Tyrannosaurus rex
|
publication ID |
https://doi.org/10.1111/j.1096-3642.2007.00334.x |
|
DOI |
https://doi.org/10.5281/zenodo.8155288 |
|
persistent identifier |
https://treatment.plazi.org/id/364187B7-FFAC-A603-70B7-14DC362AFCC0 |
|
treatment provided by |
Jeremy |
|
scientific name |
Tyrannosaurus rex |
| status |
|
We examine the influence of neck muscle topology and kinematics on feeding function of Tyrannosauridae . Cervical musculature intrinsic to the neck and craniocervical muscles from the neck to the head, are here collectively termed ‘neck muscles’. Tyrannosaurid neck muscles are reconstructed by morphological comparisons with extant archosaurs, and their function and role in feeding behaviour are inferred by newly proposed modes of kinematic, electromyographic and observational inference. To ground the subsequent explorations of functional morphology, we first summarize fundamental divisions of neck axial musculature, and introduce inferential methods in detail.
FUNCTIONAL DIVISIONS OF AMNIOTE NECK MUSCLES
Amniote axial muscle groups are functionally and anatomically classifiable by serial homology and innervation ( Tsuihiji, 2005). Transversospinalis and longissimus muscles ( Fig. 1 View Figure 1 ) are primarily involved with dorsiflexion and lateral flexion, respectively, while the iliocostalis group ( Fig. 1 View Figure 1 ) brings about ventrolateral and, to a lesser extent, ventral flexion. Unsurprisingly, muscles running along the underside of the neck are responsible for most of the ventral flexion. These include the longus colli system and m. rectus capitis anterior/ventralis, serial homologues of prevertebral muscles such as m. psoas and m. iliopsoas ( Rosse & Gaddum-Rosse, 1997).
INFERRING FUNCTION OF NECK MUSCLES OF EXTINCT VERTEBRATES
The conservatism of neck muscle development, innervation and topology in extant amniotes engenders confidence that these general characteristics are also applicable to the muscles of extinct taxa. More specific reconstruction of cervical muscle morphology in an extinct taxon requires focused comparisons with its closest extant relatives. Dissection and osteological examination of extant relatives reveal constancy and variation in muscle origins and insertions. In a formalized extension of the comparative method ( Bryant & Russell, 1992; Witmer, 1995), osteological correlates of muscle attachment common to the fossil taxon and one or more extant groups indicate the muscle’s presence in the extinct form.
An extant member of the clade of interest, and the extant sister taxon of this couplet, form the extant phylogenetic bracket (EPB) of their extinct counterpart ( Witmer, 1995). An osteological correlate of a given muscle in all three taxa provides the most robust inference of its presence in the fossil animal (a Level I inference: Witmer, 1995). Less definitive evidence of correlates in any of the taxa, such as when there is no discrete attachment scar for a muscle, leads to inference of increasing ambiguity (Level II and III inferences: Witmer, 1995). If the extinct taxon shares a correlate with one, but not both, poles of the extant bracket, a Level II inference is possible by what is herein termed ‘unipolar’ comparison or inference. Extrapolatory inference of a muscle’s morphology is still possible given a unique correlate in the extinct taxon, through explicit mechanical considerations and morphological comparison with more distantly related clades ( Bryant & Russell, 1992).
KINEMATIC AND PHYSIOLOGICAL INFERENCE OF MUSCLE FUNCTION
Comparative morphology is the foundation for a hierarchy of inference ( Witmer, 1995) about how animals use their muscles, from the kinematic action of individual muscles moving origin and insertions together, to the integration of the work and power of multiple muscles towards a desired behavioural end. A further development of Bryant & Russell’s (1992) and Witmer’s (1995) methods enables inference of a muscle’s action and function. Function incorporates aspects of a structure’s form and physiology to define what the structure does and how it works ( Bock & von Wahlert, 1965). As the basic description of what muscles do, their action in extant vertebrates often is inferred based on the relative positions of a muscle’s origin and insertion and the effect of the resulting lever mechanics on skeletal movement (e.g. Burton, 1974). This method is reliable for inferring potential muscle action, although it often ignores the stabilizing action of shunt muscles that perform little work ( Rosse & Gaddum-Rosse, 1997) or the damping action of contralaterally contracting muscles ( Cleuren & De Vree, 2000).
Applying musculoskeletal lever mechanics to the inference of action in extinct taxa is here termed ‘kinematic inference’. This is an explicit subset of extrapolatory inference proposed by Bryant & Russell (1992). Similar muscle attachment topology and leverage in extant relatives allow kinematic inference of these moment relationships in the extinct form, by extant phylogenetic bracketing. However, if the muscle’s origin and insertion are reasonably deduced, the robustness of kinematic inference is independent of differences in skeletal morphology between the extant and extinct clades.
Non-topological characteristics also contribute to a given muscle’s action. Applying these factors to deduce muscle action in extinct animals is here termed ‘physiological inference’. Physiological tests, such as electromyography (EMG), magnetic resonance imaging (MRI) and assays for muscle fibre type, can reveal patterns of muscle activation, activity and force-generating capacity in extant animals. EMG records the occurrence, timing and general intensity of muscle activity. MRI is useful for deducing fibre direction and pennation, and hence muscle architecture, which influences force-generating capacity. Fibre types dictate speed, and hence power, of contraction and fatigability of the muscle. If homologous muscles in the extant bracket display similar in vivo characteristics, physiological inference will facilitate deduction of these features in their extinct relative.
Physiological inference is more sensitive to extant comparison than kinematic inference; differing muscle physiology in the extant groups will reduce confidence in the inference. Also, differential modifications in closely related extant taxa will render the inference more tenuous. Lability of physiological influences on muscle function is likely to be higher than with gross morphology, because muscles fatigue, recover and experience different metabolic regimes.
However, high potential for disparity increases the power of bracketed physiological inference if similarities between extant taxa are established confidently. Witmer (1995) established inference Levels I′, II′ and III′ for attributes that are variably present in extant comparative clades, but that have no osteological correlates. A strong, Level I′ physiological inference is possible when both poles of the extant bracket show homologous attributes of muscle activity. If tests have been carried out on members of only one clade of the extant bracket, ‘unipolar (Level II′) physiological inference’ will tentatively indicate presence of the same physiological characteristic in the extinct taxon.
While most aspects of physiological muscle function will be difficult to bracket in fossil forms, EMG offers a clear route to physiological inference. EMG activity is an immediate, measurable correlate of muscle activity as extant animals carry out behaviour in real time. If EMG of homologous muscles in crocodilians and birds reveals electrical activity during similar functions (such as dorsiflexion and lateroflexion), bracketed inference of these functions is strengthened for homologous muscles of tyrannosaurids. Strict morphological and physiological bracketing is ideal. However, if a Level II morphological inference ( Witmer, 1997) indicates the presence of a muscle in tyrannosaurids and only one member of the phylogenetic bracket, EMG can facilitate a Level II′ physiological inference in which tyrannosaurid muscle activity is less stringently inferred.
Physiological inference is testable and falsifiable by kinematic inference. Functional bracketing by EMG might signal inference of a function in the extinct form, but its moment arm topology would render this function impossible or unrealistically limited. An example would be EMG indicating a muscle’s activity strictly during head lateroflexion in crocodilians and birds (such as m. longissimus capitis superficialis and a hypothetical avian equivalent). Morphological and physiological bracketing would indicate this function in the carnosaur Allosaurus fragilis . The insertion of the muscle is lateral to the occipital condyle, which kinematically supports the physiological bracket. However, Bakker’s (2000) analysis of osteology indicates that this muscle has a long ventroflexive lever arm as well. By kinematic inference we would deduce that the muscle was capable of strong head ventroflexion in Allosaurus as well as lateroflexion, and that Allosaurus was more versatile than extant archosaurus when employing this muscle.
Conversely, physiological inference can strongly inform, and even falsify, kinematic inferences. This will occur when moment arms indicate that a muscle would facilitate a certain action, but EMG indicates that the muscle stabilizes a joint posturally or against the actions of other muscles. One example of this is the human m. brachioradialis. Lever arms might indicate that this muscle is capable of high-geared, rapid flexion of the elbow, but EMG indicates that it acts as a shunt muscle that stabilizes the joint during both rapid flexion and extension by the biceps and triceps, respectively ( Rosse & Gaddum-Rosse, 1997). Corroborating EMG of m. brachioradialis in gibbons would allow bracketing of its shunt function in fossil apes.
BEHAVIOURAL INTERPOLATION: INFERRING THE CONSEQUENCES OF MUSCLE FUNCTION
A collective subset of extrapolatory and phylogenetic inference ( Bryant & Russell, 1992), termed ‘extant behavioural interpolation’ or EBI, expands upon deducing the basic function of muscles to facilitate inference of faculty, biological role ( Bock & von Wahlert, 1965) and behaviour. EBI involves observing a behaviour in extant groups, correlating that behaviour with a reductive muscle function, and inferring the potential for that function and behaviour in the extinct taxon. The actual behaviour can be inferred in the extinct taxon only if the extant groups carry it out, using structural and physiological capabilities observed and inferable as common to all three clades. If a muscle’s morphology and action are known for extant groups, observations of their behaviour can elucidate how the muscle contributes to a given biological role. An example would be a muscle able to dorsiflex the head that facilitates inertial feeding in birds and crocodilians. If both birds and crocodilians use a homologous muscle for inertial feeding, and the muscle is inferred as being present in tyrannosaurids, the behaviour and biological role are also inferred as potentially present in tyrannosaurids. The inference is not absolute, as the bracket cannot predict loss of a function or behaviour in the extinct taxon.
The robustness of behavioural interpolation will be greatest when the EPB indicates similarity in the extant taxa of all correlates for activity, including kinematic morphology, muscle physiology and the behaviour itself. Potential behaviours are falsifiable with refined information on structure, and inference of overly specific behaviours will not be tenable. Inertial feeding may be acceptable as an inference involving herbivorous hadrosaurian dinosaurs, employing the extant bracket of crocodiles and eagles. However, morphological considerations rule out the inference of hadrosaurs tearing flesh ( Weishampel, 1983).
A muscle’s function for a given behaviour in extant animals is deducible from analysis of morphology and kinematics, physiological data (as from EMG), and observed behaviour. The degree of corrobaration for these phenomena, ranging from initial speculation to corroboration by multiple independent authors, can be plotted along respective axes to create a box that subtends a qualitative ‘behavioural inference space’ ( Fig. 2A View Figure 2 ). The size and shape of the box, respectively, represent the degree of certainty about a muscle’s behavioural role, and the relative contributions of observation, morphology and physiology to our understanding of that utility. A similar behavioural inference space for extinct taxa is shown in Figure 2B View Figure 2 . Two axes represent inferability of morphology (to a maximum Level I inference) and physiology (maximum of Level I′ without preserved correlates). The third axis represents certainty of kinematic interpretation, which is independent of specific morphologies in the extant relatives if EPB unambiguously suggests the muscle’s presence.
EBI facilitates inference of behaviour from bracketed muscle function, but the behavioural hypothesis becomes more useful and specific when tested biomechanically. Capabilities and limits of an animal’s behavioural performance are quantifiable through biomechanical methods, such as quasi-static ( Hutchinson & Garcia, 2001) and dynamic ( Snively, 2006) modelling and finite element analysis ( Rayfield, 2004).
Figure 3 View Figure 3 presents a scheme for arriving at the initial hypothesis of EBI based on preceding inferences. Behaviour is emergent from numerous factors in addition to muscle kinematics and physiology and will remain less testable than other inferences, unless other drivers of behaviour are shown to be homologous in the extant taxa. Nevertheless, these methods facilitate, as far as possible, reconstruction of neck muscles for tyrannosaurids, inference of their action and instigation of biomechanical hypotheses, and discussion of feeding variation within Tyrannosauridae and between tyrannosaurids and other large theropods.
MATERIAL AND METHODS
MUSCLE RECONSTRUCTION
Cervical and craniocervical muscles of tyrannosaurids were reconstructed with reference to dissections and literature descriptions of extant archosaurians, lepidosaurians and mammals, and identification of muscle scars on recent and fossil bone. The guiding principles for reconstruction were EPB and extrapolatory inference ( Bryant & Russell, 1992; Witmer, 1995). Through the EPB approach, muscles present in both crocodilians and modern dinosaurs are robustly inferred as being present in tyrannosaurids. Examination of successive extant outgroups to Archosauria, such as varanid lizards and mammals, reinforces the immediate bracket if a homologous muscle is present in more distant groups, and polarizes the sequence of a muscle’s evolution and modification. When precise homologies are ambiguous in any of the taxa, less certain reconstructions are possible through higher levels of inference (by greater osteological similarity between the extinct taxon and one of the extant groups; osteological and arthrological analogues; and mechanical considerations: Bryant & Russell, 1992; Witmer, 1995). Autapomorphic loss of a muscle is considered to be a less parsimonious interpretation in the absence of osteological evidence.
Specimens for dissection and osteological examination were obtained by the University of Calgary Museum of Zoology (UCMZ), primarily through the Calgary Zoo, Alberta Department of Fish and Wildlife, and Reptile World, Drumheller, Alberta. Two specimens of Varanus dumerilii were dissected and described as part of an assessment of intervertebral flexion (our unpubl. data). Dissection of commercially obtained, preserved domestic cats and three human cadavers (generously bequeathed to the Department of Cell Biology and Anatomy, University of Calgary Faculty of Medicine: UCDA), and examination of muscle attachments on cat and human skeletal preparations (UCDA, UCMZ), introduced the context of a more distal outgroup. Dissected crocodilians were Alligator mississippiensis and Caiman crocodylus , and muscle scars were assessed on skulls and cervical vertebrae of Caiman crocodylus (two), Alligator mississippiensis (two) and Crocodylus acutus . Birds dissected were Struthio camelus (two adults and two late-stage embryos), Leptoptilos crumeniferus , Pelicanus occidentalis , Cygnus columbianus , Cygnus buccinator , Anas platyrhynchos (two), Gallus gallus (three), Falco columbarius , Aquila chrysaetos , Haliaeetus leucocephalus , Asio flammeus , Halcyon cinnamomina and Pica pica . Skeletal material consisted of cervical vertebrae and skulls of skeletonized dissection specimens, and additional skeletal specimens of numerous avian clades curated at UCMZ. The Appendix lists the examined skeletal material of extant reptiles.
Specimens of tyrannosaurids and other Mesozoic theropods were examined at numerous institutions. These included specimens of Tyrannosaurus rex (six; seven if this taxon subsumes Nanotyrannus lancencis ), Tarbosaurus bataar (two), Daspletosaurus torosus (four, possibly including other Daspletosaurus species), Gorgosaurus libratus (four) and Albertosaurus sarcophagus (two). Non-tyrannosaurid specimens included many smaller coelurosaurs, and large noncoelurosaurian theropods including Acrocanthosaurus atokensis , Allosaurus fragilis , three Ceratosaurus species, Dilophosaurus wethereli and D. sinensis , Giganotosaurus carolinii , Monolophosaurus jiangi , Sinraptor dongi and Torvosaurus tanneri . The Appendix summarizes specimen information for examined fossil theropods.
MUSCLE SYNONYMIES AND NAMING CONVENTIONS
Neck muscles are complex and have a rich history of description in multiple languages. Consequently, consistency of muscle names between taxa, or even within well-defined clades, has been difficult for researchers to maintain. Avian muscle names, in particular, conflict with those for homologues in other amniotes, but precision within the avian literature is stabilizing with the adoption of the Nomina Anatomica Avium ( Baumel et al., 1993). To avoid ambiguity between conflicting names, for a given homologous muscle or bone of insertion in birds, crocodilians and tyrannosaurids we use both the term from Nomina Anatomica Avium ( Vanden Berge & Zweers, 1993) and the synonym most commonly applied for the muscle in other reptiles (usually from Nomina Anatomica Veteri- naria: ICVGAN, 1994). However, we do list syn- onyms and include their citation when authors diverge from the ICVGAN nomenclature. For muscles present in either birds or other reptiles but not universally present in sauropsids, we apply the name given to the muscle in the respec- tive group if correlates indicate its presence in tyrannosaurids.
Neck muscles have often been described as inserting on the exoccipitals. However, the exoccipitals and opisthotics are ambiguously differentiable in many dinosaurs, and the opisthotics may be more the prominent lateral components of the occiput (A. Carabajal, pers. comm.). We therefore follow Seidel (1978) and refer to these muscles as inserting on the paroccipital processes.
There is general agreement between the names used here for muscles and those in work recently published and in preparation by Tsuihiji (2002, 2003, 2005). Tsuihiji’s research encompasses the evolution and homologies of neck muscles throughout Sauria, while the present work emphasizes analogous functions of these muscles in tyrannosaurids and their extant relatives. With its broader systematic scope Tsuihiji’s nomenclature will be more comprehensively applicable, and should be adopted in the case of conflicting names.
Muscle descriptions of extant archosaurs and tyr- annosaurids are organized by topographic and func- tional divisions. Descriptions proceed from dorsal to ventral for transversospinalis, longissimus, iliocosta- lis and longus groups, starting with superficial and/or multiarticular muscles and subsequently dis- cussing uniarticular and deeper ones. Variants of each muscle are noted in extant clades, when pos- sible including Varanus dumerilii as a comparative outgroup. Kinematic and physiological correlates of muscle function are described. The inferred mor- phologies of muscles are then described for the Tyrannosauridae , and kinematic and physiological considerations are assessed so as to facilitate infer- ence of muscle function.
RESULTS
CONTINUITIES OF OSTEOLOGICAL MUSCLE ATTACHMENTS IN EXTANT ARCHOSAURS AND TYRANNOSAURIDS
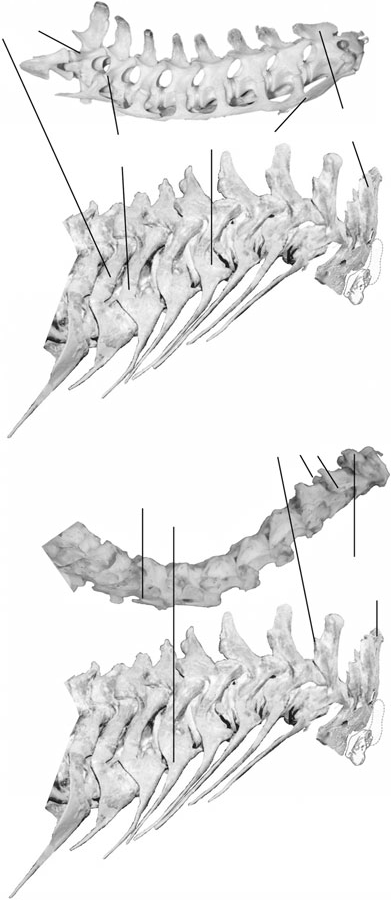
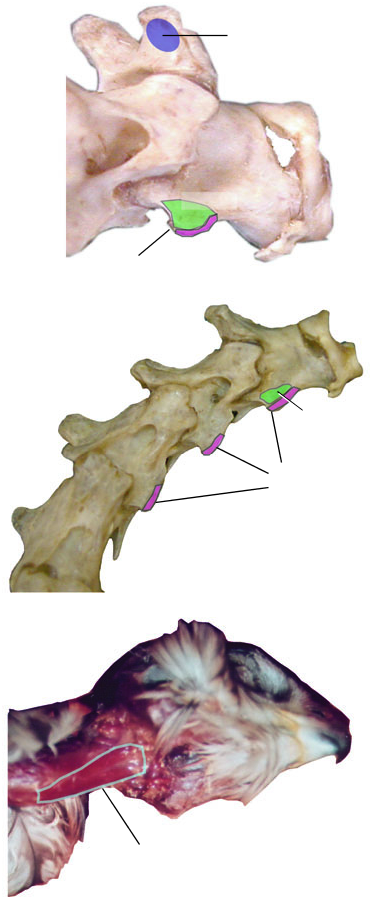
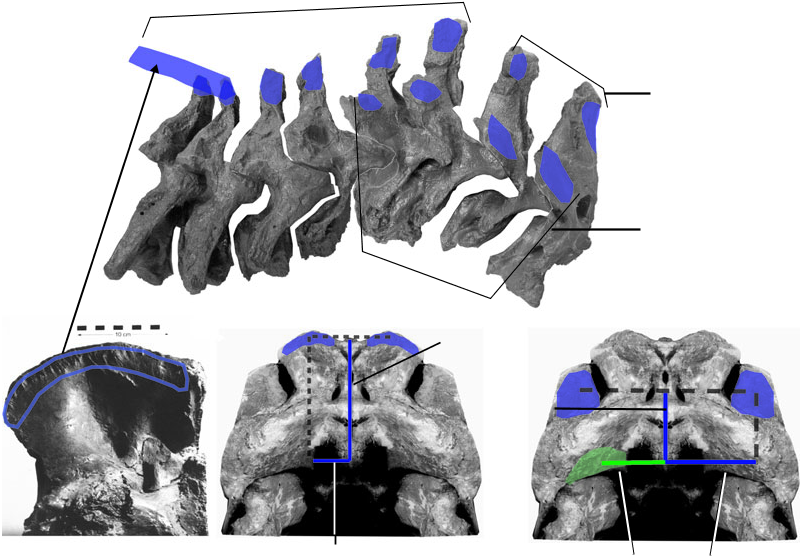
Figure 4 View Figure 4 depicts a comparison of muscle attachment sites on the cervical vertebrae of crocodilians and birds, respectively, with tyrannosaurids. Posteriorly, crocodilians and tyrannosaurids share long transverse processes with prominent diapophyseal articular surfaces for the cervical ribs. These osteological similarities indicate similar origins for muscles of the longissimus system, associated with the transverse processes. Birds are derived among archosaurs in lacking long transverse processes, which indicates modification of associated longissimus musculature. Also, unlike the condition in birds, neural spines of both crocodilians and tyrannosaurids are relatively tall throughout the cervical series, indicating similar origins for transversospinalis system components.
Conversely, anterior insertions and origins of transversospinalis derivatives are more similar in birds and tyrannosaurids than either are to crocodilians. In particular, the epipophyses (also called processes dorsales), projections above the postzygapophyses, are of the standard dinosaurian morphology in tyrannosaurids and birds, while crocodilians lack these projections. The C2 neural spine of birds and tyrannosarids is tall relative to the anteroposterior length of the centrum, again unlike the condition in crocodilians and suggesting similarity of associated muscles in the theropods.
Although tyrannosaurids share respective traits with crocodilians and birds, some aspects of their neck osteology are unlike those of either extant archosaur clade, and more like those of other large Mesozoic theropods. Shafts of the cervical ribs of most large theropods were long, attenuate structures, that extended posteriorly by the lengths of several posterior centra. In contrast, most cervical ribs of crocodilians have relatively short anterior and posterior projections that overlap each other. Only the C1–C3 ribs of crocodilians have long and posteriorly restricted shafts. In birds the cervical ribs are fused to the diapophyses as short costal processes. These differences indicate that the cervical iliocostalis muscles, associated with the cervical ribs, were distinct in tyrannosaurids and other large theropds from the condition in either clade of extant archosaurs. We now describe the neck muscles of extant reptiles, as the basis for inferring specific homologies, differences, and similarities between tyrannosaurids and their extant relatives.
EXTANT REPTILES
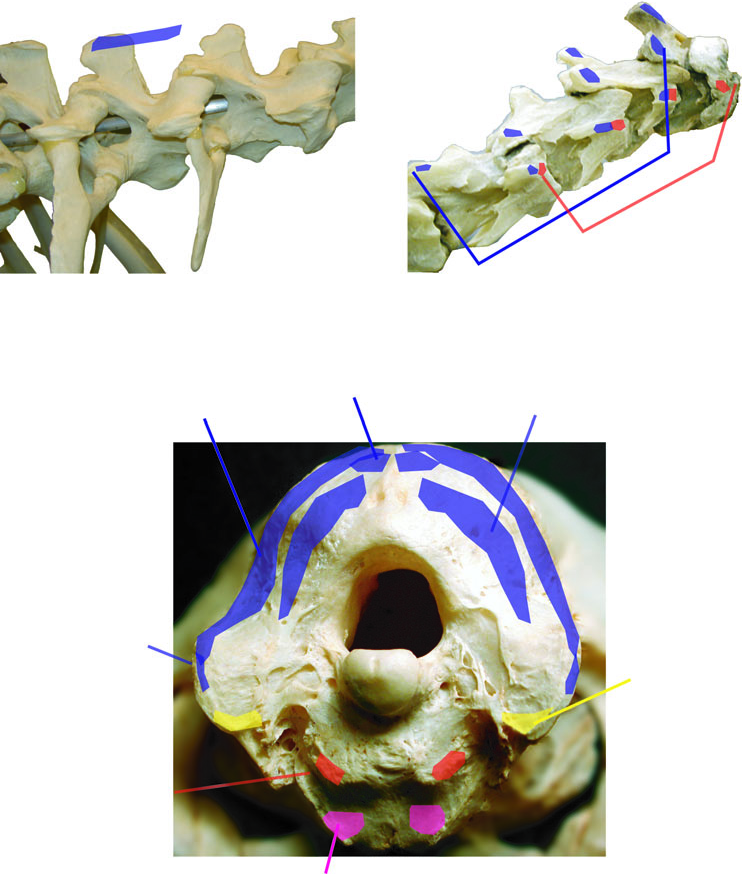
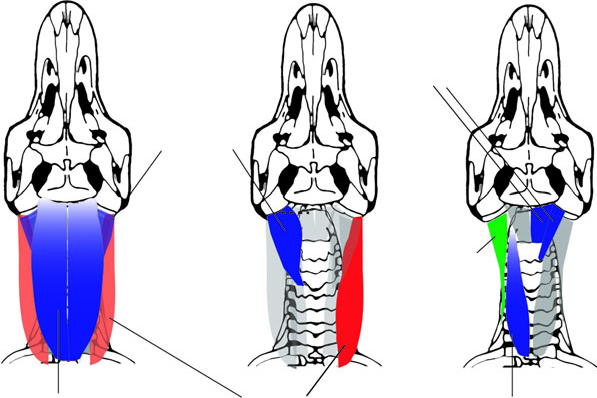
Tables 1 View Table 1 and 2 summarize origins and insertions of muscles of birds and crocodilians. Figure 5 View Figure 5 depicts attachments of muscles inserting on the occiput of birds, and Figure 6 View Figure 6 shows origins and insertions of most neck and craniocervical muscles of crocodilians. Some small, postural muscles that span intervertebral pairs in archosaurs, but that are variably present, are omitted from figures and descriptions. These include mm. interarticulares between postzygapophyses in crocodilians, and mm. interspinales that connect successive crocodilian neural spines ( Frey, 1988). Other muscles that may be uniquely configured in birds, mm. intercristales and mm. cervicales ascendentes, are treated last. The gross morphology of each muscle is figured first, and then origins and insertions are depicted. Several authors have presented excellent schematic and realistically drafed illustrations of archosaurian neck muscles ( Fisher & Goodman, 1955; Zusi & Storer, 1969; Burton, 1974; Seidel, 1978; Frey, 1988; Vanden Berge & Zweers, 1993; Cong et al., 1998; Cleuren & De Vree, 2000; especially comprehensive are those of Boas, 1929 and Tsuihiji, 2005). We present a photographic record of dissected reptiles, to highlight topographic and functional relationships between the muscles, and as as a replicative guide to corroborating or falsifying our interpretations.
A. M. transversospinalis system
M. biventer cervicis (m. biv. c.) ( Vanden Berge & Zweers, 1993). ( Aves.)
M. transversospinais capitis (m. trans. cap.) ( Seidel, 1978). ( Crocodylia .)
Origin: Each m. biventer cervicis of birds has two fusiform bellies positioned over the posterior and anterior parts of the neck that are connected by a long tendon ( Fig. 7A View Figure 7 ). The posterior belly originates ( Fig. 5A View Figure 5 ) either tendinously from the spinous processes of the posterormost cervicals, or from the medial portion of an aponeurosis in the anterior shoulder region, arising from the neural spines and transverse processes of T1, T2, and the posteriormost one or two cervical vertebrae. Because the aponeurosis is associated with the first thoracic vertebrae of the notarium, Vanden Berge & Zweers (1993) label it aponeurosis notarii. M. biv. c. is tightly attached to the portion of the aponeurosis notarii just adjacent to the neural spines.
A larger muscle lateral to the posterior belly of m. biv. c., m. longus colli dorsalis pars posterior (m. l.c.d.p.), can also originate from this aponeurosis However, its origin is more diffuse. The bellies of these two muscles are variably differentiable upon initial examination, because m. biv. c. is often small, and appears to grade laterally into m. l.c.d.p. until the fascia surrounding them are carefully dissected away. In larger birds, such as Aquila chryseatos , Leptoptilos crumeniferus and Struthio camelus , the origin of m. biv. c and its posterior belly are obvious, but their evidence is not solely size related. In Aquila chrysaetos ( Fig. 7A View Figure 7 ) the posterior belly is a long, moderately thick strap that covers the vertebrae over the posterior half of the neck. The kingfisher Halcyon cinnamomina has a large posterior belly, but the muscle is difficult to see in Pica pica and was not identifiable in an equivalently sized merlin, Falco columbarius .
.
The anterior belly of m. biv. c. originates from a long tendon ( Fig. 7A View Figure 7 ), anteriorly continuous with the posterior belly. The tendon and anterior belly run deep to the dorsal portion of a strong sheath of fascia that surrounds the dorsal neck muscles. This belly is sometimes discrete from other muscles, as in Struthio camelus , Aquila chryseatos and Pelicanus occidentalis , but in Pica pica it must be dissected free of m. complexus that runs lateral to it.
The homologous muscle in crocodilians and lizards, m. transversospinalis capitis, is large, continuous longitudinally without an intervening tendon, and defines the dorsal contour of the neck ( Fig. 7B, C View Figure 7 ). The origin of m. trans. cap. of crocodilians ( Fig. 6A View Figure 6 ) is more complex than it is in birds, and divides the muscle into lateral and deep portions. The lateral portion originates from tough fascia over the anterior thoracic vertebrae and C9, dorsal to the neural spine origins of m. rhomboideus thoracis. This fascia is probably homologous with the dorsal part of the aponeurosis notarii of birds. The medial portion of m. transversospinalis capitis originates as a set of fleshy slips from the dorsal tips of the C9–C2 neural spines. These slips coalesce laterally to form a continuous muscle belly.
Insertion: In both birds ( Fig. 5C View Figure 5 ) and crocodilians ( Fig. 6B View Figure 6 ), m. biv. c./trans. cap. usually inserts on the dorsomedial region of the occiput by a slender tendon (in some birds there is a larger fleshy insertion). The scar in birds is on the supraoccipital and medialmost portion of the posterior parietals, just lateral to the point at which the parietals often converge to a point dorsoposteriorly. In most birds the tendon dives deep to m. complexus and must be teased apart from this large muscle. The m. biv. c. tendon is visible anteriorly without dissection if m. complexus becomes slender in cross-section medially. In Pica pica the anterior belly of m. biventer cervicis merges with m. complexus, and shares the medial part of the aponeurosis of insertion for this muscle.
In crocodilians both the lateral and the medial portions of m. trans. cap. converge to insert by a tendon, in a fashion similar to the m. biv. c. insertion tendon of birds. In dissected specimens of Caiman crocodylus and Alligator mississippiensis the insertion was onto an embayment at the sutural confluence of the supraoccipital and squamosal. In the young dissected Alligator mississippiensis the tendon extended onto the posteroventral overhang of the parietals, differing slightly from the insertion usually reported ( Seidel, 1978; Cleuren & De Vree, 2000). The tendon forms the dorsalmost muscle insertion on the skull and is prominently visible, unlike in some birds where it is obscured by superficial muscle fibres of m. complexus.
Action/function: Because m. biv. c./m. trans. cap. inserts dorsal to the occipital condyle in both birds and crocodilians, it is able to dorsiflex the head relative to the level of the posterior cervicals ( Burton, 1974; Frey, 1988). Seidel (1978) and Cleuren & De Vree (2000) note subsidiary functions for lateral flexion and neck elevation in crocodilians. EMG reveals that this muscle is active during head–neck dorsiflexion for inertial feeding and opening the jaws in crocodilians ( Cleuren & De Vree, 2000), and for drinking and inertial feeding in birds ( Heidweiller, Lendering & Zweers, 1992; van der Leeuw et al., 2001).
The clear homologue of m. trans. cap. of Varanus dumerilii is morphologically similar to that of crocodilians, originating from the dorsolateral surfaces of the postatlantal neural spines and inserting medially on the posterior surface of the parietal. The muscle resembles that of crocodilians and is unlike the homologue in birds, with a fairly massive belly that is anteroposteriorly continuous. Another muscle belly (m. articulo-parietalis), inserts laterally on the parietals, but originates with components of the longissimus system.
M. complexus ( Vanden Berge & Zweers, 1993). ( Aves.)
M. transversospinalis capitis lateralis (m. trans. cap. lat.) ( Tsuihiji, 2005). M. spinocapitis posticus (m. sp. cap. post.) ( Seidel, 1978; Cleuren & De Vree, 2000), m. semispinalis capitis (m. semisp. cap.) ( Cong et al., 1998). Anterior portion: m. epistropheo-capitis lateralis (m. epist.-cap. lat.) ( Seidel, 1978; Cleuren & De Vree, 2000). m. rectus capitis dorsalis major (m. r.c.d.maj.) ( Cong et al., 1998) ( Crocodylia ). m. obliquus capitis magnus (m. obl. cap. mag.) ( Oelrich, 1956; ICVGAN, 1994; Herrell & De Vree, 1999). ( Varanidae )
Tsuihiji (2005) established homology of m. complexus of birds with m. transversospinalis capitis lateralis of crocodilians. M. trans. cap. lat. is described in the previous section. Other crocodilian muscles are described here with m. complexus, because they have similar insertions, actions and partly similar origins.
Origin: In dissected birds ( Fig. 7D, E View Figure 7 ) m. complexus originates from the epipophyses of C2–C4, and even C5 or C6 as in Gallus gallus ( Fig. 5B View Figure 5 ). In some birds (e.g. Aquila chrysaetos ), and in grebes and waders ( Zusi & Storer, 1969; Burton, 1974), m. complexus also originates from lateral tubercles associated with the transverse processes of C3–C4 or C5 ( Fig. 5B View Figure 5 ). Embryologically in chickens and quail the origins from the epipophyses of C4 and C5 are prominent, and lateral tubercle origins are ambiguous or absent ( Noden, 1983; Huang et al., 2000). Embryological studies of birds with lateral tubercle origins would shed light on coalescence of dorsal and ventral slips of the muscle in these forms.
In Caiman crocodylus and Alligator mississippiensis , m. sp. cap. post./m. semisp. cap. ( Fig. 7F View Figure 7 ) originates from the ventrolateral portions of the anterior neural arches ( Fig. 6A View Figure 6 ). M. epistr.-cap. lat./m. r.c.d.maj. ( Fig. 7F View Figure 7 ) originates from the anterodorsolateral surface of C2 ( Fig. 6A View Figure 6 ). In Varanus dumerilii m. obl. cap. mag. arises from the dorsal surfaces of the postzygapophyses, similarly to the epipophysis origins of m. complexus of birds.
Insertion: M. complexus has a fleshy or aponeurotic insertion medially on the parietals of birds ( Fig. 5C View Figure 5 ), and laterally can insert on the posterior surface of the squamosal (‘temporal’ sensu Zusi & Storer, 1969; Koch, 1973). The latter insertion represents a ventrolateral expansion of the muscle from the condition in 11-day-old chick embryos, in which the insertion is restricted to the dorsomedlal posterior face of the parietals ( Noden, 1983). The ventrolateral insertion is uniquely tendinous in Pelicanus occidentalis . In most birds dissected (Falconiformes: Aquila chrysaetos , Falco columbarius ; Pelicanus occidentalis , Struthio camelus and other large specimens), m. complexus forms a broad, dorsoventrally thick band, but in Pica pica it is more slender transversely and merges medially with m. biventer cervicis.
While m. complexus is the most superficial and prominent anterior craniocervical muscle in birds, fusion of bones in the occipital region has led to imprecise descriptions of its insertion. The posterior surfaces of adult avian squamosals are fused dorsomedially with the parietals, and ventromedially with the paroccipital processes. Hence the insertion of m. complexus has been identified as being solely on the parietals ( Fisher, 1961), or on the occipital crest, without reference to its constituent bones.
In dissected crocodilians m. sp. cap. post./m. semisp. cap. inserts via a broad tendon onto the paroccipital process ( Fig. 6B View Figure 6 ), medial to the insertion of m. longissimus capitis superficialis and dorsal to that of m. iliocostalis capitis/m. rectus capitis lateralis. The crocodilian m. epistr.-cap. lat./m. r.c.d.maj. inserts fleshily on the squamosal ( Fig. 6B View Figure 6 ), ventrolaterally to the tendon of m. spinalis capitis. M. obliquus capitis magnus inserts ventrolaterally on the squamosal of Varanus dumerilii . Similar insertions and partially similar origins indicate homology with the dorsal portion of m. complexus of birds.
Action/function: The insertion of m. complexus in birds is dorsal and dorsolateral to the occipital condyle, and this position imparts it with a long in-lever for dorsal and lateral flexion of the head relative to the axis. M. complexus is strongly active during the initial upstroke of the head in drinking in adult chickens ( Heidweiller et al., 1992), confirming the predicted dorsiflexive kinematics. Because m. complexus inserts lateral to the occipital condyle as well as dorsal to it, a kinematic role in head lateroflexion relative to the neck has been proposed ( Burton, 1974). This hypothesis has been corroborated electromyographically (our unpubl. data).
M. complexus has often been cited as the ‘piping’ or hatching muscle, through the action of which hatching birds break the shell with their egg tooth, or otherwise with their beak. Just prior to hatching in all taxa examined by Fisher (1961), the muscle distends by the infiltration of lymph rich in glycogen that is probably important for the muscle’s function. Lipar & Ketterson (2000) established that testosterone within the egg yolk of red-winged blackbirds ( Agelaius phoeniceus ) causes hypertrophy of m. complexus. Longer exposure to testosterone gives later hatchlings a larger m. complexus, and increases the effectiveness of their ‘begging’ behaviour by head dorsiflexion.
EMG of m. transversospinalis of crocodilians suggests that the lateral part of this muscle has a head dorsiflexive function homologous with that of m. complexus of birds, although EMG studies have not discriminated between lateral and medial divisions ( Cleuren & De Vree, 2000). All of the other muscles described in this section for crocodilians insert laterally and dorsally onto the occipital condyle, in such a position that they can impose more lateral flexion than dorsiflexion of the head relative to the neck. EMG of m. spinocapitis posticus/m. semispinalis capitis of crocodilians indicates that the muscle is active during multiple activities, but is especially active during lateral flexion of the cranium versus the neck ( Cleuren & De Vree, 2000).
M. splenius capitis (m. spl. cap.) ( Vanden Berge & Zweers, 1993). ( Aves)
M. obliquus capitis profundus (m. obl. cap. prof.) ( Tsuihiji, 2003), m. epistropheo-capitis medialis (m. epist.-cap. med.) ( Seidel, 1978; Cleuren & De Vree, 2000). m. altoïdius capitis ( Tsuihiji, 2005). m. rectus capitis dorsalis minor (m. r.c.d.min.) ( Cong et al., 1998) ( Crocodylia )
T. Tsuihiji (pers. comm.) was the first to establish the synonymy and homology of these muscles.
Origin: In dissected birds the origin of the medial belly of m. splenius capitis ( Fig. 7G View Figure 7 ) is demarcated by a teardrop-shaped, dorsally placed scar on the axis and sometimes C3 ( Fig. 5B View Figure 5 ). The equivalent muscle in crocodilians, m. epist.-cap. med. ( Fig. 7H View Figure 7 ), originates from the dorsal surface of the long, laterally compressed axial spinous process ( Fig. 6B View Figure 6 ).
Insertion: The insertions of these muscles in extant archosaurs are deep and medial to those of m. complexus and similarly inserting muscles. In birds m. spl. cap. has a fleshy insertion on a large area of the parietal, the dorsolateral portion of the paroccipital process, and lateral portion of the supraoccipital ( Fig. 5C View Figure 5 ). A lateral portion of this muscle, originating from C3, sometimes inserts onto the paroccipital process of birds (as in Struthio camelus : this study; Tsuihiji, 2005). In crocodilians m. epist.-cap. med. has a fleshy insertion along the suture between the squamosal and paroccipital process ( Fig. 5B View Figure 5 ), ventral to and more medially extensive than the insertion of m. epistropheo-capitis lateralis.
Action/function: M. splenius capitis of birds and m. epistropheo-capitis medialis of crocodilians insert dorsal to the occipital condyle, and lateral to the sagittal midline of the skull. Burton (1974) inferred kinematically that the muscle in birds is active during head dorsiflexion relative to the neck, and some capacity for lateral flexion is likely as well. The moment arms for both of these functions are shorter than those of the more superficial m. complexus, and m. spl. cap. may be effective in stabilizing the atlantooccipital joint. EMG of m. splenius capitis of chickens ( Heidweiller et al., 1992) confirms that the muscle is active during head dorsiflexion, although only when the muscle is unstretched. Whereas m. epist.-cap. med. of crocodilians is one of the smallest craniocervical muscles, the topology of its moment arms and presumably dorsi- and lateroflexive capacity are similar to those of m. spl. cap. of birds.
M. longus colli dorsalis (m. l.c.d.) ( Vanden Berge & Zweers, 1993). ( Aves.)
M. transversospinalis cervicis (m. trans. cerv.) ( Seidel, 1978; Cleuren & De Vree, 2000; Tsuihiji, 2005) ( Crocodylia , Aves.)
M. longissimus cervicis (m. long. cerv.) ( Frey, 1988; Cong et al., 1998). ( Crocodylia )
These muscles are clearly homologous in extant archosaurs ( Tsuihiji, 2005), but differ greatly in form and are best treated separately.
Aves: m. longus colli dorsalis
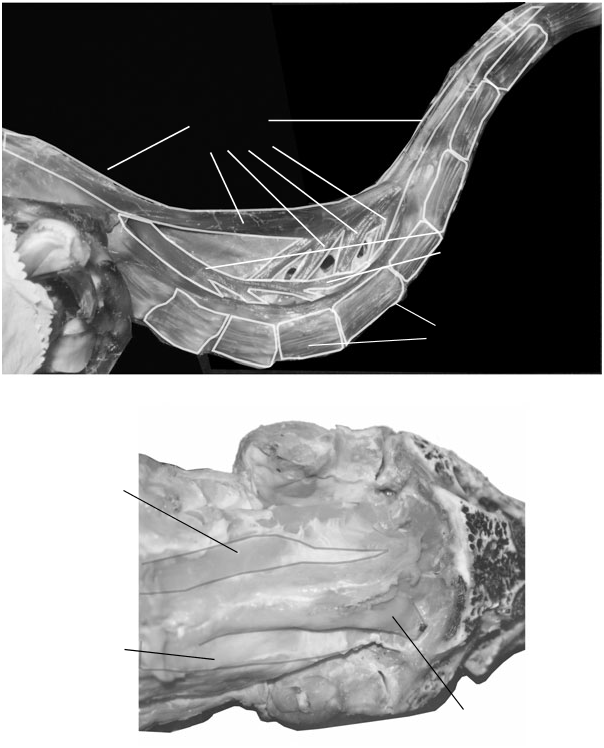
In birds m. longus colli dorsalis is a complex system involved in neck dorsiflexion ( Fig. 8A View Figure 8 ). It is divided into two major components – posterior and anterior, m. longus colli dosalis pars caudalis and m. longus colli dosalis pars cranialis. Collectively these muscles form large paired, fusiform masses that originate at the base of the neck ( Figs 8 View Figure 8 , 9B, D View Figure 9 ). Their anteriormost extents ( Fig. 9A View Figure 9 ) are the insertions of m. longus colli dorsalis pars anterior onto the epipophyses of the axis. The muscles are bound dorsally and laterally by a sheath of fascia (along with m. biventer cervicis), and contract and act on tendons of insertion within this sleeve. The profile of the posterior neck therefore follows the curve of the vertebrae, instead of filling out the entire dorsal concavity of this region.
M. longus colli dorsalis pars caudalis (m. l.c.d. caud.)
Origin: M. longus colli dorsalis pars caudalis ultimately originates from the aponeurosis notarii, arising from the neural arches and transverse processes of the anterior thoracic vertebrae and the last 1–3 cervical vertebrae, or tendinous slips arising from processes in this region ( Fig. 9D View Figure 9 ). Slips diverge from a dorsal tendon or muscle mass lateral to the m. biv. c., and are often suspended within fascia when separated from surrounding muscles.
Insertion: Usually slips of m. l.c.d. caud. insert medially to mm. cervicales ascendentes on the processes dorsales of the posterior cervicals, in the dorsally concave region of the neck ( Fig. 9D View Figure 9 ). However, they may share a tendon of insertion with a given m. cervicalis ascendens (as in the dissected Pelicanus occidentalis ) or even merge with bellies of mm. cervicales ascendentes (as in one adult specimen of Struthio camelus ).
M. longus colli dosalis pars cranialis (m. l.c.d. cran.)
Origin: M. longus colli dorsalis pars cranialis originates from the aponeurosis notarii or tendinously from neural spines of the cervicodorsal region ( Fig. 9B View Figure 9 ), and is fairly massive and fusiform where it is separable from the posterior part of m. longus colli dorsalis pars caudalis ( Fig. 8A View Figure 8 ). Other slips arise from epipophyses (processes dorsales) of cervicals from the posterior portion of the neck, medial to the insertions of m. l.c.d. caud. and mm. cervicales ascendentes ( Fig. 9C View Figure 9 ), and from epipophyses of some more anterior vertebrae.
Insertion: M. l.c.d. cran. inserts tendinously far anteriorly from its posterior origin, onto a posteriorly concave surface of the epipophysis of C2, and rarely C3 ( Fig. 9A View Figure 9 ), or onto the dorsal surface of these processes. The anterior slips of the muscle insert on this tendon as well. They thus run anterodorsally from a bony origin, contrasting with slips of m. l.c.d. caud. that run anteroventrally towards the posterior epipophyses as they diverge ventrally from the main belly of the muscle.
Crocodylia : M. transversospinalis cervicis (m. trans. cerv.) ( Tsuihiji, 2005; m. longissimus cervicis: Cong et al., 1998). M. longissimus cervicis (m. long. cerv.)/m. interarticulares ( Cleuren & De Vree, 2000; Tsuihiji, 2005)
These muscles have a confused nomenclature. They insert anteriorly at the same location, but are clearly discrete systems. They are treated together here.
Origin: In Alligator mississippiensis and Caiman crocodylus m. transversospinalis cervicis originates from the lateral surfaces of the neural spines ventral to the origins of m. transversospinalis capitis and m. spinocapitis posticus ( Fig. 6A View Figure 6 ), and just anterior to the postzygapophyses of C3–C9 ( Fig. 6A View Figure 6 ), by way of aponeuroses associated with the intermuscular septum that divides transversospinalis from longissimus systems. M. longissimus cervicis has similar aponeurotic origins from C4–C7 or C8, but also arises from the prezygapophyses of C4–C7 ( Fig. 9E View Figure 9 ).
Insertion: M. trans. cerv. and m. long. cerv. insert on the posterodorsal portion of the postzygapophysis of C1 ( Fig. 9F View Figure 9 ). M. trans. cerv. also inserts posteriorly on slightly raised scars in this position on the postzygapophyses of C3 and C4 ( Fig. 9F View Figure 9 ). The postzygapophyseal insertion is similar to the insertion of m. longus colli dorsalis pars cranialis of birds. However, these muscles of crocodilians bypass C2, the site of the most prominent insertion in birds.
The name m. longissimus cervicis associates this muscle with the longissimus system, and Frey (1988) and Cong et al. (1998) do not differentiate m. long. cerv. from m. trans cerv. These are reasonable assignations, given that the muscles run together lateral to the neural arches, and have somewhat lateral insertions. However, because the origins of both muscles are from the base of the neural arch and not the transverse processes, they belong topologically to the transversospinalis system rather than the more ventrolaterally placed longissimus group.
Action/function: In birds multiple slips of m. l.c.d promote dorsiflexion of each postatlantal cervical relative to the immediately posterior vertebra. The tendinous insertion of m. l.c.d. cran. on the axis facilitates dorsiflexion of the entire ventrally concave anterior portion of the neck. EMG activity indicates a damping function during neck ventroflexion in chickens and ducks, and strong functions in neck retraction and dorsiflexion ( van der Leeuw et al., 2001). In crocodilians m. trans. cerv. is in a position to dorsiflex the neck, an activity aided by the neck’s dorsally concave curvature. EMG activity in crocodilians confirms strong neck dorsiflexive action of this muscle ( Cleuren & De Vree, 2000). The function of m. long. cerv./m. interarticulares have not been tested by EMG.
B. M. longissimus system
M. longissimus capitis superficialis (m. long. cap. sup.) ( Crocodylia )
As discussed, the ventral portion of m. complexus of birds, arising from the lateral tubercles of the transverse processes and sometimes inserting fleshily on the paroccipital processes, may be the homologue of m. longissimus capitis superficialis of crocodilians. Unlike this muscle in crocodilians, the ventral m. complexus of birds is anteriorly restricted to C1–C4. This homology is tentative, because the ventral portion of the body of m. complexus occurs only in some birds, and is continuous with the dorsal part. In contrast the crocodilian m. long. cap. sup. is a large discrete muscle, and is described in detail here. Its morphology is depicted in Figure 10B View Figure 10 .
Origin: In Caiman crocodylus and Alligator mississippiensis , m. long. cap. sup. originates from the parapophyses and ventrolateral extent of the neural arches of C5–C9 ( Fig. 6A View Figure 6 ), dorsomedial to the origins of m. longissimus capitis profundus on C5–C7. On C8 and C9 the origins of m. long. cap. sup. origin extend ventrolaterally to cover the distal surface of the parapophysis. The origin was tendinous from C9 in the juvenile Alligator mississippiensis .
Insertion: The insertion of m. long. cap. sup. of crocodilians is tendinous onto the lateral extremity of the paroccipital process ( Fig. 6B View Figure 6 ), ventral to the origin of m. depressor mandibulae and dorsolateral to the insertion of m. rectus capitis lateralis. The scar is rugose in most specimens.
Action/function: Having the lateralmost insertion of any craniocervical muscle in crocodilians, m. longissimus capitis superficialis is kinematically positioned to impart a strong, laterally flexive moment on the skull relative to the neck. EMG reveals strong activity of m. long. cap. sup. during head lateral flexion on the ipsilateral side of the neck ( Cleuren & De Vree, 2000), with very little or no damping activity by the contralateral muscle.
M. rectus capitis dorsalis (m. r.c.d.) ( Aves). M. longissimus capitis profundus (m. long. cap. prof.) (Crododylia)
Origin: In birds, m. rectus capitis dorsalis ( Fig. 10A View Figure 10 ) typically originates from the anterolateral surface of C1, and the costal processes of the transverse processes of C1–C6 ( Fig. 5B View Figure 5 ). In crocodilians, m. longissimus capitis profundus originates from the transverse processes just dorsal to their articulations with the cervical ribs ( Fig. 6A View Figure 6 ).
Insertion: In both birds and crocodilians these muscles insert on the basioccipital, ventrolateral to the occipital condyle in crocodilians, and anteroventrolateral in birds ( Figs 4C View Figure 4 , 5B View Figure 5 ). The scar can be rugose in large crocodilians and birds, such as the dissected Caiman crocodylus and adult Struthio camelus, and on skulls of Alligator mississippiensis and Crocodylus acutus.
Action/function: In both crocodilians and birds m. r.c.d./m. long. cap. prof. insert ventral to the occipitial condyle, and are ideally positioned for head ventroflexion relative to the neck. EMG of crocodilians ( Cleuren & De Vree, 2000) confirms that the muscle is strongly active during this activity.
Mm. intertransversarii (mm. intertrans.) ( Vanden Berge & Zweers, 1993; Cong et al., 1998) ( Aves, Crocodylia , Varanidae ). Mm. inclusii ( Aves)
Mm. intertransversarii ( Figs 8A View Figure 8 , 11 View Figure 11 ) run between consecutive transverse processes in Aves, Crocodylia and Varanidae , and are here considered to be uniarticular components of the longissimus system. In this description the posterior transverse process of each pair is considered the origin, and the anterior one the insertion. This is not a universal practice in the literature ( Zusi & Storer, 1969), but the anterior vertebrae are less massive than posterior ones and are likely to be displaced more by contraction of mm. intertrans. ‘Insertion’ is a more appropriate designation for an attachment on the element that moves over greater excursion relative to the body’s centre of mass.
Mm. intertrans. vary substantially in extant sauropsids ( Figs 8A View Figure 8 , 11 View Figure 11 ). As described by Cong et al. (1998) they are dorsoventrally relatively thick bands in crocodilians, but are more dorsoventrally depressed in Varanus dumerilii . In birds these muscles are intricately subdivided and vary along the neck and among taxa. Only some of this variation is described here. Mm. inclusii, the medialmost components of this system, are present as short bellies in the posterior, dorsally concave portion of the neck.
Origin: In Aves bellies of mm. intertransversarii originate by aponeuroses from the lateral and dorsolateral tubercles ( Fig. 11A View Figure 11 ). The origins are more discrete in the anterior portion of the neck, although dorsally the aponeuroses must sometimes be dissected free of superficial aponeuroses of origin for other muscles. In the posterior part of the neck, aponeuroses of mm. inclusii originate from the anterior face of the costal prosesses of birds ( Fig. 11B View Figure 11 ). In crocodilians, the fleshy origins of m. intertrans. are deep and arise from along the anterior face of the transverse processes ( Fig. 11C View Figure 11 ). In Varanus dumerilii the origin is dorsoventrally shallower than it is in crocodilians, and is ventrolaterally restricted on the transverse process.
Insertion: In birds the insertions (anterior attachments) of m. intertransversarii are aponeurotic onto the lateral tubercles of the transverse processes ( Fig. 11A View Figure 11 ). Anteriorly, aponeuroses of mm. intertrans. to the dorsal and ventral lateral tubercles are separate, but posteriorly on the neck these aponeruroses appear to be conjoined dorsoventrally. In the anterior, dorsally convex portion of the neck in some taxa, long superficial bellies insert on the costal process of a vertebra, several cervicals anterior to the origin. This is an exception to the usually uniarticular morphology of the muscles. In the posterior portion of the neck, mm. inclusii insert on the lateral and dorsolateral crests of the posterior neural arch ( Fig. 11B View Figure 11 ). In crocodilians and Varanus dumerilii the insertions of m. intertransversarii are fleshy onto the transverse process of the anterior vertebrae of each pair ( Fig. 11C View Figure 11 ).
Action/function: In extant archosaurs and Varanus dumerilii , mm. intertrans. are positioned to laterally flex pairs of vertebrae relative to each other. The muscles are especially important for this function in birds, which lack the capacity for laterally flexing the entire neck by large superficial longissimus muscles present in other reptiles. Mm. intertrans. of birds spanning caudal vertebral joints are probably those most strongly involved in lateroflexion of the neck ( Heidweiller et al., 1992). In both crocodilians and varanids the transverse processes are laterally (and somewhat ventrally) extensive, indicating the capacity of effective intervertebral lateroflexion. Bilateral contraction might conceivably stabilize the intervertebral joints.
Interestingly, EMG of dorsal slips of anterior mm. intertrans. of adult chickens ( Heidweiller et al., 1992) shows intense activity during intervertebral dorsiflexion. This compensates for other neck dorsiflexors, which have proportionally smaller cross-sectional area than they do in juveniles ( Heidweiller et al., 1992).
C. M. iliocostalis and m. longus systems M. rectus capitis lateralis (m. r.c.l.) ( Vanden Berge & Zweers, 1993; Cong et al., 1998). Aves, Crododylia M. iliocostalis capitis (m. il. cap.) ( Seidel, 1978; Cleuren & De Vree, 2000). Crocodylia
Origin: In birds, m. rectus capitis lateralis ( Fig. 10A View Figure 10 ) originates from the lateral surface of an enlarged hypopophysis of C2 ( Fig. 12A, B View Figure 12 ) posterior to the origins of m. rectus capitis ventralis, or from the ventrolateral surface of the centrum. In dissected birds m. r.c.l. never originated from the costal process of C2, homologous with the vertebra’s cervical rib in other amniotes. This represents a dramatic medial shift from the muscle’s likely plesiomorphic origin in Archosauria that is retained in crocodilians.
In Caiman crocodylus and Alligator mississippiensis , m. iliocostalis capitis ( Cleuren & De Vree, 2000)/m. r.c.l. ( Cong et al., 1998) originates ventrally from strong white fascia surrounding ribs of C1 and C2 ( Fig. 6A View Figure 6 ). Because the C1 rib is the ventralmost of the pair, the m. iliocostalis capitis origin appears to arise strictly from the C1 rib. Palpation of the ribs and incision into the fascia reveal the fascia’s continuity with the origin of m. iliocostalis capitis.
Insertion: In both birds and crocodilians m. r.c.l./m. il. cap. usually has a flat tendinous insertion along the ventral edge of the paroccipital processes ( Figs 5C View Figure 5 , 6B View Figure 6 ). In crocodilians the muscle is a mediolaterally broad band, but in most birds (e.g. Pelicanus occidentalis ) the belly of m. rectus capitis lateralis is dorsoventrally deep where it runs lateral to m. rectus capitis dorsalis. The insertion of this muscle in Varanus dumerilii is onto the basioccipital, ventromedial to the position of insertion in extant archosaurs.
Action/function: In extant archosaurs this muscle is well positioned to impose lateral flexion of the head versus the neck. EMG of crocodilians indicates that it is also active during head rotation, and during elevation, when it may serve a damping function for head dorsiflexors ( Cleuren & De Vree, 2000).
M. rectus capitis ventralis (m. r.c.v.) ( Vanden Berge & Zweers, 1993; Cong et al., 1998). ( Aves, Crododylia )
M. longus capitis ( Cong et al., 1998). ( Crocodylia ) M. flexor colli ( Vanden Berge & Zweers, 1993). ( Aves)
These muscles are associated with the processus spinosus ventralis and ventral surfaces of the cervical centra.
Origins: In birds, lateral and medial parts of m. rectus capitis ventralis (m. r.c.v.; Fig. 12 View Figure 12 ) often originate from the ventral surface of processus spinosus ventralis (hypopophysis) of each vertebra from C2 to C5 or C6 (see Fig. 18 View Figure 18 ). These slips coalesce to form a large muscle in many birds; it is especially large relative to the size of the head in Falco columbarius .
In Caiman crocodylus and Alligator misssippiensis the origin of m. rectus capitis ventralis is restricted to the ventrolateral surfaces of the centra of C1 and C2 ( Fig. 13E View › ). The origins of m. longus capitis are from the lateral surfaces of the processus spinosus ventralis of C3–C7, forming a large, anteriorly coursing belly.
M. flexor colli of birds typically originates aponeurotically from the ventrolateral surfaces of posterior vertebrae, associated with processus caroticus, when present, or with the processus spinosus ventralis.
Insertions: In the dissected birds the insertion of m. r.c. v. was typically anteroventral to that of m. longissimus capitis profundus, onto the basitemporal plate ( Fig. 5C View Figure 5 ). In the crocodilians the insertion of m. r.c.v was onto the posteroventral surface of the basioccipital tuberosities ( Fig. 6B View Figure 6 ), ventral to the insertion of m. longissimus capitis profundus. The insertion of m. longus capitis appeared to be continuous dorsally with that of m. r.c.v. ( Fig. 6B View Figure 6 ) The insertions of m. flexor colli of most dissected birds were onto the posterior surfaces of processus spinosus ventralis on vertebrae anterior the origin.
M. r.c.v. of Varanus dumerilii is similar to that of crocodilians. M. flexor colli is similar to that of birds, but consists exclusively of uniarticular bellies running from the lateral surface of the processus spinosus ventralis of a posterior vertebra to the posterior surface of the preceding processus spinosus ventralis.
Action/function: The insertion of m. r.c.v. is ventral to the occipital condyle in crocodilians and anteroventral in birds, which indicates that the muscle ventroflexes the head relative to the vertebral column. The insertion of m. longus capitis of crocodilians is closer to the occiput than that of m. r.c.v., and, as with several such muscles in crocodilians ( Cleuren & De Vree, 2000), probably acts to stabilize the craniocervical joint during vigorous feeding behaviour.
Slips of m. flexor colli of birds (and Varanus dumerilii ) are well positioned to ventroflex intervertebral cervical joints that they cross.
M. longus colli ventralis (m. l.c.v.) ( Vanden Berge & Zweers, 1993). ( Aves)
M. iliocostalis cervicis (m. il. cerv.) ( Seidel, 1978), m. longus colli ( Cong et al., 1998). ( Crocodylia )
As with the major neck dorsiflexor (m. longus colli dorsalis/m. transversospinalis lateralis), m. longus colli ventralis/m. iliocostalis cervicis is greatly elaborated in birds relative to the condition in crocodilians. Its origins and insertions are considered separately for the extant taxa.
