Petalophthalmus armiger Willemoes-Suhm, 1875
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4444.3.4 |
|
publication LSID |
lsid:zoobank.org:pub:693E1204-5F17-4EC6-89F4-B18B9E44473A |
|
DOI |
https://doi.org/10.5281/zenodo.5988918 |
|
persistent identifier |
https://treatment.plazi.org/id/03E2878B-BA5B-C91E-62BF-60E1FB0DFD62 |
|
treatment provided by |
Plazi |
|
scientific name |
Petalophthalmus armiger Willemoes-Suhm, 1875 |
| status |
|
Petalophthalmus armiger Willemoes-Suhm, 1875 View in CoL
( Figs. 2‒9 View FIGURE 2 View FIGURE 3 View FIGURE 4 View FIGURE 5 View FIGURE 6 View FIGURE 7 View FIGURE 8 View FIGURE 9 )
Petalophthalmus armiger Willemoes-Suhm, 1875 View in CoL (partim, male only): 41, 44, pl. 7.— G.O. Sars, 1884: 34; 1885, 174, pl. 32, figs. 1‒9.— Hansen, 1888: 212; 1927: 22, pl.1, fig. 6a.— Alcock & Anderson, 1894: 144.— Faxon, 1893: 218; 1895: 221, pl. 53, fig. 2‒2c; 1896, 164.— Holt & W.M. Tattersall, 1906: 22.— W.M. Tattersall, 1911: 25; 1925: 4, pl. a, figs. 1‒3; 1939: 229; 1951: 35, figs. 3, 4.— Zimmer, 1927: 40, figs. 64‒67.— W.M. Tattersall & O.S. Tattersall 1951: 120, figs. 17‒18; Gordan, 1957: 336 (list).— Birstein & Tchindonova, 1958: 269; Pillai, 1965: 1684, fig. 1.— O.S. Tattersall, 1968: 271 ‒272 (passim).— Mauchline & Murano, 1977: 71 (list).— Kathman et al., 1986: 93, 214‒215.— Escobar Briones & Soto, 1991: 82, table 11.— Muller, 1993: 4 (list).— Casanova, 1993: 48.— Ledoyer, 1995: 614.— Anderson, 2010: 22 (list).— San Vicente et al., 2014: 88 (key).
Petalophthalmus pacificus Faxon, 1893: 218 ; 1895: 223, pl. 54, figs. 1‒1h.— Ortmann, 1905: 971.— Gordan, 1957: 336 (list).
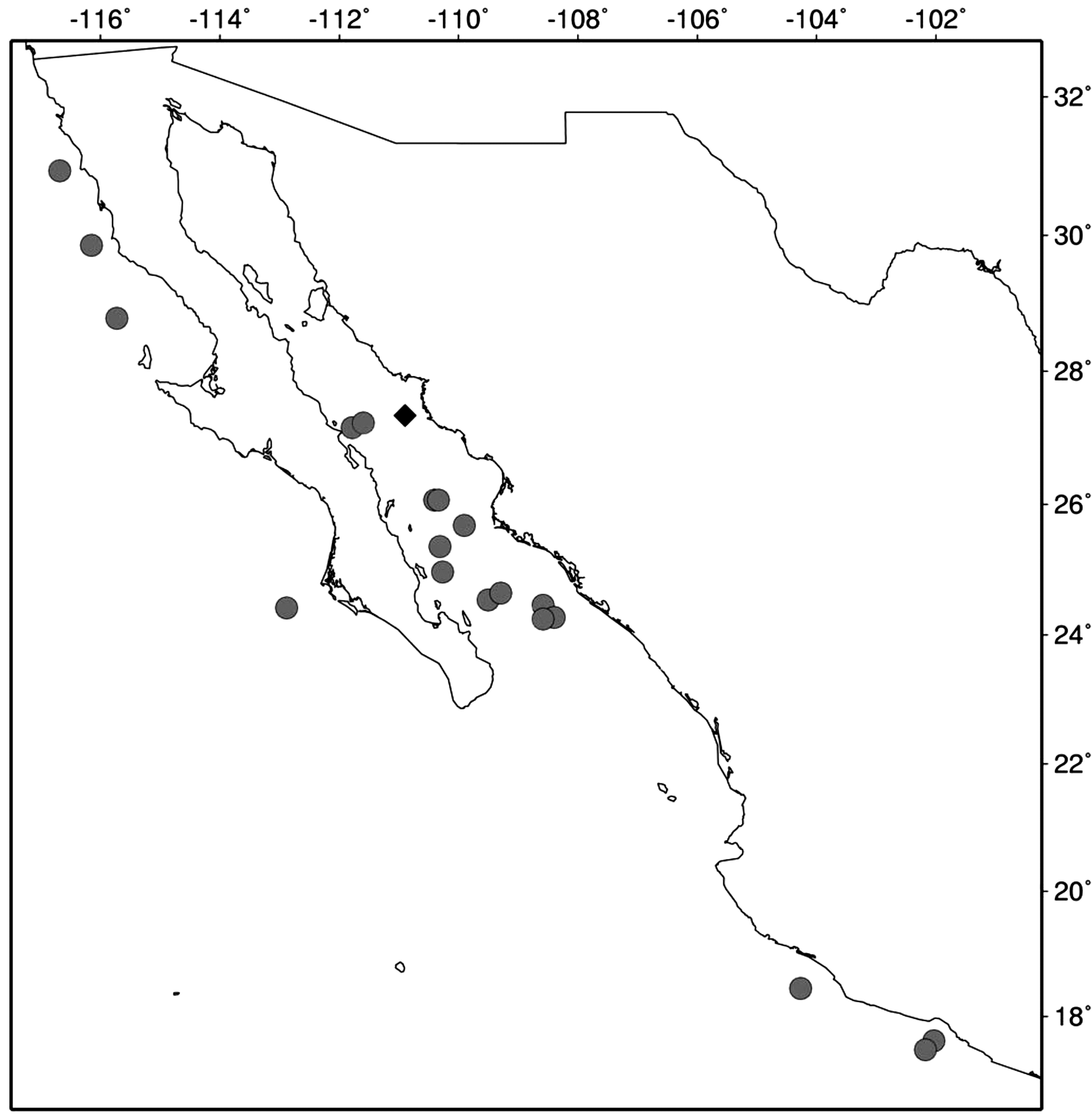
Material examined. TALUD IV. St. 20 (24°27'24" N, 108°35'16" W), August 25, 2000, 1 female (CL 8.73 mm), BS operated at 1500‒2000 m (ICML-EMU-5993); St. 34 (25°40'41" N, 109°54'24" W), August 27, 2000, 1 female (CL 7.58 mm), BS operated at 1240 m (ICML-EMU-8179-A); TALUD VII. St. 19 (24°16'12" N, 108°23'42" W), June 7, 2001, 2 males (CL 8.69‒9.42 mm) and 1 female (CL 9.37 mm), BS operated at 1160‒1180 m (ICML- EMU-6694); St. 27 (25°01'30"N, 109°12'W), June 8, 2005, 1 female (CL, 9.14 mm), BS operated at 1580‒1600 m (ICML-EMU-6694); TALUD VIII. St. 3 (24°32'36" N, 109°30'30" W), April 16, 2005, 1 female (CL 8.3 mm), BS operated at 1100 m (ICML-EMU-8185-D); St. 10, (24°58'12" N, 110°16'6" W), April 17, 2005, 2 females (CL, 8.5‒9.5 mm), BS operated at 1500 m (ICML-EMU-8185-C); St. 22 (26°03'42" N, 110°21'18" W), April 19, 2005, 2 males (CL 8.50‒9.37 mm) and 1 female (CL 8.92 mm), BS operated at 2200 m (ICML-EMU-8156-A); TALUD IX. St. 2 (24°38'42" N, 109°17'36" W), November 11, 2005, 1 female (CL 9.50 mm), BS operated at 2331‒2394 m (ICML-EMU-8253); St. 15 (25°21'27" N, 110°18'18" W), November 15, 2005, 3 females (LT 30‒38 mm), BS operated at 1985‒2220 m (ICML-EMU-8185-B); St. 22 (26°03'42" N, 110°20'36" W), November 14, 2005, 1 female (CL 10.0 mm), BS operated at 2214‒2309 m (ICML-EMU-8246); TALUD X. St. 18 (27°09'06" N, 111°46'54" W), February 12, 2007, BS operated at 1526 m, 1 female (CL 9.17 mm) (ICML-EMU-8179-C) and 2 females (CL 6.83‒9.37) (ICML-EMU-8185-A); St. 19 (27°13'30" N, 111°36'8" W), February 12, 2007, 3 females (CL 8.50‒12.40 mm), BS operated at 1786‒1816 m (ICML-EMU-8156-B); TALUD XII. St. 14 (17°36'20" N, 102°01'59" W), March 30, 2008, 1 female (CL, 7.41 mm), BS operated at 1415‒1476 m (ICML-EMU-12076); St 15 C (17°27'51"N, 102°10'43"W), March 31, 2008, 5 females (CL 3.18‒7.38 mm), MN between surface and 1530 m, 1880 m total depth (ICML-EMU-12084); St. 25 (18°26'45" N, 104°16'10" W), April 1, 2008, 1 female (CL,
8.75 mm), BS operated at 1858‒1879 m (ICML-EMU-12077); TALUD XV. St. 9 (24°25'12"N, 112°52'48"W), July 30, 2012, 3 females (CL, 8.14‒9.35 mm), 1 male (CL, 8.62 mm), BS operated at 1425‒1494 m (ICML-EMU- 12078); TALUD XVI-B. St. 4 (28°47'09"N, 115°43'78"W), May 24, 2014, 1 male (CL, 9.47 mm), BS operated at 1237‒1284 m (ICML-EMU-12079); St. 16 (29°51'04" N, 106°09'10" W), May 29, 2014, 2 females (CL, 9.10‒9.59 mm), BS operated at 1360‒1425 m (ICML-EMU-12080); St. 23 (30°56'04" N, 116°40'92" W), May 28, 2014, 1 female (CL, 9.88 mm), BS operated at 1296‒1340 m (ICML-EMU-12081).
Description (based on female unless indicated). Carapace ( Fig. 2A, C View FIGURE 2 ) long, poorly calcified, anterior margin straight, rostral projection sharp, subtriangular in lateral view, lateral corners rounded; median, rounded elevation in front of cervical sulcus; posterior margin emarginated, posterolateral lobes well marked.
Eye ( Fig. 2B View FIGURE 2 ) without stalks, leaf-like, no visual elements, a small ocular papilla on mesial margin in proximal 1/3 of eye length.
Antennule peduncle ( Fig. 2D View FIGURE 2 ) very long and slender, longer than carapace. First article longest, armed on distal margin with one cuspidate seta and three (broken) setae; second article naked; third article shorter than second, armed with two (broken) setae on distal margin; outer flagellum thinner than inner one. Male. Antennule peduncle ( Fig. 2E View FIGURE 2 ) 3-articulate, setae as illustrated. Basal portion of outer and inner flagellum similar in thickness.
Antenna peduncle 3-articulate ( Fig. 2F View FIGURE 2 ), about 5/6 of scale length; first article short, longer than broad, inner margin distally produced into sharp triangular lobe; second article about 2/5 scale length, distal margin armed with two simple setae; third article equal in length to second, distal margin with three simple setae and two short setae; flagellum multi-articulate. Antennal scale slender, about six times as long as wide, extending beyond anterior end of antennal peduncle; margins setose; apical lobe short, about 1/14 of scale length.
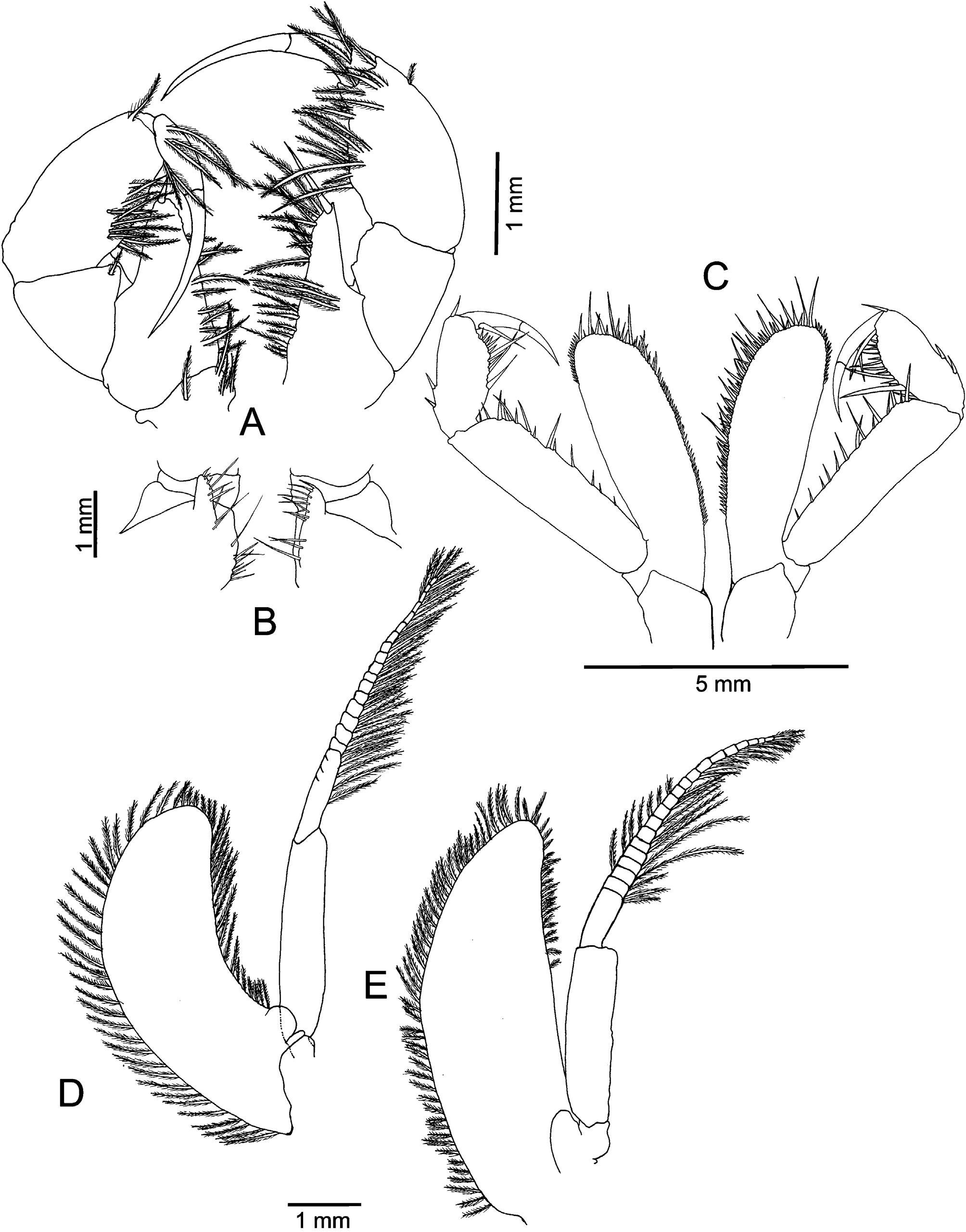
Mandibles with elongated and prehensile 3-articulate palp ( Fig. 3A View FIGURE 3 ); first article very small, armed with a small spine on the inner margin; second article about twice as long as third, 8‒12 strong spine-like setae and a few short spine-like setae on inner margin; third article with six strong spine-like setae and three strong spine-like setae on distal margin. Right mandible ( Fig. 4A‒C View FIGURE 4 ) with incisor process composed of a single chitinous ridge with four teeth: basal tooth and three apical teeth; lacinia mobilis either completely reduced or represented by a single long, stout seta between incisor and pars centralis (see note below); pars centralis conical with two apical setae; molar process concave, with strong, outer projecting process and three low chitinous ridges bearing an inner tuft of short setae. Left mandible ( Fig. 4D‒F View FIGURE 4 ) with incisor process composed of two chitinous ridges, one long ending in low blunty tooth at each end, the other short, strongly produced and separate from first by narrow deep groove; lacinia mobilis present, trifid; two closely set setae between incisor and molar processes; molar process with two strong, chitinous ridges, each bearing a tuft of short setae.
Maxillule ( Fig. 3B, C View FIGURE 3 ) small, outer lobe distally armed with seven strong cuspidate-serrate setae, each with one row of denticles; inner lobe with four pappose setae.
Maxilla ( Fig. 3D, E View FIGURE 3 ) with elongate exopod, extending to half length of endopod distal article, outer margin with series of long pappose setae, inner margin with 8‒12 pappose setae in distal half. Endopod distal article long and narrow, about three times as long as broad, series of pappose setae on inner margin, a sharp outer, small proximal spine. Coxal endites armed with pappose setae on inner margin, one long, proximal pappose seta on basal endite.
First thoracopod ( Fig. 5A View FIGURE 5 ) with long epipodite, about twice as long and almost as wide as carpus of outer and distal margins, respectively, without exopod. Endopod robust, with numerous tufts of pappose setae on inner margins of articles; basis with short lobe, row of setae on inner margin ( Fig. 5B View FIGURE 5 ); preischium very short; ischium produced into inner triangular lobe, tipped with four long pappose setae and one long simple seta; carpopropodus about as long as wide; dactylus and nail forming a curved claw armed with a few pappose setae.
Second thoracopod ( Fig. 5C View FIGURE 5 ) longer than first, without epipodite and exopodite. Endopod with preischium naked; ischium inner margin produced into a large oval lobe overreaching distal margin of merus, about three times as long as broad, with short and a few long cuspidate setae on inner margin and distal portion of outer margin; merus about four times as long as broad, inner margin armed with row of simple long and short cuspidate setae; carpopropodus about twice as long as wide, outer margin with two short proximal and one short distal cuspidate setae, inner margin with irregular row of simple long setae and short cuspidate setae; dactylus and nail forming a curved claw, dactylus with one long simple setae.
Third and fourth thoracopods ( Fig. 5D, E View FIGURE 5 ) with endopods reduced to 1-articulate naked articles; exopod 19‒21- articulate.
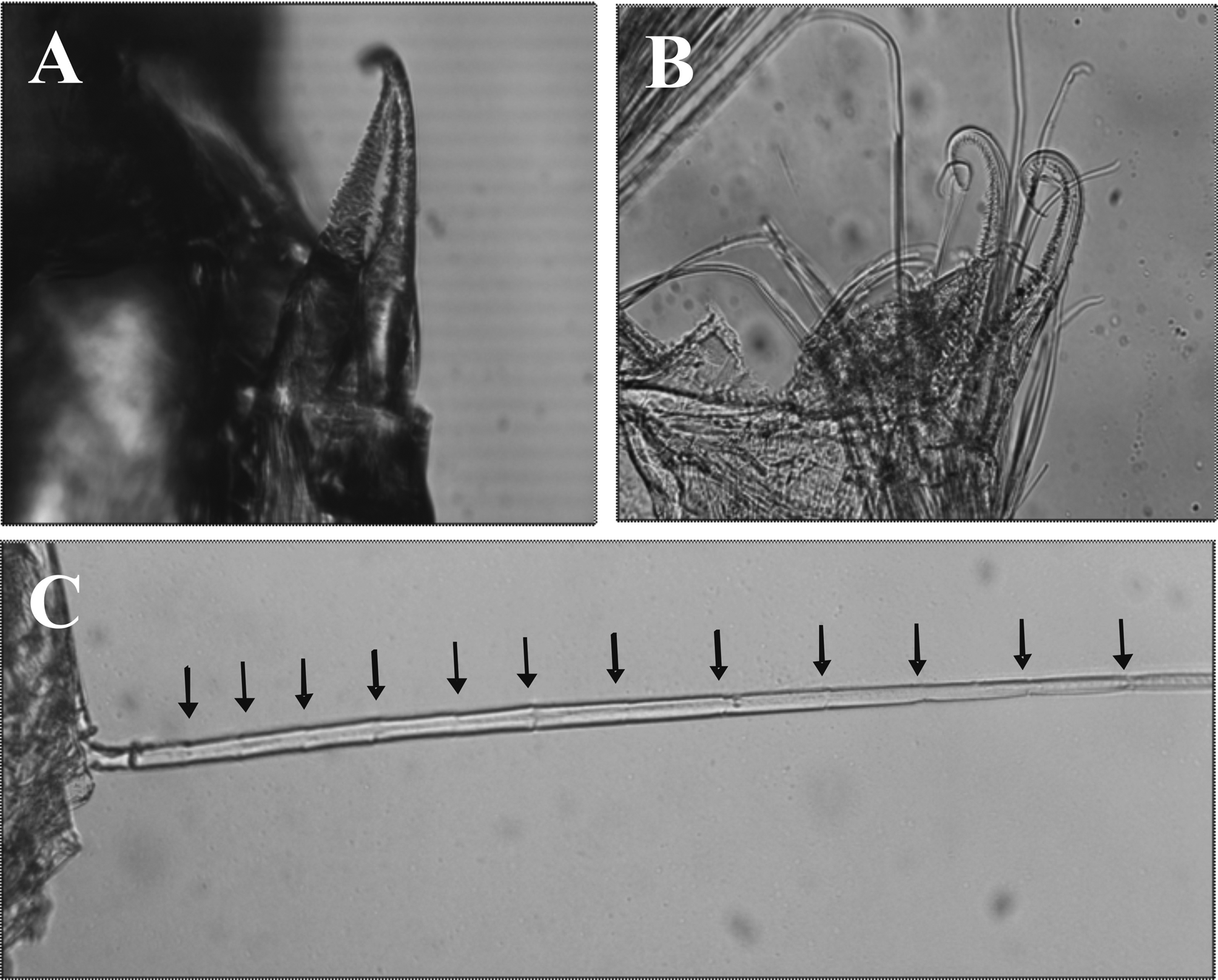
Fifth thoracopod ( Fig. 6A View FIGURE 6 ) endopod longer than other thoracopods. Preischium, ischium and merus subequal in length; carpus and propodus about 2.5 times as long as merus, propodus unarmed; dactylus very small, densely setose ( Fig. 6A View FIGURE 6 ), armed with two large partly serrate setae ( Fig. 7A, B View FIGURE 7 ). Exopod shorter than endopod, 20- articulate.
Sixth ( Fig. 6B, C View FIGURE 6 ) and seventh ( Fig. 6D View FIGURE 6 ) thoracopod endopods bearing plumose setae; eighth thoracopod ( Fig. 6E, F View FIGURE 6 ) endopod with simple setae. Preischium shorter than ischium; merus about twice as long as carpopropodus; carpopropodus undivided; dactylus short, terminating in one simple seta; a short distal nail with rounded tip. Exopods considerably shorter than endopods, 14‒20-articulate.
Penis ( Fig. 6G View FIGURE 6 ) at base of eighth thoracopod of male, moderately elongated, hemispheric apex microscopically tuberculate, with tuft of setae.
Marsupium composed of six pairs of oostegites.
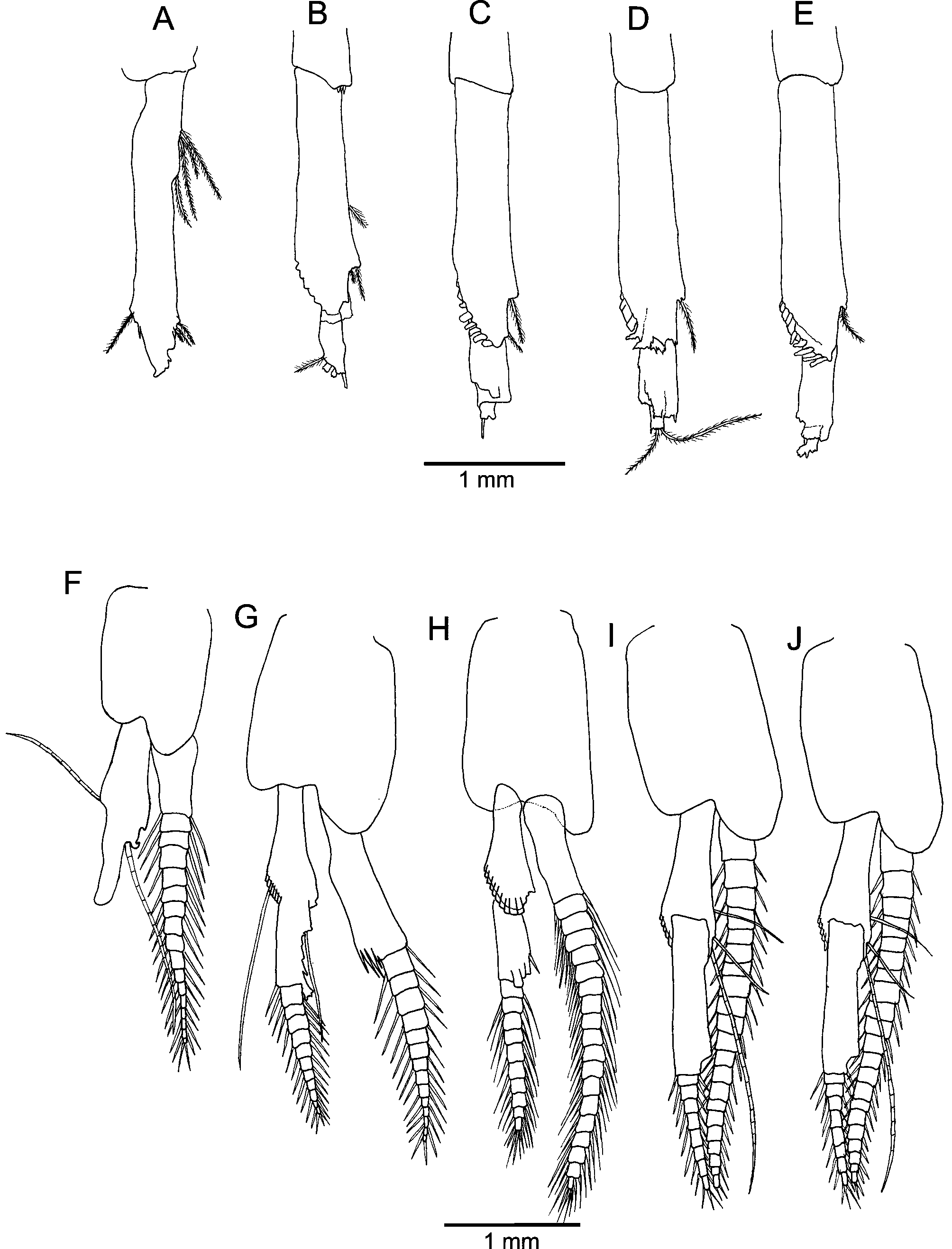
Pleopods of females ( Fig. 8A‒E View FIGURE 8 ) biramous. Endopod of first pleopod 2-articulate, distal article finger-like, two distal pappose setae, exopod 17-articulate. Exopods (14‒20-articulate) of second to fifth pleopods longer than endopods (11-articulate). Basal article of endopods with long processes, annulation-like sutures present in at least proximal section giving a multiarticulate aspect ( Fig. 7C View FIGURE 7 ). Pleopods of males ( Fig. 8F‒J View FIGURE 8 ) biramous. Endopod of first pleopod 2-articulate, distal article finger-shaped, two distal papose setae, exopod 18-articulate; endopods of pleopods 2‒5, 12-articulate; exopod of pleopods 1‒5 longer (1) or slightly longer than endopods, 14 to 20- articulate; first article of endopods distally wider, bearing long (flagellum-like) setae.
Uropod ( Fig. 9 View FIGURE 9 ) endopod slender, without statocyst, extending beyond telson apex, fully setose, inner margin straight. Uropod exopod 2-articulate, longer and broader than endopod; distal article fully setose, about one third of the basal article length; outer margin of the basal article naked, with two or three cuspidate distal setae.
Telson ( Fig. 9 View FIGURE 9 ) rectangular, about 2.3 times as long as broad; posterior half of lateral margins armed with>35 cuspidate setae. Posterior margin slightly emarginated, with five pairs of long serrate setae, one slightly shorter median serrate seta and three or four short, ventral serrate setae on both sides of median seta.
Remarks. The material of Petalophthalmus armiger examined features all the characteristics of the genus, including: the well developed rostrum; the long, slender antennular peduncle; an antennal scale entirely setose; the long, prehensile mandibular palp with third article longer than second; a long and prominent ischium lobe on the two first thoracopods; a 2-articulated uropodal exopod, with 3 small cuspidate setae near the distal angle; and a rectangular telson with proximal margin entire, armed with serrate setae ( O.S. Tattersall 1968; Bravo & Murano 1997; San Vicente et al. 2014).
