Myloplus lucienae, Andrade, Marcelo C., Ota, Rafaela P., Bastos, Douglas A. & Jégu, Michel, 2016
|
publication ID |
https://doi.org/10.11646/zootaxa.4205.6.5 |
|
publication LSID |
lsid:zoobank.org:pub:B5054D2C-47DF-4609-93E8-767F4B828268 |
|
DOI |
https://doi.org/10.5281/zenodo.5665122 |
|
persistent identifier |
https://treatment.plazi.org/id/C8450449-08C2-4A6A-A03A-292467FF9F76 |
|
taxon LSID |
lsid:zoobank.org:act:C8450449-08C2-4A6A-A03A-292467FF9F76 |
|
treatment provided by |
Plazi |
|
scientific name |
Myloplus lucienae |
| status |
sp. nov. |
Myloplus lucienae View in CoL , new species
( Figs. 1‒4 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 )
Myleus rhomboidalis View in CoL (non Cuvier): Toledo-Piza, 2002: 168, fig. 53 [rio Negro basin, listed; brief description; drawing of specimen by A.R. Wallace].
Myleus View in CoL sp.: Toledo-Piza, 2002: 180, fig. 59 [rio Negro basin, listed; brief description; drawing of specimen by A.R. Wallace]. Myleus View in CoL sp. A: Goulding, 1980: 115 (in part) [feeding behavior]; Borges, 1986: 106, figs. 3‒5 [short description; distribution].
Holotype. INPA 30717, 130.7 mm SL; Brazil: Amazonas: Novo Airão: rio Carabinani , 2°01'25"S 61°32'35"W; L. N. Carvalho, 25 Oct 2004. GoogleMaps
Paratypes. All from Brazil, Amazonas. INPA 915, 1 , 230.0 mm SL; and INPA 10215, 1 , 315.7 mm SL; São Gabriel da Cachoeira: rio Negro , 0°08'08"S 67°05'29"W; M. Goulding, 18 May 1979 GoogleMaps ; INPA 916, 4 , 279.9‒320.0 mm SL; São Pedro: rio Negro, confluence with igarapé Ibará , 0°24'50"S 65°01'09"W; M. Goulding, 23 May 1979 GoogleMaps ; INPA 917, 2 , 286.5‒ 334.3 mm SL; and INPA 3877, 4 , 266.2‒ 337.5 mm SL; Santa Isabel do rio Negro: rio Daraá , cachoeira do Aracu , 0°25'21"S 64°46'27"W; M. Goulding, 10 Feb 1980 GoogleMaps ; INPA 3682, 1 , 317.0 mm SL; and INPA 3685, 1 , 263.4 mm SL; São Gabriel da Cachoeira: rio Negro, cachoeira do Caranguejo , 0°03'15"S 67°08'56"; W. R. P. Ribeiro & R. Sotero, 7‒8 Mar 1990 ; INPA 10213, 1 , 286.8 mm SL; Manaus: rio Negro, ilha de Tamaquaré , 2°52'59"S 60°31'00"W; M. Goulding, 10 Nov 1979 GoogleMaps ; INPA 10214, 2 , 302.7‒ 312.3 mm SL; Barcelos: rio Arirará , 0°30'00"S 63°33'00"W; M. Gouding, 28 May 1979 GoogleMaps ; INPA 10216, 4 , 260.9‒ 331.3 mm SL; Barcelos: rio Negro confluence with rio Cuiuni , 0°53'41"S 62°58'21"W; M. Goulding, 3 Jun 1979 GoogleMaps ; INPA 42972, 1 , 74.9 mm SL (c&s); São Gabriel da Cachoeira: rock outcrops on rio Negro , 0°08'27''S 67°04'58''W; L. R. Py-Daniel et al., 6 Dez 2013 GoogleMaps ; INPA 49881, 4 , 211.9‒ 248.5 mm SL (234.9 mm SL, 1 skel.); São Gabriel da Cachoeira: street market (probably fished at Balaio community, rio Iá ), 0°23'22'' N 66°38'53'' W; D. A. Bastos, 20 Feb 2015 GoogleMaps ; INPA 50849, 1 , 90.2 mm SL; same data of holotype GoogleMaps . INPA 52894, 2 , 122.5‒ 123.4 mm SL; and MZUEL 14704, 1 , 137.4 mm SL; Novo Airão: Igapó no rio Negro , Parque Nacional de Anavilhanas , 2°43'10"S 60°45'18"W; J. Birindelli et al., 3 May 2016 GoogleMaps .
Diagnosis. Myloplus lucienae differs from all congeners, except M. planquettei and M. zorroi , by having anteriormost spine of prepelvic serra reaching only the middle of the abdomen between the verticals through pectoral and pelvic-fin origins (vs. reaching the vertical through pectoral-fin origin or almost so). Myloplus lucienae is distinguished from M. asterias , M. lobatus , M. planquettei , M. rhomboidalis , M. rubripinnis , M. ternetzi and M. zorroi , by having fewer scale rows between lateral line and dorsal-fin origin (30‒35 vs. 36‒61), and between lateral line and pelvic-fin origin (27‒31 vs. 32‒63). It is distinguished from M. asterias , M. levis , M. tiete and from M. torquatus , by having 18‒22 branched dorsal-fin rays (vs. 24‒27). Myloplus lucienae differs from M. arnoldi by having a relatively elongate body (body depth 53.0‒64.5% of SL vs. 69.0‒76.9% of SL). Myloplus lucienae still differs from M. ternetzi by the presence of a pair of symphyseal teeth on dentary (vs. absence), and from M. schomburgkii by the absence of a vertical black stripe on middle portion of flank (vs. presence).
Description. Morphometric data presented in Table 1 View TABLE 1 . Large-sized serrasalmid, maximum SL recorded 337.5 mm SL. Body relatively elongated. Greatest body depth at dorsal-fin origin. Snout blunt and rounded. Dorsal profile of head gently concave or straight from vertical through nostrils to tip of supraoccipital process, slightly convex from this point to dorsal-fin origin. Ventral profile of head and body slightly convex. Base of dorsal- and anal-fins gently convex.
holotypeNrangemeanSD
Mouth terminal to slightly upturned. Two rows of premaxillary teeth with 5 (28) molariform teeth in labial row, and 2 (28) in lingual row. Labial and lingual premaxillary rows interspaced by gap. First and second teeth of labial row with wide and rounded basis, more elongated anteroposteriorly, and edge with cutting crown. Dentary with 5* (26) or 6 (2) molariform teeth decreasing posteriorly in size. Pair of conical teeth at dentary symphysis. Symphyseal tooth with cutting edge on anterior margin. Maxilla edentulous.
Scales cycloid, relatively large sized for serrasalmid species. Perforated scales on lateral line imbricated on supracleithrum, and juxtaposed from humeral region to base of median caudal-fin rays. Perforated scales from supracleithrum to hypural plate end 66 (1), 67 (3), 68* (10), 69 (9), 70 (3) or 71 (2); and total perforated scales on lateral line from supracleithrum to base of median caudal-fin rays 71 (1), 72 (1), 73* (8), 74 (6), 75 (5), 76 (5) or 78 (2). Longitudinal scale rows between dorsal-fin origin and lateral line 30 (1), 31 (1), 32* (7), 33 (7), 34 (10) or 35 (2); and longitudinal scale rows between lateral line and pelvic-fin origin 27* (4), 28 (8), 29 (12), 30 (3) or 31 (1). Circumpeduncular scale rows 29* (1), 30 (7), 31 (9), 32 (9) or 33 (2).
Dorsal fin preceded by strong forward-oriented spine. Dorsal-fin rays ii* (25) or iii (3), and 18 (1), 19 (4), 20 (4), 21* (13) or 22 (6). Anal-fin rays iii* (24) or iv (4), and 31 (4), 32 (5), 33 (9) or 34* (10). Pectoral-fin rays i* (28), and 15 (1), 16* (16) or 17 (11). Pelvic-fin rays i,7* (28). Adipose fin with distal margin rounded to subrectangular. Caudal fin forked with two lobes of similar size.
Ventral keel with anteriormost spine of prepelvic serra located between verticals through pectoral- and pelvicfin origin, with series of 15 (1), 17 (1), 18 (3), 19* (5), 20 (7), 21 (5), 22 (5) or 23 (1) prepelvic spines; followed by 7* (9), 8 (14), 9 (2) or 10 (3) simple postpelvic spines; and 3 (2), 4 (19) or 5* (7) pairs of spines around anus. Total spines 28 (3), 29 (1), 31* (8), 32 (4), 33 (4), 34 (5), 35 (2) or 38 (1). First branchial arch with 24 (1), 25 (1), 27 (1), 28 (3) or 29 (1) total gill rakers; upper branch with 10 (1), 11 (1), 12 (2) or 13 (2) rakers; lower branch with 13 (2), 14 (2) or 15 (2) rakers; and 1 (6) at cartilage between upper and lower branches.
Gas bladder well-developed, two-chambered, total length 45‒50% SL, and with extrinsic muscle thin and poorly-developed (17 examined specimens). Anterior chamber smaller, cylindrical, horizontally positioned, with length 16‒19% SL. Posterior chamber larger, somewhat conical, posteriormost region narrower not very contracted, with length 26‒31% SL. Posterior chamber curved downward, lacking long appendix on its far end.
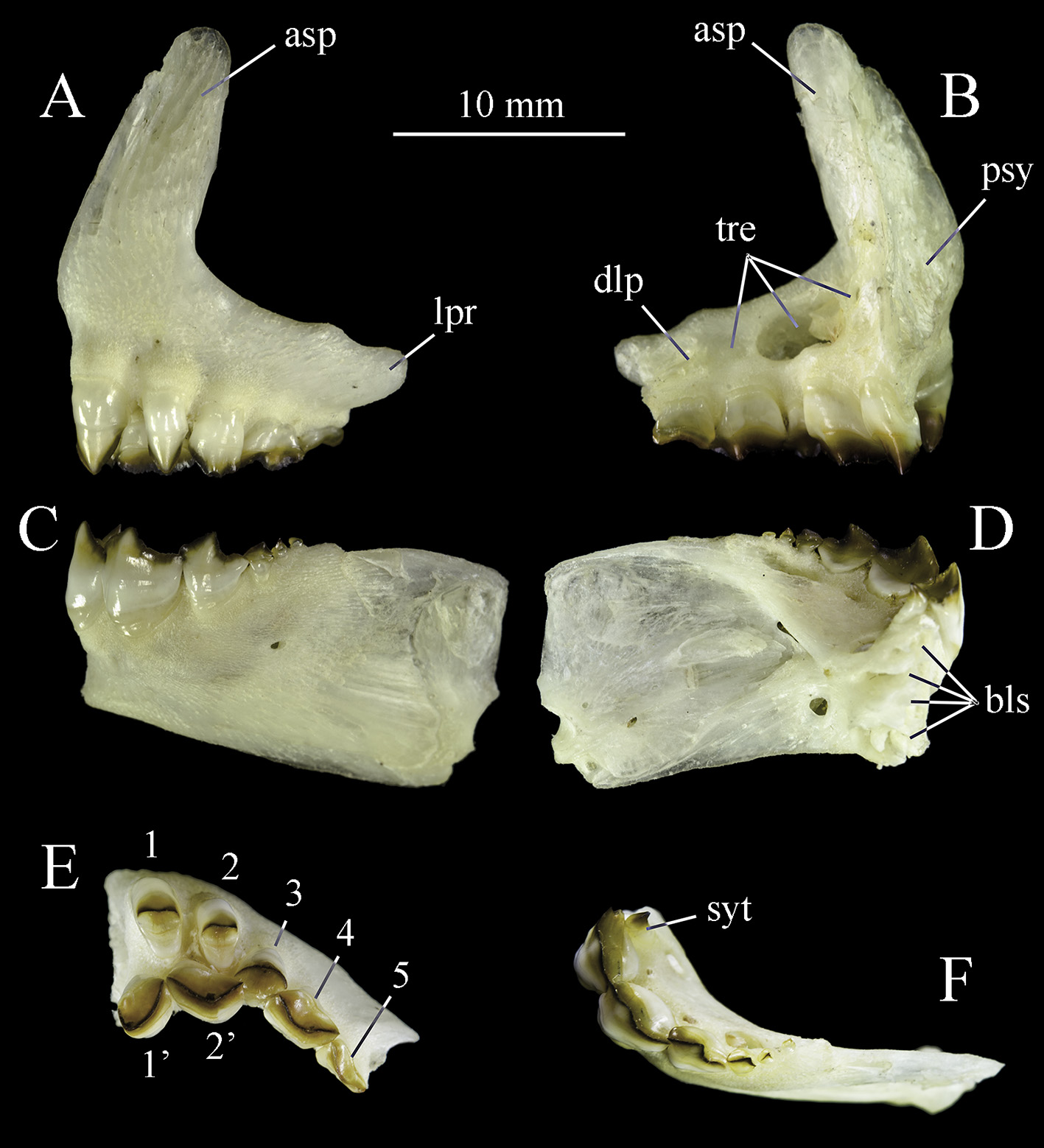
Ascending premaxillary process high, elongated with similar width from base to distal tip, and moderately inclined in relation to lateral premaxillary process ( Figs. 3 View FIGURE 3 a, b). Premaxillary lacking interdigitations at symphysis and strongly attached on mesethmoid ( Fig. 3 View FIGURE 3 b). Lateral premaxillary process lengthy, subrectangular, with dorsolateral process with concavity for maxillary insertion ( Figs. 3 View FIGURE 3 a, b). Three replacement teeth trenches on premaxillary ( Fig. 3 View FIGURE 3 b). Dentary rectangular ( Figs. 3 View FIGURE 3 c, d), slightly arched with four bony lamellae at symphysis ( Fig. 3 View FIGURE 3 d, f). Antorbital narrow and club-shaped. Supraorbital oval, narrow, with regular margins, not contacting infraorbital 6. Antorbital and supraorbital with unbranched sensory canal. Infraorbitals 1, 2, 3 and 5 with unbranched sensory canal, 4 and 6 with branched sensory canal, and 6 with Y shaped canal. Broad orbital region.
Neurocranium massif, high and triangular. Mesethmoid short, pointed and triangular anteriorly. Lateral ethmoid slender antero-posteriorly, positioned on anterior half of mesethmoid. Olfactory fossa with narrow aperture. Frontal short with oval fontanel slightly elongate and moderate concavity at epiphyseal bar. Parietal compact, narrower anteriorly, increasing slightly in width posteriorly. Supraoccipital spine well developed, its dorsal portion moderately curved posteriorly. Orbitosphenoid possessing two laterally compressed bony lamellae, anterior process wide distally and upturned, posteroventral process narrow and projecting ventrally with interdigitations on ventral margin not reaching parasphenoid. Parasphenoid lacking midventral keel with ventral aperture forming two projections parallel to each other along prootic and basioccipital ventral margins. Pterotic triangular with posterior process downward directed. Sphenotic with concave ventral margin, its anterior portion wider, narrowing posteriorly from middle portion of bone forming pointed tip.
Six (2) supraneurals, with 1st and 6th supraneurals positioned anterior to neural spine of 4th and 9th centrae, respectively. First dorsal-fin pterygiophore inserted posterior to neural spine of 11th (2) centrum. First anal-fin pterygiophore inserted behind haemal spine of 23nd (2) centrum. Forty-one (2) total vertebrae, with 20 (1) or 21 (1) precaudal and 20 (1) or 21 (1) caudal vertebrae.
Color in alcohol. Ground coloration silver-brownish hue. Dorsal portion of head and flanks darker than lower portion of head and flanks. Opercle, fourth infraorbital, and on third infraorbital cheek gap light brown. Postopercle membrane pale brown. Scales bordered distally by dark chromatophores forming overall faint reticulate pattern on body. Largest specimens with brown pectoral, pelvic, and caudal fins, posterior dorsal-fin rays and first anal-fin rays darker. Smaller specimens (131 mm SL or less) with overall coloration light brown, with pectoral and pelvic fins ranging from hyaline to hyaline with some melanophores distally. Smaller specimens with verticallyovoid dark spot on opercle, and dorsal, anal and caudal fins with distinct dark brown vertical stripe distally. Adipose fin light brown in all specimens.
Color in life. Based on freshly collected specimens (i.e. INPA 52894), the coloration in life ranging from flanks silvery with an olivaceous hue, with fins yellow reddish in young specimens ( Fig. 4 View FIGURE 4 ) to flanks silvery gray with fins black in larger specimens. Scales silvery distally with darkly pigmented base. Flanks with scattered, shapeless rosy salmon colored blotches, more concentrated at humeral region. Scales on ventral surface of body and anal-fin base slightly dark pigmented. Dorsal, adipose, anal and caudal fins with distinctly dusky indigo to black brownish coloration. Pectoral and pelvic fins with pale dark pigmentation.
Sexual dimorphism. Males of Myloplus lucienae can be distinguished from juveniles and females by having thin long filaments extending the branched dorsal-fin rays, an additional lobe formed by the projection of the middle anal-fin rays ( Fig. 2 View FIGURE 2 a), and by the presence of stiff hooks laterally divergent on distalmost lepidotrichia of each anal-fin branched ray (scarcely developed and solely recorded in the largest specimen examined, 337.5 mm SL). From ten dimorphic males (248‒337 mm SL), only four specimens presented thin filaments on dorsal fin, but all males exhibit the additional anal-fin lobe centered at the 16th branched anal-fin ray. Males of M. lucienae ( Fig. 2 View FIGURE 2 a) show overall coloration somewhat darker than females ( Fig. 2 View FIGURE 2 b). Juveniles and females lack dorsal-fin filaments or anal-fin hooks, and additionally, possesses anal fin with first 8 to 10 rays longer than posterior rays, forming a falcate anterior lobe ( Figs. 1 View FIGURE 1 , 2 View FIGURE 2 b).
Variation on body shape along ontogenetic development. Subunits of head presenting high variability along ontogeny, with three measurements among smaller analyzed specimens (i.e. 75.0‒ 130.7 mm SL) are distinct from larger specimens (i.e. 212.0‒ 337.5 mm SL). Positive allometry in postorbital distance, ranging between 28‒30% HL in smaller specimens and 30‒37% of HL in larger specimens; negative allometry in eye vertical diameter, 40‒43% of HL in smaller specimens and 30‒37% of HL in larger specimens; and positive allometry in mouth width, 30‒31% of HL in smaller specimens and 34‒39% of HL in larger specimens.
Distribution. Myloplus lucienae is so far only known from rio Negro and its main tributaries, such as the rio Arirará, rio Carabinani, rio Cuiuni, rio Curicuriari and rio Daraá, all belonging to the rio Negro basin, Amazonas State, Brazil ( Fig. 5 View FIGURE 5 ). The species is apparently widespread throughout rio Negro basin and is restricted to its blackwaters tributaries. The species is typically found in rapids, but is also recorded for slow-flowing habitats such as backwaters and lakes.
Etymology. Myloplus lucienae is named in honor of Luciene Maria Kassar Borges in recognition of her pioneer attempt to organize the knowledgement on the herbivorous Serrasalmidae from the rio Negro basin.
TABLE 1. Morphometric data of Myloplus lucienae. Range, mean, and standard deviation (SD) include values for holotype and paratypes.
| Standard length (mm) | 130.7 | 29 | 75.0–337.5 | 261.7 | * |
|---|---|---|---|---|---|
| Percentage of standard length | |||||
| Body depth | 60.6 | 28 | 53.0–64.5 | 58.9 | 2.5 |
| Head length | 24.4 | 28 | 22.5–25.4 | 24.0 | 0.8 |
| Supraoccipital process length | 29.9 | 28 | 24.0–30.7 | 27.2 | 2.1 |
| Predorsal length | 54.5 | 28 | 47.4–57.6 | 53.3 | 1.9 |
| Dorsal-fin base length | 30.4 | 28 | 23.2–32.4 | 30.1 | 1.7 |
| Interdorsal length | 13.6 | 28 | 6.6–14.1 | 12.8 | 1.4 |
| Adipose-fin base length | 6.4 | 28 | 5.5–9.2 | 6.2 | 0.7 |
| Caudal-peduncle depth | 10.6 | 28 | 9.1–10.7 | 9.9 | 0.5 |
| Anal-fin base length | 33.5 | 28 | 30.2–34.7 | 32.8 | 1.0 |
| Preanal length | 74.2 | 28 | 73.9–81.3 | 76.6 | 1.8 |
| Prepelvic length | 56.5 | 28 | 55.1–61.8 | 57.3 | 1.5 |
| Prepectoral length | 25.3 | 28 | 24.0–29.5 | 25.9 | 1.3 |
| Anal-pelvic distance | 20.5 | 28 | 19.7–27.3 | 21.7 | 1.5 |
| Pelvic-pectoral distance | 31.1 | 28 | 29.6–35.1 | 32.2 | 1.3 |
| Width of peduncle | 3.3 | 28 | 2.8–5.0 | 4.0 | 0.5 |
| Pectoral-fin length | 19.6 | 28 | 14.8–21.7 | 19.3 | 1.3 |
| Pelvic-fin length | 15.4 | 27 | 11.6–15.6 | 14.1 | 0.9 |
| First anal-fin lobe length | 22.9 | 21 | 20.1–36.3 | 28.1 | 4.5 |
| Second anal-fin lobe length | * | 6 | 14.9–19.4 | 16.6 | 1.7 |
| Dorsal-fin length | 29.2 | 27 | 20.1–32.1 | 25.4 | 3.0 |
| First oblique depth | 66.2 | 28 | 61.2–68.7 | 64.4 | 1.9 |
| Second oblique depth | 49.2 | 28 | 44.6–50.5 | 47.5 | 1.6 |
| Third oblique depth | 27.3 | 28 | 24.9–27.4 | 26.1 | 0.7 |
| Percentage of head length | |||||
| Snout length | 27.0 | 28 | 25.5–37.2 | 31.2 | 3.4 |
| Interorbital width | 52.0 | 28 | 44.8–58.5 | 53.7 | 3.6 |
| Width head | 63.1 | 27 | 58.5–73.0 | 65.9 | 3.5 |
| Postorbital distance | 30.2 | 28 | 28.1–36.9 | 32.9 | 2.4 |
| Fourth infraorbital width | 14.7 | 28 | 11.5–17.8 | 15.2 | 1.3 |
| Eye vertical diameter | 40.2 | 28 | 29.8–42.9 | 33.6 | 3.7 |
| Mouth length | 18.5 | 28 | 14.2–20.7 | 18.0 | 1.4 |
| Third infraorbital width | 9.6 | 28 | 8.1–13.9 | 11.0 | 1.3 |
| Cheek gap width | 11.0 | 28 | 8.5–13.9 | 10.5 | 1.3 |
| Mouth width | 31.0 | 28 | 29.9–38.5 | 35.5 | 2.5 |
| INPA |
Instituto Nacional de Pesquisas da Amazonia |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Myloplus lucienae
| Andrade, Marcelo C., Ota, Rafaela P., Bastos, Douglas A. & Jégu, Michel 2016 |
Myleus rhomboidalis
| Toledo-Piza 2002: 168 |
Myleus
| Toledo-Piza 2002: 180 |
| Borges 1986: 106 |
| Goulding 1980: 115 |