LUCININAE, J. Fleming, 1828
|
publication ID |
https://doi.org/10.1111/j.1096-3642.2011.00700.x |
|
persistent identifier |
https://treatment.plazi.org/id/0E297D4D-FFC4-B83E-B4FC-806AE0DE49C8 |
|
treatment provided by |
Valdenar |
|
scientific name |
LUCININAE |
| status |
|
LUCININAE View in CoL FLEMING, 1828
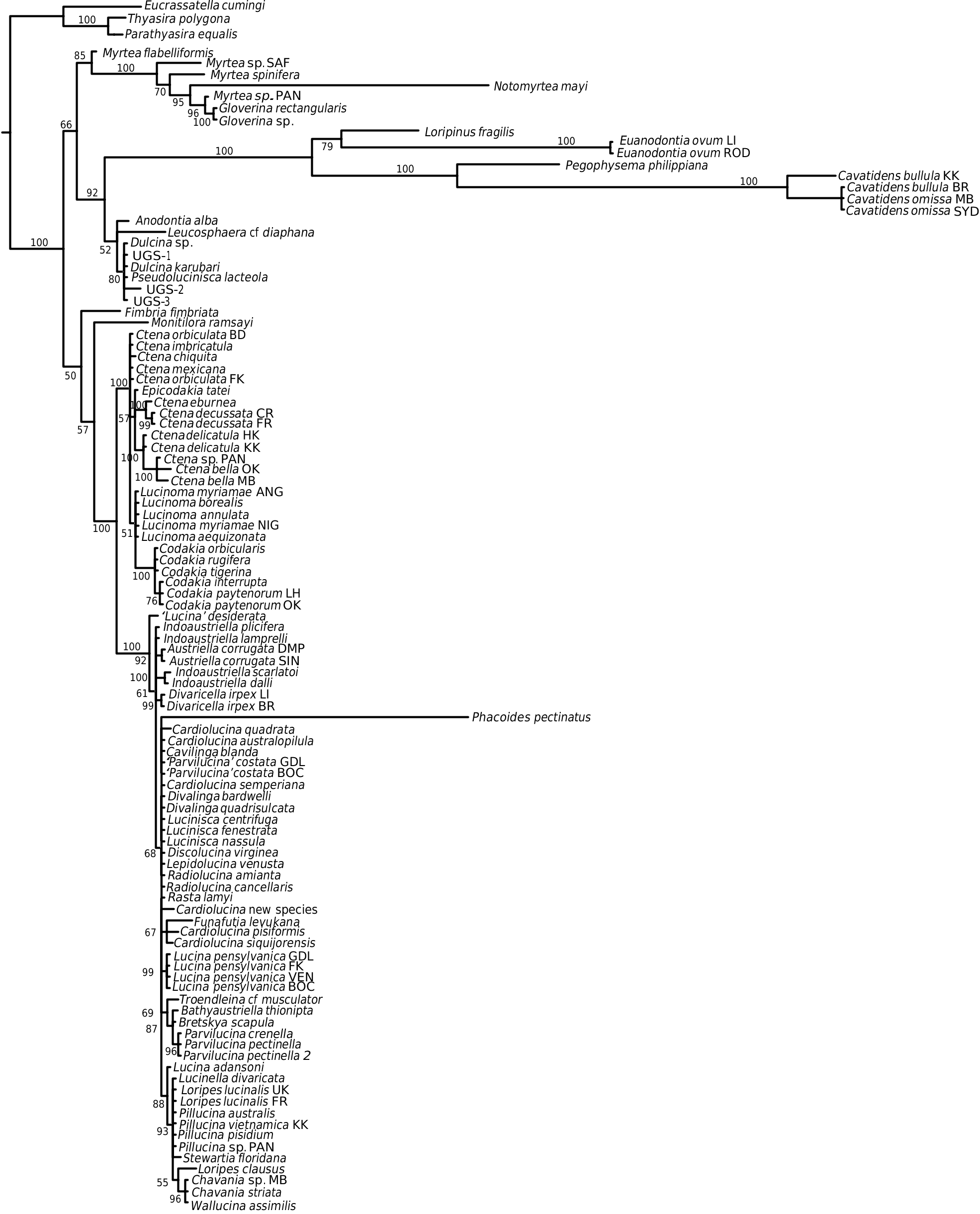
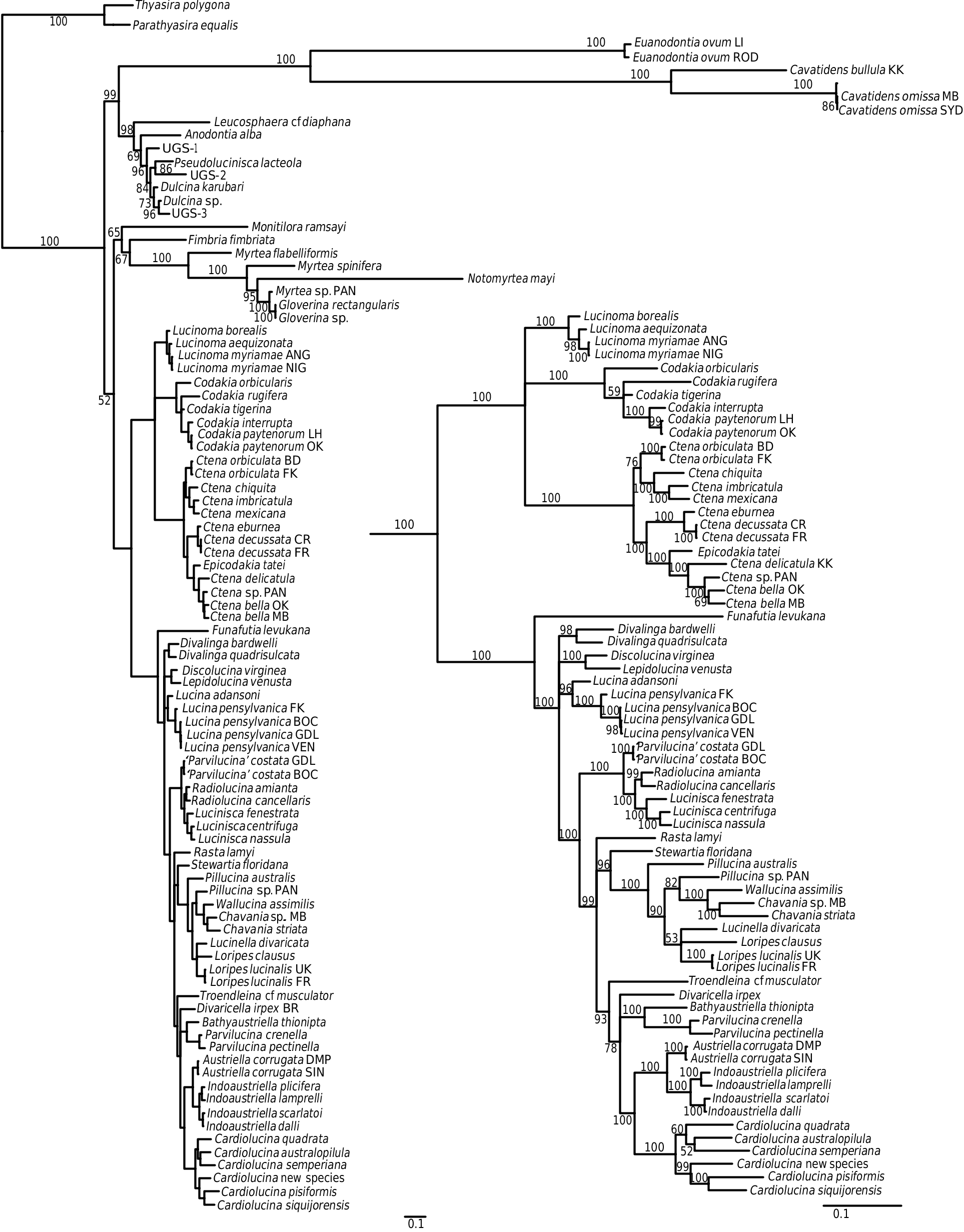
This is a large subfamily encompassing many shallow water lucinids, including Lucina , Loripes , Austriella , Phacoides , Divalinga , the speciose genera Parvilucina , Cardiolucina , and Pillucina , with a few taxa from deeper water. They are extremely disparate in shell morphology, including, for example, heavily sculptured forms with both radial and commarginal elements, and others with almost smooth shells. Most have well-developed hinge teeth, both cardinals and laterals, but there are also edentulous species. The subfamily also includes several species with divaricate sculpture that have sometimes been classified in a separate subfamily Divaricellinae . A number of well-supported subgroups can be identified, and some of these are highlighted below.
Funafutia levukana View in CoL , a small species from fore-reef habitats in the Indo-West Pacific ( Glover & Taylor, 2007 ), has an unstable position. It lies outside and sister to all other Lucininae View in CoL in the combined gene tree ( Fig. 4 View Figure 4 ), although in the 18S tree ( Fig. 1 View Figure 1 ) it lies within Lucininae View in CoL , and in the cyt b tree ( Fig. 3 View Figure 3 ) it groups as a sister to Fimbria View in CoL . We would like to have confirmed this result with a sequence from a second specimen, but no additional material was available. The species was previously thought to be related to the Loripes View in CoL subclade, as it has a short internal ligament ( Glover & Taylor, 2001 ), but does not align within this group in the present analysis, and with hindsight is not very similar in other shell characters.
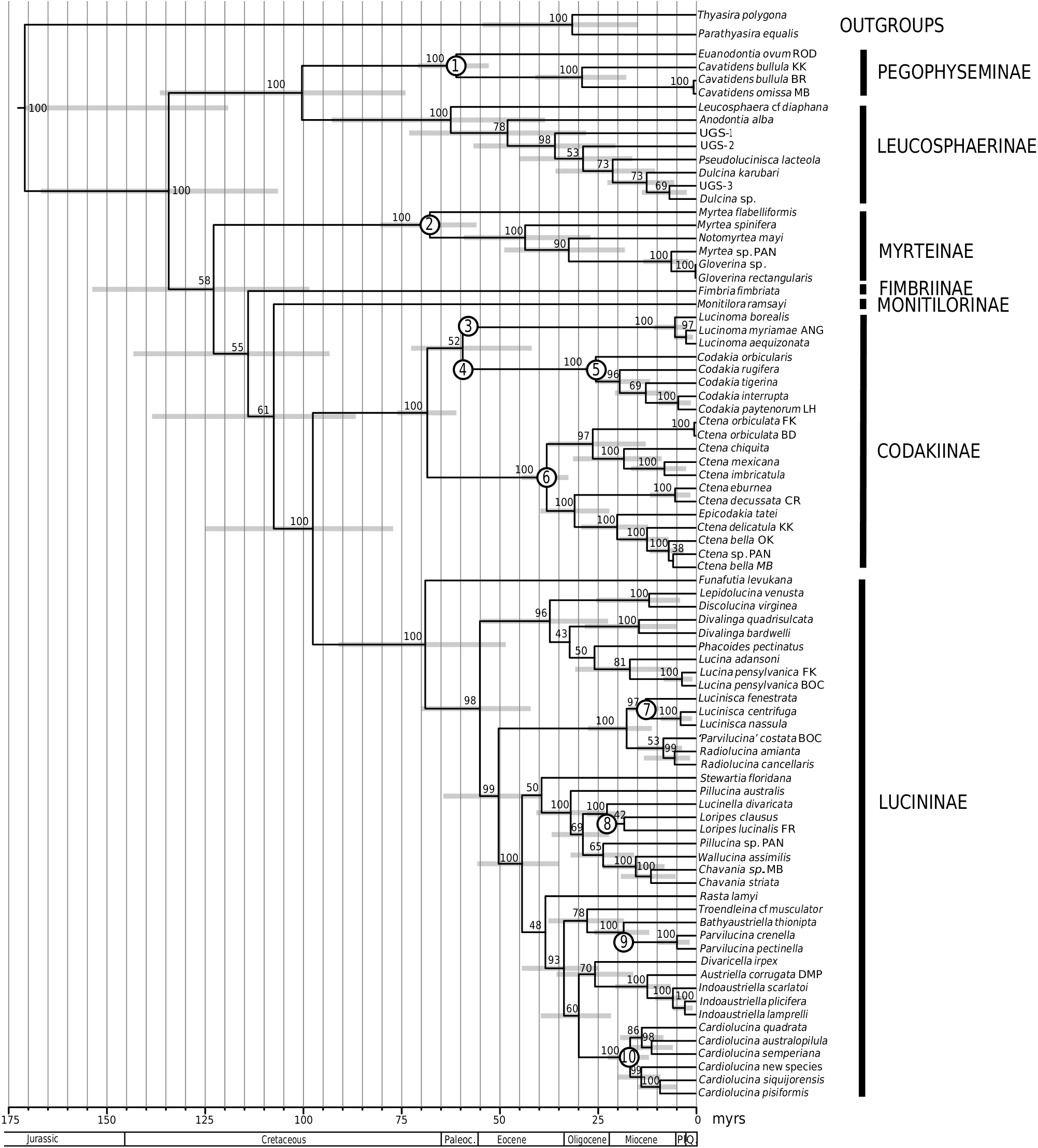
The subclade containing Lucina species also includes Divalinga View in CoL , Lepidolucina View in CoL , and Discolucina View in CoL , as well as Phacoides View in CoL ( Figs 4 View Figure 4 , 5 View Figure 5 ). Two species of Lucina View in CoL were analysed: Lucina adansoni d’Orbigny, 1839 View in CoL from the Cape Verde Islands and Lucina pensylvanica (Linnaeus, 1758) View in CoL from four locations in the western Atlantic, with the sample from Florida Keys differing from the other three sites. Morphological evidence from periostracal characters has suggested a complex of species in the Lucina pensylvanica View in CoL group of the western Atlantic ( Gibson-Smith & Gibson-Smith, 1982; Taylor et al., 2004). The earliest fossil that is usually attributed to Lucina View in CoL is Lucina carinifera Conrad, 1833 View in CoL from the middle Eocene (Bartonian) of eastern USA. Prominent later forms include the abundant Lucina columbella Lamarck, 1818 View in CoL from the Miocene of southern Europe. The chronogram ( Fig. 5 View Figure 5 ) indicates a divergence of Lucina View in CoL from the rest of the clade during the Oligocene rather than the Eocene. Lucina oligocaenica Cossmann, 1922 View in CoL from the Oligocene (Rupelian) of Gironde, France is similar in shell characters to living species.
The position of Phacoides pectinatus , a wellstudied, haemoglobin-rich species from mangrove fringes of the western Atlantic ( Frenkiel, Gros & Mouëza, 1996), is unstable on a long branch in the 18S and 28S trees, but always nests within the Lucininae , and in the chronogram within the Lucina subclade ( Fig. 5 View Figure 5 ). Despite the widespread use of the name in the past for various lucinids now consigned to other genera and subfamilies, there appears to be only one living species of Phacoides , and its relationships with other living genera are uncertain, although Bretsky (1976) classified it as a subgenus of Lucina . Chavan (1937 –1938) claimed a continuous record of Phacoides from the early Eocene, but included in his generic concept were species that are now recognized as belonging to other genera, such as Lamellolucina ( Taylor & Glover, 2002) , so that all records need re-evaluation.
From the western Atlantic and eastern Pacific, three species of Lucinisca , two of Radiolucina , and ‘ Parvilucina ’ costata form another well-supported subclade. Radiolucina amiantus ( Dall, 1901) has been associated with Cardiolucina species (formerly Bellucina ) from the Indo-Pacific by shell characters ( Britton, 1972; Bretsky, 1976), but they are distant in our molecular tree. ‘ Parvilucina ’ costata (d’Orbigny, 1842), a common western Atlantic species, has been variously placed in Codakia , Ctena , and Parvilucina (see Bretsky, 1976), but there is no molecular support for any of these possibilities. A new generic name is needed to accommodate this species and some similar fossils.
A large, highly supported (PP = 100%) subclade includes species of Loripes , Pillucina , Chavania , Wallucina , and Lucinella (hereafter Loripes subclade). Their geographical range covers the eastern Atlantic, Mediterranean, Indo-West Pacific, and southern Australia, but not the western Atlantic and eastern Pacific [with the exception of the rare Loripes brasilianus (d’Orbigny, 1842) from Brazil]. All species within this clade possess a short, obliquely inset, internal ligament that differs structurally from the internal ligament in the Pegophyseminae ( Taylor & Glover, 2005: fig. 28H), and the deeply inset resilifer of Codakia species. Shell sculpture is variable: Loripes and Wallucina have relatively smooth shells with fine commarginal lamellae; Lucinella has divaricate ribbing; and Pillucina species have both radial and commarginal elements. We have not been able to repeat our previous result (Williams et al., 2004), where a specimen of Pillucina vietnamica Zorina, 1978 from Port Douglas, Australia aligned in the Codakiinae clade. In the present analysis other samples of P. vietnamica from Thailand and the Arabian Gulf aligned within the Loripes clade. Using evidence largely from ligament morphology, Chavan (1937 –1938) thought the lucinids of the Loripes subclade were related to Ctena and Codakia , whereas Bretsky (1976) suggested a relationship between Loripes and Anodontia . From the molecular analysis there is no support for either of these suggestions, with the Loripes clade nesting within the Lucininae , and distant from both Codakia and ‘ Anodontia ’ clades.
The earliest Loripes with characters similar to the living species is Loripes dujardini (Deshayes, 1850) ( Fig. 6P) from the early Miocene (Aquitanian 23–20 Ma), and the chronogram suggests a divergence of the whole Loripes subclade in the late Eocene ( Fig. 5 View Figure 5 ). Species of the extinct genus Microloripes ( type species Loripes dentata Defrance, 1823 ) that range from the Palaeocene to the Pliocene also possessed an obliquely inset internal ligament similar to that of Loripes , and may represent a stem group of the clade. Although some lucinid species from the late Jurassic have been placed in Loripes and a subgenus Discoloripes ( Wellnhofer, 1964) , these lack the oblique internal ligament that characterizes the Loripes group, and should be excluded.
The Indo-West Pacific, mangrove-associated Austriella and Indoaustriella species form a robust clade: Austriella corrugata (Deshayes, 1843) lacks hinge teeth, whereas the four Indoaustriella species analysed possess lateral teeth (Glover et al., 2008). There is no fossil record for any of these mangrove species, but a Miocene origin is suggested by the chronogram ( Fig. 5 View Figure 5 ). In our previous analyses ( Glover et al., 2004; Williams et al., 2004), the hydrothermal vent species Bathyaustriella thionipta aligned with Austriella corrugata , with which it shares some shell and anatomical similarities. With the larger taxon set of the present analysis it groups closely with Parvilucina species ( Figs 4 View Figure 4 , 5 View Figure 5 ), but there is little obvious morphological resemblance.
The genus Cardiolucina was represented in our analysis by six species that formed a robust clade, sister to the Indoaustriella / Austriella group ( Figs 4 View Figure 4 , 5 View Figure 5 ). Cardiolucina species are abundant in the Indo- West Pacific ocean, ranging from the intertidal [ Cardiolucina pisiformis (Thiele, 1930) and Cardiolucina semperiana (Issel, 1869) ] to depths of more than 800 m [ Cardiolucina quadrata (Prashad, 1932) ], with two species known from the southern Atlantic ( Taylor & Glover, 1997). Cardiolucina (as Bellucina ) has previously been related to Parvilucina (e.g. Bretsky, 1976), but Chavan (1969) classified it as a subgenus of Lucina (as Linga ). Neither possibility is supported by our molecular analysis. The type species, Cardiolucina agassizi (Michelotti, 1839) occurs in the middle and late Miocene of southern Europe, whereas the chronogram ( Fig. 5 View Figure 5 ) suggests an Oligocene origin for the clade.
Polyphyly of Divaricellinae
Lucinids possessing divaricate sculpture species were formerly united in the subfamily Divaricellinae Korobkov, 1954 (see also Glibert & Van de Poel, 1967; Chavan, 1969), with living species classified into six genera: Bourdotia , Divalinga , Divalucina , Divaricella , Lucinella , and Pompholigina . From morphological evidence and preliminary molecular analysis, doubts have been expressed concerning the monophyly of the group ( Dekker & Goud, 1994; Williams et al., 2004). In our present analysis, Divalinga bardwelli (Iredale, 1936) and Divalinga quadrisulcata (d’Orbigny, 1842) align together, but are widely separated within the Lucininae from Divaricella irpex (Smith, 1885) and Lucinella divaricata (Linnaeus 1758) . The latter species has a hinge and an oblique internal ligament similar to taxa in the Loripes group, where it nests in the molecular analysis. For Divalinga and Divaricella , detailed morphology of the divaricate ribs reveals constructional differences, as demonstrated by Checa (2002), in addition to dissimilarity in hinge and ligament structure. Our evidence suggests a separate evolution of divaricate shell sculpture in the three groups, and we corroborate the remarks of Dekker & Goud (1994) that Divaricellinae is not a valid division of the Lucinidae . Species from other genera with divaricate sculpture, Divalucina , Bourdotia , and Pompholigina , have yet to be analysed. The fossil genus Paralucinella ( type species Lucina undulata Deshayes, 1857 ) from the Oligocene of France has a divaricate sculpture, but lacks an internal ligament, and can be excluded from a relationship with Lucinella .
Uncertain status of Milthinae
Chavan (1969) introduced the subfamily Milthinae , including eight genera with living species and 14 fossil genera, grouping together taxa with commarginal sculpture and long anterior adductor muscle scars. From our molecular analysis most of the included living genera, such as Anodontia , Pegophysema , and Austriella , align in other clades, leaving only Miltha and Eomiltha , which were not included in our analysis, being represented today by three rare living species: Miltha childreni (Gray, 1825) from the western Atlantic, Miltha xantusi (Dall, 1905) from the eastern Pacific, and Eomiltha voorhoevi (Deshayes, 1857) from the Mozambique Channel. The position of both of these genera is uncertain and, despite their names, may not be related. Both Miltha and Eomiltha have fossil records extending to the Palaeocene, with fossil species often abundant throughout the Cenozoic, with a Miltha species common in shallow water deposits of south-western Australia as recently as the early Pleistocene ( Ludbrook, 1978). The fossil genera included by Chavan in Milthinae , such as Pseudomiltha , are frequent in the Eocene, and possess very long anterior adductor muscle scars. Other fossils such as Bulacanities ( Kase & Aguilar, 2007) and Superlucina ( Taylor & Glover, 2009b) also had long adductor scars, but again their relationship to Miltha or any other lucinids is uncertain. In conclusion, the status of Milthinae as conceived by Chavan (1969) is polyphyletic, but the positions of Miltha and Eomiltha are yet to be resolved.
Incongruence of molecular results with classifications based in shell and morphological characters
Molecular analyses have demonstrated that traditional shell characters are a less satisfactory guide to suprageneric relationships, with major incongruencies apparent between the molecular trees and phylogenetic hypotheses derived from shell characters ( Chavan, 1937 –1938; Bretsky, 1970, 1976). These include the position of the ‘ Anodontia ’ group, relationships of Pseudolucinisca , the relationships of Lucinoma with the Codakia / Ctena group, the polyphyly of the shell-based Myrteinae , and non-monophyly of genera with divaricate sculpture. However, no rigorous cladistic analysis of shell characters has yet been attempted, but determining homology would be difficult, with very similar features occurring in several major clades. Hinge teeth characters have been extensively used in discussions of fossil lucinids, but their numbers and morphology seem labile, and edentulous forms appear in several different clades (Glover et al., 2008). An example of the difficulties in using shell characters is in the form of the ligament. In most lucinids the ligament is external and set in a shallow groove, but in a few species part of the ligament is internal and set in an oblique groove that extends posteriorly and laterally into the left and right valves. Such a ligament, exemplified by Loripes lucinalis ( Allen, 1960; Glover & Taylor, 2001 ), also occurs in species of Wallucina , Pillucina , Chavania , and Lucinella , which form a monophyletic subclade within the Lucininae . A similar internal ligament also occurs in Funafutia levukana , a small species from coral reef slope habitats placed within the Pillucina / Wallucina group by Glover & Taylor (2001) . However, the molecular analysis placed this species as a sister taxon to all other Lucininae and distant from the Loripes / Pillucina subclade, suggesting an independent derivation of this form of internal ligament. Some species in the Pegophyseminae clade, such as Euanodontia ovum ( Taylor & Glover, 2005: fig. 28) also possess laterally extended internal ligaments, but these differ in morphology from the Loripes type and are unlikely to be homologous. In summary, a revised and more objective study of shell characters is needed so that the rich fossil record of Lucinidae might be better incorporated into the phylogenetic analysis.
Some anatomical characters, such as the extent and type of posterior mantle fusion ( Taylor & Glover, 2006), also have potential for phylogenetic analysis, but are poorly known for the majority of taxa. Different morphologies of the mantle respiratory organs, the ‘mantle gills’, were reported by Taylor & Glover (2000, 2006). These are sporadically developed amongst the Lucinidae in Pegophyseminae , Codakia species , and, within Lucininae , in Phacoides pectinatus and Lucina species. These organs differ structurally in the different clades suggesting separate derivations. Ctenidial structure has been widely investigated by electron microscopy in several major groups of Lucinidae , but differences are minor with no clear phylogenetic signal (e.g. Frenkiel & Mouëza, 1995; Gros, Frenkiel & Mouëza, 1998; Ball et al., 2009).
Diversification history of Lucinidae
There is a good fossil record of Lucinidae throughout the Mesozoic and Cenozoic, and as demonstrated in the discussion of our recognized subfamilies it is possible to trace morphological lineages of living taxa through the Cenozoic. However, the relationships of some extinct taxa from the early Cenozoic are problematic, as is also the case for most Mesozoic lucinids.
There is only a patchy fossil record of Lucinidae through the Palaeozoic. Illionia prisca (Hisinger, 1837) , a species that was abundant in the late Silurian of Gotland, Sweden ( Liljedahl, 1991a, b), possesses convincing lucinid characters ( Taylor & Glover, 2006). Paracyclas and Phenacocyclas species are known from the Devonian ( LaRocque, 1950; Bailey, 1983; Johnston, 1993). Paracyclas species are common in some facies ( Bailey, 1983), and some of these have ventrally detached, anterior adductor muscle scars characteristic of Lucinidae ( LaRocque, 1950: pl. 12, fig. 12; Taylor & Glover, 2006: fig. 14B), but different from the elongate non-detached scar illustrated by Johnston (1993; figs 81A, 82D), who placed them in a separate family Paracyclidae . An earlier Paracyclas species from the Ordovician is claimed by Zong-Jie & Cope (2004), but the preservation is poor. Palaeolucina is widely reported from the Carboniferous of China ( Chen, 1976), but few details of morphology are available. Later, Gigantocyclus ( Boyd & Newell, 1979) from the Permian of Tunisia can be confidently placed in the Lucinidae .
For the Mesozoic there is a dearth of confirmed lucinids from the Triassic: Hautmann & Nützel (2005) suggested the small early Triassic species, Sinbadiella pygmaea Hautmann & Nützel, 2005 , as a possible lucinid, but without strong evidence. ‘ Lucina ’ deshayesii Klipstein, 1845 ( holotype, BMNH Palaeontology Department 35194) from the St Cassian Formation (late Triassic, Carnian) may be a true lucinid, but the preservation is poor. Schaflaeutlia is usually classified in the Fimbriinae ( Chavan, 1969; Hautmann, 2001), but we regard this placement as uncertain. By the late Triassic (Rhaetian), lucinids resembling modern forms are more frequent (Ivimey- Cook et al., 1999). Throughout the Jurassic and Cretaceous lucinids are more diverse, but are generally under-researched. Preservation is often poor, with many nominal species based on internal moulds, with hinge and muscle scar details often unknown. Many species are still named ‘ Lucina ’ or are ‘shoe-horned’ into the few available generic names, and most need critical study. A few studies of well-preserved faunas indicate that diversity and abundance of Mesozoic lucinids is greater than was commonly thought, with several new genera introduced over last few years ( Chavan, 1952; Fischer, 1969; Kelly, 1992; Kendrick & Vartak, 2007; Amano et al., 2008; Kiel, Campbell & Gaillard, 2010). Furthermore, only a few small species (< 1cm in length) have been described, although they undoubtedly existed as evidenced in Cenozoic and Recent faunas. At present it is difficult to assess the relationships of most Mesozoic lucinids to later Cenozoic and modern clades.
By contrast to earlier periods, Cenozoic faunas are much better documented. Nonetheless, there is a significant increase in the diversity and abundance of lucinids from the Palaeocene onwards. For example, from the Palaeocene (Danian) of Belgium, 24 species of lucinids were recorded, comprising a remarkable 19% of the total bivalve species found ( Glibert & Van de Poel, 1973). Ninety-six species in 28 genera are recorded from the Eocene of the Paris Basin (Cossmann & Pissaro, 1904–06; Pacaud & Le Renard, 1995), and similarly diverse faunas occur in the Miocene ( Sacco, 1901; Cossmann & Peyrot, 1909 –1912; Gardner, 1926). The chronogram ( Fig. 5 View Figure 5 ) demonstrates that many of the clades of living lucinids originated in the Eocene, with radiations in the Miocene. Notable appearances in the late Cenozoic were the ecologically important Codakia and Ctena species in shallow tropical habitats, and also the dominance of Lucinoma species at hydrocarbon seeps, especially in the northern Pacific ( Majima, Nobuhara & Kitazaki, 2005). Some lucinids that were abundant during the early Cenozoic have either become extinct, such as Pseudomiltha species , or, are now extremely rare, as for example Eomiltha and Miltha .
In pre-Miocene times the external sculpture of lucinids was usually in the form of low commarginal lamellae, but then species with radial and cancellate sculpture became much more frequent in the Miocene and in younger faunas. Genera with prominent radial ribbing include shallow water tropical species in the Codakiinae and Lucininae clades such as Codakia , Ctena , Cardiolucina , Lucinisca , Pleurolucina , Parvilucina , and Radiolucina .
Patterns of habitat occupation
Most lucinids occur in tropical, subtropical, or warm temperate waters, the few species inhabiting cool temperate latitudes are mainly species of Lucinoma , for example Lucinoma filosa (Stimpson, 1851) , from the eastern seaboard of North America to New Brunswick, Canada ( Britton, 1970), Lucinoma annulata (Reeve, 1850) from the eastern Pacific to 61°N in Alaska ( Coan, Valentich Scott & Bernard, 2000), Lucinoma borealis from northern Europe to the Lofoten Islands ( 68°N), Norway (BMNH collections), and Lucinoma lamellata (Smith, 1881) at Cape Horn ( 55°45′S) ( Holmes, Oliver & Sellanes, 2005). Additional higher latitude species include Myrtea spinifera (Montagu, 1803) , which ranges from the Mediterranean to 64°48′N in Norway, and Loripes lucinalis from north western Africa to southern Britain, whereas Parvilucina tenuisculpta (Carpenter, 1864) occurs from the north-eastern Pacific to southern Alaska ( 61°N) ( Coan et al., 2000). The tiny ‘ Epicodakia ’ falklandica (Dell, 1964) is recorded from the Southern Ocean ( Zelaya, 2005), but the generic placement of this species is uncertain.
Lucinids have often been reported from various marine habitats with an elevated input of organic material, such as seagrass beds or hydrocarbon seeps. In this section we address the question of whether there is any phylogenetic pattern to the habitat preferences of lucinids: are some individual clades associated with particular habitats?
Seagrass beds: There is a well-known strong association of lucinids with seagrass beds both in tropical and temperate seas (reviewed by Barnes & Hickman, 1999). Seagrass beds entrap organic detritus, and have higher rates of sulphate reduction and sulphide retention than surrounding unvegetated sediment ( Holmer, Duarte & Marba, 2003; Holmer et al., 2009). Root and rhizome masses create stable habitats and release oxygen through the roots. Notable amongst the lucinids that inhabit seagrass beds are several Codakia species [ Codakia orbicularis , Codakia punctata (Linnaeus, 1758) , Codakia tigerina (Linnaeus, 1758) , and Codakia paytenorum (Iredale, 1937) ] that are abundant in Thalassia -dominated habitats of the western Atlantic and Indo-West Pacific ( Moore et al., 1968; Taylor & Lewis, 1970; Jackson, 1972). As well as Codakia orbicularis , seagrass beds of the western Atlantic also support populations of Ctena species , Lucina pensylvanica , Anodontia alba , and Lucinisca nassula (Conrad, 1846) (J. D. Taylor & E. A. Glover, pers. observ.), and Stewartia floridana ( Fisher & Hand, 1984) . In the Indo-West Pacific, Euanodontia ovum , Cavatidens omissa Iredale, 1930 , and other pegophysemines, Pillucina species ( Nakaoka, Mukai & Chunhabundit, 2002; Meyer et al., 2008), Chavania striata (Tokunaga, 1906) , and Divaricella irpex (E. A. Glover & J. D. Taylor , unpubl. data) are widely associated with seagrass habitats. From temperate latitudes, examples of the association include Loripes lacteus (Linnaeus, 1758) , which is abundant in Mediterranean Posidonia beds ( Johnson, Fernandez & Pergent, 2002), Wallucina assimilis (Angas, 1868) from southern Australia ( Barnes & Hickman, 1999), Lucinoma borealis from southern England ( Dando, Southward & Southward, 1986; E. A. Glover & J. D. Taylor , pers. observ.), and Pillucina pisidium (Dunker, 1860) from Japan ( Uede & Takahashi, 2008). From this brief survey it is clear that although Codakia species are the largest and most conspicuous species, lucinids associated with seagrass derive from four major clades: the Pegophyseminae , Leucosphaerinae , Codakiinae, and Lucininae . Species of Myrteinae usually live at water depths below those colonized by seagrasses, but we have collected Notomyrtea mayi from an intertidal seagrass habitat in South Australia.