In his review of the Mysidacea of the United States National Museum, W.M. Tattersall (1951) cited material examined from the Caribbean (2 adult females), California (2 females) and the Bering Sea (1 adult male) (see also O.S. Tatterstall 1968). He clearly stated that no valid differences between these specimens (from three widely separated regions) were observed, except for the structure of the pleopods which was variable among the examined specimens, which he justified by the fact that "... the fully developed condition of the pleopods in the male comes very late and is coincident with the attainment of sexual maturity".
Tattersall (1951, fig. 4) figured rather short setae on the second article on endopods of males pleopods 1 and 2 of P. armiger . The presence of long and very long processes, sometimes reaching to distal end of the exopods as in our material of P. armiger , has not been emphasized in the original description of males of P. caribbeanus O.S. Tattersall, 1968 , P. macrops Tchindonova & Vereshchaka, 1991 , P. liui Wang, 1998 , and P. papilloculatus , or described for P. oculatus by Tatterstall (1951, fig. 5). Illustrations provided by Faxon (1895) for P. pacificus , and (among others) by G. O. Sars (1885) and Tatterstall (1951) for P. armiger show much shorter or no flagellum-like structures on these appendages. In the Mexican material, these processes present, at least in part of their length, annulations that closely resemble sutures of imperfectly articulated appendages ( Fig. 7C View FIGURE 7 ). These annulations are only observed under high magnification, which might explain why they have not been reported earlier.
The illustrations provided by W.M. Tattersall & O.S. Tatterstall (1951) are much more detailed than those presented by W.M. Tattersall (1951) and might have been redrawn from original illustrations. Some of these are actually taken from a much earlier publication ( W.M. Tattersall 1925). Of interest is the fact that the cutting edge (incisor) of the right mandible is proportionally longer in W.M. Tattersall & O.S. Tatterstall (1951) and that of the left mandible clearly shows two bifid "chitinous ridges", which are not visible in the figure by W.M. Tattersall (1951) although mentioned in the text.
Examination of the mandibles using a SEM clearly shows that one of the two chitinous ridges illustrated by W.M. Tattersall & O.S. Tattersall (1951) actually corresponds to a well developed lacinia mobilis in the right mandible ( Fig. 4E, F View FIGURE 4 ), similar to the lacinia mobilis described for P. papuensis by San Vicente & Corbari (2015). Additionally, there are two closely-set setae between incisor and molar processes instead of only one, and the molar process features two strong, slightly curved ridges instead of three in W.M. Tatersall (1951) illustrations. The left mandible is also different in some aspects when compared with W.M. Tattersal (1951) illustrations. The lacinia mobilis is reduced or may be represented by a single, stout seta between incisor and pars centralis; it is unclear, however, if this seta is not part of the adjacent conical pars centralis.
It is unclear if W.M. Tatterstall was able to examined the type material of R. Willemoes-Suhm collected in the Atlantic Ocean (a male of ca 37 mm total length; 01°22'N, 26°36'W). G.O. Sars (1885: 174) made the following statement in which he obviously referred to P. armiger : "Of this interesting form I have only had opportunity of examining a single male specimen, apparently that described by the late Dr. v. Willemoes-Suhm. That author has also described the female [...] resembling rather the species of the genus Boreomysis ". This was later confirmed by Hansen (1888). Hansen (1927) stated that " ... En 1888, j'ai établi que le spécimen décrit et figuré par Willemoes- Suhm comme étant la femelle de Petalophthalmus armiger appartenait en réalité au genre Boreomysis ...". The illustrations provided by Willemoes-Suhm (1875) for the male of P. armiger are of very poor quality, and details are difficult to appreciate. The lateral view of the male specimen is particularly bad as it is not possible to see the typical rostrum of the species and the low, anterior carapace process or bump illustrated by subsequent authors (e.g., Faxon 1895; Tattersall 1925). The description of the eye-stalk by Willemoes-Suhm (1875: page 44) stated "Eyes wanting. Eyestalks with spherical concave terminations at the place where one would expect to find eyes". Indeed, the figure proposed (Pl. VII, 2) shows a perfectly spherical "eye".
San Vicente et al. (2014) recently described a new species of Petalophthalmus from the NE Atlantic. According to these authors, there is some similarity between P. papilloculatus San Vicente, Frutos & Cartes, 2014, and P. armiger . These authors provided eight differences that allow separating the two species. The Mexican material examined allows us to specify the following:
1 There is, indeed, an acute triangular rostrum in P. armiger vs. no rostrum in P. papilloculatus .
2 An ocular papilla is present in P. armiger , as in P. papilloculatus .
3 The antennal scale features a well marked apical lobe, as in P. papilloculatus .
4 There are, indeed, three chitinous ridges on the right molar process of the mandible (vs. two on the left mandible).
5 Indeed, there is no rounded lobe with two simple setae on the male exopod (first article) of the second pleopod.
6 In P. armiger , the three cuspidate setae on the distal corner are rather irregular in size, not always lengthening from inner to outer one.
7 The number of setae along the telson lateral margin is variable with size according to W.M. Tattersall (1951: 40) who stated that "The number of spines on the lateral margins increases as growth proceeds".
8 The telson of P. armiger is armed with five pairs of serrated setae on the posterior margin, plus a shorter median serrate seta, and three to five pairs of short serrate setae, only visible in ventral view; in P. pappiloculatus these short setae (or teeth) are smooth.
It would therefore appear that some differences used by San Vicente et al. (2014) to separate P. papilloculatus from P. armiger are not valid (see 2, 6, 8 above), in particular the presence vs. the absence of an ocular papilla used to separate P. papilloculata from P. armiger , as stated in the identification key to species of the genus. This ocular papilla was not described by G.O. Sars (1885: fig. 3), or by Tatterstall (1951: fig. 3) and—as far as we can seenever addressed in any contribution dealing with P. armiger . Also, the apical lobe observed on the antennal scale is clearly present in our material, although the suture is weak. No apical lobe was represented by G.O. Sars (1885), Hansen (1927), or by Tattersall (1951) in their illustrations of P. armiger antennal scale.
San Vicente et al. (2014) included an illustration of the penis of P. papilloculatus . This appendage has not been illustrated for P. armiger and it is reproduced herein ( Fig. 6G View FIGURE 6 ). Comparing with P. papilloculatus it is considerably shorter, and the hemispheric apex is microscopically tuberculate and bears a distal tuft of setae (smooth and without setae in P. papilloculatus ).
Considering the six other described species of Petalophthalmus , three obvious character states are observed in the new material that indicate an identity of P. armiger : 1) the typical triangle-shaped rostrum with an acute process (no rostrum in other species); 2) the leaf-like eye without visual elements (vs. globular, functional eye in P. oculatus , P. caribbeanus , P. macrops , P. liui , and P. papuaensis ); 3) the three chitinous ridges on the right molar process of the mandible vs. one chitinous ridge in P. papilloculatus .
Geographic distribution. Widely distributed in the tropical Atlantic; Gulf of Aden (Indian Ocean); off Hawaii (Pacific Ocean); Southern Ocean to 46°S. In the eastern Pacific it has been recorded from the Bering Sea, off California and in the Gulf of Panama ( Tattersall 1951; San Vicente & Corbari 2015), and in the type locality of P. pacificus , in the Gulf of California ( Faxon 1895).
New records. The only previous record off western Mexico is the one corresponding to the type locality of P. pacificus , in the Gulf of California: "Albatross station" 2637, off Guaymas ( Fig. 1 View FIGURE 1 ). The present survey adds 18 new localities in western Mexico, recording the presence of P. armiger throughout the central and southern Gulf of California, off SW Mexico, and off the west coast of the Baja California Peninsula ( Fig. 1 View FIGURE 1 ). Based on present records, P. armiger appears to be a common species in western Mexico. Lack of previous records in the area could be linked to the lack of sampling at depths where this species occurs.
Bathymetric distribution. According to San Vicente & Corbari (2015), P. armiger has been recorded in depths between 900 and 4572 m. Sampling depths at which pelagic species occur, however, are often erroneously reported when bottom sledges and trawls, non-closing mid-water gear or non-closing plankton nets are used. Hendrickx (2016) analyzed the depth distribution of Plesionika sanctaecatalinae Wicksten, 1983 , off western Mexico noting that maximum depth at which sampling gear is deployed is often cited as the maximum depth of occurrence for some species. However, there is no possible way to know at which depth specimens enters the net, when the gear cannot be opened and closed at a precise depth. Closing-opening mid water gear have been used routinely only recently and many early records should be treated with caution, carefully revising which type of gear was used. On the other hand, the use of an opening-closing suprabenthic sledge can provide reliable information on the benthic habitat of some species, as in the case of P. papilloculatus ( San Vicente et al. 2014). All specimens, except one lot (ICML-EMU-12084), examined herein were captured with a bottom sledge operating at depths between 1100 and 2309 m and were either captured close to bottom or captured during the ascent of the gear through the water column. Thus, they could have entered the net at any depth between the bottom and the surface. During the same survey, however, many samples of benthos and pelagic shrimps were obtained in shallower water (roughly in areas with maximum depth between 500 and 1000 m) using the same bottom sledge. No specimen of P. armiger were found at these sampling stations, thus indicating that this species might indeed inhabits very much offshore, with waters deeper that 1000 m. This is reinforced by the fact that not a single specimen of P. armiger was collected during a similar research cruise (i.e., same gear was used) in the northern Gulf of California where almost all stations were sampled in less than 1000 m total depth. The one lot not collected with the benthic sledge was obtained with a mid water micronekton net sampling the entire water column from surface to 1530 m, in a locality where total depth was 1880 m, 350 m above bottom. This would therefore indicate that P. armiger , at least occasionally, inhabits a water fringe far away from the bottom. The only previous record of P. armiger (as P. pacificus ) in the Gulf of California was from a presumably non-closing midwater tow-net operating from surface to 1280 m, in a locality with a total depth of 1413 m ( Faxon 1895).
Due to lack of discrete deep-water samples in the water column, it is difficult to assess the influence of the wide and intensive Oxygen Minimum Zone (OMZ) that occurs off western Mexico. In some areas (e.g., southern part of the California Current area, southern Gulf of California, SW Mexico), this OMZ is up to 800‒1000 m wide, starting in very shallow water (as shallow as 50 m), and features an anoxic to almost anoxic core ( Hendrickx and Serrano 2010; Serrano 2012). The absence of specimens of P. armiger in samples taken close to shore, in depths shallower than 1000 m, and the scarcity of previous records might indicate that it inhabits below the OMZ core. It could also live close to the bottom, and be just another eurybathic species of Petalophthalmus (see San Vicente & Corbari 2015: 251). In future studies the use of opening-closing bottom and mid water samplers, operating in a wide depth range, will prove useful in determining the habitat and depth range of P. armiger .
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Petalophthalmus armiger Willemoes-Suhm, 1875
| Hendrickx, M. E. & Hernández-Payán, J. C. 2018 |
Petalophthalmus armiger
| G.O. Sars, 1884 : 34 |
| Hansen, 1888 : 212 |
| Alcock & Anderson, 1894 : 144 |
| Faxon, 1893 : 218 |
| Holt & W.M. Tattersall, 1906 : 22 |
| W.M. Tattersall, 1911 : 25 |
| Zimmer, 1927 : 40 |
| W.M. Tattersall & O.S. Tattersall 1951 : 120 |
| Gordan, 1957 : 336 |
| Birstein & Tchindonova, 1958 : 269 |
| Pillai, 1965 : 1684 |
| O.S. Tattersall, 1968 : 271 |
| Mauchline & Murano, 1977 : 71 |
| Kathman et al., 1986 : 93 |
| Escobar Briones & Soto, 1991 : 82 |
| Muller, 1993 : 4 |
| Casanova, 1993 : 48 |
| Ledoyer, 1995 : 614 |
| Anderson, 2010 : 22 |
| San Vicente et al., 2014 : 88 |
Petalophthalmus pacificus
| Faxon, 1893 : 218 |
| Ortmann, 1905 : 971 |
| Gordan, 1957 : 336 |