gender dimorphism. All males examined differed from females as commonly observed in Lamiinae via longer antennae and narrower bodies. Nevertheless, the following differences were noted: white pubescence on the male head and vertical sides of the elytra; the maculae with brownish-black setae on the elytra were smaller than that in females (maculae on pronotum were usually smaller but could have a similar size seen in females). However, the position of the pronotal and elytral maculae were similar in all examined males and females from Bolivia and Brazil (including sympatric males and females from Goiás in Brazil and Tarija in Bolivia), which led us to the conclusion that the examined males and females were con-specific. Despite the gender colour dimorphism, the key to species of Phoebe from Martins & Galileo (2014) permitted recognizing P. ornator and was determined not needing modification.
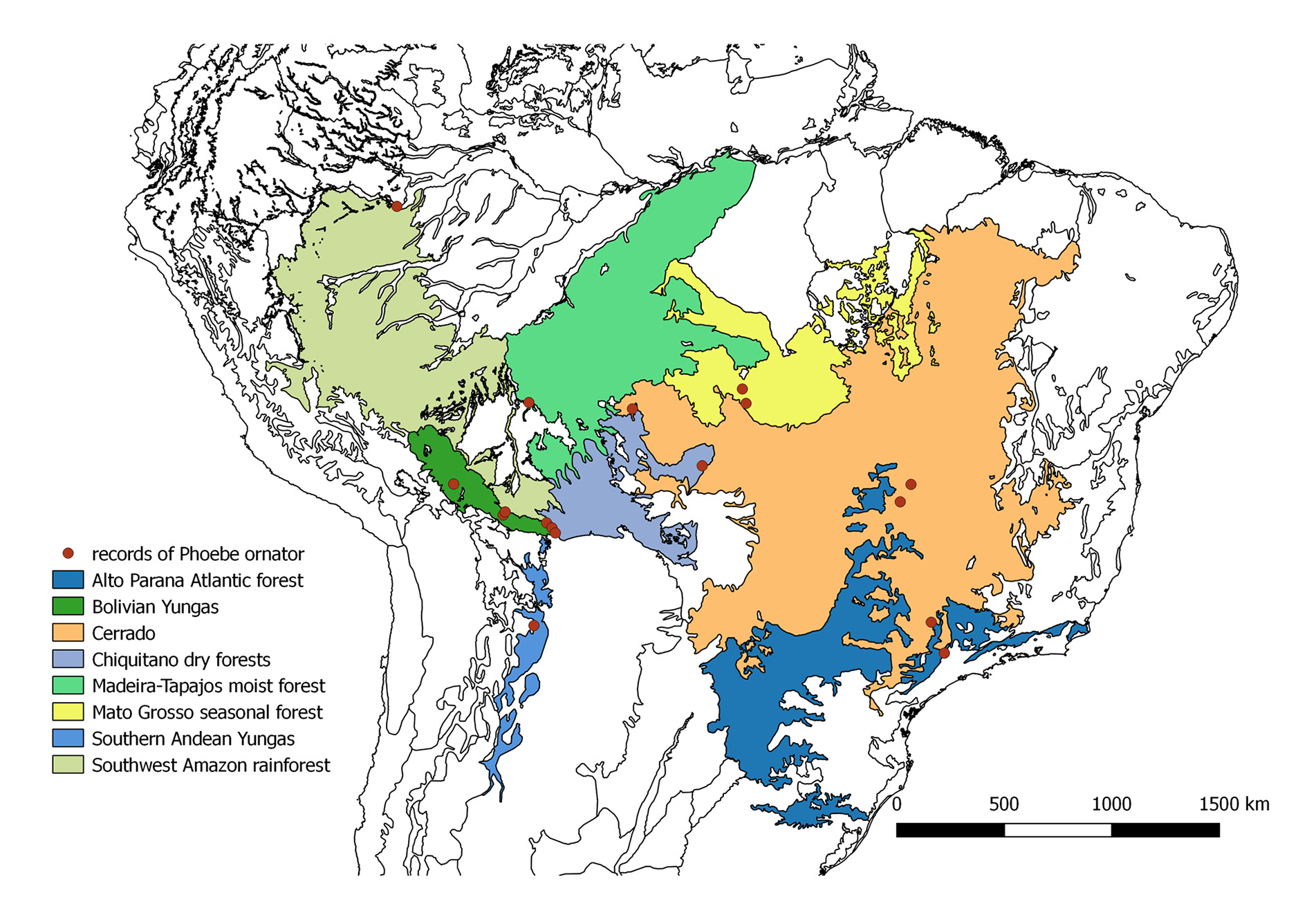
Biogeography. Records for seventeen locations could be obtained from the literature and collections ( Fig. 7 View FIGURE 7 ). All records excepting one (Tabatinga, Amazonas state, Brazil) were situated within a hypothetical rectangle of 1200 km in height (between 11.84°S and 22.9°S) and 2200 km in width (between 67.56°W and 47.06°W). Tabatinga is located another 1100 km north of the next northern most location. GoogleMaps
According to the biogeographic regionalization of Olson et al. (2001), P. ornator has been observed in eight ecoregions. Five records of P. ornator are situated in the Bolivian Yungas (the forests in Villa Tunari, Buena Vista and Potrerillo del Guenda are considered as sub-Andean Amazon rainforest by Navarro & Ferreira 2011), four in Cerrado, and two in the Mato Grosso seasonal forest and the Chiquitano dry forest, respectively (Table 1, Fig. 7 View FIGURE 7 ). Single records were obtained for southwest Amazon rainforest (Tabatinga), Madeira-Tapajos forest (Forte Principe da Beira), Alto Parana Atlantic forest (Campinas) and southern Andean Yungas (Salinas) (this forest is considered Bolivian Tucuman forest by Navarro & Ferreira 2011).
TABLE 1. Known records of Phoebe ornator , coordinates (decimal degrees), (E) elevation (m a.s.l.), ecoregion and (P) mean annual precipitation; *new ecoregion records; Southern Andean Yungas ( Olson et al. 2001) are considered Bolivian Tucuman forest by Navarro & Ferreira (2011); the forests in Villa Tunari , Buena Vista and Potrerillo del Guenda are considered as sub-Andean Amazon rainforest by Navarro & Ferreira (2011). 1 Lavado et al. 2012; 2 Vourlitis et al. 2002; 3no data available, data for Sinop ( Vourlitis et al. 2002), close to Vera ; 4no data available, data for Costa Marques ( Franca 2015), close to Forte Principe da Beira; 5 SUDAM 1984 ; 6no data available, data for Goiânia ( Sousa et al. 2010), close to Leopoldo Bulhões; 7 Landers 2007; 8 Santos et al. 2001; 9 Martins et al. 2011; 10 Gonfiantini et al. 2001; 11 Abrahamczyk et al. 2013; 12no data available, because abiotic conditions in mountainous areas often shift rapidly over short distances, no data for close locations is provided; 13 SENAMHI 2007; 14CEPAGRI 2010.
The mean annual precipitation of the 15 locations with available data is 1770 mm. The mean annual rainfall of 14 of the 15 locations with available data ranges between 1085 and 2101 mm (Tab. 1); Villa Tunari (Cochabamba department, Bolivia) has a mean annual precipitation of 3710 mm.
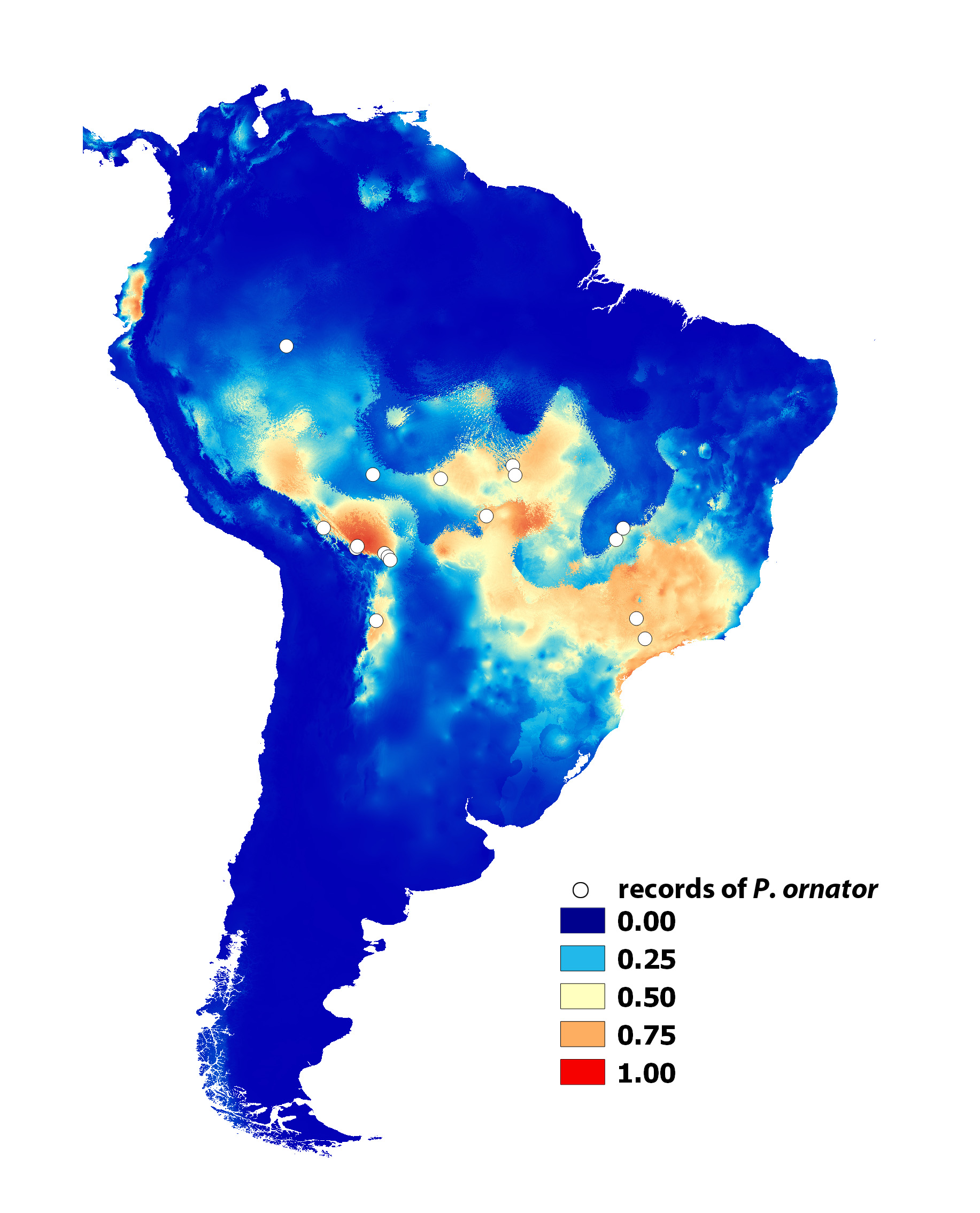
Abiotic variables and potential distribution range. According to the MaxEnt model, the most important abiotic variables contributing to the occurrence of the species were the precipitation of the warmest quarter of the year (62%), the temperature annual range (12%) and the isothermality (10%). The AUC (0.930) suggested a high score for model performance.
Areas with actual records and high probability of suitable conditions according to the MaxEnt model were in the ecoregions of the Bolivian Yungas and southwest Amazon rainforest, northwestern Cerrado, Eastern Alto Parana Atlantic forest, Serra do Mar coastal forests and Eastern portions of the Mato Grosso seasonal forest (see Olson et al. 2001) ( Fig. 8 View FIGURE 8 ). Conditions typical of those where the species was found were predicted mainly in the southwest Amazon rainforest, Mato Grosso seasonal forest, Cerrado, Alto Parana Atlantic forest, Ucayali moist forest, and, to a certain extent, the Bolivian and Peruvian mountain forests.
Areas without actual records but with suitable conditions for P. ornator were predicted in the northwestern Andean montane forests, Western Ecuador forest (both in Ecuador), southern portions of the Peruvian Yungas, and Iquitos varzea/ Ucayali moist forest. Low predicted probability of suitable conditions were indicated for Caatinga, Gran Chaco, the northeastern portion of Cerrado, and most of the Amazon rainforests (wide parts of Madeira- Tapajos moist forest, Purus varzea, Tapajos-Xingu moist forest, Xingu-Tocantins-Araguaia moist forest, Tocantins / Pindare moist forest, and forests north of the Amazon River ) ( Fig. 8 View FIGURE 8 ).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Phoebe ornator ( Tippmann, 1960 )
| Perger, Robert, Santos-Silva, Antonio & Guerra, Fernando 2017 |
Phoebe ornator
| Monne 2016: 773 |
| Martins 1993: 113 |
Adesmus ornator
| Tippmann 1960: 204 |