Daphnephila urnicola Chiang, Yang & Tokuda
|
publication ID |
https://doi.org/ 10.11646/zootaxa.3955.3.5 |
|
publication LSID |
lsid:zoobank.org:pub:D8BFF6D1-FDF7-44EB-B8E7-DA9261834E7D |
|
DOI |
https://doi.org/10.5281/zenodo.5675637 |
|
persistent identifier |
https://treatment.plazi.org/id/0D249D6A-707A-A33B-3FF7-FCE24DA3CA90 |
|
treatment provided by |
Plazi |
|
scientific name |
Daphnephila urnicola Chiang, Yang & Tokuda |
| status |
sp. nov. |
Daphnephila urnicola Chiang, Yang & Tokuda sp. nov.
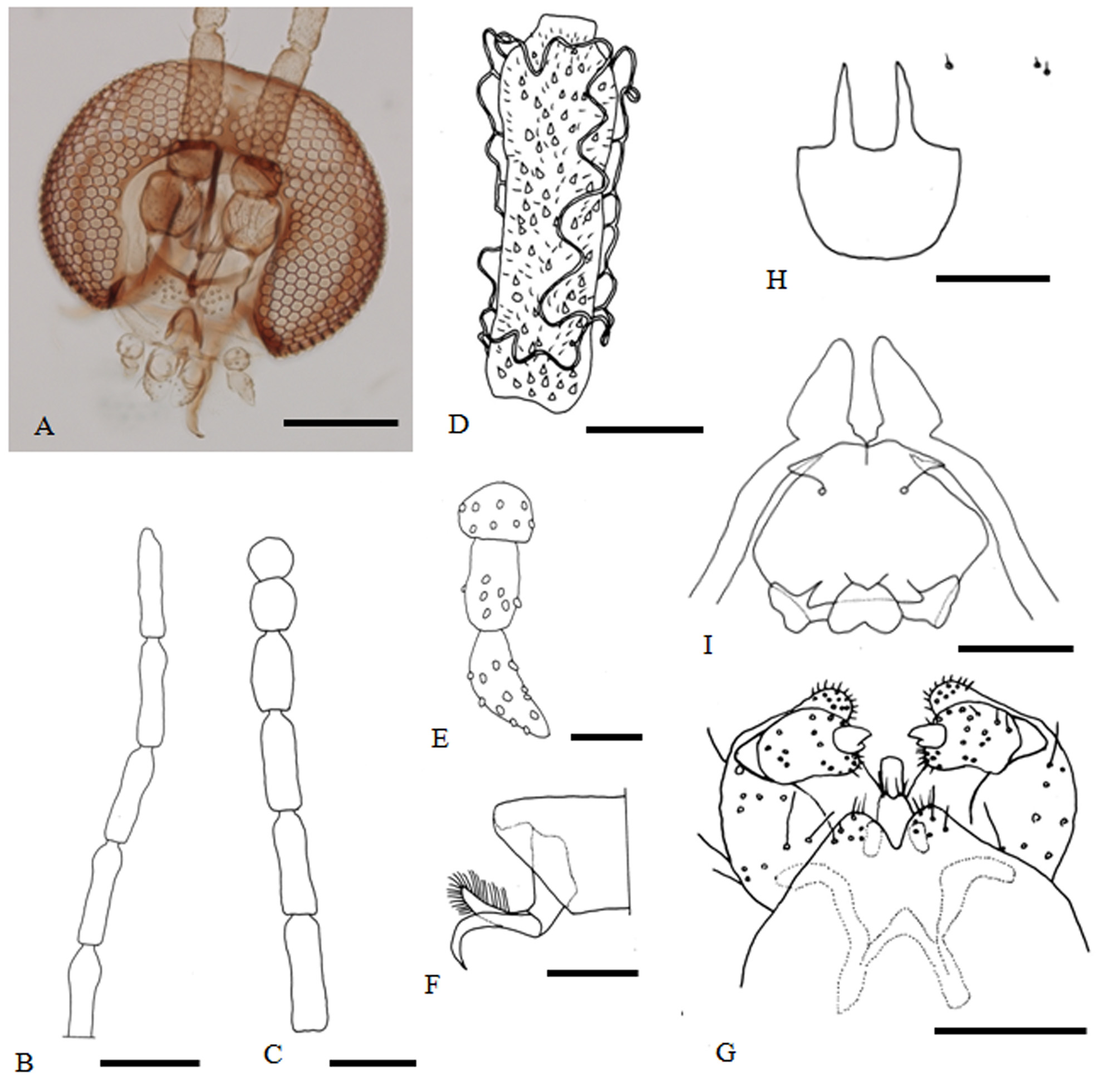
MALE ( Fig. 4 View FIGURE 4 B, D–G) Eye bridge 3–4 facets long medially. Among the twelve flagellomeres, first flagellomere 175–190 µm long, 3.1–3.5 times as long as wide, 1.1–1.2 times as long as second; fifth flagellomere 160–175 µm long, 3.2–3.5 times as long as wide; twelfth flagellomere 140–145 µm long, 2.8–4.3 times as long as wide. Frontoclypeus with 26–27 setae. Labella hemispherical in lateral view, with 32–37 setae. Palpus three-segmented, first segment globose in ventral view, about 29 µm long; second about 1.5 times as long as first; third about 1.2 times as long as second; distal two palpal segments sometimes more or less fused.
Wing length about 3.0 mm long, 2.3–2.4 times as long as wide. Scutum with two rows of dorsocentral setae; anteriorly with 38–39 dorsolateral setae, posteriorly with 44–50 dorsolateral setae. Anepisternum with 15–17 scales, mesepimeron with 44–75 setae.
First through seventh abdominal tergites rectangular, eighth tergite not sclerotized. Second through seventh abdominal sternites 0.42–0.50 times as long as wide; eighth sternite small, 0.79 times as wide as seventh sternite, about 0.60 times as long as wide.
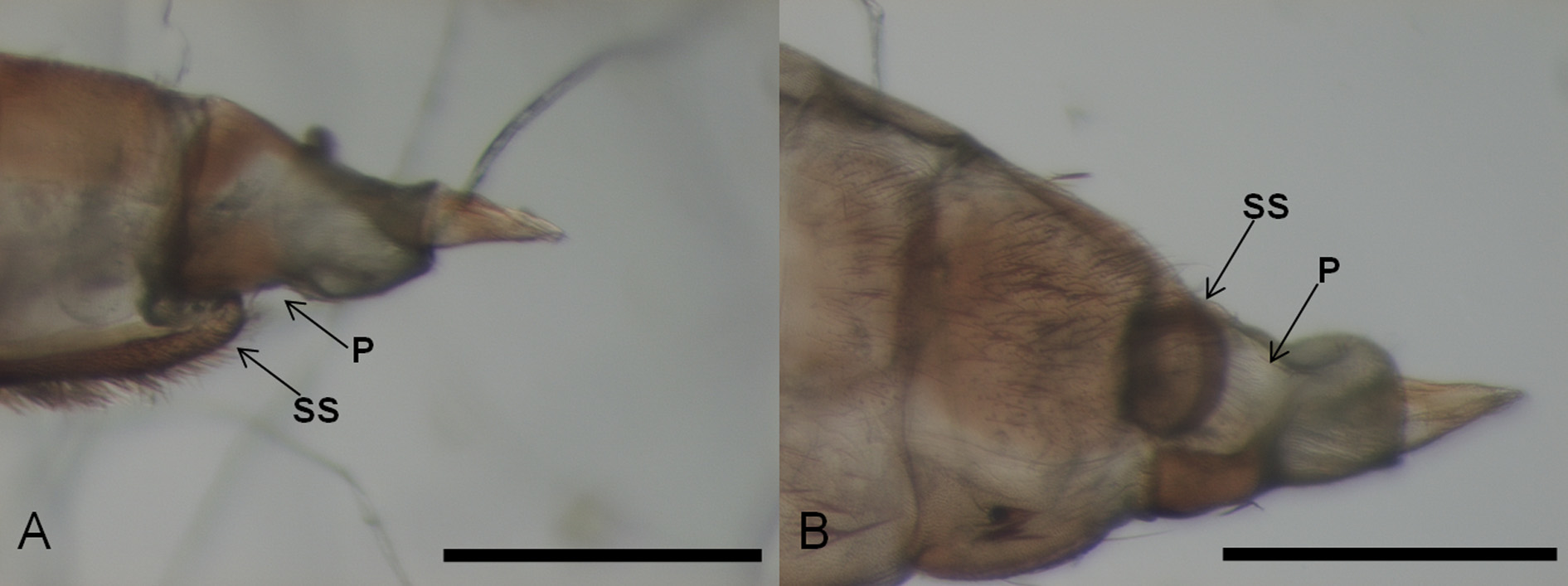
Terminalia: hypoproct incised by shallow or deep emargination, forming a pair of lobes, each lobe with a few apical setae; mediobasal lobe shorter than cerci; aedeagus nearly parallel sided, truncate apically.
FEMALE ( Fig. 4 View FIGURE 4 A, C). Frontoclypeus with 17–23 setae. First flagellomere 120–200 µm long, 2.4–3.6 times as long as wide, about 1.1–1.2 times as long as second; fifth flagellomere 133–145 µm in length, 2.9–3.2 times as long as wide; distal flagellomeres shortened and terminal one subglobular.
Wing length about 2.3 mm, 2.3–2.5 times as long as wide. Anterior dorsolaterally with 35–51 setae, posterior dorsolaterally with 48–58 setae. Anepisternum with 17–20 scales, mesepimeron with 43–60 setae.
First through seventh abdominal tergites and second through sixth sternites as in male. Seventh abdominal sternite setose, 520–560 µm long, about 1.1–1.3 times as long as wide.
Ovipositor short, with many long setae ventrally on apical half; cerci-like structure bilobed, situated on anterior of intersegmental membrane between eighth and ninth abdominal segments.
MATURE LARVA ( Fig. 4 View FIGURE 4 H). Body light yellow. Second antennal segment short, subglobular; two cervical papillae each with a minute seta. Spiracle present on prothorax and first through seventh abdominal segments, invisible on eighth abdominal segment; four dorsal papillae present on thoracic segments, each with a minute seta; two dorsal papillae present on first through seventh abdominal segments, each with a minute seta; terminal papillae not apparent. Sternal spatula semicircular, anteriorly with two slender, acutely pointed lobes, each lobe 60–70 µm long; two lateral papillae present on thoracic segments, each with minute seta. First through seventh abdominal segments with two posterior ventral papillae, each with a minute seta; terminal papillae not apparent.
PUPA ( Fig. 4 View FIGURE 4 I). Body length 3.1–4.1 mm. Length from the base of antennal sheath to the tip of apical horn 295–320 µm long; prothoracic spiracle 145–230 µm long; abdominal spiracles on second to seventh segments about 22–35 µm long; second through eighth abdominal segments with 5–7 transverse rows of rather long spines on anterior half of dorsal surface, each spine 16–38 µm long.
Material examined. Holotype: ♂ (on slide, NCHU 2013004-001, deposited in NCHU) emerged on 18 March 2008 from a gall on M. zuihoensis collected by T. C. Chiang on 17 March 2008 from Lung-Ding Ancient Road (323m asl.), Changhua, Taiwan.
Paratypes: 8 ♂ & 1 ♀ (on slides, NCHU 2013004-002 to 010; NCHU 2013004-002 (1♂) and 0 10 (1♀) deposited in NMNS and others deposited in NCHU), same data as holotype; 4 ♀ (on slides, NCHU 2013004-011 to 0 14, deposited in NCHU), Lung-Ding Ancient Road (374m asl.), Changhua, Taiwan, same data as holotype; 2 mature larvae (on slides, NCHU 2013004-015 to 0 16, deposited in NCHU), Lung-Ding Ancient Road (323m asl.), Changhua, Taiwan, same data as holotype; 2 mature larvae (on slides, NCHU 2013004-017 to 18, deposited in NCHU), collected by Y.T. Hong on 15 January 2008 from same locality as holotype; 2 ♂ & 2 ♀ (on slides, NCHU 2013004-019 to 0 22, NCHU 2013004-019 (1♂) deposited in ELKU and others deposited in NCHU), collected by T. C. Chiang on 16 March 2008 from Chushui Lane (349m asl.), Changhua, Taiwan; 3 ♂ (wet in 70% Ethanol, NCHU 2013004-023 to 025; deposited in NCHU), 2 ♀ (wet in 70% Ethanol, NCHU 2013004-026 to 027; deposited in NCHU), 1 ♂ (on a slide, NCHU 2013004-028; deposited in NCHU) & 1 ♀ (on a slide, NCHU 2013004-029; deposited in NCHU) from galls on M. zuihoensis collected by T. C. Chiang on 16 March 2008 from Sihu (395m asl.), Miaoli, Taiwan; 2 ♀ (on slides, NCHU 2013004-030 to 031; deposited in ELKU and NCHU), from galls on M. zuihoensis collected by T. C. Chiang & W. N. Chen on 26 March 2009 from Da-Han woodland path (640m asl.), Pingtung, Taiwan; 1 mature larva & 2 pupae (on slides, NCHU 2013004-032 to 034; deposited in NCHU) from galls on M. zuihoensis collected by T. C. Chiang & W. N. Chen on 26 March 2009 from Lilishan (882m asl.), Pingtung, Taiwan; 1 ♀ & 1 pupa (on slides, NCHU 2013004-035 to 036; deposited in NCHU) from galls on M. zuihoensis collected by T. C. Chiang on 20 April 2008 from Lilishan (873m asl.), Pingtung, Taiwan; 1 mature larva (on a slide, NCHU 2013004-037; deposited in NCHU) from a gall on M. zuihoensis collected by T. C. Chiang on 16 February 2010 from Pi-Lu Zen Temple (295m asl.), Houli, Taiwan; 1 ♂ & 1 mature larva (on slides, NCHU 2013004-038 to 039; deposited in NCHU) from galls on M. mushaensis collected by T. C. Chiang on 19 March 2008 from Dasyueshan (1393m asl.), Taichung, Taiwan; 1 ♂ & 1 mature larva (on slides, NCHU 2013004-040 to 041; deposited in NCHU) from galls on M. mushaensis collected by T. C. Chiang on 19 March 2008 from Dasyueshan (1418m asl.), Taichung, Taiwan; 1 pupa (on a slide, NCHU 2013004- 042; deposited in NCHU) from a gall on M. mushaensis collected by T. C. Chiang on 0 2 March 2010 from Gao- Feng Lane (1234m asl.), Nantou, Taiwan.
Distribution. [ Taiwan] 300–1300 asl. primary and secondary forest ( Fig. 2 View FIGURE 2 ).
Host plants. Machilus zuihonensis Hayata and M. mushaensis Lu (Lauraceae) .
Galls. Urn-shaped, with 3–7 vertical ridges along the outer surface, hypophyllous ( Fig. 1 View FIGURE 1 ), remain green from young to dehiscent stages, single chambered.
Etymology. The specific name, urnicola , refers to the urn-shaped (urna) gall that the species induces and dwells (cola) in.
Remarks. In terms of morphology, the new species can be distinguished from the other Daphnephila as follows. The Indian D. haasi Kieffer , D. glandifex Kieffer and D. linderae Kieffer were described as having the posterior margin of the hypoproct entire ( Yukawa 1974), whereas in D. urnicola the hypoproct has a shallow incision. The new species is distinguishable from five Taiwanese Daphnephila associated with M. thunbergii by the following characters: from D. ornithocephala Tokuda, Yang & Yukawa by the four-segmented palpus; from D. stenocalia Tokuda, Yang & Yukawa by the acute pupal antennal horns; from D. sueyenae Tokuda, Yang & Yukawa by the longer mediobasal lobe; from D. taiwanensis Tokuda, Yang & Yukawa by the presence of setose pupal apical papillae and longer female seventh abdominal sternite; from D. truncicola Tokuda, Yang & Yukawa by the shape of the larval sternal spatula, setose pupal apical papillae, and wider aedeagus. Daphnephila machilicola from Japan differs from D. urnicola in having pupal lower facial papillae ( Yukawa 1974). In addition, it should be noted that unlike other species, spiracles of the larval eighth abdominal segment are not apparent in D. urnicola . This character may be useful for future taxonomic studies of this genus.
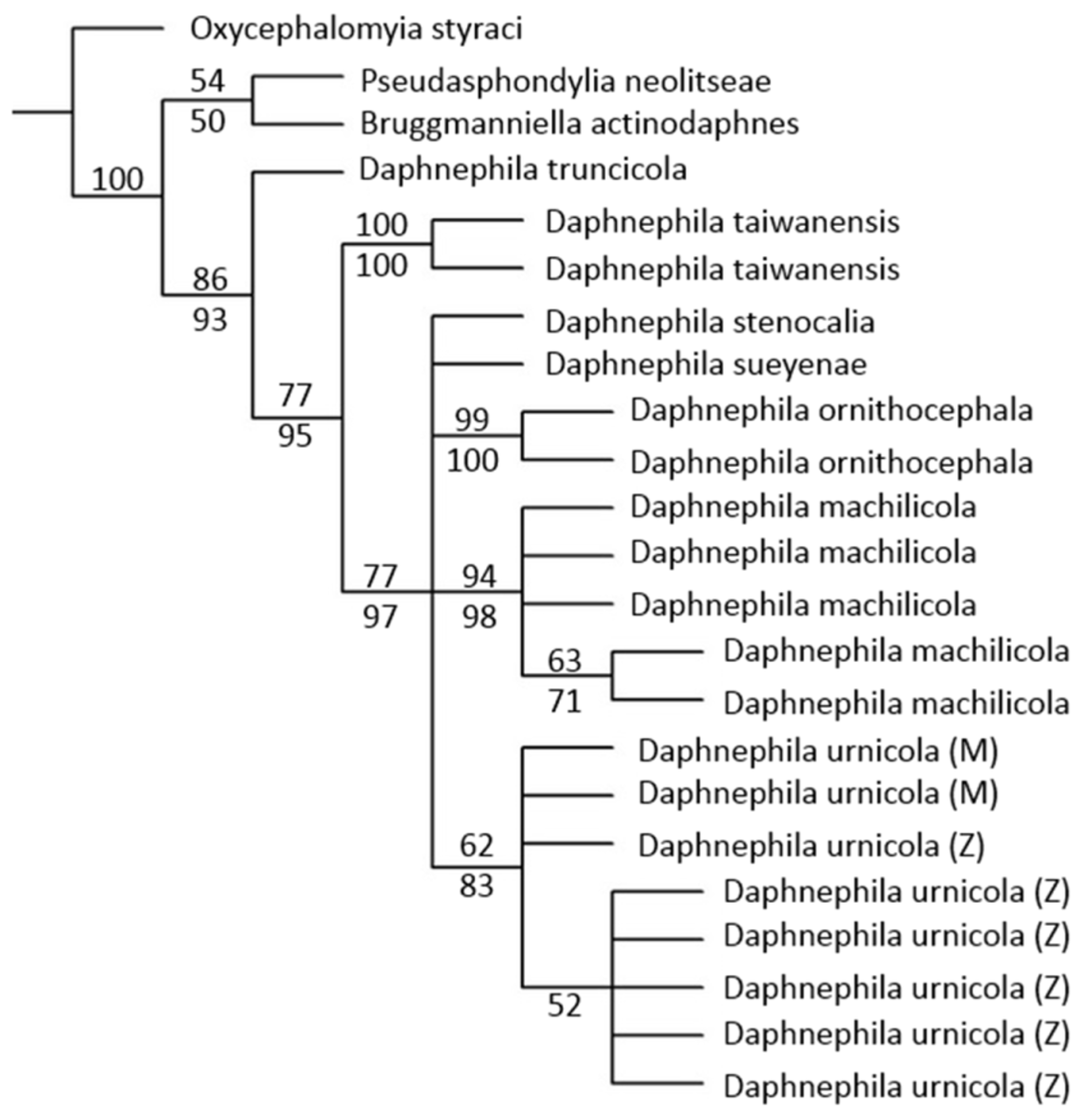
Both the NJ and the MP trees showed a similar topology with higher resolution in the NJ tree. Here we present the most parsimonious inference, the consensus from 3 trees of length of 223, with the bootstrapping values of both NJ and MP clustering method ( Fig. 5 View FIGURE 5 ). The taxa of the M. zuihonensis and M. mushaensis populations formed a monophyletic group and were supported by relatively high bootstrap values (83%, Fig. 5 View FIGURE 5 ). The minimum and maximum pairwise distances were 0 and 0.47%, respectively inside the D. urnicola group ( Table 1 View TABLE 1 ). The sister taxon of D. urnicola populations from M. zuihoensis and M. mushaensis is D. stenocalia , which forms longitudinal club-shaped galls on M. thunbergii in NJ tree.
Biology. Life cycle and development of gall tissues. Both M. zuihonensis and M. mushaensis populations of D. urnicola were univoltine, inducing single-chambered galls along the abaxial surfaces of the lamina, and completing their annual life cycle on either M. zuihoensis or M. mushaensis . These urn-shaped galls have never been found on other Machilus spp. in Taiwan, such as M. thunbergii and M. kusanoi , which usually coexist with M. zuihoensis or M. mushaensis in broad-leaved evergreen forests.
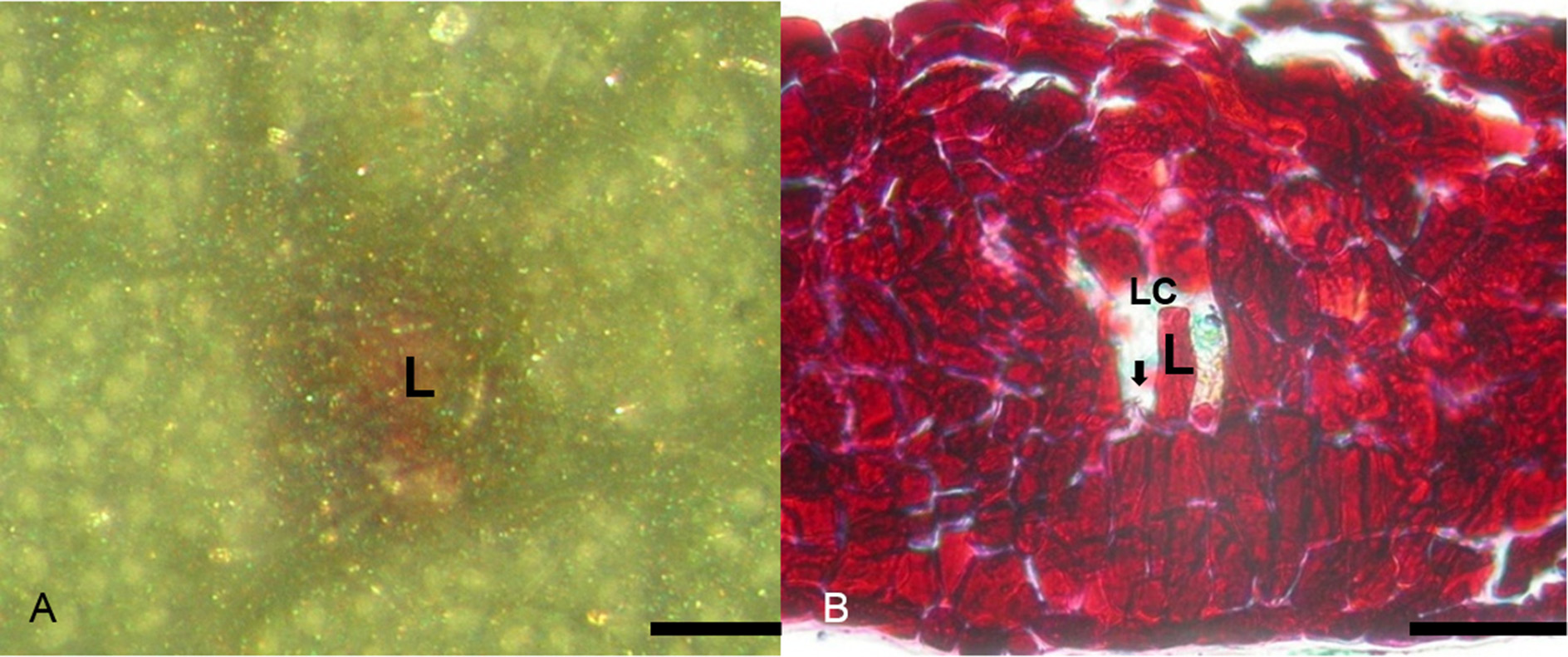
In field situations, mating usually occurred in the morning, and oviposition occurred between 1500 and 1700 h. Females walked along primary veins of freshly expanding leaves of M. zuihoensis and M. mushaensis and laid eggs in inter-vein locations between primary and secondary veins on the under surface. Occasionally oviposition occurred along the under surface of the tip of newly opened leaf buds. In 4–7 days, the neonate first-instar larvae penetrated