Origins: M. longus colli ventralis is divided in birds into multiple sets of interconnected origins, intervening tendons, bellies and insertions, each spanning several vertebrae ( Fig. 13 View › ). The posteriormost extent of a given system is usually a fleshy origin from the processus spinosus ventralis or sublateral process of a vertebra ( Fig. 13B View › ). (The hypopophyses are usually large and medially placed fusions of processes caroticales, while the sublateral processes are smaller and paired.) More than one slip complex of m. l.c. v. can arise from a given origin, and more anteriorly originating, smaller bellies can insert on intervening tendons of a given slip. Posteriorly slips can originate from the body of the centrum. In Leptoptilos crumeniferus large fusiform bellies arise from the ventral surfaces of mid and anterior thoracic vertebrae of the notarium, deep in the thoracic region ( Fig. 13A View › ).
In crocodilians, parts of the m. iliocostalis cervicis/m. longus colli (m. il. cerv.) system originate from the hypopophyses and ventral surfaces of the centra of the anterior thoracic and posterior cervical vertebrae ( Fig. 13D, E View › ). Dorsal origins occur on the capitula of the ribs; Seidel (1978) reported origins from the anterior portion of the myoseptum associated with each rib.
Insertions: Slips of m. l.c.v. insert by long tendons, 3–10 vertebrae anterior to their origin, with slips situated anteriorly crossing fewer joints than more posterior slips. These tendons run anterolaterally to insert on the cervical ribs ( Fig. 13C View › ) if these are well developed, or on the postlateral processes. Origins and insertions are difficult to tease apart in smaller birds.
In crocodilians, m. il.cerv. inserts ventrally onto the posterior processes of the cervical ribs ( Fig. 13D View › ), and dorsally by short slips onto the myosepta of ribs just anterior to their origin. The anteriormost insertion is by a white aponeurosis onto the rib of C1.
Action/function: The multiple slips of m. l.c.v. of birds potentially allow complex intervertebral ventroflexion. Anterior and posterior portions of the system are active during both approach and retraction phases of pecking and filter feeding, acting to ventroflex appropriate portions of the neck relative to posterior vertebrae, but also to damp the actions of dorsiflexive muscles ( van der Leeuw et al., 2001). In ducks the anterior and posterior slips fire asynchronously during approach, assisting with the complex rolling neck kinematics of feeding ( van der Leeuw et al., 2001).
Because insertions of m. il. cerv. in crocodilians lie lateral to the midline of the neck, kinematically the muscle complex appears capable of neck lateral flexion versus the trunk by unilateral contraction. The insertions are ventral to the intervertebral articulations, indicating the potential for neck ventroflexion as well. EMG of m. iliocostalis cervicis of crocodilians does indicate intense muscle activity during lateral flexion of the neck ( Cleuren & De Vree, 2000). However, the muscles are not active during ventroflexion, and instead show damping and stabilizing activity during dorsiflexion ( Cleuren & De Vree, 2000).
D. Complex and uniarticular muscles
Birds have several neck muscles with a distinct morphology that span one or more intervertebral joints.
Mm. intercristales ( Vanden Berge & Zweers, 1993)
Mm. intercristales appear quite large in birds, covering much of the dorsal surface of each post-C2 vertebra lateral to the spinous processes ( Fig. 14A, B View Figure 14 ). They may be medially displaced homologues of mm. interarticulares of crocodilians, which strictly span the postzygapophyses, while mm. intercristales run between transverse oblique crests of vertebral pairs.
Origins: Each m. intercristalis originates from the anterior surface of the crista transversoobliqua of the posterior vertebra of each pair ( Fig. 14C View Figure 14 ). Sometimes part of the origin is from the epipophysis as well.
Insertions: Each m. intercristalis inserts onto the posterior edge of the crista transversoobliqua of each vertebra pair, medial to the epipophysis ( Fig. 14C View Figure 14 ).
Action/function: The insertions of mm. intercristales are dorsal and slightly lateral to the probable respective centres of rotation of each intervertebral pair. They are kinematically positioned for intervertebral dorsiflexion, and may play a role in stabilizing the intervertebral joints.
Mm. cervicales ascendentes (mm. cerv.
asc.)
( Vanden Berge & Zweers, 1993). ( Aves)
Tsuihiji (2002) identified mm. cervicales ascendentes ( Fig. 14B View Figure 14 ) as being homologous with the most lateral occurrence of the m. transversospinalis system of the trunk of crocodilians, the m. tendinoarticularis trunci (probably synonymous with m. multifidis spinae of Cong et al., 1998), subsumed into m. transversospinalis cervicis in the crocodilian neck. Dissection confirms that these muscles do not occur discretely in the neck of Alligator mississippiensis or Caiman crocodylus , nor in Varanus dumerilii . In birds, slips of each m. cervicalis ascendens are traceable posteriorly from their common insertion back to their origins at least two vertebrae posteriad.
Origins: In birds slips of these muscles originate from the dorsal surfaces of the transverse processes just ventral to the postzygapophyses, or aponeuroses associated with the transverse processes ( Fig. 14C View Figure 14 ). The anteriormost extent of the origins is typically caudal to the strong anterior ventral curvature of the neck.
Insertions: Slips of each m. cervicalis ascendens converge onto a processus dorsalis/epipophysis at least two vertebrae anterior to its origin ( Fig. 14C View Figure 14 ). The insertion is sometimes shared with an insertion of m. longus colli dorsalis pars cranialis, and sometimes is medial to the epipophysis, onto the transverse oblique crest connecting the postzygapophysis to the neural spine.
Action/function: The insertions of mm. cerv. asc. are positioned dorsal and lateral to the likely centres of rotation of each vertebral pair. Heidweiller (1992) reports that mm. cervicales ascendentes contract during neck dorsiflexion in adult chickens, compensating for the m. longus colli dorsalis that is relatively smaller than it is in juveniles. Mm. cerv. asc. are extraordinarily massive in the posterior part of the neck of Aquila chrysaetos . Their large cross-sectional areas indicate forceful dorsiflexion of this part of the neck relative to the trunk, expected in a raptorial bird that tears carcasses of large prey. Other dorsiflexors, of both the neck and the cranium, are large in this taxon as well.
RESULTS: RECONSTRUCTED NECK MUSCULATURE OF TYRANNNOSAURIDAE
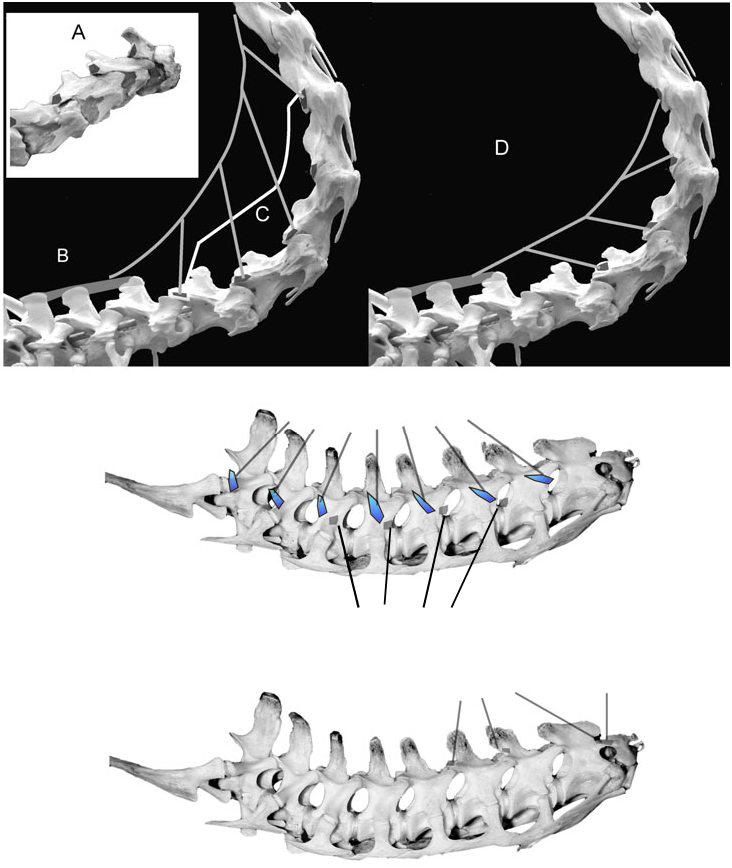
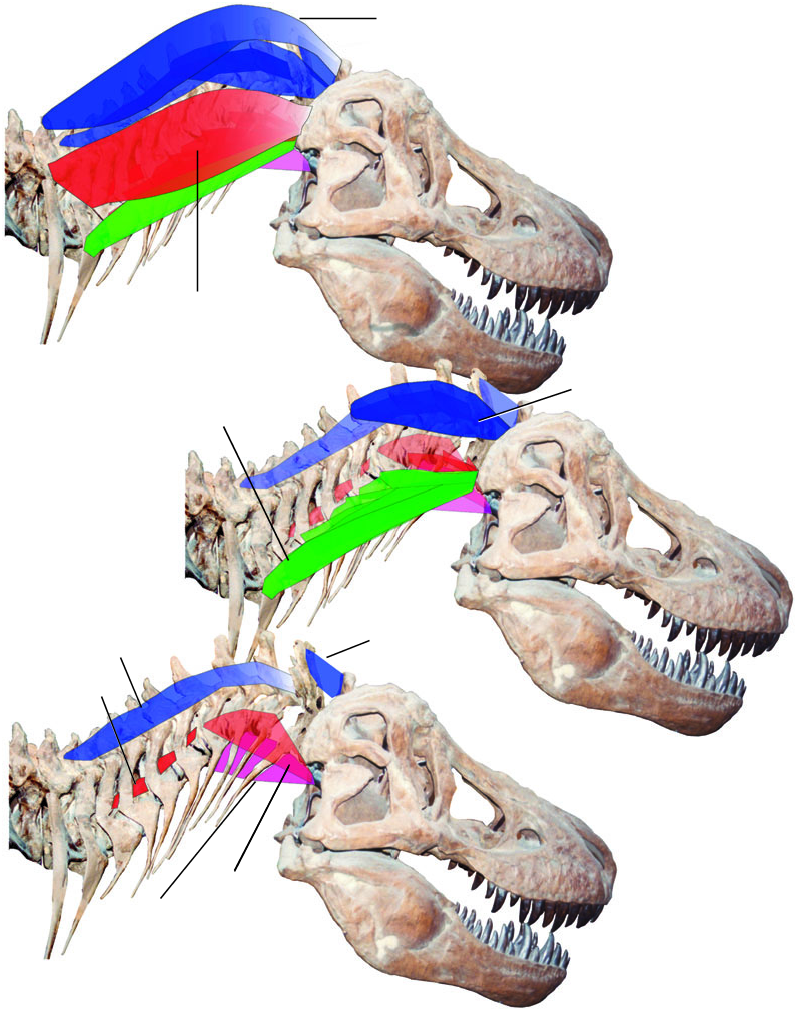
Presence of most neck muscles in tyrannosaurids is inferable by bracketing between birds and crocodilians. For the sake of continuity, their description and illustration proceeds as for the muscles of extant archosaurs in the previous section. The tyrannosaurid neck muscles are first figured schematically and then reconstructed on a photograph of the skeleton of Tyrannosaurus rex . These restorations are justifiable given strong inference of the occurrence of these muscles, but the accuracy of their appearance between origin and insertion must be viewed with caution. Muscle attachments are then mapped onto photographs of tyrannosaurid skeletal elements. When attachments are ambiguous, the likely alternatives are figured. Figures 15 View Figure 15 and 16 View Figure 16 depict the muscles in lateral and dorsal views, and Figures 17 View Figure 17 , 18 View Figure 18 and 23 View Figure 23 show details.
A. M. transversospinalis system
M. biventer cervicis (m. biv. c) ( Vanden Berge & Zweers, 1993). ( Aves)
M. transversospinais capitis (m. trans. cap.) ( Seidel, 1978). ( Crocodylia )
Origin: Close association of the m. biventer cervicis/m. transversospinalis capitis (m.biv. c./trans. cap.) origin with the posterior cervical and anterior thoracic neural spines of extant archosaurs indicates a similar origin in tyrannosaurids ( Fig. 19A View Figure 19 ). The anterior thoracic spines are broad and rugose in tyrannosaurids, and the m. biv. c./trans. cap. origins in this region were probably commensurately large and strong.
The rugose tips of the neural spines of C4–C10 contributed to the provision of origin of m. biv. c./trans. cap., similarly to the origin of the medial portion of the muscle in crocodilians ( Fig. 19A View Figure 19 ). In adult Daspletosaurus torosus and Tyrannosaurus rex the posterior cervical neural spines are relatively taller than they are in adult Gorgosaurus libratus , and appear more robust in Tyrannosaurus rex . This may indicate more extensive origins for m. biv. c./trans. cap. in the tyrannosauroids. In all tyrannosaurids, the strong dorsally convex curvature of the neck from C1 to C5 placed the neural spine of C4 at the highest point of the neck, allowing a slip from this vertebra to descend over the anterior neural spines towards the insertion.
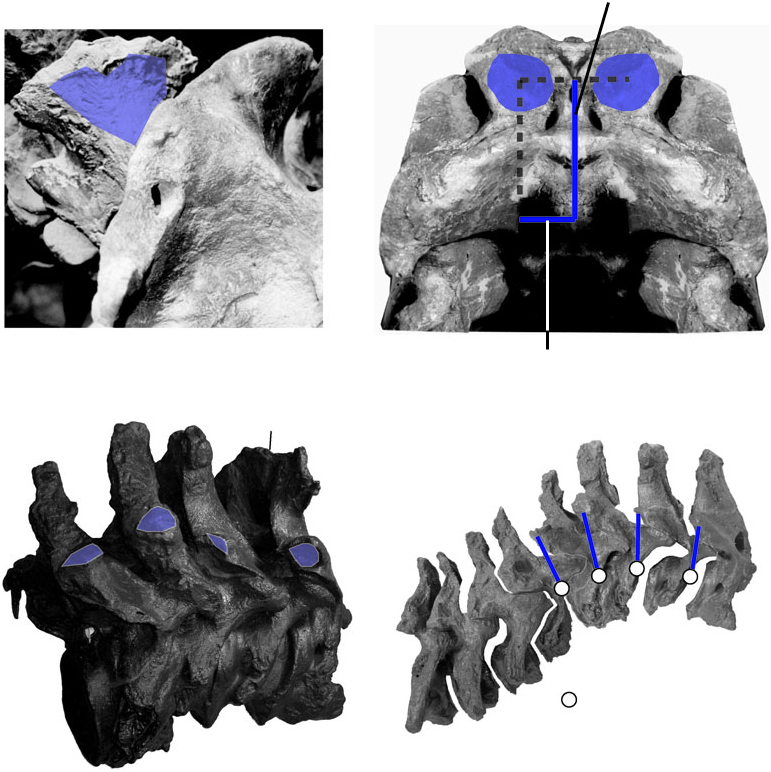
Insertion: In tyrannosaurids the general location of the inferred m. biventer cervicis/m. transversospinalis capits (m. biv. c./m. tr. cap.) insertion is similar to that of birds and crocodilians, but with a different bony attachment ( Fig. 19B, C View Figure 19 ). The posterodorsal surface of the parietals in tyrannosaurids forms a double arch dorsal to the inferred insertion of m. splenius capitis. These arches are often rugose ( Fig. 19B View Figure 19 ), indicating mineralization of Sharpey’s fibres parallel to the line of muscle pull, and a strong tendinous insertion for m. biv. c./m. tr. cap. In specimens of Tyrannosaurus rex these rugosities have extraordinarily high relief ( Fig. 19B View Figure 19 ). The m. biv. c./m. tr. cap. tendon ran dorsal to the spinous process of the axis, and m. splenuis capitis, before inserting on the parietal scar.
Action/function: By kinematic inference, the high dorsal position of the m. biventer cervicis/ transversospinalis capitis (m. biv. c./m. trans. cap.) insertion relative to the occipital condyle of tyrannosaurids indicates that the muscle bilaterally dorsiflexed the parietal (and thus the head) relative to the axis neural spine ( Fig. 19C View Figure 19 ). EMG confirms the muscle’s activity during this action in extant archosaurs ( Heidweiller et al., 1992), and a Level I′ physiological inference indicates that the muscle experienced the same activity in tyrannosaurids (26 C). Behavioural interpolation from observations of birds ( Gussekloo, 2000) and crocodilians ( Seidel, 1978), coupled with physiological inferences, implies that tyannosaurids employed dorsiflexion by m. biv. c./m. tr. cap. during inertial feeding ( Gussekloo, 2000) and drinking ( Heidweiller et al., 1992).
M. complexus ( Vanden Berge & Zweers, 1993). ( Aves.)
M. spinocapitis posticus ( Seidel, 1978; Cleuren & De Vree, 2000), m. semispinalis capitis ( Cong et al., 1998). Anterior portion: m. epistropheo-capitis lateralis ( Seidel, 1978; Cleuren & De Vree, 2000). m. rectus capitis dorsalis major ( Cong et al., 1998) ( Crocodylia ). m. obliquus capitis magnus ( Oelrich, 1956; ICVGAN, 1994; Herrell & De Vree, 1999). ( Varanidae ) ( Figs 15 View Figure 15 , 16 View Figure 16 , 17, 19 View Figure 19 )
Origin: Tyrannosaurids lack discrete lateral tubercles anterodorsal to the parapophyses for the cervical ribs, the position expected if they shared this origin of m. complexus with birds ( Zusi & Storer, 1969). However, C2–C5 have large anterodorsal surfaces of the epipophyses similar to those of birds (anterior to where the mm. cervicales ascendentes often insert), with a rugose ridge lateral to these surfaces on C2–C4. A system of tendons or aponeuroses arising from these surfaces was probably the origin for m. complexus in tyrannosaurids ( Fig. 19A View Figure 19 ), forming several slips that would coalesce anteriorly.
Slips of a muscle homologous with m. spino-capitis posticus/m.semispinalis capitis of crocodilians may have originated from lateral portions of the rugose posterior cervical neural arches, lateroventral to origins of m. transversospinalis capitis on the spinous processes. This origin (and therefore the presence of the muscle) is ambiguous in tyrannosaurids, as these surfaces may have served as origins and insertions of mm. intercristales or m. transversospinalis cervicis ( Tsuihiji, 2005).
Insertion: The insertion of the equivalent of the dorsal avian m. complexus was most likely along the posterodorsolateral edge of the squamosal, and was probably excluded from the parietals ( Fig. 19D View Figure 19 ). This is similar to the insertion pattern of the crocodilian m. epistropheo-capitis lateralis, and the m. obliquus capitis magnus of Varanus dumerilii . This inferred insertion is more discrete in Daspletosaurus torosus than in some other tyrannosaurids. In one wellpreserved Tyrannosaurus rex specimen (TMP 81.6.1), the left squamosal bears a long, narrow scar running ventrolaterally that may be the insertion for m. complexus. The insertion is preserved as a welldemarcated ridge of bone in the non-tyrannosaurid coelurosaurs Saurornithoides mongoliensis and Velociraptor mongoliensis .
By comparison with crocodilians, the insertion of m. spinocapitis posticus in tyrannosaurids, if present, would have been on the paroccipital processes medial to the insertion of m. longissimus capitis superficialis and dorsal to the insertion of m. iliocostalis capitis. There is a large area here where the muscle could have inserted by a fleshy attachment, or perhaps a broad yet dorsoventrally low aponeurosis, as seen in crocodilians. A discrete scar is not visible at this location in tyrannosaurids, but neither is it present on examined crocodilian skulls.
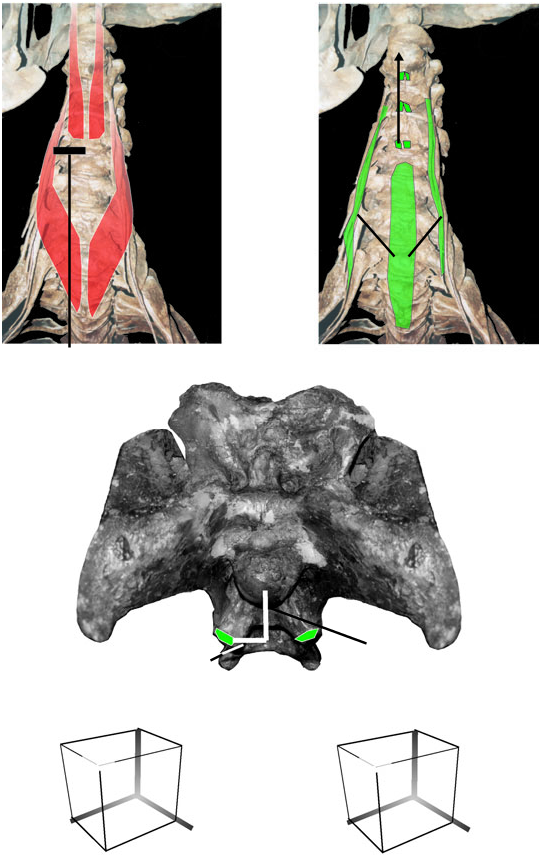
Action/function: The insertion of m. complexus is dorsolateral to the occipital condyle in tyrannosaurids, well positioned for dorsolateral and dorsal flexion of the skull relative to the axis (with uni- and bilateral contraction, respectively) ( Fig. 19C View Figure 19 ). Tyrannosaurid skulls are taller relative to their length than those of crocodilians, and the moment arm from the level of the occipital condyle to the squamosal is proportionally greater. Kinematic inference therefore indicates proportionally advantageous leverage for head dorsiflexion by m. complexus of tyrannosaurids, relative to the similarly inserting m. epistropheocapitis lateralis of crocodilians. Strict lateral flexion by m. complexus may have been relatively less effective in tyrannosaurids than in some birds, which have a more ventrally placed lateral insertion of this muscle. EMG shows that m. complexus of chickens is active during head dorsiflexion ( Heidweiller et al., 1992), and by unipolar, Level II′ physiological inference the same is likely in tyrannosaurids ( Fig. 19D View Figure 19 ).
M. spinocapitis posticus of tyrannosaurids, if present and inserting on the same region of the paroccipital processes as in crocodilians, would insert dorsolateral to the occipital condyle but medial to the insertion of m. longissimus capitis superficialis. By kinematic inference this would position the muscle for high-geared lateral flexion of the head relative to the cervical vertebrae. Unipolar physiological inference by EMG of crocodilians supports the likelihood of this utility in tyrannosaurids ( Cleuren & De Vree, 2000).
If present separately from the m. complexus, the curvature of the tyrannosaurid neck would enforce an unusual relationship between m. sp. cap. post. and m. transversospinalis cervicis/m. longus colli dorsalis (m. l.c.d.). The origins of m. sp. cap. post., from the neural arches of C6 or C7 to C10, would lie dorsomedial to the origins, posterior insertions, and posterior portion of the mass of m. l.c.d. Anterior to C6 or C7, m. sp. cap. post. of tyrannosaurids would diverge laterally, enabling m. l.c.d to continue a course (towards its insertion on C2) dorsomedial to that of m. sp.cap.post.
M. splenius capitis ( Vanden Berge & Zweers, 1993). ( Aves)
M. obliquus capitis profundus ( Tsuihiji, 2003), m. epistropheo-capitis medialis ( Seidel, 1978; Cleuren & De Vree, 2000). m. altoïdius capitis ( Tsuihiji, 2005). m. rectus capitis dorsalis minor ( Cong et al., 1998) ( Crocodylia ) ( Fig. 17A, C View Figure 17 )
Origin: The anterior face of the neural spine of the axis of tyrannosaurids bears a teardrop-shaped scar, identical in outline to the scar related to the origin of m. splenius capitis in birds ( Figs 19A View Figure 19 , 20A View Figure 20 ). A similar scar occurs on the axis of nearly all examined dinosaurs, including all birds and other tetanuran theropods, the basal theropods Coelophysis bauri , Dilophosaurus wetherelli and Ceratosaurus nasicornis , and the sauropods Camarasaurus grandis and Diplodocus longus. The origin in tyrannosaurids may have extended onto dorsolateral projections of the neural spines of C2 and C3 (forming the spine table of the axis: Gauthier, 1986), which is common in large tetanuran theropods.
Insertion: The insertion of the m. splenius capitis was probably on a large depression on the parietal, just lateral to the supraoccipital ( Fig. 19B View Figure 19 ). In some Tyrannosaurus rex specimens this area is convex, and the muscle may have inserted partly on the squamosal ventromedial to the insertion of m. complexus. The muscle is unlikely to have inserted along the entire opisthotic–squamosal suture, as it does in crocodilians, because a pneumatic foramen perforates this area ( Brochu, 2003). M. spl. cap. conceivably inserted lateral to this foramen, along the dorsal edge of the paroccipital process and ventral portion of the squamosal.
Action/function: As with birds, the line of action of m. splenius capitis of tyrannosaurids lies dorsal and somewhat lateral to the occipital condyle ( Fig. 19B View Figure 19 ). Kinematic inference therefore indicates that the muscle bilaterally imparted dorsiflexion, and unilaterally some lateroflexion, to the cranium relative to the axial neural spine. Dorsolateral flexion of the head, and stabilization of the craniocervical joint, would have been possible as well. By Level II′ physiological inference, head dorsiflexion by m. splenius capitis in chickens ( Heidweiller et al., 1992) corroborates the kinematic inference of dorsiflexion in tyrannosaurids ( Fig. 19B View Figure 19 ). However, if m. spl. cap inserted along the opisthotic–squamosal suture lateral to the pneumatic foramen, lateral flexion may have been more pronounced, and head dorsiflexion less effective, than if the muscle inserted more dorsally.
M. longus colli dorsalis ( Vanden Berge & Zweers, 1993). ( Aves.)
M. transversospinalis cervicis ( Seidel, 1978; Cleuren & De Vree, 2000; Tsuihiji, 2002) ( Crocodylia , Aves.)
M. longissimus cervicis ( Frey, 1988; Cong et al., 1998). Crocodylia ( Fig. 17A View Figure 17 )
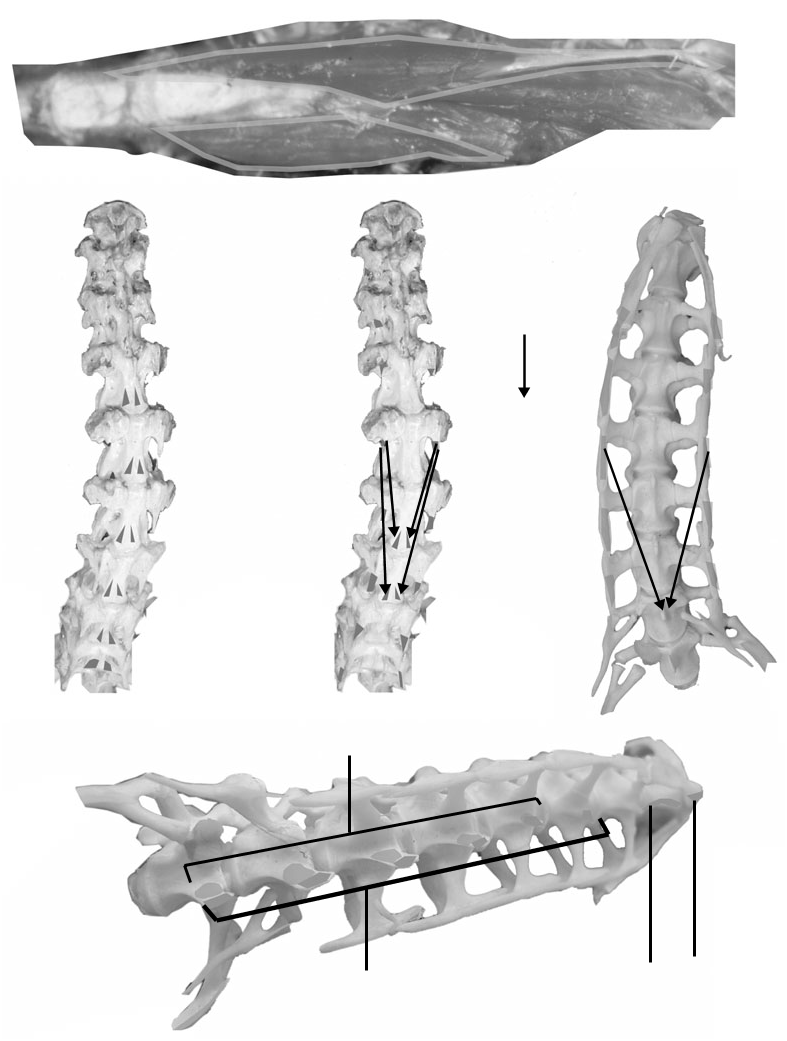
Origin: The origin of m. longus colli dorsalis/m. transversospinalis cervicis (m. l.c.d./trans. cerv.) is ambiguous in tyrannosaurids. Tyrannosaurids have several bony correlates that undefinitively resemble origins for the muscle in crocodilians and birds. Tyrannosaurids have rugose areas dorsal to the postzygapophyses of posterior cervicals, possibly homologous to the m. l.c.d./trans. cerv. origin in crocodilians. Dorsomedial to these rugosities, tyrannosaurid neural arches C6–C10 bear ridges running ventrolaterally. It is possible that these ridges and the postzygapophseal rugosities anchored an aponreurotic origin of m. l.c.d./ trans. cerv. similar to the medial portion of the aponeurosis notarii of birds. The identification of this muscle’s origin in tyrannosaurids can be no more precise than an association with the neural spines ventral to m. transversospinalis capitis, and dorsomedial to origins of m. longissimus capitis profundus and superficialis.
Insertion: In contrast to its origin, the insertion of m. l.c.d./tr. ans. cerv. is well circumscribed in tyrannosaurids ( Fig. 20C View Figure 20 ). The epipophysis/processus dorsalis of C2 of tyrannosaurids has a posterolaterally concave scar similar to the tendinous m. l.c.d. insertion of some birds, except that the concavity in tyrannosaurids appears proportionally deeper than in avians. Unlike in birds, the epipophysis of C3, and sometimes C4 and C5, in tyrannosaurids also bears a posterolaterally concave scar, indicating other insertions similar to that on C2. Under this interpretation, insertion tendons of this muscle would have attached to the processes dorsales posteroventral to the origins of m. complexus, as occurs in some birds.
There are some probable differences between tyrannosaurids and birds in the insertions of m. l.c.d./ trans. cerv. In tyrannosaurids insertions may have extended onto the epipophyses of C6 and C7. Many birds lack a posteriorly concave scar altogether on C2, and the insertions of all slips of m. longus colli dorsalis pars anterior (and the axial insertion of m. l.c.d.) are onto the dorsal surface of the C2 processus dorsalis, as for the other cervicals. Tyrannosaurids have dorsolateral projections of the atlas that are not present in birds. This may indicate an anterior extension of m. l.c.d./m. trans. cerv. in tyrannosaurids, somewhat like that of crocodilians.
Action/function: Because the insertions of m. longus colli dorsalis/transversospinalis cervicis pars lateralis (m. l.c.d./trans. cerv.) lie dorsal to its origin in tyrannosaurids, kinematic inference indicates that the muscle dorsiflexed the entire neck relative to the trunk ( Fig. 20D View Figure 20 ) by bilateral contraction. The sinusoidal curve of the neck would impart complexity to this dorsiflexion. The epipophysis insertions on C2 and C3 are dorsal to the centres of dorsiflexive rotation between these vertebrae and the posteriorly adjacent one. Kinematic inference thus also indicates that the muscle dorsiflexed C2 relative to C3, and C3 relative to C4. As C2 and C3 were retracted, vertebrae in the posterior, dorsally concave portion of the neck would be passively dorsiflexed relative to each other, additively raising the anterior vertebrae.
The homologous muscles in crocodilians and birds show strong EMG activity during neck dorsiflexion ( Cleuren & De Vree, 2000; van der Leeuw et al., 2001); a Level I′ physiological inference thus clearly supports neck dorsiflexion by this muscle in tyrannosaurids ( Fig. 18B′ View Figure 18 ).
B. M. longissimus system
M. longissimus capitis superficialis ( Crocodylia ) ( Fig. 17B View Figure 17 )
Origin: There are proximal scars on the parapophyses of C7–C9 of large tyrannosaurids that may have served as origins for m. longissimus capitis superficialis ( Fig. 21A View Figure 21 ). As with C8 of crocodilians, the posterior origins of m. long. cap. sup. probably extended onto the lateral portions of the parapophyses. Discrete ventrolateral scars are not present on the neural arches at the expected position on C6–C4, but fleshy origins are possible.
The broad, rounded parapophyses of C10 and the anteriormost dorsal vertebrae of tyrannosaurids have lateral scars similar to m. longissimus capitis origins on these vertebrae in Homo sapiens , lateral to areas of attachment for m. longissimus thoracis. It is possible that these scars were the location of stout tendinous origins of m. longissimus capitis superficialis in tyrannosaurids. However, differentiation is ambiguous between possible scars for this muscle, m. longissimus capitis profundus and m. longissimus thoracis.
Insertion: As in crocodilians, the insertion of m. longissimus capitis superficialis in tyrannosaurids was probably tendinously onto the lateral portion of the paroccipital process ( Fig. 21C View Figure 21 ), just medial or dorsal to the origin of m. depressor mandibulae on the lateralmost surface of the process.
Action/function: Kinematic inference implicates multiple functions for m. longissiumus capitis superficialis in tyrannosaurids. Its lateral insertion, distal to the occipital condyle, would provide a long moment arm for lateral flexion of the head versus the neck, especially in the tyrannosaurines and most markedly in Tyrannosaurus rex ( Fig. 21C View Figure 21 ). Unipolar (Level II′) physiological inference by EMG in crocodilians supports the lateral flexion role for m. long. cap. sup. ( Fig. 17B′ View Figure 17 ). However, in tyrannosaurids the insertion of m. long. cap. sup. was positioned dorsally relative to the centres of intervertebral rotation in the posterior part of the neck. This would result in a moment arm for dorsiflexion of the head relative to the base of the neck, should both muscles contract synchronously ( Fig. 21B View Figure 21 ). Because the insertions are lateral to the occipital condyle, the rostrum would not tip upwards under this activity; instead, the entire head would be retracted posterodorsally.
M. rectus capitis superior/dorsalis ( Aves)
M. longissimus capitis profundus (Crododylia) ( Fig. 18D View Figure 18 )
Origin: Following the extant phylogenetic bracket, m. rectus capitis superior/m. longissimus capitis profundus (m.r.c.s./m.l.c.p.) of tyrannosaurids could have originated from the parapophyses of C1–C7 ( Fig. 21A View Figure 21 ). Because the neck is ventrally concave anteriorly, as in birds, the origins of m.r.c.s./m.l.c.p. may have been restricted to the parapophyses of C1–C6 for an unobstructed line of pull. It is possible that (as occurs in birds) tyrannosaurids possessed multiple bellies of m.r.c.s./m.l.c.p., each originating from its respective parapophysis.
Insertion: The insertion of m. rectus capitis dorsalis/m. longissimus capitis profundus of tyrannosaurids is less ambiguous than its origin. The basioccipital has large ventrolaterally projecting tuberosities, and m.r.c.s./m.l.c.p. probably inserted along their lateral margins, as in crocodilians ( Fig. 21D View Figure 21 ). Multiple bellies of the muscle would have converged on the same insertion, as in birds.
Action/function: The insertion of m. rectus capitis superior/m. longissimus capitis profundus is ventrolateral to the occipital condyle of tyrannosaurids, with a short moment arm relative to the length of the skull ( Fig. 18D View Figure 18 ). Kinematic inference ( Fig. 18D′ View Figure 18 ) indicates that the muscle was used for rapid lateral or ventrolateral flexion of the head versus the anterior cervicals. Bilateral contraction of m.r.c.s./m.l.c.p. would ventroflex the head relative to the neck.
Mm. intertransversarii ( Fig. 18E View Figure 18 )
Origin: M. intertransversarii of tyrannosaurids probably originated from the anterior surfaces of the parapophyses of C10–C6 ( Fig. 22A View Figure 22 ). Because the parapophyses are robust in this region, there may have been superficial and deep divisions of m. intertransversarii, as seen in Heloderma ( Herrell & De Vree, 1999). Anterior cervicals in some tyrannosaurids display a groove posteroventral to the prezygapophyses that may have served as origin for m. intertransversarii in this region.
Insertion: Posterior cervical insertions of m. intertransversarii in tyrannosaurids would have been on the posterior parapophyseal surface of the anterior vertebra of each pair, from C9 to C5 ( Fig. 22A View Figure 22 ). Insertions on anterior vertebrae are ambiguous, and may have been onto fascia attaching to the posterior parapophyses of C4–C1 Action/function: The laterally extensive posterior transverse processes of tyannosaurids suggest long moment arms for lateroflexion of vertebrae relative to each other in this region ( Figs 18B View Figure 18 , 22B View Figure 22 ). The slight opisthocoely of the centra in this region suggests limited displacement, and possible functions of damping and stabilization of lateral flexion by contralateral muscles. The anterior mm. intertransversarii are bracketable as being present in tyrannosaurids. They had shorter lever arms, and were kinematically less suited for intervertebral lateroflexion than were the posterior bellies.
.
C. M. iliocostalis and m. longus systems
M. rectus capitis lateralis ( Vanden Berge & Zweers, 1993; Cong et al., 1998). Aves, Crododylia M. iliocostalis capitis ( Seidel, 1978; Cleuren & De Vree, 2000). Crocodylia ( Fig. 17D View Figure 17 )
Origin: The origin of this muscle is ambiguous in tyrannosaurids ( Fig. 22A View Figure 22 ). The morphology of their cervical ribs and ventral portion of the anterior cervicals differ from those of both extant archosaur clades. Large non-avian theropods lack the large axial hypopophysis that serves as the site of origin of m. rectus capitis lateralis in birds. The cervical rib shafts of archosaurs primitively are long, slender structures in which the anterior rib overlaps the succeeding one ventrally. The shafts are especially long in large theropods, and in Tyrannosaurus rex run ventrolateral to the centra of up to five vertebrae posterior to that of the rib’s attachment. Except for the atlantal rib, the ribs form large proximal plates lateral to the parapophyses and diapophyses ( Fig. 22A View Figure 22 ).
This morphology contrasts markedly with the poles of the extant bracket, and complicates identification of the muscle’s origin in tyrannosaurids. In crocodilians only the altantal and axial cervical ribs have long and overlapping shafts. In birds cervical ribs are absent or reduced to a costal process. Bakker, Williams & Currie (1986) and Bakker (2000) propose that in large theropods m. rectus capitis lateralis/m. iliocostalis capitis (which they termed m. longissimus transversarius) originated from a tendinous sheath that surrounded the overlapping shafts of the anterior cervical ribs. This is similar to the origin from C1 and C2 ribs in crocodilians, which represent an anterior restriction of multiple overlapping cervical ribs that probably occurred ancestrally in archosaurs ( Bakker, 2000).
However, because the cervical rib shafts are proportionally much more slender in tyrannosaurids than are the C1 and C2 ribs of crocodilians, an origin of m. rectus capitis lateralis (m. r.c.l.) from a sheath surrounding the ribs may have been relatively weak. This conflicts with a large possible insertion for this muscle, which indicates a large cross-section and high force-generating capacity. An alternative possibility is that m. rectus capitis lateralis of tyrannosaurids was dorsally displaced and originated from the robust heads of the cervical ribs ( Fig. 22A View Figure 22 ), and that m. iliocostalis cervicis inserted on a tubular sheath of fascia that enveloped the shafts. Because this arrangement differs from that of crocodilians and birds, it must be considered tentative, but can be tested by histological examination of the rib heads for tendon attachment.
Insertion: In both birds and crocodilians, and in tyrannosaurids by EPB, the m. rectus capitis lateralis inserts onto a distinct ridge on the ventral margin of the paroccipital processes ( Fig. 19D View Figure 19 ). The insertion is broader dorsoventrally in crocodilians than in birds. In tyrannosaurids a large triangular concavity, broad medially and tapering laterally, occurs dorsal to the ventral edge of the paroccipital processes ( Fig. 19D View Figure 19 ). It is possible that m. rectus capitis lateralis extended dorsally onto this entire indentation, but a ventrally restricted insertion is more parsimonious given its occurrence in extant archosaurs.
Action/function: M. rectus capitis lateralis/m. iliocostalis capitis of tyrannosaurids inserts laterally and somewhat ventrally to the occipital condyle, indicating primarily lateral flexion of the head versus the neck by each muscle ( Fig. 19D View Figure 19 ), and some capacity for head ventroflexion. Kinematic inference of the muscle’s action in Tyrannosauridae is consistent with crocodilian EMG showing activity during head lateral flexion and rotation, as well as damping activity during head dorsiflexion ( Figs 17D View Figure 17 , 19D View Figure 19 ).
M. rectus capitis ventralis. ( Vanden Berge & Zweers, 1993; Cong et al., 1998). ( Aves, Crododylia ) M. longus capitis ( Cong et al., 1998). ( Crocodylia ) M. flexor colli ( Vanden Berge & Zweers, 1993). ( Aves) ( Fig. 23A View Figure 23 )
Unlike in many extant archosaurs, the ventral spinous processes of tyrannosaurids are small, which complicates inferences about these muscles. The only one that can be readily bracketed is m. rectus capitis ventralis. The presence of m. longus capitis, as seen in crocodilians, and of m. flexor colli, as in birds, are equivocal in tyrannosaurids; hence these muscles are not discussed here.
Origin: The small, anterior ventral spinous processes of tyrannosaurids bear surfaces visible in anterior view that may have served as origins for m. rectus capitis ventralis ( Fig. 23B View Figure 23 ). The origins could also have extended onto the ventral surfaces of the anterior cervical centra, as in crocodilians.
Insertion: The insertion of m. rectus capitis ventralis of tyrannosaurids could have been either onto the ventral surface of the basioccipital tuberosities ( Fig. 23B View Figure 23 ), as in crocodilians, or onto the basioccipital, as seen in birds.
Action/function: Regardless of whether either likely insertion was present, m. rectus capitis ventralis would have attached ventrally to the occipital condyle of tyrannosaurids. Kinematic inference indicates head ventroflexion relative to the neck ( Fig. 23C, E View Figure 23 ).
M. longus colli ventralis. ( Vanden Berge & Zweers, 1993). ( Aves.)
M. iliocostalis cervicis ( Seidel, 1978), m. longus colli. ( Cong et al., 1998). ( Crocodylia .) ( Fig. 23A View Figure 23 )
As for the origins of m. rectus capitis lateralis/m. iliocostalis capitis, origins and insertions of m. logus colli ventralis/m. iliocostalis cervicis (m. l.c.v./m. il. cerv.) of tyrannosaurids are bracketable between extant archosaurian morphologies.
Origins: Tyrannosaurids lack large hypopophyses, such as those present on the posterior cervicals of crocodilians and many birds, so the medial origins of m. l.c.v/m. il. cerv. were probably from the ventral surfaces of the anterior thoracic and posterior cervical vertebrae ( Fig. 23B View Figure 23 ).
Insertions: If present, dorsal components of m. l.c.v./m. il. cerv. of tyrannosaurids may have inserted on the posterior edges of the proximal cervical ribs ( Fig. 23B View Figure 23 ). As mentioned in the description of m. iliocostalis capitis, the insertions of medially originating components of m. l.c.v/m. il. cerv. may have been aponeurotic onto fascia surrounding bundled cervical rib shafts ( Fig. 23B View Figure 23 ).
Action/function: As in both crocodilians and birds, the position of possible m.l.c.v./m. il.cerv. insertions of tyrannosaurids are ventral to the intervertebral articulations, which, by kinematic inference, supports a function of head ventroflexion relative to the neck. The insertions are also lateral to the centres of the centra, which indicates that head lateral flexion was possible ( Fig. 23C View Figure 23 ).
Level II′, unipolar physiological inference with reference to crocodilians ( Fig. 23D View Figure 23 ) indicates a laterally flexive function, and, as in birds, the possibility of the muscles’ activity during protraction of the head. The bundled shafts of the cervical ribs, and concomitantly ambiguous insertions, preclude kinematic assessment of the preponderance of these respective functions. However, by both kinematic and physiological bracketing between extant archosaurs, m. l.c.v./m. il. cerv. of tyrannosaurids were most likely active in damping dorsiflexion of the neck.
D. Complex and uniarticular muscles
Mm. intercristales. ( Vanden Berge & Zweers, 1993). ( Aves.)
Origins: Tyrannosaurids have prominent transverse oblique crests, especially on C5–C10, from the dorsally projecting neural spines running laterally to the postzygapophyeses ( Fig. 22A View Figure 22 ). It is possible that mm. intercristales originated from the anterior surfaces of these crests, but they could serve as origins for large components of the m. transversospinalis system.
Insertions: Mm. intercristales could have inserted on the posterior surface of the crista transversoobliqua of each intervertebral pair, from C2 posteriorly (Fig. 34A).
Action/function: If present, these muscles would be kinematically positioned for slight intervertebral dorsiflexion, as well as more for lateral flexion than in birds.
Mm. cervicales ascendentes. ( Vanden Berge & Zweers, 1993). ( Aves.)
In configuration these muscles are probably unique to birds ( Tsuihiji, 2005), and there are no discrete attachment sites in tyrannosaurids that would contradict this assessment.
DISCUSSION
EXAMPLES OF COMMON AND DIVERGENT PATTERNS OF NECK MUSCLES IN EXTANT ARCHOSAURS
By the criterion of position, most neck muscles are recognizable as homologous between birds and crocodilians. For example, m. rectus capitis lateralis is establishable as homologous in extant archosaur clades, despite differing origins. M. rectus capitis lateralis originates from an enlarged hypopophysis of the axis in birds, or from the lateral surface of the centrum, and in crocodilians from the proximal portion of the C2 rib and an aponeurosis of the ventral surfaces of the C1 and C2 cervical ribs. Mm. intertransversarii span the transverse processes in specimens of both birds and crocodilians, although these muscles are greatly subdivided and elaborated in birds.
Other muscles are less readily identified as homologous, or vary substantially between modern Aves and Crocodylia . M. longus colli dorsalis inserts onto a posteriorly facing concavity of the processus dorsalis of the axis of birds. Its crocodilian homologue (m. transversospinalis lateralis) inserts by a flat tendon primarily onto the atlas, but in Alligator sinensis this tendon has also been reported to insert onto the axis, in a position ventral to the postzygapophysis ( Cong et al., 1998). M. longus colli dorsalis is enormously complex in birds. Multiple, long heads of m. l.c.d. insert onto the processes dorsales (epipophyses) above the postzygapophyses of anterior cervical vertebrae, and slips run from the posterior cervicals to the long tendon, inserting on the processus dorsalis of C2.
Several issues of homology have been controversial, and some muscles are exclusive to either birds or crocodilians. M. complexus of birds is most difficult to reconcile with positionally similar muscles of other reptiles, such as m. spinocapitis posticus, m. epistropheo-capitis lateralis and m. obliquus capitis magnus. Fusion of dorsomedial and ventrolateral portions of m. complexus of some birds bestows on the muscle a configuration unique to birds among extant amniotes. In crocodilians, Varanus dumerilii and partially in some adult birds, these muscles originate from the neural arches, spines, and from flat surfaces (crocodilians and V. dumerilii ) or epipophyses (birds) dorsal to the postzygapophyses. These origins indicate affinity with the transversospinalis system, associated with portions of vertebrae dorsal to the centrum and transverse processes. Insertions dorsal to the paroccipital processes in both taxa support the transversospinalis assignment (including a dorsomedial insertion in midstage bird embryos: Noden, 1983). Tsuihiji (2005) noted that a spinal nerve ventral to m. complexus allied it with m. transversospinalis capitis lateralis of crocodilians. However, in some birds the anteroventral origins of m. complexus arise from the lateral tubercles of the transverse processes, as expected of derivatives of the longissimus system. The position of the lateral tubercles on the transverse processes of birds indicates that ventral slips contributing to m. complexus may be homologous with origins of m. longissimus capitis superficialis of crocodilians. M. complexus of some birds may form from a unique dorsoventral fusion of transversospinalis and longissimus derivatives; Tsuihiji’s (2005) neuroanatomical evidence will provide a guide for falsification in each case.
The breadth and superficial placement of m. complexus in birds led some workers to associate it with m. cucularis ( Fisher & Goodman, 1955; Goodman & Fisher, 1962), a subcutaneous muscle derived from branchial levator musculature. Craniocervical insertions and origins, and primary motor innervation by the first six spinal nerves ( Gross, 1985), rather than the accessory ( Vanden Berge & Zweers, 1993), have indicated the muscle’s homology with axial musculature in other amniotes. Interestingly, however, Gross (1985) discovered motor efferents from the accessory and even the facial cranial nerves serving the complexus, and afferents to the tangential nucleus of the brain. Because the tangential nucleus also receives inputs from the vestibulocochlear apparatus and sends motor signals to the spinal nerves, this circuit shows how sensory input from spindles of complexus provide feedback for modulating this muscle’s own action.
While homology between m. rectus capitis dorsalis and m. longissimus capitis profundus is less problematic than the issues surrounding m. complexus, several factors introduce ambiguity. Origins of m. rect. cap. dors. are restricted to the anterior cervicals of birds, and, unlike in birds, m. long. cap. prof. does not originate from the atlas in crocodilians. M. long. cap. prof. is unitary in crocodilians, but in birds the origins of m. rect. cap. dors. are discrete slips that converge towards the insertion.
However, the apparent anterior restriction of the muscle in birds is a consequence of the posteriorly elongate neck. In both extant archosaur clades the muscles originate from the anterior portions of the transverse processes of about six vertebrae; the set is displaced posteriorly by one vertebra in crocodilians. These muscles share a similar insertion on the basioccipital of birds and crocodilians, dorsolateral to the insertion of m. rectus capitis ventralis on the basioccipital and medial to the paroccipital process insertion of m. rectus capitis lateralis.
The main differences between bird and crocodilian neck muscles involve consequences of the elongation and sinusoidal curvature of the neck in birds, and the heavier head skeleton of crocodilians. For example, with m. longus colli dorsalis and ventralis of birds, the complexity of dorsi- and ventroflexors is greatly increased over the crocodilian, and probably original archosaurian, morphology. Long tendons connect multiple bellies of these muscles to multiple insertions, often following the curvature of the neck within long fascical sheaths. Conversely, crocodilians retain heavy musculature that runs along the entire neck (m. longissimus capitis superficialis) for turning the head.
MORPHOLOGICAL AFFINITIES AND UNIQUE PATTERNS OF TYRANNOSAURID NECK MUSCULATURE
Generally tyrannosaurids combined the curvature and some of the muscular complexity of bird necks with the bulkiness of crocodilian neck musculature. In tyrannosaurids the insertions of m. longus colli dorsalis/m. transversospinalis cervicis, and the origin of m. splenius capitis, are strikingly similar to those of birds. However, m. longissimus capitis superficialis was present, and probably massive, as in crocodilians. Tyrannosaurids have features of the cervical series and occiput not found in either clade of the extant bracket, and probably had muscular morphologies not seen in extant archosaurs.
The spinous processes of C2 and C3 in tyrannosaurids may reflect the morphology of the m. biv. c./m. trans. cap. insertion. Gauthier (1986) noted dorsolateral projections of the C2 neural spine in tyrannosaurids and some other large theropods. He collectively termed the two projections the spine table of the axis for their flat, wide appearance in anterior or posterior view. Lateral projections of the spinous process of C3 in tyrannosaurids have a similar morphology, although they are narrower and form more of a W-shape than a T-shape in coronal section. The insertion tendon of m. biv. c./m. trans. cap., or the collagenous ventral portion of the perimysium that enveloped the muscle, ran over the C3 and spine table of the axis. The breadth of these structures suggests decreased pressure (force/area) on the ventral surface of the tendon or fascla.
In tyrannosaurids the posterior origin of m. longissimus capitis superficialis may have been caudally displaced relative to the condition in crocodilians, arising from the parapophysis of the anteriormost dorsal, instead of from C7 as in crocodilians. This surface in tyrannosaurids is large and knob-like, suggesting a strong attachment and a bulky, forcefully contracting m. long. cap. sup. This is a logical interpretation of muscle size, but is based on an extrapolatory, Level II inference ( Bryant & Russell, 1992; Witmer, 1995) of attachment position.
Conversely, the muscle’s moment arms are more strongly inferable, both lateral to the occipital condyle and dorsal to the centre of sagittal rotation of the entire neck. The breadth of the skull of the tyrannosaurine tyrannosaurids Daspletosaurus torosus ( Russell, 1970) , Tarbosaurus bataar ( Hurum & Sabath, 2003) and especially Tyrannosaurus rex ( Brochu, 2003) provided m. longissimus capitis superficialis with long moment arms for lateral flexion of the cranium. The posterior origins of this muscle on the laterally inclined parapophyses of C10 and T1 are dorsal to the likely rotation centre of the neck between C10 and C9. This vertical moment arm indicates the capacity for dorsiflexion of the neck and retraction of the head when left and right m. long. cap. sup. contracted synchronously.
This rectractive function was the consequence of bird-like neck curvature, a crocodilian-like m. longissimus capitis superficialis, and tall vertebrae in the cervicodorsal region. The bird-like posterior U-curve of the neck and tall posterior centra of tyrannosaurids resulted in an unusual dorsiflexive moment arm for m. l. cap. sup. not present in either extant clade.
BEHAVIOURAL INTERPOLATION OF TYRANNOSAURID FEEDING ACTIVITY: EXAMPLES FROM BEHAVIOUR OF EXTANT ARCHOSAURS AND CORROBORATIVE BIOMECHANICS
With physiological and kinematic inferences of muscle function possible for tyrannosaurids, is it feasible to interpolate between extant taxa to deduce correlations between muscle use and behaviour? The most likely broad behaviours would involve dorsiflexive and even laterally flexive muscles for inertial feeding, assisted by the inertia of a non-muscular tongue at it was moved by hyolingual muscles ( Cleuren & De Vree, 2000; Tomlinson, 2000).
EMG upholds the muscular/behaviour bracket for inertial feeding in crocodilians and palaeognathous birds ( Heidweiller et al., 1992; Cleuren & De Vree, 2000; Gussekloo, 2000; Tomlinson, 2000). Crocodilians often transport pieces of flesh or bodies of smaller prey towards the pharynx by tossing the head dorsally and laterally, throwing and catching the food until it reaches the proper orientation for swallowing. The animals are versatile and adaptable with these inertial movements, which are less stereotyped than mammalian intraoral transport ( Cleuren & De Vree, 2000). Palaeognaths employ a throw and catch method of inertial transport similar to that of crocodilians ( Gussekloo, 2000). The smaller relative size of the food items of palaeognaths necessitates less intricate modulation of the movements than in crocodilians.
The similarities of muscle activation patterns during these behaviours are striking, given the differences between crocodilian and avian neck osteology and details of musculature. This makes behavioural interpolation of this style of inertial feeding particularly strong for tyrannosaurids. The divergences in anatomy between tyrannosaurids and their extant relatives signal some caution in restricting the animals to inertial feeding seen in the extant bracket, but hypotheses of somewhat different actions will be testable by biomechanical methods.
Other behaviours are less confidently inferable in tyrannosaurids, but are possible to deduce through unbracketed comparisons with extant archosaurs. The curvature of theropod necks suggests an approach phase to food common to Mesozoic and extant forms. The long, sagittally sinusoidal necks of birds, and similar cervical motor patterns in birds with diverse diets ( Zweers & Vanden Berge, 1997), indicate that pecking and retracting the head to procure food were present in their common ancestor ( Zweers, 1992). Birds employ similar motions when drinking, although these are more complex in anseriforms, which have large numbers of cervical vertebrae ( Heidweiller et al., 1992; van der Leeuw et al., 2001).
In tyrannosaurids, elevation and retraction by neck dorsiflexors, and the unusual kinematics of m. longissimus capitis superficialis, would facilitate extension and retraction behaviour to procure food. This provides a mechanism for puncture-and-pull feeding recorded by tooth marks in bone ( Erickson & Olson, 1996; Erickson et al., 2004; Rayfield, 2004), but more critically allows inference of bird-like musculoskeletal kinematics during drinking, which would not leave traces in the fossil record.
Parallels between tyrannosaurid and crocodilian neck musculature may indicate feeding behaviours shared by these taxa that birds generally do not undertake. Crocodilians sweep their heads sideways when attacking prey and tearing meat ( Busbey, 1989; Cleuren & De Vree, 2000), using m. longissimus capitis superficialis and m. spino-capitis posticus as the main lateroflexors ( Cleuren & De Vree, 2000). The large m. long. cap. sup. inferred in tyrannosaurids would have enabled powerful lateral flexion of the head and neck, augmented by m. complexus. Thus, kinematically and by unipolar physiological inference, tyrannosaurids were capable of lateral strikes at prey with a combination of, respectively, crocodilian- and bird-like muscles.
These inferences seem obvious, but become informative about specific behaviours through quantitative testing. Calculations from 3D dynamic modelling of an adult Tyrannosaurus rex indicate rapid accelerations for both dorsi- and lateroflexion, with high moment-generating capacity overcoming rotational inertias of the head and neck ( Snively, 2006; E. Snively & A. P. Russell, unpubl. data; E. Snively et al., unpubl. data). Rotational inertias are proportional to the fifth power of linear size while muscle force is proportional to the square. Thus, even with much lower moment-generating capacity, smaller tyrannosaurids could probably strike even more quickly than adults of the largest taxon.
Shaking of the head to subdue smaller prey is seen in chickens (with muscle use documented in EMG studies: Snively, 2006), scleoglossan lizards ( Schwenk, 2000b) and even in some crocodilians when feeding on fish ( Thorbjarnarson, 1993). Lingham-Soliar (1998) and Fowler & Sullivan (2006) suggest that large tyrannosauirds engaged in shake-feeding to excise flesh or dispatch prey. Behavioural interpolation of this activity is more tenuous than the others proposed here, as it is based on observations and not physiological bracketing between extant taxa. More importantly, shake-feeding by tyrannosaurids to kill prey is unsupported by quantitative data. However, as with inference of rapid lateral strikes and gaze shifts, biomechanics of flesh extraction has been tested quantitatively.
For example, lateroflexive neck muscles of adult Tyrannosaurus rex had enormous capacity to perform work of fracture on food ( Snively, 2006). Section moduli indicate that T. rex maxillary teeth were were coevally strong in lateral and anteroposterior bending ( Henderson & Weishampel, 2002; Snively et al., 2006), and much stronger in lateral bending than teeth of other large theropods ( Farlow & Brinkman, 1994; Snively et al., 2006). Muscular capacity and tooth strength suggest that T. rex could tug alternately with lateroflexors, the ipsilateral teeth producing puncture-and-pull tooth marks roughly parallel with the carinae ( Erickson & Olson, 1996; Erickson et al., 1996), while the contralateral teeth remained relatively stationary. This alternate tearing action would be similar to behaviour seen in varanid lizards, canids, and even in predatory birds as they grip and tear soft tissue of prey. The lateral component of this tearing action may have been less vigorous in smaller tyrannosaurids, which had weaker teeth ( Farlow & Brinkman, 1994; Snively et al., 2006) and shorter lateroflexive moment arms ( Snively, 2006) than adult T. rex . However, linear retraction by simultaneous contraction of certain dorsiflexors, lateroflexors and anterior ventroflexors in all tyrannosaurids is supported by kinematic and physiological inference, and observation of these behaviours in both carnivorous birds ( Snively, 2006) and crocodilians. EBI and biomechanics thus refine the picture of puncture-andpull feeding in tyrannosaurids ( Erickson & Olson, 1996; Erickson et al., 1996; Rayfield, 2004), and predict that bilateral tooth drag marks of tyrannosaurids will be found on fossil bones large enough to record scoring by left and right tooth rows.
CONCLUSIONS
Our methods are potentially applicable to reconstructing many functional systems in extinct animals, and the archosaurian neck provides a morphologically grounded and complexly challenging trial. This study presents an example of both soft-tissue and behavioural inference in a well-represented fossil clade, and demonstrates the correlative necessity of understanding musculoskeletal systems in extant vertebrates. Neck musculature of birds and crocodilians is highly divergent in detail. However, morphological, physiological and behavioural commonalities highlight the power of archosaurian homologies to elucidate unpreserved attributes of extinct relatives. Tyrannosaurid bite forces and skull strengths were among the most formidable of any archosaurs ( Erickson et al., 1996; Meers, 2003; Therrien et al., 2005; Snively et al., 2006), and the neck muscles of adults exerted higher forces than in other theropod clades ( Snively & Russell, 2007). EBI and biomechanics applied to the tyrannosaurid neck yield a rigorous, yet expansive and vivid picture of feeding and other craniocervically mediated behaviours in these powerful carnivores.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |