Catasetum × grasineideae D.R.P.Krahl, Krahl, Chiron & J.B.F.Silva, 2023
|
publication ID |
https://doi.org/ 10.11646/phytotaxa.594.2.1 |
|
DOI |
https://doi.org/10.5281/zenodo.7892015 |
|
persistent identifier |
https://treatment.plazi.org/id/03E187AE-0206-FFED-FF12-F9C7FAA5FA73 |
|
treatment provided by |
Plazi |
|
scientific name |
Catasetum × grasineideae D.R.P.Krahl, Krahl, Chiron & J.B.F.Silva |
| status |
sp. nov. |
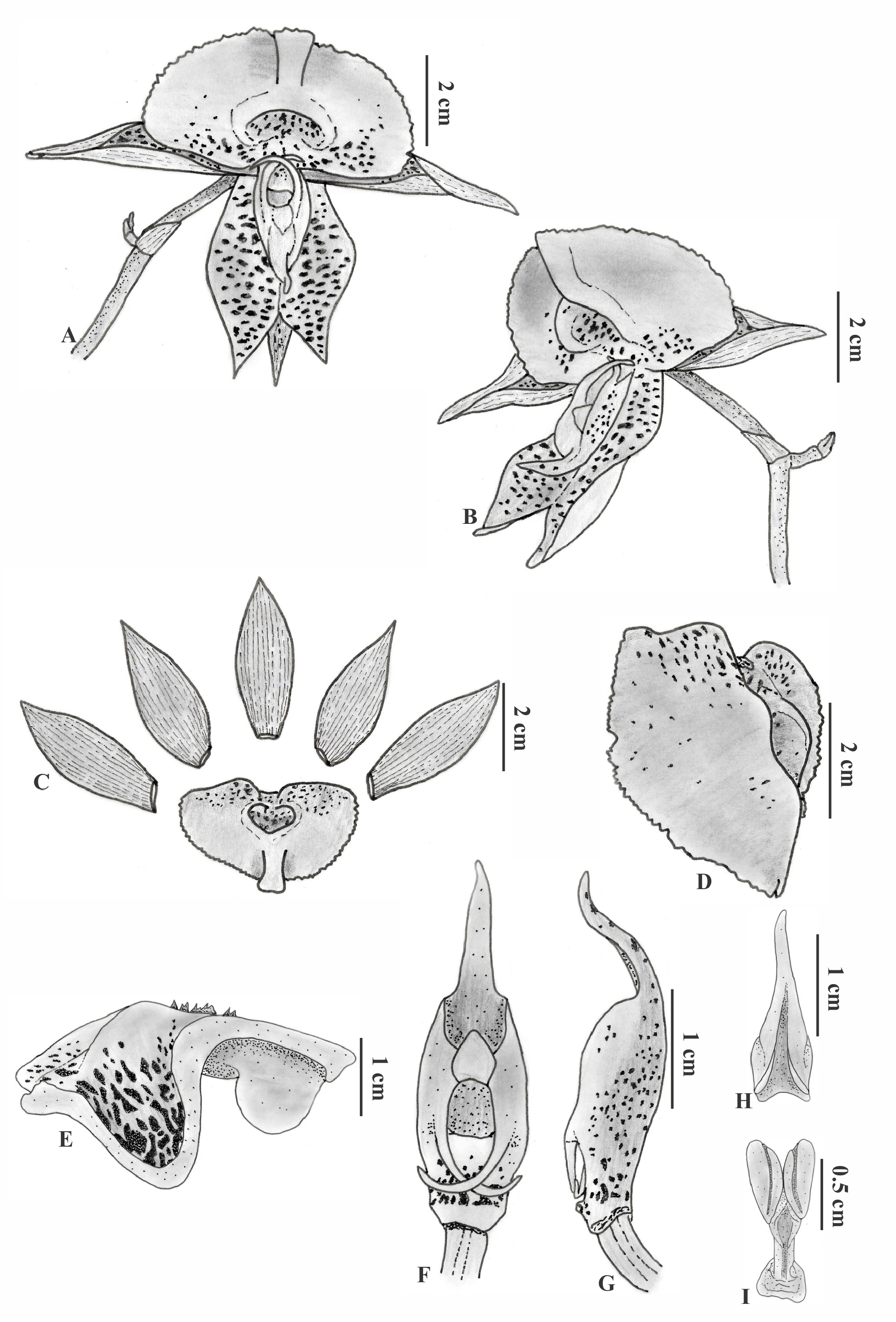
Catasetum × grasineideae D.R.P.Krahl, Krahl, Chiron & J.B.F.Silva , nothosp. nov. ( Figures 2–3 View FIGURE 2 View FIGURE 3 ).
Type:— BRAZIL. Amazonas : Careiro Castanho , Castanho Lake, epiphyte in Igapó, 3°44’08.35”S; 60°26’41.44”W, 8-10 m, 12 January 2022, A.H. Krahl & D.R.P. Krahl 1635 (holotype: INPA!) GoogleMaps .
Hoc taxon naturalis hybrida inter C. gnomus et C. saccatum est, characteres intermedios efficiens. Non-resupinatos flores, ellipticolanceolata sepala petalaque, integrum suborbiculatum labellum com denticulato-serrato margine apice reflexo, parte centrali sacciformi conica, ostio latiore quam longo et duabus parvis protuberationibus in columnae proximali margine.
Description:— Epiphytic caespitose plant. Rhizome short inconspicuous. Pseudobulb 3.7–7.5 × 1.0– 1.5 cm, fusiform, erect, clustered, 4-5–leaved, covered by leaf sheaths. Leaves 4.9–19.8 × 1.5–2.7 cm, linear-lanceolate, plicate, 3- 5–nerved, entire and slightly undulate margin, apex acute. Male inflorescence ca. 21.2 cm long, lateral, racemose, 1-5–flowered, erect then arched under the flower weight; peduncle cylindrical, greenish lightly purplish; floral bract ca. 0.7 × 0.6 cm, triangular, lightly greenish, margin entire, apex acute to obtuse. Male flower yellowish with brownish spots, especially on the sepals and petals, non-resupinated, pedicelled; pedicel ca. 3.3 cm long, cylindrical, erect and apically curved, purplish; sepals ca. 4.0 × 1.5 cm, elliptic-lanceolate, symmetrical, concave, margin entire, apex acute; petals ca. 4.1 × 1.6 cm, elliptic-lanceolate, symmetrical, margin entire and reflexed, apex acute; lip 3.0 × 4.5 cm, entire, subtrilobed, suborbicular, margins reflexed near the apex, denticulate to serrate, central part sacciform and conical; ostium ca. 0.9 × 1.4 cm, ca. 1.3 cm deep, opening somewhat flattened (transverse diameter longer), with two little protuberances on the edge nearest to the column; column ca. 3.2 × 1.0 cm, apex rostrate; antennae ca. 1.1 cm long, crossed; anther cap ca. 1.7 × 0.5 cm, rostrate; viscidium 0.25 × 0.35 cm, whitish, sticky; stipe ca. 0.6 × 0.2 cm, lamellate, rolled up, dark; pollinia 2, ca. 0.6 × 0.4 cm, obovate, thick, compressed, sulcate, yellowish. Female inflorescence and fruit not seen.
Distribution and ecology:—the new taxon was found and collected on a phorophyte on the shore of the Castanho lake, in an igapó forest, in the municipality of Careiro Castanho, AM, Brazil. Based on personal observations, the parental species have already been observed in this place and in neighboring municipalities (e.g. Careiro da Várzea), especially along the BR 319 road.
According to Petini-Benelli (2022), C. gnomus occurs in the states of Amazonas, Pará and Rondônia and C. saccatum in the entire North-Brazilian region as well as in the states of Mato Grosso and Mato Grosso do Sul. Thus, the geographical distribution of both species overlaps, including in the state of Amazonas where the new hybrid has been found.
The new natural hybrid has been observed in bloom in January, a time when the parental species can flower. Based on author’s personal observations and on herbarium registers (e.g. CRIA database, 2022), we know that C. gnomus flowering generally begins in December and stops in April whereas C. saccatum is in bloom from August up to February. Consequently, the blooming times of both species overlap during at last three months (December, January and February), making possible their crossed pollination.
As for the flower visitors, we already observed visit of male Eulaema bees on both parental species, which, once more, makes a genetic flux between them possible. These bees visit the flowers looking for volatile compounds (perfume) present on the lip.
Etymology:—the specific epithet is given in honor to Maria Grasineide Gomes Passos, mother of the first author, orchid lover and great incentive to orchid study.
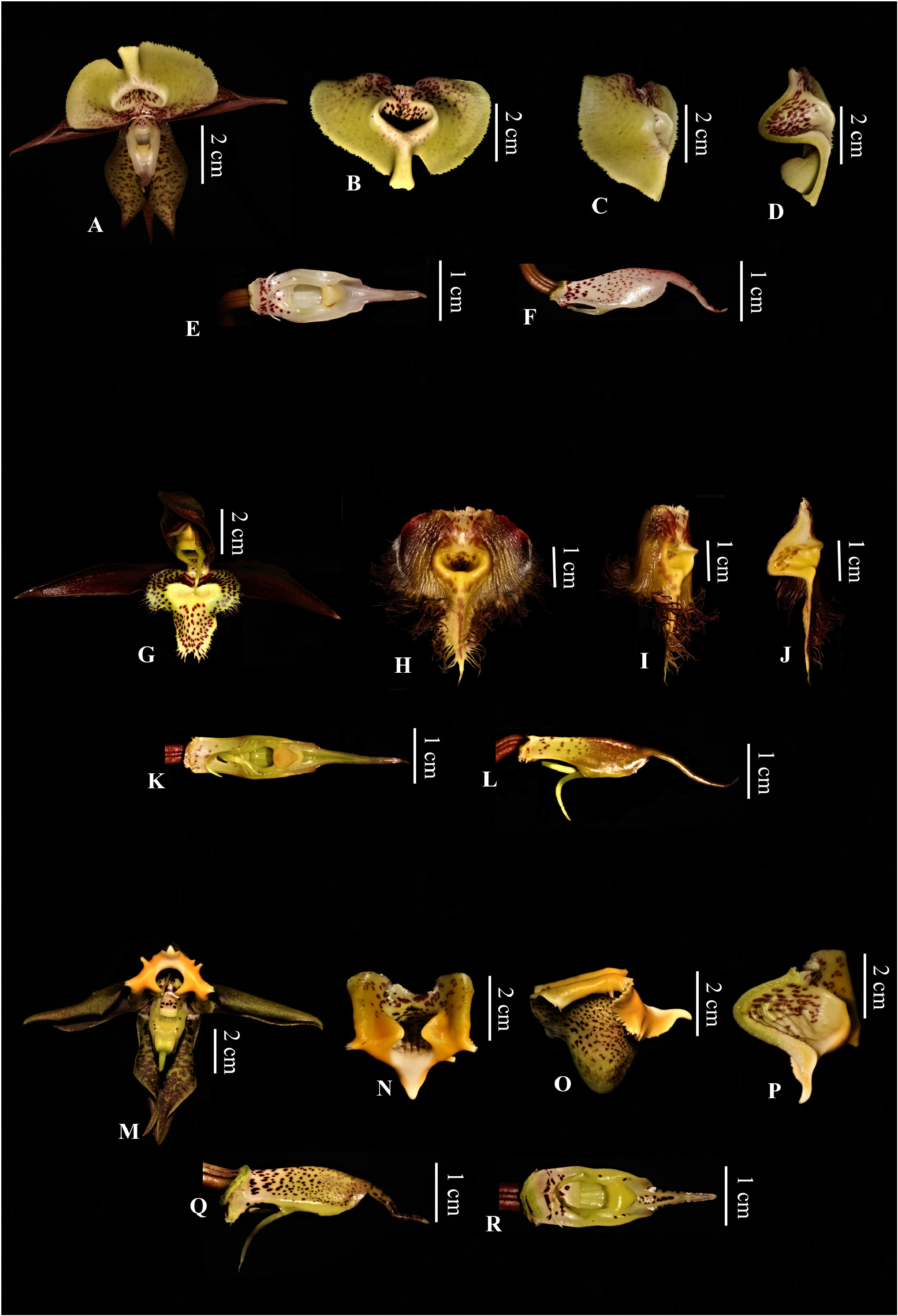
Taxonomic Discussion:—the new entity shows characters intermediate between C. gnomus and C. saccatum ( Figure 3 View FIGURE 3 ), which allowed us to infer its taxonomic position as a nothospecies. As in most species within the genus it is impossible to define diagnostic vegetative features because these species practically share the same characters ( Holst 1999, Walker-Larsen & Harder 2000).
As for the floral morphology, especially in male flowers, we can mention various characters shared with the parental species. Flowers are not resupinate, as in C. gnomus (vs. resupinate in C. saccatum ). Floral segments are elliptic-lanceolate as in C. saccatum (vs. obovate-lanceolate and linear-lanceolate in C. gnomus ) (Pettini-Benelli 2022). The lip is entire and subtrilobed (vs. trilobed in C. gnomus and C. saccatum ), but suborbicular as in C. saccatum , with the margin next to the apex reflexed and overall denticulate to serrate as in both parents (serrate in C. gnomus and fimbriate, laciniate or denticulate in C. saccatum ). The central part of the lip is sacciform and conical with a transverse ostium as in C. saccatum and with two small protuberances on the edge next to column as in both parents, however less acute as in C. saccatum . The column shows a more rounded shape and a beak shorter and broader as in C. gnomus (vs. column narrower and oblong with a beak filiform in C. saccatum ) (see Figure 4 View FIGURE 4 ) ( Lindley 1840a, Linden & Reichenbach 1870, Pessoa et al. 2015, Petini-Benelli 2017, 2022, Krahl 2020).
We propose here a key to all the natural hybrids present in the Brazilian state of Amazonas ( Figure 5 View FIGURE 5 ), i.e. C. × grasineideae ( C. gnomus × C. saccatum ); C. × issanense Pabst (1975: 405) ( C. pileatum Reichenbach (1882: 492) × C. longifolium Lindley (1839: 94)) ; C. × louiseae Krahl et al. (2020: 216) ( C. discolor ( Lindley 1835: t. 1735) Lindley (1844: Misc. 34) × C. macrocarpum Kunth (1822: 331)) ; C. × roseo-album ( Hooker 1840: t. 3796) Lindley (1840b: 65) ( C. discolor × C. longifolium ); and C. × tapiriceps Reichenbach (1888: 133) ( C. macrocarpum × C. pileatum ) (according to Krahl et al. 2020 and Petini-Benelli 2022). It should be noted that C. × sheyllae Krahl, Cantuária & J.B.F.Silva in Cantuária et al. (2021) perhaps occurs in Amazonas but it is not confirmed. It occurs in fact in a border region between Amazonas and Pará (see Cantuária et al. 2021).
We treated C. × roseo-album as a nothospecies, so disagreeing with Barbarena (2021) who treats it as an independent species. We have a large field knowledge as for the occurrence of C. × roseo-album in localities of Brazilian Amazon where C. discolor and C. longifolium are clearly sympatric species a few meters away from each other (eg. Pessoa et al. 2015). Moreover, the nothospecies clearly presents intermediate features between the parental taxa, what we already could observe in artificial crossings. In this way we agree with the opinion of Romero & Carnevalli (1989).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |