Potamotrygon wallacei, De Carvalho, Marcelo R., Rosa, Ricardo S. & De Araújo, Maria Lúcia G., 2016
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4107.4.5 |
|
publication LSID |
lsid:zoobank.org:pub:8218A512-8DD5-4C79-944B-36D235AA00E4 |
|
DOI |
https://doi.org/10.5281/zenodo.5621383 |
|
persistent identifier |
https://treatment.plazi.org/id/59315067-FF92-FF9D-FF7C-CCB49CB6FD0C |
|
treatment provided by |
Plazi |
|
scientific name |
Potamotrygon wallacei |
| status |
sp. nov. |
Potamotrygon wallacei View in CoL , n. sp.
( Figs. 1–10 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 View FIGURE 5 View FIGURE 6 View FIGURE 7 View FIGURE 8 View FIGURE 9 View FIGURE 10 , Tables 1–2 View TABLE 1 View TABLE 2 )
Potamotrygon schroederi Fernandez-Yépez, 1957 View in CoL [in part].— Castex & Yagolkowski, 1970: 1 –13 (redescription); Rosa, 1985: 302 –314 (revision).
Disceus thayeri Garman, 1913 View in CoL .— Nieuwenhuizen, 1974: 370 (in aquarium).
Potamotrygon laticeps Garman, 1913 View in CoL [in part].— Wheeler, 1975: pl. 10 (text book).
Potamotrygon View in CoL sp.— Géry, 1976: 2 (fig.); Wallace, 2002: 70, pl. 22 (notes, illustration); Duncan et al., 2009: 20 –31 (figs. 3, 4, 7–10) (gill functional morphology); Shibuya et al., 2010: 869 (fig. 3C) (lateral-line canals); Kodra et al., 2014: 15 –21 (fig. 2) (osmoregulation); Duncan et al., 2015: 128 –134 (fig. 1B) (gill physiology); Fontenelle & Carvalho, 2016: 6 (morphology of brain).
Potamotrygon hystrix View in CoL [in part].— Axelrod et al., 1986: 105 (fig.); Lambert, 1994: 204; Axelrod et al., 1997: 216 (fig. 1), 217 (figs. 1, 2); Fenner, 1997: 18, 20, 28 (figs.).
Potamotrygon histrix (Müller & Henle, 1836) View in CoL [in part].—Kuroiwa, 1994; Driedzic & Almeida-Val, 1996; Rabut & Fasquel, 1998: 42; Ross & Schäfer, 2000: 84, 85, 150 (figs.).
Potamotrygon cf. histrix View in CoL .—Zaiden et al., 2012: 1–8 (physiology of male reproductive system); Brinn et al., 2012: 139 –144 (physiology, stress response); Magro et al., 2015 (on-line publ., pathogenic agents).
Potamotrygon View in CoL sp. C.— Araújo, 1998: 10 –150 (figs.) (reproduction, fisheries).
Holotype. MZUSP 117809 (adult female, 371 mm TL, 247 mm DW, 264 mm DL), field no. RN11.19, col. F. Marques et al. ( Fig. 1 View FIGURE 1 ).
Type locality. Rio Negro, municipality of Barcelos, Amazonas, Brazil, 0° 57' 30.24"S, 062° 56' 14.697"W.
Paratypes. (21 specimens). BMNH 1893.4.24.42 (female, 250 mm DL), Manaus, Amazonas, Brazil, 24 April 1893; MNRJ 3532 (3 specimens, female 231 mm DL, male 219 mm DL, male, 144 mm DL), Uipiranga, Rio Negro, Amazonas, 1941; MZUSP 10290 (female, 324 mm TL, 206 mm DL), vicinity of Manaus, Rio Negro, Amazonas, Brazil, 15–23 November 1967; MZUSP 19209 (female, 217 mm DW), Rio Negro, at mouth of Rio Cuieiras, above Manaus, AM, Brazil, January 1977; MZUSP 104962 (adult female, 345 mm TL), lake adjacent to mouth of Rio Demeni, Rio Negro, Barcelos, Amazonas, Brazil, 0° 45' 52.9986"S, 062° 56' 34.998"W, field no. RN04.07, col. M.L.G. de Araújo et al.; MZUSP 104966 (female, 320 mm TL), Comunidade Marabá, Rio Negro, Barcelos, Amazonas, Brazil, 0° 58' 37.9992"S, 062° 56' 34.998"W, field no. RN04.58, col. M.L.G. de Araújo et al.; MZUSP 104977 (adult male, 282 mm TL), Praia do Tabuleiro, Rio Negro, Barcelos, Amazonas, Brazil, 0° 54' 24.0006"S, 062° 58' 20.9994"W, field no. RN05.65, col. M. Domingues et al.; MZUSP 117804 (female, 325 mm TL), Rio Negro, Barcelos, Amazonas, Brazil, 0° 57' 30.24"S, 062° 56' 14.697"W, field no. RN11.01, col. F. Marques et al.; MZUSP 117805 (adult male, 250 mm TL), field no. RN11.08, same data as MZUSP 117804; MZUSP 117806 (adult male, 367 mm TL), mouth of Rio Demeni, Rio Negro, Barcelos, Amazonas, Brazil, 0° 46' 31.8"S, 062° 56' 14.28"W, field no. RN04.33, col. F. Marques et al. ( Fig. 2 View FIGURE 2 ); MZUSP 117807 (adult female, 380 mm TL), same data as MZUSP 117804; MZUSP 117808 (adult female, 343 mm TL), same data as MZUSP 117804; MZUSP 117810 (adult female, 331 mm TL), same data as MZUSP 117804; MZUSP 117811 (adult female, 335 mm TL), same data as MZUSP 117804; MZUSP 117812 (adult male, 315 mm TL), field no. RN11.03, same data as MZUSP 117804; UFPB 3543 (male, 175 mm DL), Barcelos, Rio Negro, Amazonas, Brasil, 25 October 1996; UFPB 4866 (female, 107 mm DL), Rio Itu, tributary of Rio Negro, Amazonas, Brasil 00º56’S, 62º54’W, October 2001, col. M.L.G. de Araújo; UFPB 4867 (male, 107 mm DL), same data as UFPB 4866; UFPB 9017 (two females, 225 and 281 mm TL), Rio Itu, tributary of Rio Negro, ca. 70 km upstream from Barcelos, Amazonas, Brazil, 00°28’39’’S, 63°15’30’’W, 3 January 1999, col. M.L.G. de Araújo.
Other specimens examined. (50 specimens). MZUSP 27473 (juv. male, 93 mm DL), Baia Grande, Rio Jufari, upper Rio Negro, Caracaí, Amazonas, Brazil, 1982; MZUSP 73650 (adult female, 263 mm DW), Rio Itu, tributary of Rio Negro, ca. 70 km upstream from Barcelos, Amazonas, Brazil, 00°28’39’’S, 63°15’30’’W; MZUSP 116094 (data as in MZUSP 104692); UFPB 3543 (male, 175 mm DL), Barcelos, Rio Negro, Amazonas, Brasil, 25 October 1996; UFPB 4865 (36 specimens), same data as UFPB 4866; UFPB 5215 (3 specimens), Rio Itu, tributary of Rio Negro, ca. 70 km upstream from Barcelos, Amazonas, Brazil, 00°28’39’’S, 63°15’30’’W, 3 January 1999; UFPB 5214 (4 specimens), Igarapé Zamula, tributary of Rio Negro ca. 20 km downstream from Barcelos, Amazonas, Brazil, 00°51’54’’S, 62°46’41’’W; UFPB 5215 (3 specimens), same data as UFPB 9017; UFPB 5216 (adult male), same data as UFPB 9017; UFPB 5217 (dissected female, 240 mm DW), same data as UFPB 9017; UFPB 6100 (dissected male, 174 mm DW), same data as UFPB 9017.
Diagnosis. A species of Potamotrygon endemic to the Rio Negro drainage, diagnosed by its unique color pattern, composed of a light brown dorsal disc surface with blackish irregularly-shaped, few and broad vermiculations on central disc, delimiting a light brown amphora- or Ω-shaped figure or large light brown reniform blotches at disc center, with subcircular to reniform ocellated light brown spots towards disc margins, a darkbanded tail on its sides, and small size (smallest known species of Potamotrygon , maturing at 160–190 mm DW, largest examined specimen 310 mm DW). The following combination of characters further distinguishes this species from congeners: enlarged spines on dorsal midtail in one to three irregular rows, few in number, low, and not extending posteriorly to caudal stings, ending near mid length of tail; denticles on posterior mid-disc and tail base Y-shaped, with a central, anterior, bulbous cusp and usually two posterior pairs of smaller, rounded cusps; anterior angular cartilage well-developed, lacking posterior angular cartilage (both angulars present and well developed in most species of Potamotrygon , except P. tigrina , P. schroederi , P. histrix , P. schuhmacheri , P. orbignyi , P. hu m e ro s a, P. signata , and P. marinae ; P. wallacei , n. sp., is easily distinguished from these species by its unique color pattern, arrangement of enlarged spines on dorsal tail, and smaller size); and clasper skeleton with a single basal segment (almost all species of potamotrygonids have two segments).
Description. Small sized stingray, largest examined specimen 290 mm DL, 263 mm DW. Disc markedly oval, disc length 1.1–1.2 times DW; greatest disc width anterior to its mid-length. Disc slightly tapers posteriorly. Anterior snout with small knob-like protuberance, present also in adults. Anterior disc margin broadly convex, especially in larger adult specimens. Head not greatly protruding above disc, and not particularly broad. Eyes relatively large, somewhat bulging; eye length 1.6–4.8 in interorbital distance, 1.1–1.9 in interocular distance. Preocular distance 3.0– 3.8 in DW, 1.0–1.5 times interocular distance. Spiracles relatively large, rhomboidal, but more slender in preserved specimens. Spiracle length 1.2–2.4 times eye length, and 1.9–3.1 in interspiracular distance. Spiracles extending anterolaterally to about mid-length of eyes.
Mouth relatively small, its width about equal to internarial distance. Nasal curtain more broad posteriorly, trapezoidal, with small median indentation, and covering almost all of mouth; posterior margin of nasal curtain fringed in some specimens. Nostrils very slender, elongate. Posterior margin of mouth with small median notch. Very weak, shallow grooves at lower jaw corners. Floor of mouth with five buccal papillae. Preoral distance 4.0– 5.2 in DW. Teeth relatively small, loosely set in quincunx, in 24–58/24–53 total longitudinal rows (holotype with 36/38), and 4–7 exposed teeth in median row of lower dental plate. Tooth crown reniform in apical view, with concave anterior margin, without prominent cusps except in mature males. Teeth sexually dimorphic, adult and subadult males with pointed central cusp and two small accessory cusps; females with blunt teeth. Branchial basket relatively wide, distance between first pair of gill slits 3.7–4.9 in DW; distance between first and fifth gill slits 5.3–6.7 in DW. Fifth gill slits about half of width of first gill slit; gill slits weakly sinuous.
Pelvic fins relatively broad and somewhat rounded at apices, partially covered by disc and only slightly protruding beyond disc posteriorly. Claspers in adult males slender and elongate, extending posteriorly to about one-third tail length. Tail relatively short and slender, tail length about equal to disc width in intact juvenile specimens; other examined specimens with broken tails. Tail width at base about one-half to three-fourths interorbital distance. Lateral tail ridges well-developed, present from tail base to close to sting origin. Tail in crosssection somewhat flat dorsoventrally. Tail with well-developed dorsal and ventral finfolds posterior to caudal stings, especially dorsal finfold; height of dorsal finfold 1.5–2.5 in tail height. Caudal sting slender, sting length 1.3–3.3 times tail width at base. Usually one or two caudal stings present, with lateral serrations more developed at sting posterior third, near sting tip; lateral serrations not very well developed.
Coloration. Dorsal surface of disc light brown, brown or grayish-brown with irregularly-shaped dark brown to black figures and vermiculations, delimiting light brown, large reniform or brain-shaped areas at disc center, and subcircular light brown areas towards disc margins ( Figs. 1–5 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 View FIGURE 5 ). The black pigment forms a light brown amphora- or Ω-shaped figure on mid-disc ( Fig. 4 View FIGURE 4 g), starting immediately behind eyes and spiracles and continuing posteriorly on disc. The blackish pigment is easily washed away in captured or preserved specimens, but is usually retained around the spiracles and anterior disc. Light brownish irregular spots on disc periphery usually have darker centers forming weakly ocellated spots. The outer margins of these spots usually have lighter, yellowish spots. These smaller spots may be reniform or cerebriform, and decrease in size toward disc margins; on posterolateral disc they may be fused into lighter streaks. Orbits surrounded by yellowish spots delimited by a fine, darker vermiculate pattern ( Fig. 6 View FIGURE 6 ). Sides of tail with alternate light brown and intense black bars that do not connect dorsally, and occurring from tail origin to just posterior to caudal sting origin. Tail finfolds black with slender, lighter vertical lines. Ventral surface of disc white with broad, dark brown disc margins, and creamy to grayish center. Ventral anterior snout region and tail region with grayish-brown, faded spots. Small orange blotches sometimes present on ventral tail base, anterior snout and disc sides, mixed with grayish-brown spots. Occasionally a ventral dark brown spot present on center of disc in adult specimens. Pelvic fins ventrally white with dark brown posterior margins and claspers. Ventral surface of tail dark brown, mottled with light brown and grayish blotches.
Dermal denticles. Dorsal surface of disc not prickly, with few small, not densely distributed denticles. Denticle crowns form a Y-shape in apical view on mid-disc, with an anterior, central rounded or bulbous cusp and two or sometimes three pairs of smaller, rounded accessory posterior cusps ( Fig. 7 View FIGURE 7 ). Small denticles more numerous on posterior mid-disc and tail base. Enlarged spines (or thorns) just posterior to pelvic girdle and anterior to tail origin on posterior mid-disc and on dorsal tail midline. Tail with up to 30 middorsal thorns in adult and subadult specimens, in one to three irregular and more or less parallel rows; usually more thorns present over tail base and posterior disc, fewer thorns posteriorly on midtail. Thorns in multiple rows on tail base, and usually in one row on midtail. Thorns with slightly rounded bases, posteriorly pointed and raked apices, and usually greater in size at posterior level of pelvic fins. Thorns extend posteriorly to terminate well anterior to caudal stings, at near half tail length. Juvenile specimens with less developed and fewer middorsal spines, and almost no smaller denticles on dorsal surface. No tubercles developed dorsally or laterally on tail. Larger female specimens with moderately developed lateral spines on tail.
Ventral lateral-line canals. Suborbital loop (sol) slender, curved medially ( Fig. 8 View FIGURE 8 ), extending toward anterior disc margin close to hyomandibular canal (hyoc); suborbital loop formed by infraorbital canal (ioc). Medial component of infraorbital canal undulated; posterior portion of infraorbital canal extends to close to first gill slit, forming small, subtriangular infraorbital loop (iol). Orbitonasal component of supraorbital canal (soc) extends in parallel to medial portion of infraorbital canal, closer to nostrils, and is also highly undulated, forming a very slender prenasal loop (pnl). Mandibular canal (man) short, somewhat arched, and just posterior to mouth. Anterior jugular loop (ajl) slender; posterior jugular loop (pjl) broader, rectangular, formed medially by jugular canal (jug). Subpleural component of hyomandibular canal (hyoc) extends laterally at branchial slits, somewhat undulated. Initial portion of infraorbital canal (ioc) sometimes with slight undulations. Anterior subpleural tubules (ast) present anteriorly, branching from anterior portion of hyomandibular canal. Posterior subpleural tubule (pst) single, extending posteriorly past anterior pelvic fin margin. Subpleural loop (spl) slender, formed by posterior segment of hyomandibular canal.
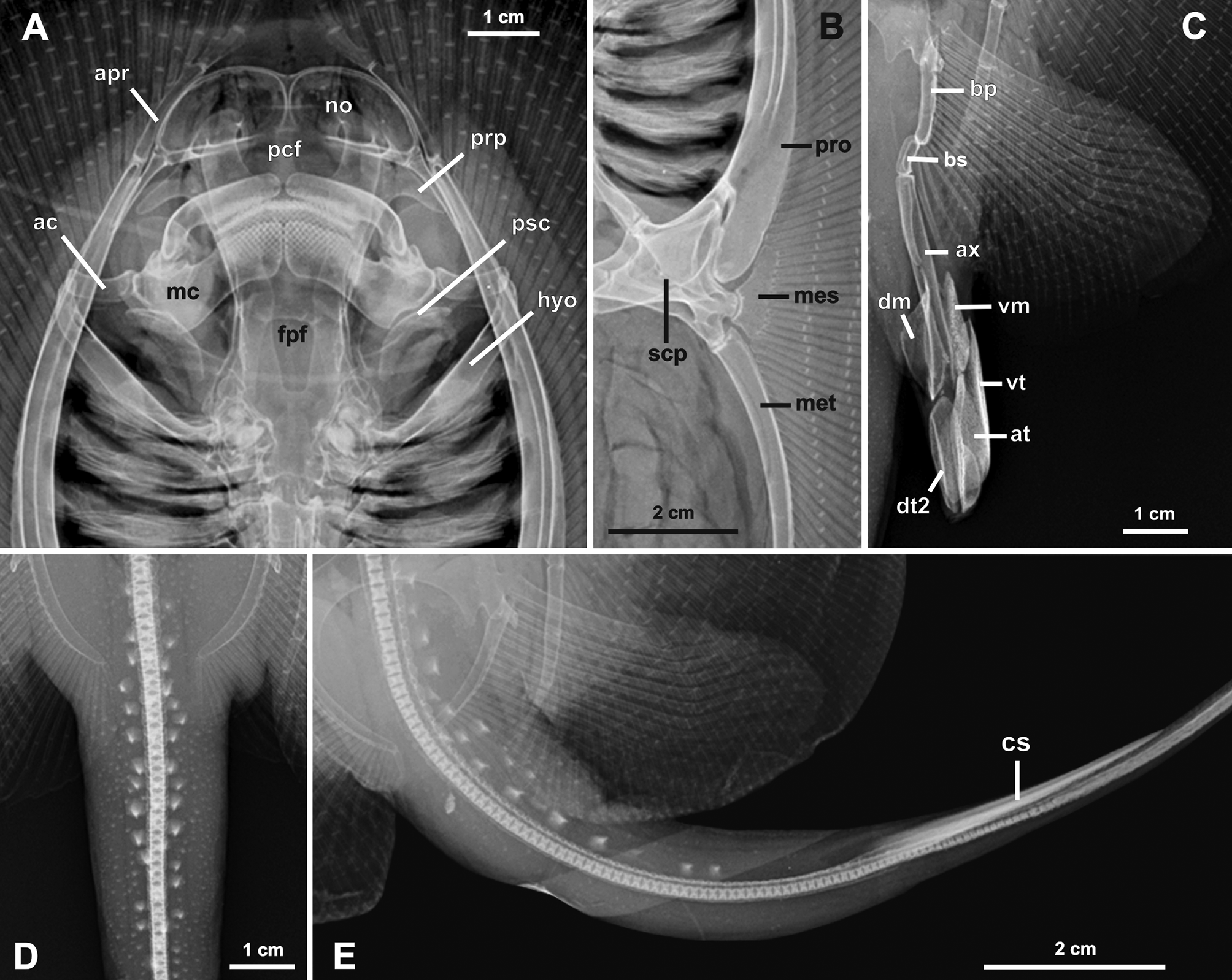
Skeletal features. Neurocranium about as twice as long as wide, with greatest width at level of pre- and postorbital processes ( Figs. 9 View FIGURE 9 , 10 View FIGURE 10 ). Nasal capsules broadly rounded, without strong anterior median indentation; internasal septum very slender. Nasal apertures (no) broad, much wider than long. Preorbital processes (prp) posterolaterally directed, slender, and broadly triangular. Postorbital processes (pop) projecting anterolaterally, rectangular, broad and relatively robust. Anterior to postorbital process, triangular supraorbital process (sp) well defined. Neurocranium most slender at posterior third of orbit. Otic capsule short, straight in dorsoventral view, and wider than orbital region. Precerebral (pcf) and frontoparietal (fpf) fontenellae about three-fourths of neurocranial length. Precerebral fontanelle very circular, occupying first third of neurocranial length; frontoparietal fontanelle slender and elongate, constricted medially and tapering posteriorly, terminating at mid-level of postorbital processes. Epiphysial bar separating both fontanelle incomplete but present even in larger adults. Parietal fossa (paf) very shallow. Antorbital cartilage very slender in dorsoventral view, wider at articulation with posterolateral aspect of nasal capsule; antorbitals laterally compressed, extending posteriorly beyond level of preorbital processes to level of articulation between palatoquadrate and Meckel’s cartilage. Prespiracular cartilages (psc) slender, weakly calcified, relatively elongate, and laterally very concave.
Meckel’s cartilages (mc) much stouter than palatoquadrates, with well-developed dorsally projecting lateral processes; palatoquadrates very slender. Hyomandibulae (hyo) relatively slender, especially at articulation with ventrolateral aspect of otic capsule. Hyomandibulae extend laterally to more or less level of propterygium. A single, robust (anterior) angular cartilage (ac) present; angular cartilage anteriorly concave and about one-third length of hyomandibulae; posterior angular possibly vestigial, not seen in radiographs or dissected and clear-andstained specimens. Anterior angular positioned perpendicular to neurocranial length, not obliquely oriented.
Cervicothoracic synarcual about three-fourths length of neurocranium, its greatest width about equal to greatest width of orbit; lateral stays not projecting laterally to great extent. Spinal nerve foramina ranging from 26–29 on each side. Two to three vertebrae incorporated into cervicothoracic synarcual. Thoracolumbar synarcual very slender, not strongly calcified, and relatively short. Individual vertebral centra occurring caudally to about one-half length of caudal sting. Caudal to individual centra, a weakly calcified notochordal extension (cartilaginous rod) present distally. Transition from mono- to diplospondyly occurs at about third to fourth centrum posterior to rear margin of pelvic girdle.
Propterygium (pro) posteriorly wide, tapering anteriorly, much more stout at base than meso- and metapterygium, and with a slender anterior propterygial segment (apr) articulating with six to seven radial elements lateral to nasal capsule. Metapterygium (met) only slightly more broadly arched than propterygium, with four posterior segments decreasing in size caudally. Mesopterygium (mes) more elongated anteriorly, slender, slightly convex externally but with slight indentation at about its mid-length. Pectoral radial elements sometimes fused at base, especially at posterior propterygium close to mesopterygium and at anterior metapterygium. Pectoral radials slender close to pectoral basals, slightly widening and shortening toward mid-disc, and slender again distally, bifurcating at 8th, 9th or 10th segment depending on location in disc; a total of up to 18 radial segments present at mid-disc.
Median prepelvic process (pp) of pelvic girdle slender, elongate and longer than maximum width of pelvic girdle. Pelvic girdle with concave anterior margins lateral to median prepelvic process. Lateral prepelvic processes markedly triangular, well developed. Iliac processes (ip) extending caudally beyond ischial processes (isp), somewhat irregular and broad distally. Ischial process broadly triangular. Three obturator foramina (of) present. Posterior margin of puboischiadic bar highly concave. Basipterygium stout and relatively short, articulating with pelvic radials except first enlarged radial (articulates to lateral aspect of iliac projection of pelvic girdle directly) and two posteriormost enlarged radials (articulate with beta cartilage and basal segment of clasper). Proximal pelvic radial element much longer than other radial elements; a total of six radial segments present from basal elements to outer disc margin, bifurcating at fourth segment from basals.
Clasper skeleton ( Figs. 9 View FIGURE 9 c, 11) consists of a single basal segment, beta cartilage, axial, dorsal marginal, ventral marginal, dorsal terminal 2, accessory terminal, and ventral terminal cartilages. Basal segment (bs) curved in apical view, with convex inner margin. Beta cartilage (be) slender, proximally articulating with basal segment and with posteriormost pelvic radials, distally tapering and articulating with axial cartilage at its proximal third. Axial cartilage (ax) elongate and nearly straight in ventral view. Dorsal marginal nearly trapezoidal in dorsal view, tapering anteriorly and with a depression along midline. Ventral marginal (vm) elongate, anteriorly tapering. Dorsal terminal 2 (dt2) elongate with rounded distal margin. Accessory terminal (at) elongate with convex outer margin. Ventral terminal (vt) elongate, posteriorly broad in ventral view, and with concave outer margin.
Geographic distribution. Potamotrygon wallacei is endemic to the Rio Negro drainage in Amazonas, Brazil, occurring from Santa Isabel down to Rio Cuieiras in the vicinity of Manaus ( Fig. 12 View FIGURE 12 ).
Common name. The species is known as “raia cururu” in Brazil; “cururu” is a common name for the cane toad, Rhinella marina , to which the dorsum of P. wallacei bears some resemblance.
Etymology. The specific name honors Alfred Russel Wallace (1823–1913), the first naturalist to collect, observe and illustrate this species while on his travels in the Rio Negro in the early 1850's ( Wallace, 2002).
Remarks. Historically, this species has been known at least since Alfred Russel Wallace’s journey to the Amazon in the mid 19th Century (1848–1852). Wallace figured and provided a short descriptive account on this species in plate 22 of his manuscript, under the vernacular name “raia” (Portuguese name for ray) ( Wallace, 2002), mentioning its color, branchial slits, and caudal stings, and noting that young specimens have markings that are "more finer and fainter". His remarkably precise illustration is reproduced here ( Fig. 13 View FIGURE 13 ); its only somewhat inaccurate aspect is the enlarged spines (thorns) on dorsal tail, which appear exaggerated. Unfortunately, all his biological materials were lost upon the sinking of the vessel Helen during his return to England, and no formal description was subsequently provided.
Potamotrygon wallacei View in CoL has been pictured several times in aquarium fish books and magazines, cited either as Potamotrygon View in CoL sp. or misidentified as P. histrix View in CoL (or P. hystrix View in CoL ), P. laticeps View in CoL , P. motoro View in CoL or Disceus thayeri . Some of the most commonly used names are included above in the synonymy. One of the most widely used names for the species is P. histrix View in CoL (e.g. Kuroiwa, 1994; Driedzic & Almeida-Val, 1996; Ross & Schäfer, 2000). As pointed out by Rosa (1985), Carvalho et al. (2003), and Rosa et al. (2008), P. histrix View in CoL is endemic to the Paraná-Paraguay river basin and does not occur in the Amazon region, while P. laticeps View in CoL is a junior synonym of P. m o t o ro. Disceus thayeri is considered a junior synonym of Paratrygon aiereba ( Rosa, 1991) View in CoL and is clearly distinct from species of Potamotrygon View in CoL . The scientific literature has also misidentified this species. Castex & Yagolkowski (1970) and Rosa (1985) mistakenly treated P. wallacei View in CoL as juvenile specimens of P. schroederi View in CoL due to the small number of specimens, including adults of P. wallacei View in CoL , available for study at that time.
Comparisons with congeners. Potamotrygon wallacei View in CoL is easily distinguished from congeners by its color pattern, size, distribution of enlarged spines (thorns) on posterior disc and anterior dorsal tail, morphology of small dermal denticles on posterior mid-disc and tail base, number and morphology of angular cartilages, and presence of a single basal segment in clasper. Other, smaller distinctions in bodily proportions and dentition also appear to be diagnostic but require further comparisons. The single basal segment in the clasper skeleton of P. wallacei View in CoL ( Figs. 9 View FIGURE 9 c, 11) contrasts with the condition in other potamotrygonids; P. histrix View in CoL also has a single basal segment, but two basal segments are present in P. schuhmacheri View in CoL , P. f a l kn e r i, P. scobina View in CoL , P. limai View in CoL , P. schroederi View in CoL , P. orbignyi View in CoL , P. humerosa View in CoL , P. m o t o ro, and in species of Plesiotrygon View in CoL , Paratrygon View in CoL and Heliotrygon View in CoL (further comparisons are in progress).
No other species of Potamotrygon View in CoL , except perhaps P. magdalenae View in CoL , sexually matures and maintains such a small size over its lifetime. Potamotrygon wallacei View in CoL clearly matures smaller than 200 mm DW and the largest female specimen examined was 310 mm DW (another female of 263 mm DW measured 440 mm TL with intact tail); hundreds of specimens have been examined by us over a period of many years, and the largest females are usually well below 310 mm DW. Potamotrygon magdalenae View in CoL in known to reach larger sizes, with large females reported to reach 470 mm DW ( Lasso et al., 2014). Both species have markedly oval discs (especially in smaller specimens) and slender, proportionally elongate claspers in adult males. Potamotrygon magdalenae View in CoL further differs from P. wallacei View in CoL in having a much longer tail, two equally-sized angular cartilages, usually a finely vermiculate color pattern, and is endemic to the Magdalena and Atrato rivers of northern Colombia.
The specific design of the dermal denticles on mid-disc and tail base in P. wallacei View in CoL is conspicuous among species of Potamotrygon View in CoL . The Y-shaped design of the denticles, composed of an anterior, central, larger and bulbous cusp, and two or three posterior pairs of smaller, rounded cusps, has also been found in an undescribed species of Potamotrygon View in CoL from the Rio Tapajós, which has other distinctive types of denticles as well (Carvalho & Lemos, unpubl.). A survey of denticles among almost all congeners found this pattern only in P. wallacei View in CoL and the endemic Tapajós species, but further comparisons are underway.
The number and morphology of angular cartilages are important features that may greatly aid in distinguishing species of Potamotrygon View in CoL (e.g. Carvalho et al., 2004, 2011; Loboda & Carvalho, 2013; Fontenelle et al., 2014). Potamotrygon wallacei View in CoL has a single angular element in between the hyomandibulae and lower jaws (the anterior angular); its length is about one-third hyomandibular length. A single angular cartilage associated to each hyomandibula in Potamotrygon View in CoL is also found in P. tigrina View in CoL , P. schroederi View in CoL , P. histrix View in CoL , P. schuhmacheri View in CoL , P. orbignyi View in CoL , P. humerosa View in CoL , P. signata View in CoL , and P. marinae (Carvalho et al., 2011) View in CoL . The angular cartilage in P. wallacei View in CoL is unlike the extremely elongated, anterior angular of the Rio Parnaíba basin endemic P. signata View in CoL , which is close to one-half of hyomandibulae length ( Carvalho et al., 2004). The angular in P. wallacei View in CoL is similar in dimensions to that of P. orbignyi View in CoL and P. hu m e ro s a, and appears to be slightly larger proportionally than in P. m ar i n ae and P. tigrina View in CoL .
Potamotrygon wallacei View in CoL is most similar to the much larger and more widely distributed northern South American species P. orbignyi View in CoL in color pattern, as both species have a brown to grayish-brown dorsal color covered with darker brown to blackish markings. However, these dorsal markings differ substantially in both species (see Silva & Carvalho, 2015 for P. orbignyi View in CoL ), as P. wallacei View in CoL does not have a complete or incomplete reticulate pattern. Although there is some variation among specimens of P. wallacei View in CoL in color pattern, this is not nearly as great as in P. orbignyi View in CoL ; both species, however, have similarly colored tails. These species also differ in their arrangement of enlarged dorsal tail spines ( P. orbignyi View in CoL usually has a single row of wider-based spines extending to caudal stings) and denticles ( P. orbignyi View in CoL has more intense denticulation on disc and tail). Both species co-occur in the Rio Negro, and small or neonate specimens may be confused, even though their specific characteristics in color pattern and spines and denticles are already present.
Similar differences separate P. wallacei View in CoL from P. humerosa View in CoL and P. m ar i na e, species with color patterns that resemble P. orbignyi View in CoL (especially P. humerosa View in CoL , less so in P. marinae View in CoL ) but that are very distinct from the color pattern of P. wallacei View in CoL . Both of these species have enlarged spines on dorsal tail extending posteriorly to caudal stings. Potamotrygon humerosa View in CoL has many well developed spines, also present on lateral tail, whereas P. marinae View in CoL has more slender spines in few rows, but very distinct from the arrangement of spines in P. wallacei View in CoL , and also has small lateral tail spines in larger specimens. Potamotrygon humerosa View in CoL occurs in the mid Rio Amazonas basin in Brazil, and P. marinae View in CoL is endemic to French Guiana (for further data on these species, see Silva & Carvalho, 2015).
From the northern South American species P. t igrina and P. schroederi View in CoL , P. wallacei View in CoL is easily distinguished in color pattern, as both former species have ornate patterns with rosettes, cerebriform markings, spots and elaborate broad vermiculations, and have the posterior tail with broad and intense cream-colored and brown alternating bands. These species also have relatively larger dorsal tail spines with broad bases that reach posteriorly to the caudal stings, unlike in P. w a l l a ce i. Potamotrygon schroederi View in CoL also occurs in the Rio Negro, but P. tigrina View in CoL is so far restricted to the upper Rio Amazonas basin in Peru (Carvalho et al., 2011).
The Paraná-Paraguay basin species P. histrix View in CoL and P. schuhmacheri View in CoL are easily differentiated from P. wallacei View in CoL by their color pattern, comprised dorsally of slender, small vermiculate and rosette-like markings on a grayish-olive to dark brown background, and ventrally mostly gray with unpigmented areas near anterior and central disc. Potamotrygon histrix View in CoL and P. schuhmacheri View in CoL also have markedly prickly discs and tails with numerous closely packed and sharp denticles, with denticles also on disc margins and lateral tail, and usually have a single row of enlarged spines that are much greater than in P. wallacei View in CoL and extend posteriorly to the caudal stings (Carvalho, unpubl.). Both species are also small sized, sexually maturing at about 210 mm DW, but achieve greater sizes compared to P. wallacei View in CoL (larger specimens of P. histrix View in CoL and P. schuhmacheri View in CoL attain sizes greater than 350 mm DW).
Habitat and movements. Potamotrygon wallacei View in CoL occurs in black waters with low pH and dissolved oxygen levels ( Duncan & Fernandes, 2010); hence, even though it is present in the lower and upper Rio Negro and in some of its direct effluents, it does not enter the Rio Branco (a white water river that enters the Rio Negro but with a higher pH). For example, P. wallacei View in CoL appears to remain near the mouth of the Rio Demeni, a river with murkier waters that flows into the Rio Negro on its left bank just above Barcelos, where there is a strong influx of water from the Rio Negro. It also inhabits small black water tributaries of the Rio Negro drainage (“igarapés”), with sandy bottoms and abundant litter cover, including leaves and fallen tree trunks from the flooded forest (“igapó”) ( Fig. 14 View FIGURE 14 ). Individuals show limited seasonal movements within the habitat, following flood and ebb cycles, when they respectively enter the flooded forest or congregate around islands in the main channels. The longest traveled distance by a tagged male specimen was 15 km (Araújo, unpubl.). No movement patterns on a daily basis were observed in the field. The rays remain on or under the litter cover during the day and become active at night.
Food habits. Newborn stingrays subsist on yolk reserves for the first three weeks of life and then start feeding on aquatic insects amidst the litter; sub-adults and adults shift their diet to shrimp and small characiform fishes ( Shibuya et al., 2009; Araújo, unpubl.).
Reproduction. Potamotrygon wallacei View in CoL , as with other stingray species, exhibits matrotrophic viviparity with lipid histotrophy, the embryos being nourished by means of yolk sac and trophonemata. Sexual maturity occurs between the sizes of 160–190 mm DW, both for males and females. Fecundity ranges from 1–5 embryos (mode = 2) per female. Gestation is completed in three months (one of the shortest known for elasmobranchs) and newborns range from 60–95 mm DW. Adult females reproduce during their second year of life, much earlier than other Potamotrygon View in CoL species, which usually take 4–5 years (Araújo, unpubl.), in agreement with its small size. Parturition occurs between the months of October and January, which corresponds to the dry season and beginning of the floods in the Rio Negro.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
ParvPhylum |
Chondrichthyes |
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Potamotrygon wallacei
| De Carvalho, Marcelo R., Rosa, Ricardo S. & De Araújo, Maria Lúcia G. 2016 |
Potamotrygon cf. histrix
| Brinn 2012: 139 |
Potamotrygon histrix (Müller & Henle, 1836)
| Ross 2000: 84 |
| Rabut 1998: 42 |
Potamotrygon
| Araujo 1998: 10 |
Potamotrygon hystrix
| Axelrod 1997: 216 |
| Fenner 1997: 18 |
| Lambert 1994: 204 |
| Axelrod 1986: 105 |
Potamotrygon
| Fontenelle 2016: 6 |
| Duncan 2015: 128 |
| Kodra 2014: 15 |
| Shibuya 2010: 869 |
| Duncan 2009: 20 |
| Wallace 2002: 70 |
| Gery 1976: 2 |
Disceus thayeri
| Nieuwenhuizen 1974: 370 |
Potamotrygon schroederi Fernandez-Yépez, 1957
| Rosa 1985: 302 |
| Castex 1970: 1 |