Seagrass-dominated shallow water communities became widespread during the Eocene ( Brasier, 1975), and the association with lucinids dates from this time. Various Eocene deposits of the Paris Basin have been interpreted as representing seagrass-dominated communities ( Murray & Wright, 1974), with lucinids found frequently amongst the molluscs ( Pacaud & Le Renard, 1995). From the Eocene (late Lutetian) Selsey Formation of southern England lucinids occur in association with plant remains of seagrasses ( Collinson, 1996; Tracey et al., 1996). Additionally, the giant Superlucina megameris ( Dall, 1901) from the Eocene, White Limestone of Jamaica, has been interpreted as an inhabitant of seagrass communities ( Taylor & Glover, 2009b). Such associations have continued through the Cenozoic, as for example in the early Oligocene of the Etampes area of the Paris Basin ( Gitton, Lozouet & Maestrati, 1986), and in the Roe Calcarenite of south-west Australia (late Pliocene– early Pleistocene), which has been interpreted as a seagrass-dominated deposit ( James & Bone, 2007), where Miltha hamptonensis Ludbrook, 1969 was common, along with other lucinids ( Ludbrook, 1978).
Mangrove association: Mangrove communities are another shallow water marine ecosystem with a high organic input. In the present day, only a few lucinids are closely associated with mangrove environments. Notable amongst these are Austriella corrugata and Indoaustriella species from the central Indo-West Pacific (Glover et al., 2008), Pegophysema philippiana from the Indo-West Pacific ( Lebata & Primavera, 2001; Taylor & Glover, 2005), Phacoides pectinatus from the western Atlantic, which inhabits black muds amongst mangroves ( Frenkiel et al., 1996), and Keletistes rhizoecus (Oliver, 1986) from West Africa, which lives in mangrove root peat in the Niger Delta ( Zabbey, Hart & Wollf, 2010). In South-East Asia, lucinids can also be abundant in communities peripheral to mangrove stands ( Meyer et al., 2008). Although organic-rich, the suboxic sediments are a less favourable habitat than seagrass beds for burrowing bivalves, because the low pH causes extensive corrosion of shells and the substrata are often very soft. In summary, mangrove-associated lucinids derive from two main clades, Pegophyseminae and Lucininae , and within the latter the well-defined Austriella / Indoaustriella subclade and Phacoides pectinatus . Mangrove systems diversified during the Palaeocene, and by the mid to late Eocene were widespread in the tropics and subtropics ( Plaziat et al., 2001). Although some mangrove-associated gastropods are recorded from Cenozoic deposits ( Reid et al., 2008), lucinids have not been documented.
Deeper water habitats: Most lucinid species have been described from shallow water habitats, generally from water depths of less than 100 m. Until recently, relatively few lucinids were known from deeper water, but increased sampling of bathyal habitats in the tropics and hydrocarbon seeps is revealing an unexpected diversity, with lucinids now known to depths of 2570 m ( Cosel, 2006; Cosel & Bouchet, 2008; Taylor & Glover, 2009a). The relationships of some of these newly described taxa are uncertain, but they most likely can be classified in Leucosphaerinae or the Myrteinae clades. Deep-water lucinids that have been analysed molecularly derive from: Myrteinae ( Myrtea and Gloverina ); Leucosphaerinae , with genera such as Dulcina and several undescribed species; the Lucinoma clade of the Codakiinae; and a few species from the Lucininae such as Troendleina musculator Cosel & Bouchet, 2008 , Bathyaustriella thionipta , and Cardiolucina quadrata . The deepest recorded lucinid is Elliptiolucina labeyriei Cosel & Bouchet, 2008 from 2570 m in the Sulu Sea. Nevertheless, Lucinoma species are the lucinids most widely reported from deeper water habitats, and several have been found in association with hydrocarbon seeps and mud volcanoes. These include Lucinoma atlantis Mclean, 1936 ( Cordes et al., 2007), Lucinoma anemiophila Holmes, Oliver & Sellanes, 2005 , Lucinoma yoshidai Habe, 1958 ( Okutani & Hashimoto, 1997), Lucinoma kazani Salas & Woodside, 2002 , and Lucinoma myriamae ( Cosel, 2006) from off West Africa, included in our analysis from 2050 m. Additionally, two species, Lucinoma aequizonata (Stearns, 1890) and Lucinoma gagei Oliver & Holmes, 2006 are known from sediments of oxygen minimum zones ( Cary et al., 1989; Oliver & Holmes, 2006). Other lucinids recorded as associated with hydrocarbon seeps include Graecina karinae Cosel, 2006 , Graecina colombiensis Taylor & Glover, 2009a , Meganodontia acetabulum Bouchet & Cosel 2004 , Mesolinga soliditesta Okutani & Hashimoto 1997 , Myrtea amorpha ( Olu-Le Roy et al., 2004), and Jorgenia species from the Louisiana Slope ( Taylor & Glover, 2009a). The only lucinid recorded as yet from a hydrothermal vent is Bathyaustriella thionipta from the Kermadec Ridge off New Zealand ( Glover et al., 2004). As well as hydrocarbon seeps, Cosel & Bouchet (2008) suggest that around the Philippines and Indonesia the many semi-enclosed basins with poor water circulation and accumulations of decomposing continental plant debris create favourable habitats for chemosymbiotic animals.
The fossil record indicates that lucinids have been present at putative hydrocarbon seeps since at least the late Jurassic ( Gaillard et al., 1992; Kiel et al., 2010), through the Cretaceous with Ezolucina , Nipponothracia , and Nymphalucina species ( Speden, 1970; Kauffman et al., 1996; Kelly et al., 2000; Amano et al., 2008; Kiel, Amano & Jenkins, 2008), and in the Cenozoic with, for example, species of Cryptolucina , Nipponothracia , Megaxinus , and Lucinoma ( Taviani, 1994; Saul, Squires & Godert, 1996; Majima et al., 2003; Campbell, 2006; Kase, Kurihara & Hagino, 2007).
Symbionts and co-evolution
The new lucinid phylogeny will provide a framework to examine the distribution of the bacterial symbiont phylotypes. No comprehensive molecular analysis of symbionts and their hosts has yet been attempted, although details of some lucinid symbionts are available, mainly from western Atlantic host species ( Distel, Felbeck & Cavanaugh, 1994; Durand & Gros, 1996; Dubilier, Bergin & Lott, 2008). All symbiont phylotypes recorded from lucinid ctenidia to date belong to the gamma group of proteobacteria. Experimental evidence shows that symbionts are acquired by environmental transmission from the sediment ( Gros et al., 1998), suggesting that a close co-evolutionary history between host and symbiont is unlikely. Indeed, several lucinids from seagrass beds around Guadeloupe shared the same symbiont ( Gros et al., 2003). Although few symbiont sequences from lucinids have yet been published, preliminary results ( Cavanaugh et al., 2006; Dubilier et al., 2008; Ball et al., 2009) show some differentiation between hosts, so that the symbionts of Phacoides pectinatus , Euanodontia ovum , and Pegophysema schrammi (as Anodontia philippiana in the cited papers) are distinct from the symbionts associated with other lucinids (e.g. Lucinisca , Codakia , Stewartia , and Lucinoma ) that group more closely together. The addition of further taxa from different host clades and from a wider range of geographical locations and habitats should result in an improved understanding of host–symbiont relationships and the diversity of symbionts.
Phylogeography
In the present analysis, Ctena species provide the most informative data set for phylogeographic analysis. The species form two distinct groups, one consisting of both western Atlantic [ Ctena orbiculata (Montagu, 1808) and Ctena imbricatula (Adams, 1845) ; note the latter species is usually synonymized with Ctena orbiculata , but molecular data shows that they are distinct species] and eastern Pacific species [ Ctena chiquita ( Dall, 1901) and Ctena mexicana ( Dall, 1901) ], and the other comprising West African [ Ctena eburnea (Gmelin, 1791) ], Mediterranean [ Ctena decussata ( Costa, 1829) ], and Indo-West Pacific species [ Ctena bella (Conrad, 1834) , Ctena delicatula (Pilsbry, 1904) , Epicodakia tatei , and Ctena sp. ]. Albeit with only limited sampling, this phylogeny suggests an Atlantic origin with later radiations in the Indo-West Pacific, consistent with the pattern observed in many other taxa (e.g. Williams & Reid, 2004). The chronogram ( Fig. 5 View Figure 5 ) estimates that the western Atlantic/eastern Pacific clade diverged from the eastern Atlantic/Mediterranean and Indo-West Pacific clades in the late Eocene. In the western Atlantic/eastern Pacific clade, Ctena orbiculata is sister to the eastern Pacific species and Ctena imbricatula , with a divergence in the middle Oligocene. In the other clade, the Atlantic and Mediterranean species ( Ctena eburnea and Ctena decussata ) are sister to the Indo-West Pacific species, with a divergence in the early Oligocene, earlier than estimates of the closure of the Tethys Seaway, approximately 19–18 Ma ( Vrielynck, Odin & Dercourt, 1997; Rögl, 1998; Harzhauser et al., 2002, 2007), although 95% HPD ranges suggest a more recent separation is also possible. The Indo-West Pacific clade diversified in the early Miocene, consistent with age estimates of many other tropical, shallow water Indo-West Pacific radiations (e.g. Williams & Duda, 2008).
Codakia View in CoL is the only other genus with sufficient interspecific sampling to make any biogeographic comment. In this clade the western Atlantic Codakia orbicularis View in CoL occupies a position sister to the other species from the Indo-West Pacific that may have separated in the late Oligocene, slightly later than that of Ctena View in CoL and Epicodakia View in CoL (although the 95% HPD ranges overlap). Divergence times between eastern Pacific/Atlantic and Indo-West Pacific clades have been noted to differ in several studies (e.g. Frey & Vermeij, 2008). Some of this variation may reflect uncertainty in molecular clock analyses or real differences, resulting from multiple vicariant events or the fact that the constriction of the Tethys Seaway was not an instantaneous event, but extended over several millions of years ( Bellwood, van Herwerden & Konow, 2004). The latter seems particularly pertinent, given a similar pattern of protracted speciation events between marine species either side of the Isthmus of Panama ( Lessios, 2008). Other possible explanations for disparate estimates include the extinction of true sister lineages ( Paulay, 1997) or the role of alternate vicariant events ( Reid, Dyal & Williams, 2010).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
LUCININAE
| Taylor, John D., Glover, Emily A., Smith, Lisa, Dyal, Patricia & Williams, Suzanne T. 2011 |
Lepidolucina
| Glover & Taylor 2007 |
Discolucina
| Glover & Taylor 2007 |
Divalinga
| Chavan 1951 |
Epicodakia
| Iredale 1930 |
Lucina oligocaenica
| Cossmann 1922 |
Ctena
| Morch 1861 |
Phacoides
| Gray 1847 |
Lucina adansoni d’Orbigny, 1839
| d'Orbigny 1839 |
Lucina carinifera
| Conrad 1833 |
Lucininae
| J. Fleming 1828 |
Lucininae
| J. Fleming 1828 |
Lucina columbella
| Lamarck 1818 |
Fimbria
| Muhlfeld 1811 |
Lucina
| Bruguiere 1797 |
Lucina
| Bruguiere 1797 |
Lucina
| Bruguiere 1797 |
Loripes
| Poli 1791 |
Codakia
| Scopoli 1777 |