Aploparaksis (Aploparaksis) brachyphallos ( Krabbe, 1869 ) Fuhrmann, 1908
|
publication ID |
https://doi.org/ 10.1080/00222930601114168 |
|
persistent identifier |
https://treatment.plazi.org/id/03F787BE-F04F-7339-A7CC-D0DD5874FD1C |
|
treatment provided by |
Felipe |
|
scientific name |
Aploparaksis (Aploparaksis) brachyphallos ( Krabbe, 1869 ) Fuhrmann, 1908 |
| status |
|
Aploparaksis (Aploparaksis) brachyphallos ( Krabbe, 1869) Fuhrmann, 1908 View in CoL
( Figures 1–3 View Figure 1 View Figure 2 View Figure 3 ) Synonyms: Aploparaksis diminuens von Linstow, 1905, pp. 8–9 , Figures 29–31. Diorchis serpentata von Linstow, 1905 (in part), p. 7, Figures 23–25. Skorikowia clausa von Linstow, 1905, pp. 10–11 , Figures 38–40.
Aploparaksis larina Fuhrmann, 1921, pp. 518–520 View in CoL , Figures 114–117, new synonymy. Aploparaksis uelcal Spassky and Yurpalova, 1969, pp. 69–72 , Figures 23–25, new synonymy.
Hosts. Gulls: Larus argentatus Linnaeus , L. dominicanus Lichtenstein , Rissa tridactyla (Linnaeus) , Stercorarius longicaudus Vieillot , Xema sabini Vigor ; other hosts: numerous shorebirds of the genera Arenaria Brisson , Calidris Merrem , Heteroscelus Baird , Gallinago Brisson , Limnodromus Wied-Neuwied , Phalaropus Brisson , Pluvialis Brisson and Tringa Linnaeus (Charadriiformes) and Alauda arvensis Linnaeus (Passeriformes) .
Intermediate hosts. Stilodrilus sp. and Lumbriculus olgae Morev (Oligohaeta) , natural and experimental infection.
Metacestode. Caudate diplocyst ( Bondarenko 1973, 1975; Bondarenko & Konrimavichus 1976a, 1976b).
Localities. Alaska, Antarctic, Greenland, Iceland, Faroe Islands, Russia (White Sea, Kaliningrad Region, western Taimyr Peninsula, the lowlands of Enisey and Ob’ Rivers, Kamchatka, Chukotka, Islands of Wrangel, Bering, Karaginskiy and Paramushir, the coast of the Sea of Okhotsk), Ukraine, Turkmenistan.
Material studied. Aploparaksis rissae (USNPC 47086, type, Rissa tridactyla , Alaska); MHNG: A. brachyphallos of Baer (1962) Nos. 121/79–81, Tringa totanus , Iceland, 1 specimen and sections; A. diminuens Nos. 24/77 and 27/78, types, Phalaropus fulicarius, West-Taimyr , sections; A. larina Nos. 62/15–19, types, Larus dominicanus, No. 17, clarified anterior part of a strobila mounted in For Berlise fluid, Nos. 15, 16, longitudinal and transverse sections of a strobila, and Nos. 18, 19 whole mounted in Canada balsam strobiles; Diorchis serpentata Nos. 24/79–81, types, Calidris maritima, Enisey (Dickson Island), scolex and sections of strobila; Skorikowia clausa Nos. 24/83–85, types, Calidris sp. , longitudinal and transverse sections; A. brachyphallos from Dr R. Rausch’s collection, No. 26210, Xema sabini of Alaska (Yukon); two mature specimens (ZIN collection, voucher A. brachyphallos No. 86 from Kulachkova’s collection and voucher No. 131 from T. A. Ginetzinskaja’s collection (identified as A. pseudofilum in parts), both from Arenaria interpres from White Sea (Kandalaksha); our material: two specimens from Stercorarius longicaudus from Chaun Bay, Chukotka. The description of specimens of A. brachyphallos in Calidris alpina from Chukotka and its life cycle have been previously published ( Bondarenko 1975; Bondarenko & Kontrimavichus 1976a, 1976b). Treatment and clarification of the numerous definitions of A. brachyphallos recorded in shorebirds will be published separately.
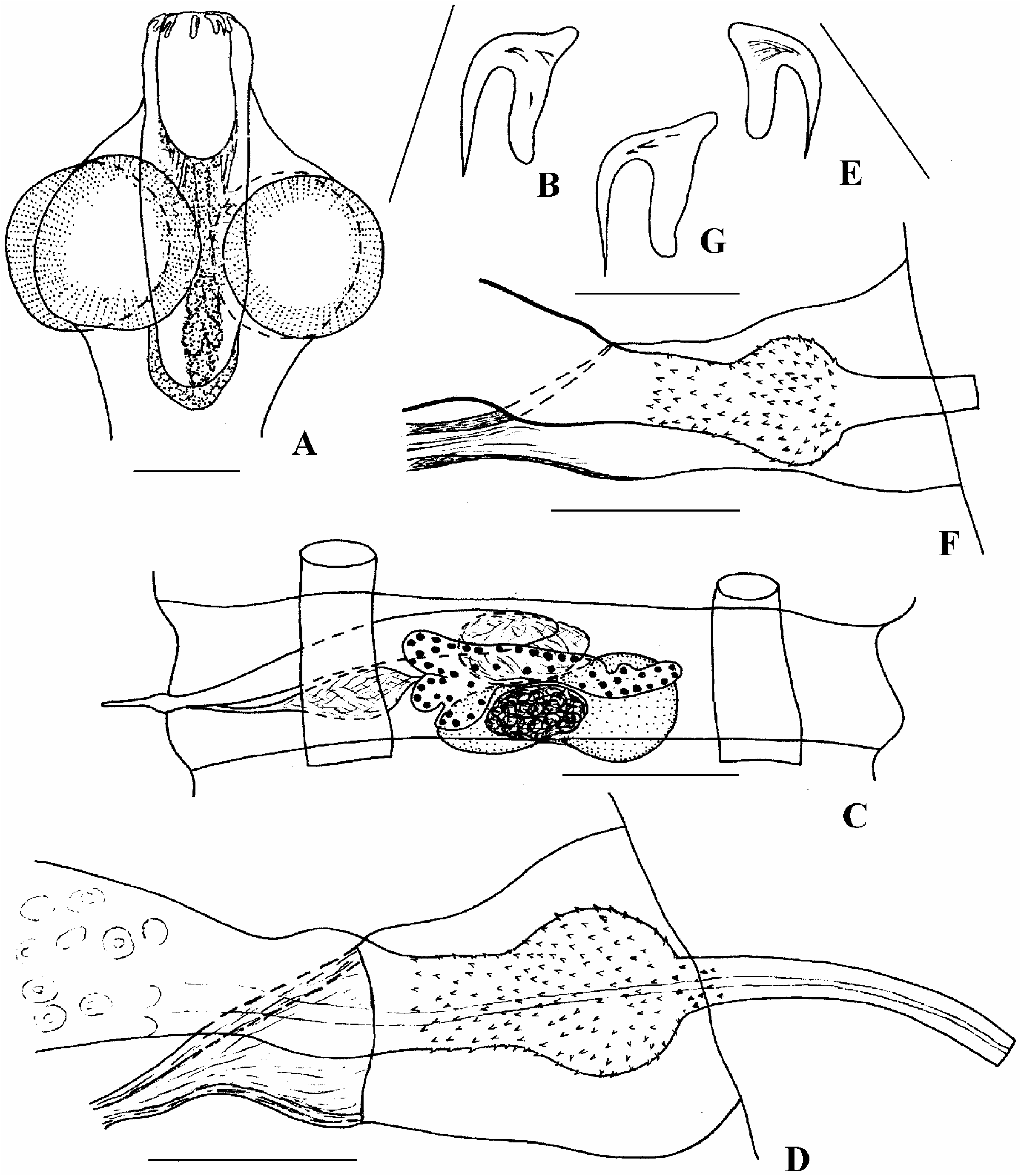
Description (based on two specimens from S. longicaudus from Chaun Bay, Chukotka). Length of pre-gravid specimen 38 mm. Maximum width of strobila 0.52 mm at level of last proglottides. Scolex ( Figure 1A View Figure 1 ) with evaginated rostellum 197× 168 mm. Suckers round, prominent, 80 mm in diameter. Rostellum 69×37, rostellar sheath 197× 53 mm, passes beyond level of posterior margins of suckers. Rostellar gland clustershape, located within cavity of rostellar sheath between its proximal part and proximal part of rostellum. Rostellar hooks 10, with thin blade and rudimentary handle ( Figure 1B View Figure 1 ). Total length of hook 16.6–17.4 mm (length of blade 10, length of base with guard 12–14, length of handle about 2 mm). Strobila flat, proglottides numerous, trapezoidal. Genital pores unilateral; alternation was only marked in one specimen in which the last five proglottides had indiscernible margins. Neck 410 mm long and 61 mm wide immediately posterior to scolex. Diameter of osmoregulatory canals: ventral to 45 mm, dorsal 4–5 mm in hermaphroditic proglottides. Transverse anastomoses not observed.
Testis oval or egg-shaped, rarely slightly lobed, relatively large, 90–102× 47–57 mm, in centre of median field ( Figure 1C View Figure 1 ), sometimes it is displaced close to antiporal osmoregulatory canals. Cirrus sac cigar-shaped, 184–270× 25–37 mm, thick-walled, with a well-developed layer of muscular fibres orientated diagonally. In male proglottides, cirrus sac reaching or crossing median line. Internal seminal vesicle fills almost entire cavity of cirrus sac. External seminal vesicle egg-shaped, to 80× 49 mm, opposite antiporal region of cirrus sac or overlapping it dorsally. Fully evaginated cirrus ( Figure 1D View Figure 1 ) small, 45–57 mm long, with parabasal spherical swelling. Basal part of cirrus 10–12× 8 mm and parabasal swelling 12–16 mm in diameter covered with dense, small spines; with distal part up to 29 mm long and 4 mm wide at base, and 3 mm at distal part, unarmed.
Ovary bi-winged, lobed, up to 180 mm wide. Each wing of ovary can form two to three lobes. Vitellarium compact or slightly lobed, 41–53× 25 mm, posterior and median to ovary. Seminal receptacle elongate-oval, 65–70× 29–37 mm. Vagina tubular, 45–61 mm long; distal end of copulatory part funnel-shaped, 29× 18 mm. Young uterus sacciform, lobed, overlaps longitudinal osmoregulatory canals. Proglottides with fully-developed uterus and ripe eggs not available.
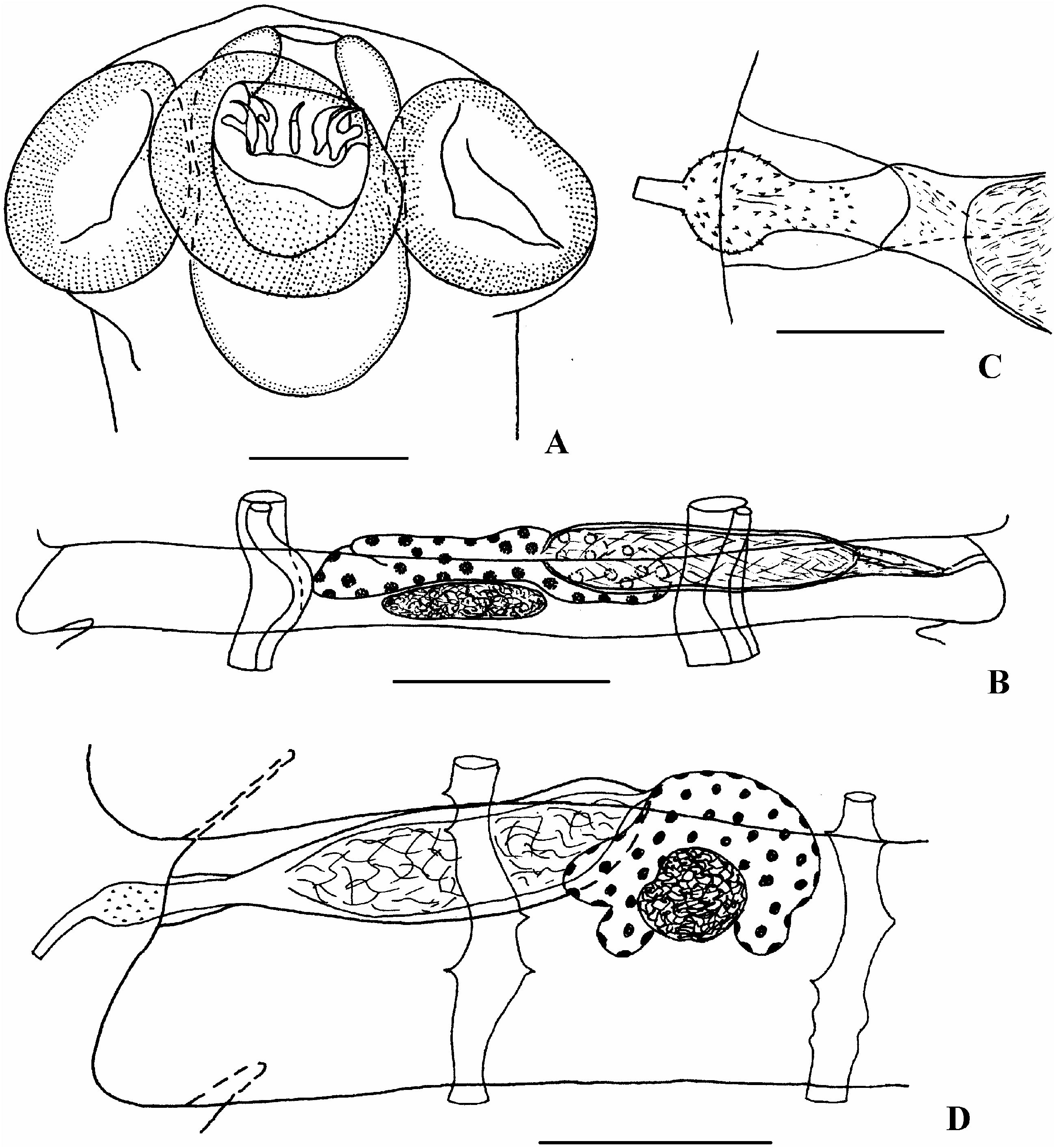
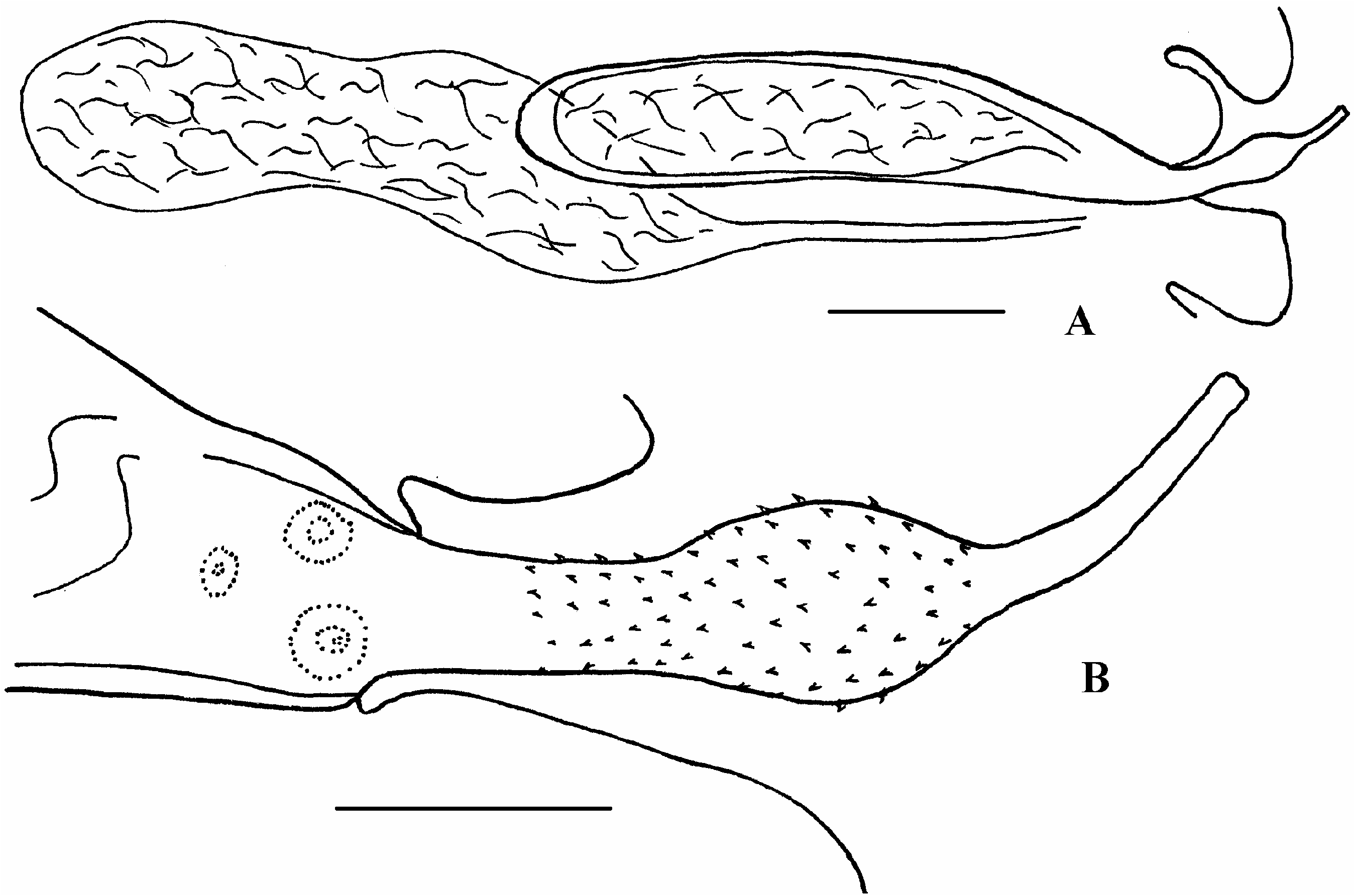
Morphological variability. A comparison of the morphological features of A. brachyphallos from various definitive hosts from various parts of the Holarctic showed certain differences. They relate to the length of rostellar hooks, and size of the testis and the cirrus sac. In specimens from sandpipers ( Figure 1E and F View Figure 1 ) the hooks are 16–18.2 mm long, whereas their length in X. sabini ( Figure 1G View Figure 1 ) is 19.8–21 mm. The testis in specimens from Calidris alpina (Linnaeus) , Limnodromus griseus (Gmelin) , Phalaropus fulicarius (Linnaeus) and Stercorarius longicaudus is considerably larger than that from Arenaria interpres (Linnaeus) ( Bondarenko 1975) . The specimens with shorter cirrus sac and somewhat different configuration of cirrus ( Figure 2D View Figure 2 ) were also observed by us from the latter host, especially in the White Sea population (see Table I). The base of the cirrus of these specimens is slightly elongated with gradual transition into the parabasal swelling. The cirrus of the specimens from Larus dominicanus from Antarctica ( Figure 3 View Figure 3 ) has an elongated base of the cirrus too. The cirrus of the specimens from other shorebirds ( Figure 2C View Figure 2 ) is shorter, and the bulbar swelling is clearly expressed (i.e. identical to those described above from S. longicaudus ).
Remarks. Aploparaksis brachyphallos is here recorded for the first time in the Laridae . It is one of the Aploparaksis spp. most frequently recorded in shorebirds. It was briefly described by Krabbe (1869) as a species of Taenia on the basis of material from sandpipers Calidris maritima (Brunnich) ( Greenland) and C. alpina (the Faroe Islands). The description lacks information on the morphology of the proglottides and the armament of the cirrus. The type material of A. brachyphallos was not available for our study. Nevertheless, we believe that the spherical swelling on the cirrus and the form and length of the rostellar hooks are characters typical of this species; these features are clearly seen in the Figures 193 and 194 in the original description ( Krabbe 1869). Moreover, Baer (1962) recorded A. brachyphallos from Tringa totanus (Linnaeus) in Iceland, i.e. close to the type localities. Comparison of the morphology of Baer’s specimens (MHNG 121/79–81) ( Figures 2A–C View Figure 2 ) with that of the specimens from our collection revealed their identity.
We had the opportunity to re-examine the type-materials of two species allocated by von Linstow (1905) to the genera Diorchis Clerc, 1903 and Skorikowia von Linstow, 1905 , and confirmed that Fuhrmann (1908) was right in regarding both these species as synonymous with A. brachyphallos . The specimen of D. serpentata , whose transverse sections are present on the slide No. 24/79, is an exception; we identified it as A. xemae ( Figure 8D View Figure 8 ).
Examination of the type of A. rissae by Bondarenko (1993) showed that the original description ( Schiller, 1951b) is composite, and includes the characters of both A. rissae and A. brachyphallos .
After re-examination of the type material A. uelcal Spassky and Yurpalova, 1969 (INPA from Phalaropus fulicarius from Chukotka) and A. diminuens Linstow, 1905 (MHNG 24/77- 78 from Ph. fulicarius from Taimyr), these species were also designated as synonyms of A. brachyphallos by Bondarenko and Kontrimavichus (1976a) and Bondarenko (1980).
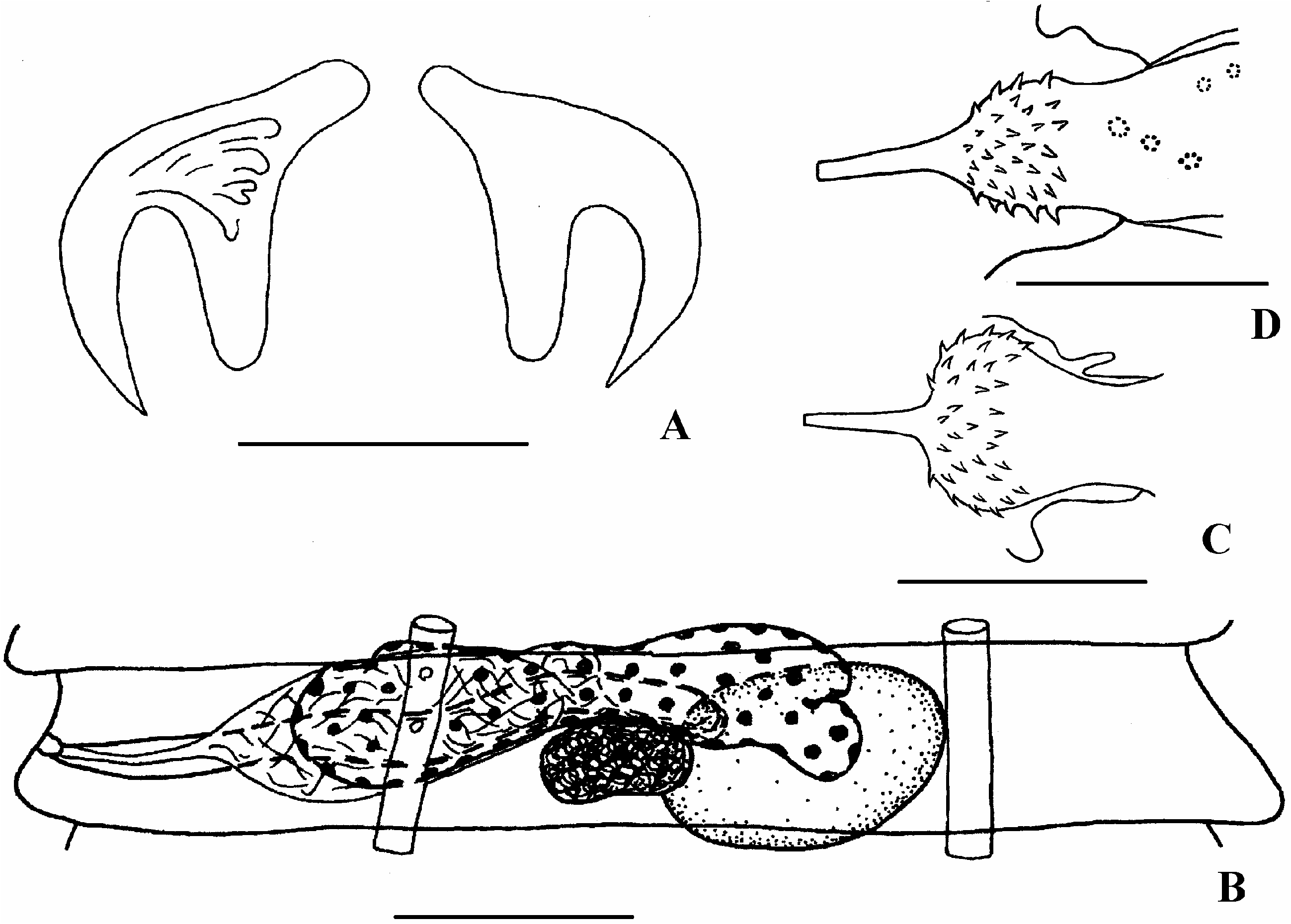
The comparative study of the morphology of A. brachyphallos from shorebirds and Holarctic gulls and specimens of the type-material of A. larina from Antarctic L. dominicanus (MHNG 62/15–19) gave us good reasons to identify these two species as synonymous. We failed to find differences in the cirrus morphology of the above-described specimens of A. brachyphallos ( Figures 1 View Figure 1 and 2 View Figure 2 ) and Fuhrmann’s specimens ( Figure 3 View Figure 3 ). Hence we established that A. brachyphallos has a bipolar distribution. However, we did not study Prudhoe’s (1969) specimens of A. larina from Antarctic L. dominicanus or those of Johnston (1937) from the same bird, from Marquarie Island.
It should be noted that even after detailed investigation of the material available, the question about morphological variability of A. brachyphallos remains unresolved. This species is a rare parasite of gulls. In addition to the records in L. dominicanus , from Antarctica and in S. longicaudus from Chukotka, East Siberia, we also found A. brachyphallos in L. argentatus on Paramushyr Island, Kurils and in X. sabini from the Yukon River, Alaska ( Bondarenko & Kontrimavichus 1999).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Aploparaksis (Aploparaksis) brachyphallos ( Krabbe, 1869 ) Fuhrmann, 1908
| Bondarenko, Svetlana & Kontrimavichus, Vytautas 2006 |
Aploparaksis uelcal
| Spassky and Yurpalova 1969: 69 - 72 |
Aploparaksis larina
| Fuhrmann 1921: 518 - 520 |