Metaconchoecia, Chavtur & Angel, 2011
|
publication ID |
https://doi.org/ 10.11646/zootaxa.2857.1.1 |
|
persistent identifier |
https://treatment.plazi.org/id/03DC87FD-EC02-FFC1-6FDD-FF72A490FCF6 |
|
treatment provided by |
Felipe |
|
scientific name |
Metaconchoecia |
| status |
|
Key to species of the genus Metaconchoecia View in CoL
Males (the key excludes M. sp. nov. 1 and M. sp. nov. 6 of Chavtur 1992, which are yet to be described, and also the two forms of M. acuta Gooday 1981 ).
1a. Copulatory appendage strongly curved, carapace small (<1.1 mm)...................................... M. australis
1b. Copulatory appendage straight or almost straight, carapace length> 1.1mm in most species (0.85–1.73 mm)............ 2
2a. LAG opens>15%CL behind tip of rostrum....................................................... M. discoveryi
2b. LAG opens <15%CL behind tip of rostrum................................................................ 3
3a. Length ≥ 1.7 mm, dorsal margin of carapace shallowly V–shaped with indentation just posterior to posterior end of the A2 protopodite, g-seta of A2 about 2.5x the length of first endopodite segment.................................. .. M. ampla View in CoL
3b. Length <1.7 mm, dorsal margin of carapace straight, g-seta of the A2 three times length of first endopodite segment....... 4
4a. Length> 1.3 mm ...................................................................................... 5
4b. Length <1.3 mm ..................................................................................... 7
5a. RAG opens very close to the posterior dorsal corner; capitulum of frontal organ extends well beyond boundary between second and third segments of A1............................................................................... 6
5b. RAG opens on the posterior margin ~10% height below hinge line; capitulum of frontal organ barely reaches or does not extend beyond distal end of second A1 segment................................................... M. skogsbergi
6a. Carapace short and round in lateral view (height>50% of length); c-seta of A1 stout 3%CL, i.e. shorter than combined length of segments 3 to 5; e-seta of A2 large, stout, claw-like................................................... M. alta View in CoL
6b. Carapace more cylindrical (height usually <50% of length); c-seta of A1 slim and long 6%CL, i.e. similar to combined length of segments 3 to 5; e-seta of A2 a tiny, thin, peg.................................................... M. longiseta View in CoL
7a. A1 c-seta relatively long>4%CL; i.e. similar to the combined lengths of segments 3–5; A1 e-seta is double the length of the limb.............................................................................................................................................................................................. M. wolferi
7b. A1 c-seta relatively short <4%CL; i.e. less then than combined length of segments 3 to 5; A1 e-seta <x1.5 length of the limb 8
8a. a-seta of A1 noticeably swollen at base and longer than the limb....................................... M. crassiseta View in CoL
8b. a-seta of A1 unswollen or barely swollen at base, subequal to or shorter than the limb.............................. 9
9a. Carapace very broad in ventral view (breadth>50% of length)................................................. 10
9b. Carapace rather slim in ventral view (breadth ≤50% of length)................................................ 11
10a. Capitulum of frontal organ broad with short proximal spines, A2 e-seta relatively large................. M. inflata inflata View in CoL
10b. Capitulum of frontal organ slim with long proximal spines, A2 e-seta minute........................... M. inflata lata View in CoL
11a. Armature of e-seta of A1 composed of short non-overlapping spines...................................... M. obtusa
11b. Armature of e-seta of A1 composed of long, overlapping spines............................................... 12
12a. Carapace length <0.9 mm ..................................................................... M. rotundata View in CoL
12b. Carapace length> 0.9 mm ..............................................................................13
13a. Carapace length ≤ 1.1 mm; A1 - a-seta of quite short, 25%CL, does not extend to base of limb; e-seta armature consists of 9–12 pairs of spines...................................................................................... 14
13b. Carapace length ≥ 1.1 mm, A1- a-seta of long,>40%CL, extends to base of limb; e-seta armature consists of 12–15 pairs of spines.............................................................................................. 15
14a. Carapace length 0.85–1.0 mm; A1 e-seta armature consists of 10–12 pairs of spines; in ventral view anterior margin below incisure is acutely angled.......................................................................... M. acuta (inclusive of M. aff. acuta 1 and M. aff. acuta 2 Gooday 1981).
14b. Carapace length 0.95–1.20 mm, A1 e-seta armature consists of 9–11 pairs of spines; in ventral view anterior margin below incisure is bowed and rounded................................................................. M. subinflata
15a. Carapace length 1.10–1.28 mm, mean height 41 ± 1.3%CL.............................................. M. fowleri
15b. Carapace length 1.28–1.40 mm, mean height 45.5 ± 2.2%CL..................................... M. fowleri Form A
Females (excludes M. sp. nov. 1 and M. sp. nov. 6 of Chavtur 1992).
1a. LAG opens ≥17%CL behind tip of rostrum....................................................... M. discoveryi
1b. LAG opens ≤15%CL behind tip of rostrum................................................................. 2
2a. Exopodite of A2 long, ~80% length of protopodite........................................................... 3
2b. Exopodite of A2 ≤70% length of protopodite............................................................... 4
3a. Carapace length <1.3 mm, height>50% of length, width>45% length; terminal setae of endopodite of A2 are half the length of exopodite................................................................................. M. inflata lata View in CoL
3b. Carapace length> 1.6 mm, height <50% of length, width 40% length; terminal setae of endopodite of A2 are about 80% length of exopodite................................................................................... M. ampla View in CoL
4a. Terminal setae of endopodite of A2 40% length of protopodite and 60% length of the exopodite................. M. acuta
4b. Terminal setae of endopodite of A2 ≥50% length of protopodite and>60% length of exopodite....................... 5
5a. Carapace length <0.9 mm ..................................................................... M. rotundata View in CoL
5b. Carapace length> 0.9 mm .............................................................................. 6
6a. Carapace length ≥ 1.3 mm ...............................................................................7
6b. Carapace length <1.3 mm .............................................................................. 10
7a. RAG opens close to the posterior dorsal corner (<5% carapace height below hinge-line)............................. 8
7b. RAG opens well below posterior dorsal corner (10% carapace height below hinge-line).................. M. skogsbergi
8a. Carapace length> 1.4 mm, second segment of A1 has minute dorsal seta......................................... 9
8b. Carapace length <1.4 mm, second segment of A1 lacks a dorsal seta............................... M. aff. skogsbergi
9a. Capitulum of frontal organ is fused to shaft or separated by indistinct suture; first segment of A1 bare, length of first endopodite segment of maxilla is ~1.5x its height........................................................... M. alta View in CoL
9b. Capitulum of frontal organ is divided from shaft by distinct suture; first segment of A1 is armed with dorsodistal spines; length of first endopodite segment of maxilla is similar to its height.......................................... M. longiseta View in CoL
10a. e-seta of A1 ≥2.5x length of limb; terminal setae of A2 exopodite are ≥80% length of exopodite...................... 11
10b. e-seta of A1 ≤2x length of limb; terminal setae of A2 exopodite <80% length of exopodite......................... 12
11a. Carapace length ≥ 1.1 mm .............................................................................. 16
11b. Carapace length ≤1.0 mm....................................................................... M. obtusa
12a. Exopodite of A2 rather long, 80% length of protopodite.......................................... M. inflata inflata View in CoL
12b. Exopodite of A2 rather short ≤70% length of protopodite..................................................... 13
13a. e-seta of A1 2x the lengths of limb and sensory setae........................................................ 14
13b. e-seta of A1 <2x the lengths of limb and sensory setae.............................................. M. crassiseta View in CoL
14a. Height and breadth of carapace respectively>50 and>40%CL........................................ .. M. australis
14b. Height and breadth of carapace respectively ≤50 and ≤40%CL................................................. 15
15a. Carapace elongate (height 45%CL); capitulum of frontal organ straight, tip rounded.......................... M. wolferi
15b. Carapace less elongate (height 48%CL); capitulum of frontal organ curved, tip pointed.................... M. subinflata
16a. Carapace length 1.12–1.28 mm, mean height 37.5% ± 1.1% CL.......................................... M. fowleri
16b. Carapace length 1.28–1.38 mm, mean height 40.5% ± 2.8 %CL................................... M. fowleri form A
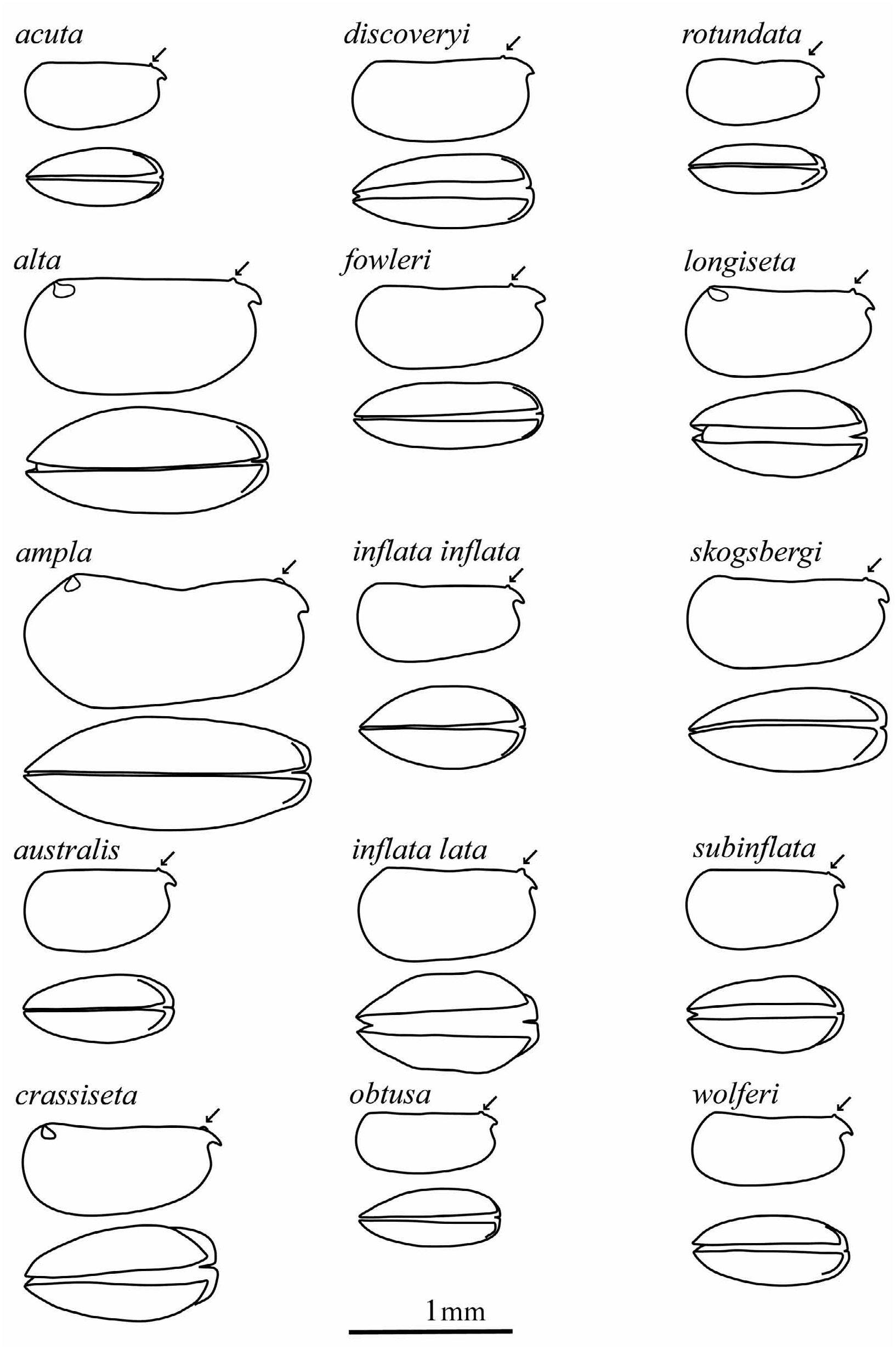
With practice, all these species can generally be determined on the basis of carapace shape and size (although carapace shapes do vary intraspecifically ( Gooday 1981). To facilitate such identifications, Figure 3 View FIGURE 3 reproduces the carapace outlines of the females of all the known Metaconchoecia species drawn to the same scale.
Remarks. Metaconchoecia species are all very similar. Those species that have been described during the last two decades have been recognized as a result of analyses of large collections of depth-stratified samples from wide geographical ranges. Initial separation of these species resulted from the measurements of large numbers of specimens revealing distinct size groupings ( Gooday 1981). Size is generally a very conservative character in halocyprid species, and measurement of large populations can often reveal the existence of possible cryptic species. Also the careful examination of similarly sized populations may reveal subtle, but consistent, differences in the shapes of the carapace, which detailed dissections may again show are discrete populations. Importantly these discretely sized populations often prove to have distinct zoogeographical and/or bathymetric distributions, albeit with some overlap. These overlaps are, in part, artifacts which are the result of sampling errors (e.g. leakage of shallow living specimens into deep samples), or bathymetric sampling ranges being too wide. When examining specimens from under-sampled regions, Metaconchoecia species can often be difficult to identify with any confidence. So rather arbitrary decisions have to be taken whether to broaden the current species concept, or to describe the specimens as novel species. Such dilemmas may only be fully resolved through molecular studies, and some of these are under way ( Bucklin et al. 2010).
To illustrate some of these difficulties, when Gooday (1981) first described Conchoecia fowleri , he distinguished C. fowleri form A. This form differs from his type by having a slightly longer and broader carapace, somewhat longer setae on the male A1, and longer swimming setae on the female A2. Gooday stated (1981, p.148), “This form…does not have a sufficiently distinct depth distribution to be regarded as a subspecies. The broad spectrum of length values for specimens taken at depth of 1000–1500m near the equator, suggests that the two forms are hybridizing here....” We have not been able to clarify the status of this form herein.
Gooday (1981) examined some of the Dana material from the South Pacific that had been identified by Poulsen (1973) as Conchoecia rotundata . Generally he decided that these specimens belonged to his new species Conchoecia acuta , but described two forms, one “narrow”, the other “broad”, that differ slightly from his type material from the subtropical Atlantic. The females of the Conchoecia aff. acuta ‘ broad form’ tend to be broadest posterior to midlength ( Gooday 1981: Figs. 35 C–Q, 36 M–DD), whereas the type form is broadest at midlength. Conchoecia aff. acuta narrow form is not only slimmer but is more parallel-sided ( Gooday 1981: Figs. 35 A, B, 36 A–C). Gooday also identified the narrow form in some of Müller’s (1908) Gauss material from the South Atlantic.
Metaconchoecia skogsbergi also has populations with distinctive size ranges, which otherwise show no other clear distinguishing morphological characters that have yet been identified. Skogsberg’s (1920) thorough description of Conchoecia rotundata from the Antarctic, was cited by Iles (1951) as the type description of Conchoecia (Metaconchoecia) skogsbergi . Skogsberg recorded the lengths of both sexes to be 1.45–1.60 mm. Angel (1968a) described and figured a single female specimen of Metaconchoecia from the Norwegian Sea, which he tentatively attributed to Conchoecia skogsbergi (= Metaconchoecia skogsbergi ), since it was distinct from all the other species in the rotundata group, which were recognized at the time. Gooday (1981) re-examined this specimen and hesitantly agreed with Angel’s initial identification, but in the absence of a male specimen he felt unable decide whether or not it is conspecific with M. skogsbergi . This specimen shows some clear differences from Skogsberg’s (1920) typical form; it is smaller (1.34 mm) and the RAG opens closer to the posterior dorsal corner of carapace ( Angel, 1968a: Fig. 1 View FIGURE 1 ). Skogsberg’s illustration (1920 Fig. CXXII–3), shows the RAG displaced 10% of the height down the posterior margin. So here we suggest that Angel’s Norwegian specimen is best provisionally identified as M. aff. skogsbergi . Other specimens that are similar to this Norwegian specimen occur regularly in small numbers in samples collected from depths> 1000 m in the central and North Atlantic; when these specimens can be critically evaluated we expect them to prove to be a different species.
Gooday (1981) also examined a range of specimens of Metaconchoecia skogsbergi collected by Discovery and Gauss from southern latitudes in the Southern, Indian and Atlantic Oceans. All the Gauss specimens had originally been identified by G.W.Müller as Conchoecia rotundata . Gooday found the size range of females to be 1.34–1.64 mm (n=111), and males to be 1.31–1.62 mm (n=51). He decided there were three distinct size ranges, but meristic measurements of these three size categories failed to reveal any other distinguishing features ( Gooday, 1981: his table 7, p.167). One of us (MVA) measured the sizes of a large sample of a population of M. skogsbergi sampled from a single tow at a depth of 600–700m at 59°N, 20°E (Discovery Station 9969). The mean size of the females was 1.40 ± 0.030 mm (range 1.34–1.50, n = 159), and of males was 1.37 ± 0.027 mm (range 1.32–1.46, n = 160), which are consistent with the size ranges for Gooday’s smaller species, and are slightly smaller than the range of 1.45–1.60 mm given in Skogsberg’s (1920) type description. Once again we expect that critical examination will show these to be different species.
Leung (1972, 1973, 1975) also reported Conchoecia (= Metaconchoecia ) skogsbergi from the Arctic Ocean; his specimens are larger than typical material, and so may belong to yet another species. Thus the present species concept may well include a number of cryptic species that may prove difficult to resolve using a traditional morphological approach.
In his analysis of specimens collected during the Cruises 39 and 45 of the Vityaz from many regions of the North Pacific , Chavtur (1992) identified some specimens as new species, M. species nov.1 and M. species nov.6. These two putative species are yet to be described, and so their morphological characteristics have neither been included in our descriptions nor in the compilation of the tables. Current studies of ostracods from the Celebes Sea show that they include at least three forms of Metaconchoecia , none of which can be confidently attributed to any of the current species .
We have accepted Gooday’s (1981) assumption that the small species found in the N. Atlantic outside the Straits of Gibraltar is conspecific with the small form described by Müller (1894) from the Mediterranean, which in turn is dubiously considered to be conspecific with the species from the Pacific first described as Conchoecia rotundata by Müller (1890). If Mediterranean specimens are critically examined and found to be distinct, then the species from the North Atlantic, herein regarded as M. rotundata , will need to be designated as a new species.
Thus the complexity of the Metaconchoecia species swarm is yet to be resolved fully. The reasons for this complexity are far from being understood. Does it result from a recent or even on-going species radiation? Or, conversely has there been a collapse of barriers to genetic interchange that has lead to recent hybridization? The possibility remains that some of these forms are ecotypes of the same species with flexible developmental responses to local environmental conditions. Gooday (1981) illustrated the range of variation that can be observed within some of the Metaconchoecia species , although some of his variations may have resulted from distortion during collection and preservation. At least some of these issues can be addressed in the future by DNA sequencing.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.