into the leaf tissue, where they entered summer diapause ( Fig. 6 View FIGURE 6 A). There was no active proliferation of cells, but hyphae could be found inside the gall chamber at this stage ( Fig. 6 View FIGURE 6 B).
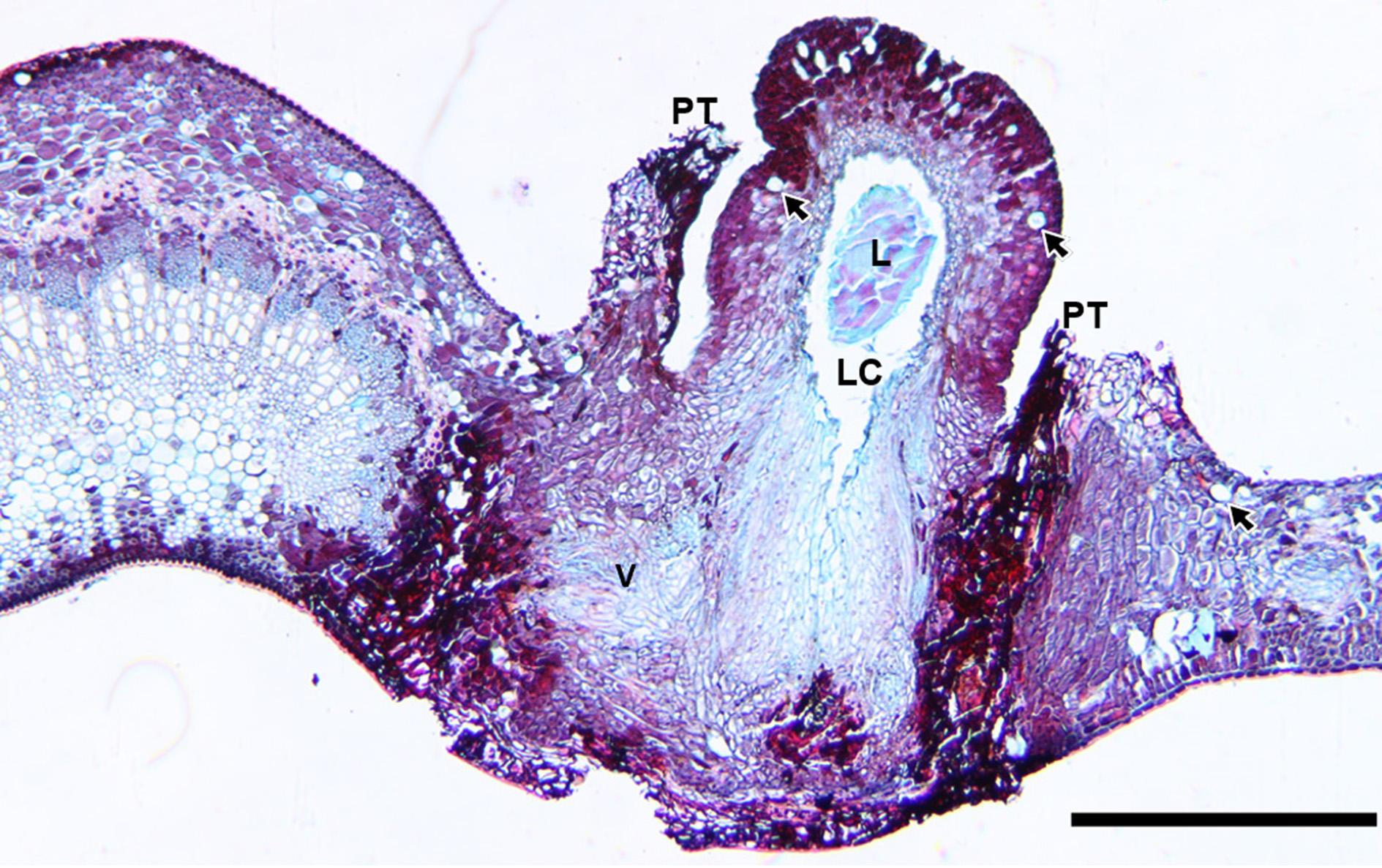
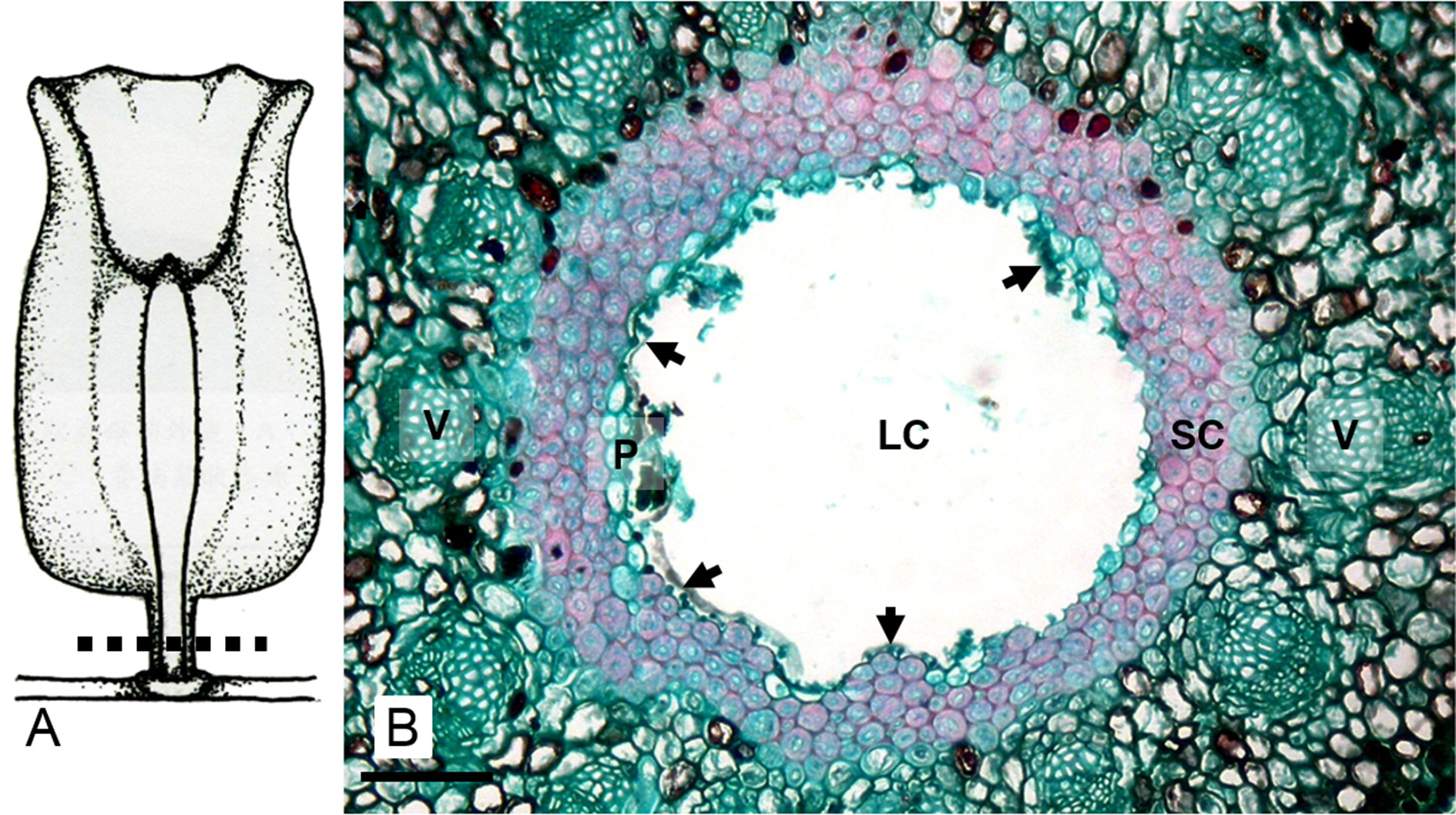
The developmental processes of galls induced by D. urnicola on M. zuihonensis and M. mushaensis are similar. Galls started to develop and protruded from the abaxial side of leaves in autumn. They developed rapidly and broke through leaf tissue when the larvae molted into the second instars ( Fig. 7 View FIGURE 7 A & B). Gall tissue was about 15 cell-layers in width and could be differentiated into (1) epidermis, (2) cortex, (3) vascular traces, and (4) nutritive tissue including the parenchyma layer and fungus layer within ( Fig. 8 View FIGURE 8 ). The parenchyma cells in the cortex were filled with tannin and some secretory cells existed among them. The galls grew along the vertical axis, transforming the spherical projection beyond the leaf surface to grow like a column ( Fig. 7 View FIGURE 7 C & D). The second instar larva remained at the basal part of the gall.
Most D. urnicola reached third instars in the galls during winter ( Fig. 7 View FIGURE 7 E & F). Exceptionally a few individuals from the southern populations remained as second instar larvae and a few from the northern populations reached the pupal stage. Galls were 40–50 cell-layers in width, with an organization similar to that during the occupation of the second instar larva, except for the differentiation of sclerenchyma between vascular bundles and the nutritive tissue ( Fig. 9 View FIGURE 9. A ). At the end of the growth and differentiation phase, around January, cells at the base of galls grew along the vertical axis of the gall, and those at the top of galls grew along the lateral axis. The differentiated gall tissues were heterogeneous at the basal part of the gall, sclerenchyma occurred between vascular bundles and the nutritive tissue, whereas at the terminals, only parenchyma cells and a few secretory cells occurred.
In late winter (January) and early spring (February), as the third instars pupated within galls ( Fig. 1 View FIGURE 1 ), sclerenchyma cells filled nearly half of the basal part of galls and the parenchyma layer inside the sclerenchyma layer was undetectable. Meanwhile, the shape of the operculum (the apical part of the gall), which opens at the time of emergence, was acute in angle on M. mushaensis ( Fig. 10 View FIGURE 10 D) compared to those on M. zuihonensis ( Fig. 10 View FIGURE 10 C). The number of parenchyma cell layers in this part of D. urnicola galls was 7–12 on M. zuihonensis ( Fig. 10 View FIGURE 10 A) compared to 16–18 on M. mushaensis ( Fig. 10 View FIGURE 10 B).
Adults emerged in March—April (spring) leaving the pupal exuviae at the opercular points of the gall ( Fig. 10 View FIGURE 10 E & F). The gall tissue gradually dried and detached from the leaf. The life span of D. urnicola adults emerging in the laboratory and kept in vials was 4.13±1.66 (63 females) and 2.14±0.86 days (50 males) from M. zuihonensis ; and 3.40±1.76 (53 females) and 2.49±0.73 days (72 males) from M. mushaensis . The longest life span observed was seven days for females from both hosts.
Measurements of galls. The D. urnicola galls on M. mushaensis were significantly larger than those on M. zuihonensis (ANOVA; F=14.57, p <0.001 for gall length and F=4.54, p <0.05 for gall width). The average length of galls on M. zuihonensis was 12.35 ± 2.76 mm, average width 6.61 ± 1.72 mm, and length–width ratio 1.87 (n=318). The average length of galls on M. mushaensis was 14.13 ± 1.90 mm, average width 7.23 ± 1.19 mm, and length–width ratio 1.95 (n=183).
Associated fungi. Botryosphaeria dothidea was isolated from almost all chambers of the D. urnicola galls induced on M. zuihonensis (98.68%) and M. mushaensis (97.37%). Phomopsis sp. (1.33%) was also isolated from galls on M. zuihonensis , and Nigrospora sp. (1.32%) and Pestalotia sp. (1.32%) from galls on M. mushaensis . The pouch on the females of D. urnicola is covered by an enlarged sheath on the 7th abdominal sternite ( Fig. 11 View FIGURE 11 ). However, under microscopic examination neither spores nor hyphae were found in the mycangia (pouch) of females just after emergence.
Parasitoids. Three endoparasitoids, Leptacis sp., Platygaster sp. (both Hymenoptera : Platygastridae ), and Gastrancistrus sp. ( Hymenoptera : Pteromalidae ), and six species of ectoparasitoids, Bracon sp., Simplicibracon curticaudis (both Hymenoptera : Braconidae ), Sigmophora sp. ( Hymenoptera : Eulophidae ), Eupelmus sp. ( Hymenoptera : Eupelmidae ), Ormyrus sp. ( Hymenoptera : Ormyridae ), and a pteromalid species were obtained from D. urnicola galls on M. zuihonensis . In contrast, only Platygaster sp. and Eupelmus sp. were obtained from galls on M. mushaensis .
TABLE 1. The minimum and maximum pairwise distances within D. urnicola and between D. urnicola and other Daphnephila species.
| pairwise distances | minimum | maximum |
|---|---|---|
| D. urnicola | 0.00% | 0.47% |
| D. stenocalia | 0.70% | 1.24% |
| D. machilicola | 1.64% | 2.50% |
| D. ornithocephala | 2.61% | 3.02% |
| D. sueyenae | 2.83% | 3.51% |
| D. taiwanensis | 6.03% | 6.65% |
| D. truncicola | 11.21% | 11.90% |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |