Colossopus grandidieri Saussure, 1899
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4341.2.2 |
|
publication LSID |
lsid:zoobank.org:pub:05152B19-56AA-4CCD-A3C6-53EA3369A54C |
|
DOI |
https://doi.org/10.5281/zenodo.5611865 |
|
persistent identifier |
https://treatment.plazi.org/id/533F87C4-FF9D-733B-26E2-FEE8FE03CA44 |
|
treatment provided by |
Plazi |
|
scientific name |
Colossopus grandidieri Saussure, 1899 |
| status |
|
Colossopus grandidieri Saussure, 1899 View in CoL
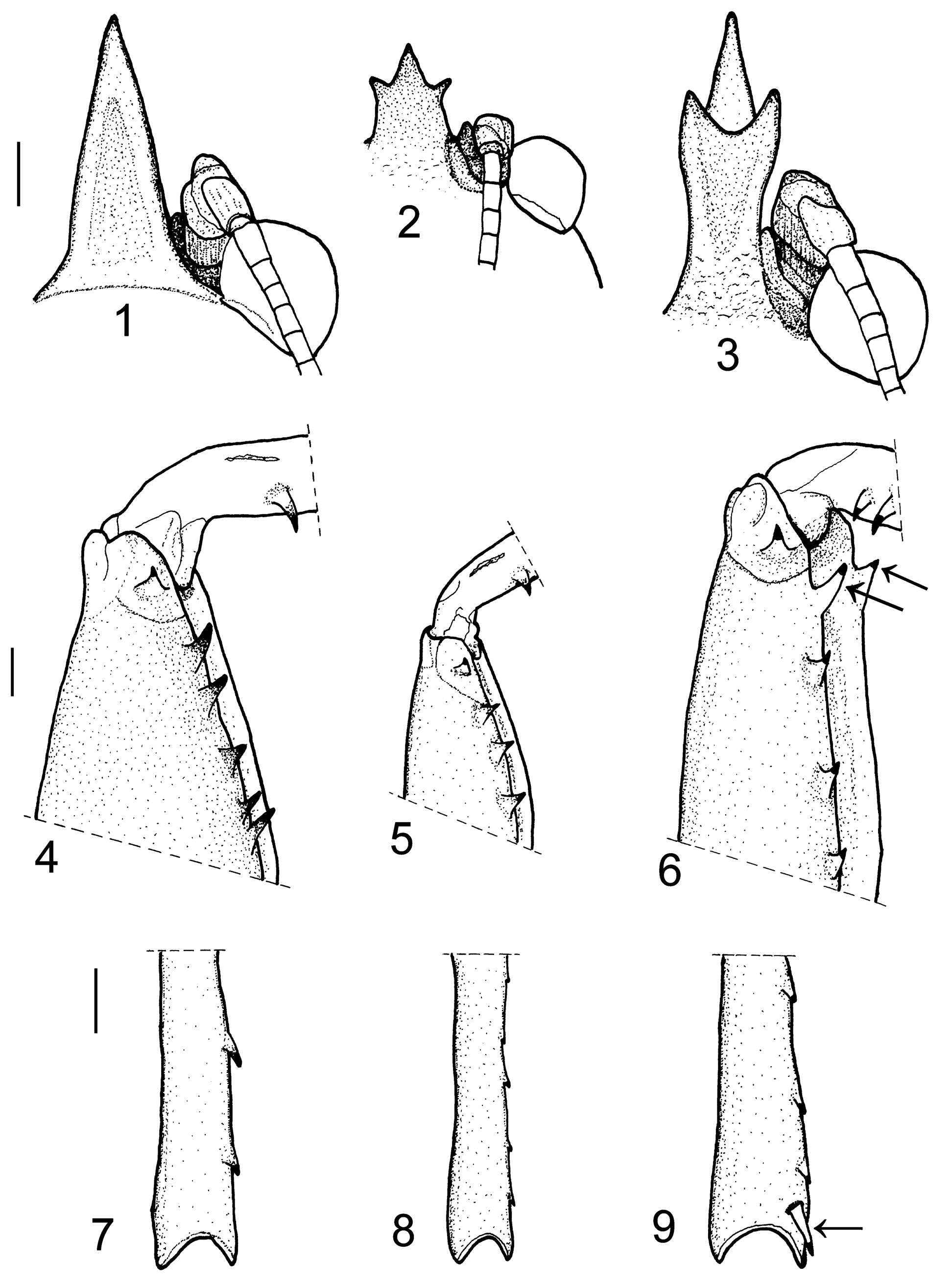
( Figs. 3, 9 View FIGURES 1 – 9 , 76–86 View FIGURES 76 – 86 , 111 View FIGURES 109 – 111 , 119–127 View FIGURES 112 – 119 View FIGURES 120 – 129 )
http://lsid.speciesfile.org/urn:lsid: Orthoptera .speciesfile.org:TaxonName:15119
Colossopus grandidieri: Saussure 1899: 628 View in CoL ; Kirby 1906: 270; Karny 1912: 39; Otte 1997: 27; Ünal & Beccaloni 2008: 35; Hollier & Heads 2015: 310.
Type locality. Madagascar. Lectotype male ( MHNG).
Redescription. Male: Body size large ( Tab. 4 View TABLE 4 ). Fastigium of vertex ( Figs. 3 View FIGURES 1 – 9 , 76 View FIGURES 76 – 86 ) forming a long horn with 3 arms, upper 2 arms short and equal, lower one long, placed between the upper arms, slightly downcurved; its basal part 1.7 times narrower than diameter of eye and 1.1 times wider than antennal scape. Fastigium of frons ( Fig. 77 View FIGURES 76 – 86 ) with a simple, spiniform tooth. Face strongly flattened with distinct irregular tubercles. Pronotum ( Figs. 76, 77 View FIGURES 76 – 86 ) large, almost as long as wide; 3.8 times longer than high; anterior margin convex, posterior margin with a deep angular median incision; prozona 2.8 times longer than metazona. Tegmina ( Figs. 76, 77 View FIGURES 76 – 86 ) protruding from under pronotum, reaching 2/3 of the way down metanotum; its upper surface wavy. Prothoracic auditory spiracle ( Fig. 79 View FIGURES 76 – 86 ) almost rounded, its dorsal half concealed under lateral lobe of pronotum in lateral view. Mesothoracic auricle ( Fig. 79 View FIGURES 76 – 86 ) long, its inner gap oval without or with an indistinct ventral lobe. Prosternum with 2 long spines, distinctly V-shaped. Fore femur ( Fig. 76 View FIGURES 76 – 86 ) with a large inner spine on anterio-lateral margin; with a large inner and a large outer apical spine ventrally (in one male with an additional ventral spine on outer side apically); with 4–5 additional ventral spines on inner side. Fore tibia ( Fig. 76 View FIGURES 76 – 86 ) with 5 very large inner and 6–7 smaller outer spines ventrally. Mid femur with 2–4 and hind femur with 2–4 ventral spines on outer side. Hind tibia with 5–7 dorsal spines on inner side, with a single apical spur on inner side dorsally ( Fig. 9 View FIGURES 1 – 9 ); with 4 apical spurs ventrally, inner two shorter than outer ones. Last abdominal tergite ( Fig. 80 View FIGURES 76 – 86 ) broad, posterior part elongated backward, with a deep narrow median incision. Cercus ( Fig. 81 View FIGURES 76 – 86 ) long, narrow and slender, apical third sharply narrowed with a large, slightly incurved apical tooth. Subgenital plate ( Fig. 82 View FIGURES 76 – 86 ) almost as long as wide; its styli long, much longer than depth of posterior incision.
Female: Head as in male, relatively slightly larger. Fastigium of vertex ( Figs. 83, 84 View FIGURES 76 – 86 ) as wide as half eye diameter, its basal part 1.1 times wider than antennal scape. Fastigium of frons ( Fig. 84 View FIGURES 76 – 86 ) with a simple, large, slightly downcurved tooth. Pronotum ( Figs. 83, 84 View FIGURES 76 – 86 ) short and broad, anterior margin convex, posterior margin broadly concave; prozona 1.4 times narrower and 3.9 times longer than metazona. Tegmina strongly reduced, like a very small scale. Dorsal half of prothoracic auditory spiracle covered by pronotum. Mesothoracic auricle not covered by pronotum. Fore femur ( Fig. 83 View FIGURES 76 – 86 ) with a large inner spine on anterio-lateral margin; with a large inner and a large outer apical spine ventrally; with 5–6 additional inner spines ventrally. Fore tibia ( Fig. 83 View FIGURES 76 – 86 ) with 5 inner and 6–7 outer strong spines ventrally. Mid femur with 3 and hind femur with 1–3 outer spines ventrally. Hind tibia with 6–8 dorsal spines on inner side, with a single inner apical spur dorsally; with 4 apical spurs ventrally, inner 2 slightly shorter than outer ones. Last tergite very short and broad, posterior margin with a narrow, median incision. Cercus conical, pointed at apex like a spine. Subgenital plate ( Fig. 86 View FIGURES 76 – 86 ) short and broad, with a wide median carina, its lateral lobes with transverse ridges. Ovipositor ( Fig. 85 View FIGURES 76 – 86 ) almost straight, 2–2.3 times longer than hind femur and 1.5–1.6 times longer than abdomen; apical part of ventral valve with small teeth.
Colour (dry specimens). Body dark, black and dark brown. Face dark reddish brown; labrum light brown; mandibles and upper surface of head black. Pronotum black with some irregular dark brown spots. Male tegmina, mesonotum and metanotum in both sexes yellowish milky brown. Forelegs reddish brown and reddish black. Mid and hind tibia yellowish milky brown. Abdominal tergites black or dark reddish brown in some specimens. Sternites yellowish milky brown in male, darker in female. Ovipositor black.
Diagnosis. The shape of fastigium of the vertex and the forelegs indicates that this species is closely related to C. redtenbacheri . It differs from it in the shapes of the pronotum, the male cercus and the ovipositor, in the ratios of ovipositor/hind femur and ovipositor/abdomen, in its distinctly darker body colour and also its larger size. It differs from C. parvicavus sp. nov. by its very larger body size, dark colouration, widely open prothoracic auditory spiracle, the shape of male pronotum, and the presence of ventral spines on the hind femur.
Material examined. Madagascar, 3 females (leg. M. Grandidier) (Paralectotypes); Prov. De Tulear, Bas Fiherana [ Fiherenana ], 1906, 1 male, 2 females (leg. F. Geay) ; Madagascar (S.O.), Forest du Fiherana [ Fiherenana ], 1905, 1 male, 1 male nymph (leg. F. Geay) ; Madagascar sud, 14.5. 1963, 1 female nymph (leg. D. Wintrebert) ; Madagascar, sud-wat, Efeda , 29.4. 1963, 1 female nymph (leg. D. Wintrebert) ; Madagascar, Fianarantsoa, Anpamaherana [ Ampamaherana ], station forestire, 1 female ; Madagascar, Tulear, Manombo , 10.5. 1956, 1 male (all in MNHN) . Madagascar, S. Centr. Mdg., 2 males; Toliara Province, Forest near village of Mangily, c . 27 km N of Toliara (-23.15, 43.60) [23 ° 8'60'' S, 43 ° 36'0'' E], 19.11. 2004, Coll. G.W. Beccaloni, 2 males, 1 female (all in NHMUK) . 1 female (det. Karny), without locality label (NMW). Madagascar, Toliara Prov., Forét de Tsinjoriaky 6.2 km 84 E Tsifota , elev. 70 m, 6– 10 Mar. 2002, 22 ° 48'8" S, 43 ° 25'14" E, 1 female (leg. Fisher, Griswold et al.), in spiny forest thicket GoogleMaps ; Madagascar, Toliara Prov., Lake Ranobe , elev. 20 m, 23 ° 02' 524" S, 43 ° 37' 214" E, 13– 21 May 2003, 1 male, 1 female (leg. Frontier, Wilderness), spiny forest ; Madagascar, Toliara Prov., Mahafaly Plateau, 6.2 km 74 ENE Itampolo , elev. 80 m, 21– 25 Feb. 2002, 24 ° 39'13" S, 43 ° 59'48" E, 1 female (leg. Fisher, Griswold et al.), in spiny forest thicket ( CAS). In addition, 2 adult females were photographed but not collected in spiny forest near Mangily, north of Toliara, Toliara Province by Martin Bader on 15.3.2015 (photographs and video footage were examined by the authors). They were found during the day hiding under bark attached to dead trees.
Measurements (mm). Lectotype (male): Body length: 53; pronotum length: 14; width of pronotum: 12.5; fore femur length: 16.9; width of fore femur: 6.2; fore tibia length: 14; hind femur length: 18. See Table 4 View TABLE 4 for the range of measurement of the other specimens including female.
Distribution. South-west and south-central Madagascar ( Fig. 111 View FIGURES 109 – 111 ). Toliara Province: Toliara, Fiherenana, Mangily, Efeda, Manombo, Tsifota, Lake Ranobe, NE Itampolo. Fianarantsoa Province: Anpamaherana [Ampamaherana].
Habitat. This species was collected in spiny forest. The specimens collected by the second author were found under loose bark still attached to dead trees.
Phenology. This species has been collected or observed between February and May: in November and March as adults, and in April and May as nymphs.
Remarks. The record for Anpamaherana [Ampamaherana] is dubious since this locality is located in the subhumid bioclimatic zone ( Cornet 1974), rather than the subarid zone like all of the other collecting localities for this species (see remarks for O. janetae ).
Life history. C. grandidieri has been bred in captivity by several hobbyists in Europe and to our knowledge it is the only one of the species discussed in this paper that has been studied in this respect. What follows is a compilation of information from several people who have reared this species.
C. grandidieri View in CoL is omnivorous, feeding on a wide range of leaves and fruit, as well as living and dead insects, and manufactured food, e.g. lettuce, Rubus and Quercus View in CoL leaves, rose flowers, zucchini, cucumber, banana ( Röhrs 2017), living crickets, cockroaches, moths and Zophobas morio larvae ( Fritzsche 2008), dried Tenebrio molitor larvae, dog food and fish flakes (Guillermo González, pers. commun., 2017). All instars of both sexes have raptorial forelegs, which are used to grasp living prey as well as being used for defence (see below).
The pale brown cigar-shaped eggs are deposited singly into soil ( Röhrs 2017) and females can lay 150–200 during their lifetime ( Tänzler 2012). Eggs measure c. 6mm in length when first laid ( Fig. 119 View FIGURES 112 – 119 right) and they increase in volume over several weeks ( Tänzler 2012), presumably by absorbing water from the surrounding soil ( Figs. 119 View FIGURES 112 – 119 left, 120). The eggs hatch in ca. 3 months, although some may take more than a year to emerge ( Röhrs 2017). Hatching appears to be triggered by alternating dry and wet conditions ( Röhrs 2017), so it is possible that the eggs which took a year to hatch did not receive the appropriate wet/dry regime.
First instar nymphs ( Fig. 121 View FIGURES 120 – 129 ) measure ca. 8mm in body length ( Tänzler 2012), are dark brown/black, and lack the turquoise markings on their hind- and midlegs which third instar and older nymphs and adults have. Third instar and older nymphs have pale markings on their pronota, legs and tergites ( Figs. 122–123 View FIGURES 120 – 129 ), whereas, in adults these markings are replaced by black ( Figs. 124, 126 View FIGURES 120 – 129 ). Nymphs take 4–5 months to reach adulthood and adults can live for about six months ( Tänzler 2012). It is unclear how many nymphal instars there are.
C. grandidieri is nocturnal and first and second instar nymphs hide singly in holes in dead bark during the day (Guillermo González, pers. commun., 2017). Older nymphs and adults will rest together during the day in groups, but smaller individuals may be eaten by larger ones ( Tänzler 2012). In their natural habitat adults of several species, including C. grandidieri , have been observed to rest by day as male-female pairs, never in groups of more than two individuals (see Discussion), so the aggregations seen in captivity may be the result of a lack of harbourages.
Adult males and females have very similar colouration, except that the labrum of the male is yellow-orange ( Fig. 127 View FIGURES 120 – 129 ), whilst that of the female is orange-red ( Fig. 125 View FIGURES 120 – 129 ). When disturbed adults and larger nymphs rear up, open their mandibles and raise and spread their forelegs ( Fig. 125 View FIGURES 120 – 129 ). Adult males stridulate loudly when in this defensive position (George Beccaloni, pers. obs.; Tänzler 2012; Guillermo González, pers. commun., 2017), whereas the adult females do not make any audible sound (Martin Bader, pers. commun., 2017; Guillermo González, pers. commun., 2017). If an aggressor approaches closely enough the insect will attempt to grab it with its forelegs and bite it. The threat display of only one other species, O. brongniarti , has been observed ( Fig. 115 View FIGURES 112 – 119 ). It was an adult female and the defensive display was the same as that of the female C. grandidieri (Martin Bader, pers. commun., 2017).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Colossopus grandidieri Saussure, 1899
| Ünal, Mustafa & Beccaloni, George W. 2017 |
Colossopus grandidieri: Saussure 1899 : 628
| Hollier, J. & Heads, S. W. 2015: 310 |
| Unal, M. & Beccaloni, G. W. 2008: 35 |
| Karny, H. H. 1912: 39 |
| Kirby, W. F. 1906: 270 |
| Saussure, H. 1899: 628 |