Renicola sp.
|
publication ID |
https://doi.org/ 10.11646/zootaxa.3784.5.5 |
|
publication LSID |
lsid:zoobank.org:pub:95E7BE61-021D-476D-89A9-73F22B39759A |
|
DOI |
https://doi.org/10.5281/zenodo.5062505 |
|
persistent identifier |
https://treatment.plazi.org/id/387F87C7-E410-EE63-FF34-0A68AD81A11B |
|
treatment provided by |
Felipe |
|
scientific name |
Renicola sp. |
| status |
|
Renicola sp. “martini”
( Figs. 3–5 View FIGURE 3 View FIGURES 4–5 )
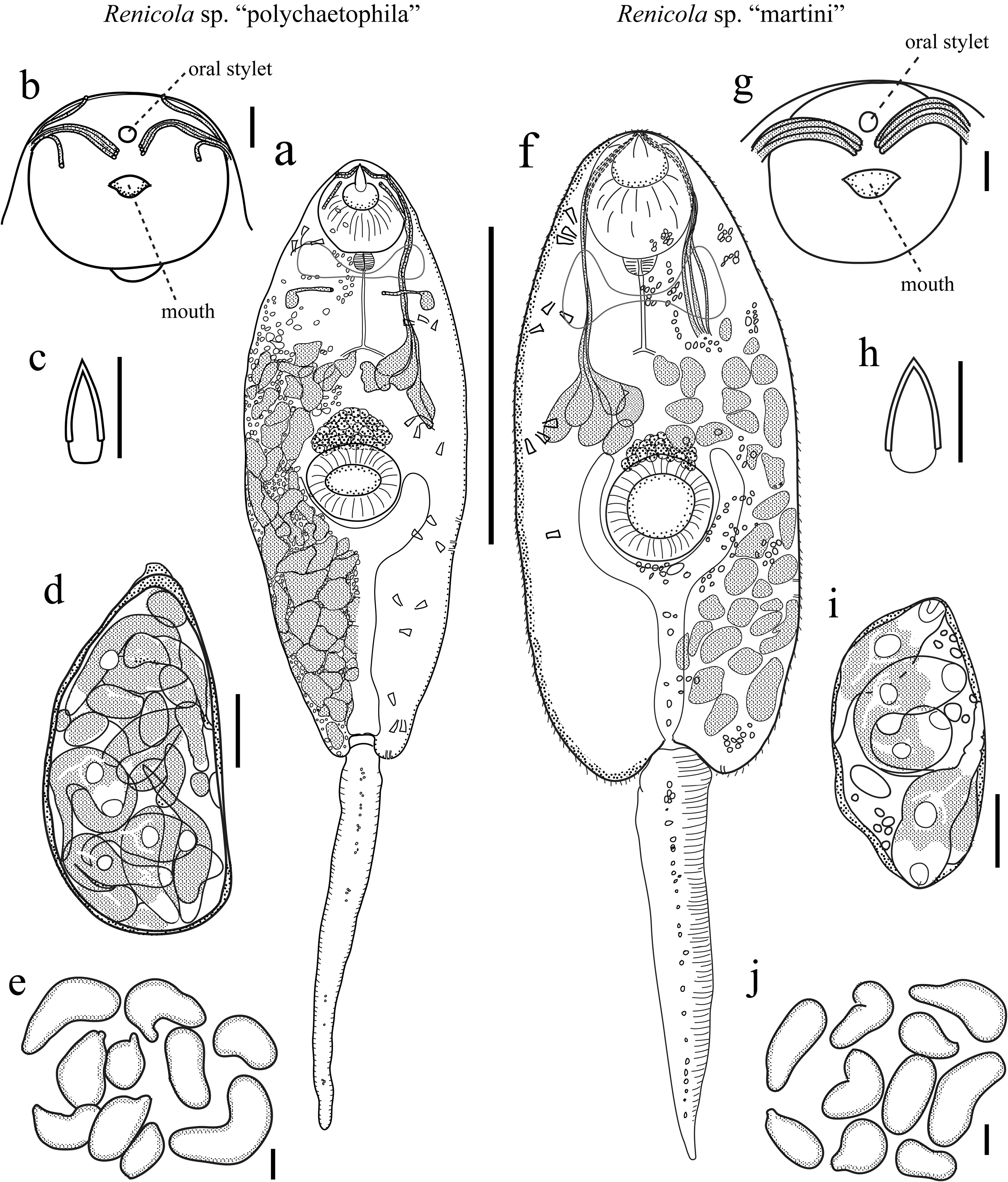
Diagnosis: Distome, leptocercous xiphidiocercariae with all penetration-gland ducts opening in single cluster at anterior oral sucker, with fine tegmental spines, with a simple, bullet-shaped stylet that is sclerotized for 50–80% of its length, and with an obscuring cystogenous-gland field with an anterior-most extent about half way between oral and ventral suckers. Cercariae produced by daughter sporocysts that comprise a colony that resides in the gonadal and digestive gland regions of first intermediate host.
Host: Cerithidea californica (Haldeman)
Location in host (daughter sporocysts): gonadal and digestive gland regions
Prevalence: 0.3% in 5392 snails dissected in 2012
Locality: Carpinteria Salt Marsh , Santa Barbara County, California, USA (34.4°N, 119.54°W) GoogleMaps
Habitat: Estuaries (intertidal flats, pans, channels)
Dates of collection: September–October 2009
Deposited material: Cercariae and sporocysts from 7 infections (USNPC nos. 107307–107318).
GenBank accession numbers: COI KF512553 View Materials – KF512560 View Materials ; ITS1 KF512561 View Materials – KF512568 View Materials
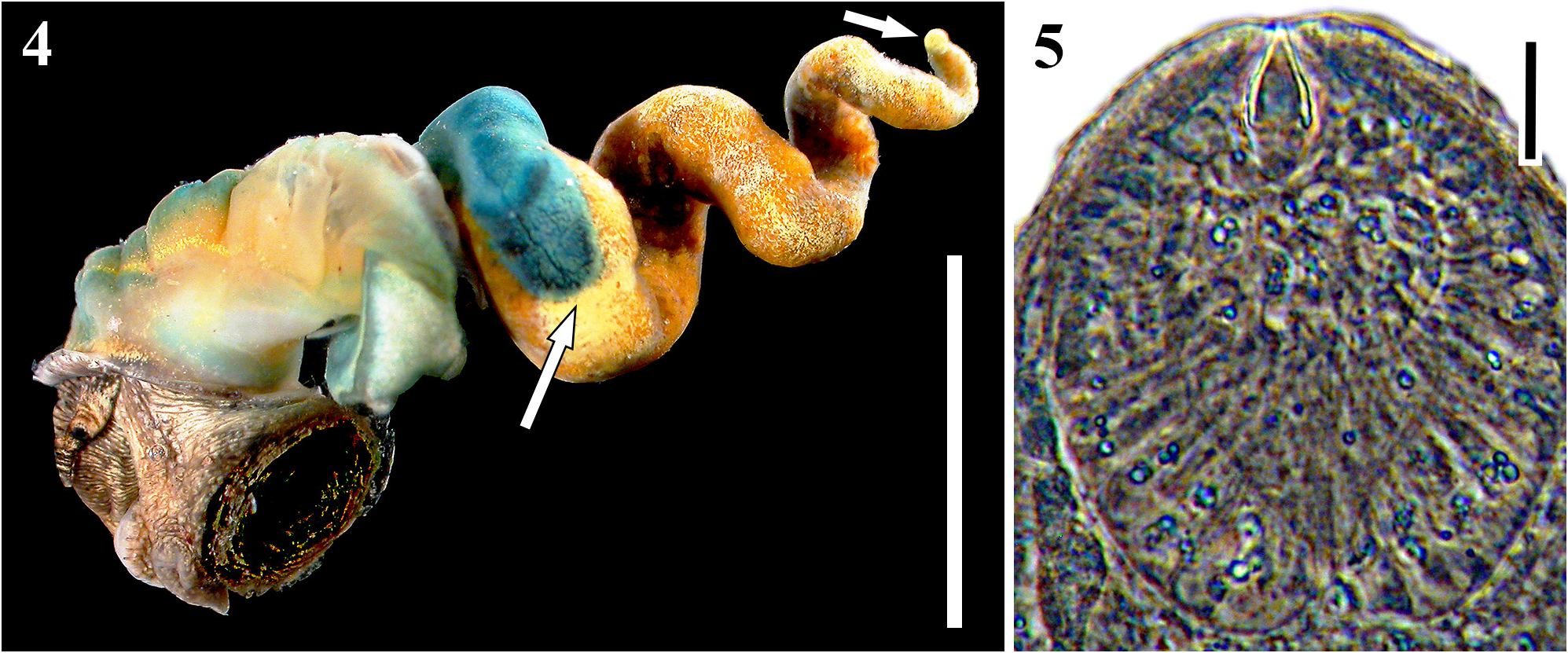
Description: Daughter sporocysts. Body immobile, shape variable, elongate to pyriform to spheroidal, sometimes pinched at one end to form a nipple or neck that appears to mark the birth pore. Tegument translucent, typically covered with thicker paletot that gives sporocysts lemon-cream to orangish appearance and causes them to stick together, and lined on interior by tissue that likely represents germinal cells. Ripe sporocysts with cercariae in various stages of development; germinal balls to developed cercariae distributed in sporocyst with no clear spatial polarity. Developed cercariae impart a whitish coloration to sporocysts under reflected light that mottles with the orange and clear tegument color. Distribution of sporocysts includes the snail gonadal space with some infiltration of digestive gland space. Sporocysts exhibit seasonal variation, wherein sporocysts in developed infections physically regress in winter, becoming thinner-bodied, somewhat flaccid, more orangish, with zero to few developed cercariae; sporocysts ripen up again during spring, becoming more plump, filled with more developed and developing cercariae, and together form a more compact mass. Morphometrics and meristics are presented in Table 1 View TABLE 1 , and are based on 30 sporocysts from three infections while cercaria counts are of the welldeveloped cercariae from 30 sporocysts from four infections.
Cercariae. Distome, leptocercous xiphidiocercaria. Body elliptic-elongate-ovate in dorsal view, dorso-ventrally flattened, maximal width and dorso-ventral height usually just anterior to ventral sucker; with refringent, varying sized spherules scattered throughout most of interior. Cystogenous glands fill posterior ~¾ of body, densely packed, filled with refringent granules, reflect much white light, obscure fine internal anatomy. Oral sucker welldeveloped, round, large, equal-subequal to ventral sucker; with small stylet; stylet embedded horizontally at anterior oral sucker, dorsal to mouth, small, usually appears less than ¼ length of oral sucker, sclerotized for 50- 80% of its length, bullet-shaped, tapering anteriorly from near base of sclerotized portion, difficult to carefully observe unless cercaria is flattened. Mouth subterminal; prepharynx absent; pharynx small, ovoid-spheroid, abutting posterior margin of oral sucker; esophagus thin and thin-walled, branching into two ceca approximately midway between oral and ventral suckers near anterior margin of cystogenous gland field; ceca thin and thinwalled, posterior-most extent obscured by cystogenous glands. Ventral sucker positioned at mid-body, well developed, round, protrudes from ventral surface, lacking spines. Tegument thin, with tegmental spines on ventral, dorsal, and lateral surfaces; tegmental spines fine, most readily apparent on non-flattened or slightly flattened specimens, distributed from anterior to posterior edge of body, particularly apparent laterally and dorsally, increase in size posteriorly. Penetration gland bodies extremely difficult to observe given obscuring cystogenous glands, but are primarily clustered ~2/5 into body from anterior edge, anterior extent of cluster close to anterior extent of cystogenous gland field; potentially more than seven pairs of unknown number of types. Penetration gland ducts pass anteriorly from glands in a single bundle per side; bundles pass around lateral margins of oral sucker, contain at least three individual ducts, empty anteriorly just ventral to oral stylet tip. Two pairs of lateral gland ducts of unknown type inconsistently observed opening at lateral body margins posterior to ventral sucker; gland bodies not reliably observed. One pair of posterior gland ducts of unknown type inconsistently evident opening at posterior body margin; gland bodies not reliably observed. Excretory bladder prominent, Y-shaped; stem longer than arms; arms embracing posterior half or more of ventral sucker margin; excretory opening at posterior edge of bladder stem. Flame cell arrangement likely 2 [(3+3+3) + (3+3+3)] = 36 bilateral arrangement, but posterior four groups typically obscured by cystogenous glands and posterior 3 groups never entirely observed. Other details of collecting ducts not observed. Cerebral ganglion just posterior of and extending laterally to pharynx. Genital primordium a compact mass of cells, adjacent to dorso-anterior margin of ventral sucker, very evident upon acetocarmine staining. Tail attached terminally at posterior body margin in socket, simple, unadorned, slender, cylindrical in cross-section, gradually tapering, can extend and contract, approximately equal in length to body when extended, with finely annulated tegument overlying muscle layers, with scattered, refringent spherules along mid-line. Morphometrics are presented in Table 1 View TABLE 1 and are based on 30–40 individuals from four infections.
Behavior: Cercariae swim by widening and flattening the mid- and posterior-body, flexing posterior body ventrally to partly overlap ventral sucker, often also slightly flexing or protruding anterior body, and lashing the tail left to right to move in a general anterior direction. Cercariae do not clearly respond to light.
Other biology: The trematode appears to usually infect the host by crossing the mid-gut, as expected given its taxonomic affiliation, and as evidenced by residual sporocyst or germinal material in the parenchymous tissue surrounding the mid-gut.
Developed infections parasitically castrate the host and appear usually to almost completely replace snail gonadal tissues. However, the seasonal (winter) regression, where cercaria production is greatly decreased and sporocysts become somewhat flaccid, is associated with infiltration of the parthenita colony by snail digestive gland tissue and sometimes a small amount of snail gonadal tissue regeneration (but there is no sign that snail reproduction takes place). These observations are consistent with the information provided in Hechinger et al. (2009) indicating that the aggregate sporocyst mass takes about 20% of the infected host soft tissue mass in summer and 16% in winter. That study may have pooled together this species and Renicola sp. “polychaetophila”, but the two species appear to be very similar concerning the way they grossly use host space.
Infections are sometimes encountered that are in the process of being invaded by other trematode species, particularly those with rediae, which can be observed ingesting the sporocysts and cercariae of Renicola sp. “martini”. This is consistent with the relatively low ranking of this species in the interspecific dominance hierarchy characterizing the guild of trematodes infecting the California horn snail ( Kuris 1990; Sousa 1993).
Field surveys and preliminary laboratory exposure experiments indicate that the cercariae encyst as metacercariae in the tissues of gastropods and potentially the hemocoel of various estuarine polychaetes (Hechinger and Smith (unpublished data); see list of possible hosts in Hechinger et al. (2011a)). The use of gastropods is further evidenced by cercariae encysting as viable-appearing metacercariae in the mantle tissues of snails with daughter sporocyst infections, particularly when those snails have been maintained out of water for days to weeks.
The final host species are uncertain, but almost certainly include birds that prey on polychaetes.
Geographic distribution: This species may occur throughout the entire range of the California horn snail, from North of San Francisco Bay to northern Peru ( Keen 1971; Miura et al. 2010). However, is has been positively identified from snails from Carpinteria Salt Marsh (the north part of southern California, USA) to the Estero de Punta Banda (northern Baja California, Mexico). In addition, preliminary genetic data indicates that this species occurs in the Atlantic Cerithidea pliculosa Menke (sister species to the California horn snail), in the Gulf of Mexico and the Yucatan Peninsula (Miura et al. unpublished data).
Etymology: The potential specific epithet, “martini”, represents an honorific for Dr. Walter Martin, who provided much of the taxonomic and lifecycle groundwork for the trematodes using the California horn snail as first intermediate host.
Remarks: Renicola sp. “martini” fits well within the diagnosis for renicolid cercariae (above), and the molecular results (below) further indicate that it is embedded within Renicola .
Table 2 lists attributes of previously described renicolid xiphidiocercariae. Renicola sp. “martini” is similar to most renicolid xiphidiocercariae in having a spinose tegument and having all penetration gland ducts opening very close to the stylet. It appears to be distinguishable from those similar species by having a combination of (1) possessing a simple, bullet-shaped stylet that is sclerotized for 50-80% of its length, and (2) in having the anteriormost extent of the cystogenous-gland field being about half way between the oral and ventral suckers.
Renicola sp. “martini” likely corresponds to what Hunter (1942) provisionally termed “Cercaria cerithidia 23” in her unpublished dissertation.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |