Canis aureus Linnaeus, 1758
|
publication ID |
https://doi.org/ 10.1093/mspecies/sey002 |
|
publication LSID |
lsid:zoobank.org:pub:9FC747F8-BAA4-4E2A-BC5B-53A20A0A03FA |
|
DOI |
https://doi.org/10.5281/zenodo.4591937 |
|
persistent identifier |
https://treatment.plazi.org/id/03D187ED-AE11-263D-1864-512DFBEE0CD6 |
|
treatment provided by |
Felipe |
|
scientific name |
Canis aureus Linnaeus, 1758 |
| status |
|
Canis aureus Linnaeus, 1758 View in CoL
Golden Jackal
Canis aureus Linnaeus, 1758:40 View in CoL . Type locality “oriente;” restricted to “Benná Mts., Laristan, S. Persia ” by Thomas (1911:135).
Lupus aureus Linnaeus, 1758:40 View in CoL . No type locality. From a pre- 1758 name.
Vulpes indiae orientalis Linnaeus, 1758:41 . No type locality. From a pre-1758 name.
[ Canis] View in CoL anthus F. Cuvier, 1820:plate. Type locality “ Senegal.”
Canis variegatus Cretzschmar, 1826:31 , plate 10. Type locality “Nubien. Oberes Egypten;” preoccupied.
Canis syriacus Hemprich and Ehrenberg, 1830 :sig z, plate 16. Type locality “e monte Lebano.”
Canis lupaster Hemprich and Ehrenberg, 1833 View in CoL :sig ff. Type locality “Fayum, Egypt ” (not seen cited in Allen 1939:194).
Canis sacer Hemprich and Ehrenberg, 1833 :sig ff. Type locality “Fayum, Egypt ” (not seen cited in Allen 1939:194).
Canis riparius Hemprich and Ehrenberg, 1833 :sig ff. Type local- ity “Coast of Abyssinia, near Arkiko” (not seen cited in Allen 1939:195).
Canis aureus indicus Hodgson, 1833:237 View in CoL . Type locality “Himalaya.”
Canis aureus var. moreotica I. Geoffroy Saint-Hilaire, 1835 View in CoL :plate 1. Type locality “de Morée.”
Thous anthus: Hamilton Smith, 1839:195 . Name combination.
Thous variegatus: Hamilton Smith, 1839:198 . Name combination.
Thous sengalensi Hamilton Smith, 1839:201 , plate 13. Type locality “ Gambia and Senegal.”
Sacalius aureus: Hamilton Smith, 1839:214 . Name combination. Sacalius barbarus Hamilton Smith, 1839:218 . Type locality “ Tunis;” preoccupied by Canis barbarus Shaw, 1800:311 synonym for Vulpes vulpes View in CoL .
Sacalius indicus: Hamilton Smith, 1839:219 . Name combination.
C [anis]. dalmatinus Wagner, 1841:383. No type locality; Dalmatia implied.
Canis graecus Wagner, 1841:383 . Type locality “Europaei.” Renaming of C. aureus moreotica I. Geoffroy Saint-Hilaire, 1835 View in CoL .
C [anis]. aureus View in CoL vulgaris Wagner, 1841:383. No type locality.
Canis aureus algirensis Wagner, 1841:384 View in CoL . Type locality “d’Alger” from ‘chacal d’Alger’ I. Geoffroy Saint-Hilaire, 1835:22.
C [anis]. aureus View in CoL tripolitanus Wagner, 1841:384. No type locality; Tripoli, Tunis implied.
Canis aureus typicus Kolenati, 1858:96 . No type locality; Persia, Georgia, Turkey implied.
Canis aureus typicus var caucasica Kolenati, 1858:96 . No type locality; Caucasus implied.
Canis aureus var syriaco Kolenati, 1858:96 . Unauthorized emendation of syriacus Hemprich and Ehrenberg, 1830 .
Canis aureus var indica Kolenati, 1858:96 . Unauthorized emendation of indicus Hodgson, 1833 .
Canis aureus var nubica Kolenati, 1858:96 . Renaming of Canis variegatus Cretzschmar, 1826 but Kolenati attributes the name to Rüppell without giving a date.
Canis aureus var algira Kolenati, 1858:96 . Unauthorized emendation of algirensis Wagner, 1841.
Canis aureus var senegalensis: Kolenati, 1858:96 . Name combination.
Canis aureus var barbarus: Kolenati, 1858:96 . Name combination.
Canis aureus balcanicus Brusina, 1892:317 . Type locality “Nardu — nadomak Valpova — na obali Drave, Slavonija.”
Canis hadramaticus Noack, 1896:356 . Type locality “Hadramaut in Arabien.” A jackal-wolf composite. The jackal was chosen as the lectotype (Morrison-Scott 1939)
Canis mengesi Noack, 1897:518 . Type locality “Ostafrikanische.”
Canis lupus minor Mojsisovico, 1897:202 , 241, 244. Preoccupied by Canis spelaeus minor Wagner, 1829 according to Kretzoi, 1947.
Canis cruesemanni Matschie, 1900:145 . Type locality “ Siam.”
Canis anthus soudanicus Thomas, 1903:295 . Type locality “El Obeid,” Kordofan, Sudan.
Canis somalicus Lorenz, 1906:306 . Type locality “Ireso bei Agada.”
Canis gallaënsis Lorenz, 1906:307 . Type locality “Ginea (Arussi).”
Canis doederleini Hilzheimer, 1906a:116 . Type locality “Oberägypten.”
Canis thooides Hilzheimer, 1906b:364 . Type locality “Sennaar.”
Canis lupaster grayi Hilzheimer, 1906b:367 . Type locality “Nordafrika.”
Canis studeri Hilzheimer, 1906b:368 . Type locality “ Tunis.”
C [anis]. mengesi lamperti Hilzheimer, 1906b:371 . No type locality.
Thos aureus bea Heller, 1914:5 . Type locality “Loita Plains, British East Africa,” Kenya.
Canis indicus kola Wroughton, 1916:651 . Type locality “Palanpur,” India.
Canis naria Wroughton, 1916:651 . Type locality “Coorg,” west coast of India.
Canis lanka Wroughton, 1916:652 . Type locality “Mankeni,” Sri Lanka.
Thos lupaster maroccanus Cabrera, 1921:263 . Type locality “ Mogador ( Marruecos).”
Th [os]. l [upaster]. Algirensis: Cabrera, 1921:263. Name combination.
Th [os]. a [ureus]. Riparius: Cabrera, 1921:264. Name combination.
Th [os]. aureus nubianus Cabrera, 1921:264. Renaming of variegatus Cretzschmar, 1826 .
Canis aureus hungaricus Éhik, 1938:11 . Type locality “Tyukod (Marshy-land Ecsed), Comitat Szatmár, Hungary;” Preoccupied according to Kretzoi, 1947.
Canis aureus minor: Éhik, 1938:13 . Name combination; preoccupied.
Thos aureus algirensis: Allen, 1939:194 . Name combination.
Thos aureus anthus: Allen, 1939:194 . Name combination.
Thos aureus lupaster: Allen, 1939:194 . Name combination.
Thos aureus maroccanus: Allen, 1939:195 . Name combination.
Thos aureus soudanicus: Allen, 1939:195 . Name combination.
Thos gallaensis: Allen, 1939:195 . Name combination.
Thos lamperti: Allen, 1939:195 . Name combination.
[ Canis aureus View in CoL ] moreoticus Kretzoi, 1947:287. Unjustified emendation of moreotica I. Geoffroy Saint-Hilaire, 1835.
Thos aureus ecsedensis Kretzoi, 1947:287 . Renaming of C. a. hungaricus Éhik, 1938.
Canis lupaster doederleini: Saleh and Basuony, 2014:49 View in CoL . Name combination.
Canis aureus qattarensis Saleh and Basuony, 2014:49 View in CoL . Type locality “northern region of the Egyptian Western Desert.”
CONTENT AND CONTEXT. Order Carnivora , family Canidae , subfamily Caninae , genus Canis . As many as 13 subspecies of C. aureus are distinguished through Europe and Africa ( Allen 1939; Ellerman and Morrison-Scott 1951; Coetzee 1977). However, these subspecies may be part of a 3-species complex (see “Nomenclatural Notes”). Unfortunately, much of the literature does not specify geographic location or subspecies. Here, C. aureus is used for the global range from Africa into Eurasia (Wozencraft 2005). Therefore, we list the historical 13 subspecies of C. aureus ( Heller 1914; Allen 1939; Ellerman and Morrison- Scott 1951; Coetzee 1977; Wozencraft 2005). The newly named subspecies, qattarensis Saleh and Basuony, 2014, is tentatively treated as a synonym of syriacus . Subspecies designations for the orientalis Linnaeus, 1758 and studeri Hilzheimer, 1906b are not known.
C. a. algirensis Wagner, 1841. See above; synonyms are algira Kolenati, 1858; barbarus ( Hamilton Smith, 1839) ; grayi Hilzheimer, 1906b; tripolitanus Wagner, 1841.
C. a. anthus F. Cuvier, 1820 View in CoL . See above; synonym is senegalensis ( Hamilton Smith, 1839).
C. a. aureus Linnaeus, 1758 View in CoL . See above; synonyms are balcanicus Brusina, 1892; caucasica Kolenati, 1858; hadramauticus Noack, 1896; maroccanus ( Cabrera, 1921); orientalis ( Linnaeus, 1758); typicus Kolenati, 1858; vulgaris Wagner, 1841.
C. a. bea ( Heller, 1914). See above.
C. a. cruesemanni Matschie, 1900 . See above.
C. a. ecsedensis ( Kretzoi, 1947). See above; synonyms are hungaricus Ehik, 1938; minor Mojsisovico, 1897 (preoccupied).
C. a. indicus Hodgson, 1833 . See above; synonyms are indica Kolenati, 1858; kola Wroughton, 1916.
C. a. lupaster Hemprich and Ehrenberg, 1833 View in CoL . See above; synonym is sacer Hemprich and Ehrenberg, 1833 .
C. a. moreotica I. Geoffroy Saint-Hilaire, 1835. See above; synonyms are dalmatinus Wagner, 1841; graecus Wagner, 1841 .
C. a. naria Wroughton, 1916 . See above; synonym is lanka Wroughton, 1916 .
C. a. riparius Hemprich and Ehrenberg, 1833 . See above; synonyms are gallaënsis Lorenz, 1906 ; hagenbecki Noack, 1897; lamperti Hilzheimer, 1906b ; mengesi Noack, 1897 ; somalicus Lorenz, 1906 .
C. a. soudanicus Thomas, 1903. See above, synonyms are doederleini Hilzheimer, 1906a ; nubianus ( Cabrera, 1921); nubica Kolenati, 1858; thooides Hilzheimer, 1906b ; variegatus Cretzschmar, 1826 (preoccupied).
C. a. syriacus Hemprich and Ehrenberg, 1830 . See above, synonym are qattarensis Saleh and Basuony, 2014; syriaco Kolenati, 1858.
NOMENCLATURAL NOTES. The above-named list may represent 3 taxa of canids: Canis anthus (African wolf), C. aureus , and C. lupus (gray wolf). Genetic analyses of C. aureus and C. lupus suggest that C. aureus from Africa and Eurasia are distinct clades and should be considered to be separate species, that is C. anthus (Africa) and C. aureus (Eurasia—Koepfli et al. 2015) . As noted above, the literature does not distinguish these species and we include both as C. aureus .
In addition, large C. aureus in Egypt ( lupaster ) may be a cryptic subspecies of C. anthus ( Rueness et al. 2011; Gaubert et al. 2012). Canis aureus lupaster (Hemprich and Ehrenberg 1833) is classified as a golden jackal; however Ferguson (1981) suggested that the taxon C. aureus lupaster , which is present in arid areas of Egypt and Libya (Osborn and Helmy 1980), may actually represent a small C. lupus rather than a large jackal. Mitochondrial DNA analyses suggest that C. a. lupaster represents an ancient wolf lineage that may have colonized Africa prior to the northern hemisphere radiation ( Rueness et al. 2011). Mitochondrial DNA analyses assigned individuals from Algeria, Mali, and Senegal to C. lupus lupaster ( Gaubert et al. 2012) and identified C. lupus lupaster mtDNA haplotypes in C. aureus from Senegal questioning the genetic differentiation between the proposed C. anthus and C. aureus . We retain C. aureus lupaster as a form of the golden jackal not as a subspecies of C. lupus following Wozencraft (2005— Qumsiyeh 1996; Ferguson 2002).
The specific name is Latin for golden. Common names include Asiatic jackal, common jackal, ibn awa or ibn awee (classic Arab), wa wie (spoken Arab), cakalli (Albanian), čagalj (Croatian), šakal obecný (Czech), sjakal (Danish, Swedish), jakhal (Dutch), šaakal (Estonian), sakaali (Finnish), Sjakalur (Faeroese), chacal doré (French), Goldschakal (German), tơɑκɑλ (Greek), aranysakál (Hungarian), sciacallo dorato (Italian), chagal, turg (Kurdish), zeltainais šakālis (Latvian), xakali (Maltese), gullsjakal (Nowegian), shoghal (Persian), szakal zlocisty (Polish), chacal-dourado (Portuguese), şakal (Rumanian), šakal obyčajný (Slovakian, Slovenian), chacal (Spanish), çakal (Turkish—Hatt 1959; P. D. Moehlman, in litt.). Indigenous names include Amharic: tera kebero ( Ethiopia); Fulani: sundu; Hausa: dila; Hindi: Giddhad; Kanada: nuree; Kiswahili: bweha wa mbugani, bweha dhahabu ( Tanzania); Marathi ( India): kolha; Nepali ( Nepal), Bengali, Gujarati and Kutchi ( India): shiyal; Singhelese: nariya; Songhai: nzongo; Tamil ( India): peria naree; Wolof: Tili (P. D. Moehlman, in litt.).
DIAGNOSIS
Canis aureus occurs sympatrically with the silver-backed jackal ( Canis mesomelas ) and the side-striped jackal ( Canis adustus ) in parts of East Africa. All have skull lengths of 141– 147 mm with minimal divergence in size (van Valkenburgh and Wayne 1994). C. mesomelas is distinguished by reddish flanks and limbs, and the skull may have a less developed parietal crest (Clutton-Brock et al. 1976), the angle of its ears which are more upright, and, usually, the prominent dark “silver” saddle composed of black and white hairs (Walton and Joly 2003). C. aureus may have a dark saddle seasonally, which is not prominent. C. adustus characteristically has shorter ears set more to the side of the skull, a pale side-stripe, and a white-tipped tail (Moehlman and Jhala 2013). C. lupus is much larger with a heavier body and variable pelage coloring. The skull is much larger with a welldeveloped interparietal crest, strong jaw, and large canine and carnassial teeth (Clutton-Brock et al. 1976).
GENERAL CHARACTERS
Canis aureus ( Fig. 1 View Fig ) is a medium-sized canid, considered the most typical representative of the genus Canis (Clutton- Brock et al. 1976). Basic coat color is golden but varies from pale creamy yellow to a dark tawny hue on a seasonal basis. The pelage on the back is often a mixture of black, brown, and white hairs, such that they can appear to have a dark saddle similar to the C. mesomelas (Walton and Joly 2003; Moehlman and Jhala 2013). The long contour hairs have an extensive black tip and a wider penultimate white band ( Pocock 1938). In winter, a copious under wool is present ( Pocock 1938). C. aureus inhabiting rocky, mountainous terrain may have a grayer coat shade ( Sheldon 1992). The belly and underparts are a lighter pale-ginger to cream. The tail is bushy with a tan to black tip. Females have 7–8 mammae ( Poché et al. 1987), comprising 4 pairs ( Sheldon 1992). Eye color varies from pale yellow to amber ( Lewis et al. 1968).
Means and parenthetical ranges of external measurements (mm) from 6 males and 3 females, respectively, in Gujarat, India (Y. V. Jhala, in litt.) were: length of head and body, 793 (760–840), 760 (740–800); length of tail, 220 (200–240), 205 (200–210); length of ear, 76 (68–90), 80 (75–85); body mass (kg) of 6 male and 4 females was 8.8 (7.6–9.8) and 7.3 (6.5– 7.8), respectively. In Tanzania, the external measurements (mm or kg) from 2 males in the Serengeti (Moehlman and Jhala 2013) were: length of head and body, 740, 785; length of tail, 270, 280; length of ear, 110, 110; mass, 6.3, 7.7. In Egypt, means and parenthetical ranges (mm) for 9 adult C. a. lupaster of unknown sex were: length of head and body, 872 (822–893); length of tail, 312 (290–347); length of hind foot, 200 (190–212); length of ear, 112 (104–121); and body mass (kg) of 4 adults was 13 (10–15—Osborn and Helmy 1980). In Lebanon, external measurements (mm) of 3 adult C. a. syriacus were: total length, 988.3 (950–1015); length of tail, 275 (265–290); length of head and body, 713.3 (680–750); length of hind foot, 156 (140–170); length of ear from notch, 74.7 (70–80— Lewis et al. 1968). In Bangladesh, mean measurements (mm, SD, n) of females and males, respectively, were: total length, 1009.3 (33.67, 6), 1058.6 (20.30, 7); length of tail, 220.1 (17.16, 7), 245.0 (21.41, 6); length of hind paw, 58.0 (4.93, 7), 56.3 (4.83, 7); length of hind foot, 146.2 (24.66, 5), 162.6 (8.52, 7); length of ear, 69.4 (4.80, 5), 74.1 (5.49, 7); mass (kg), 8.5 (0.73, 7), 10.3 (0.95, 7— Poché et al. 1987). Mean adult mass (kg) of 5 females and 3 males, respectively, was 10.0 and 12.0 ( Palmqvist et al. 2002). On average, females are 12% lighter than males: 5.8 kg versus 6.6 kg (Moehlman and Hofer 1997).
Mean cranial measurements (mm, SD; Fig. 2 View Fig ) on 10 mandibles and upper jaws of adult C. aureus in Iran were: length of skull, 169, 31.9; length of cranium, 101, 23.7; nasal length, 67, 16.6; cranial width, 72, 4.4; length of mandible, 112, 34.6; lateral alveolar root to mental foramen, 25, 1.7; mental foramen to caudal mandibular border, 87, 3.8; mandibular foramen to base of mandible, 12, 0.4; caudal border of mandible to below mandibular foramen, 11, 0.8; maximum mandibular height, 60, 5.8 ( Mofared 2013). Mean measurements (mm, range) for 3 Lebanese adults were: greatest length of skull, 148.6 (135.2–157.4); condylobasal length, 142.9 (131.9–149.1); zygomatic breadth, 83.9 (73.3–90.2); breadth of braincase, 48.8 (46.0–50.3); interorbital constriction, 29.4 (25.5–32.7); maxillary toothrow, 62.9 (60.4– 65.0); mandibular toothrow, 72.2 (58.2–74.9); mandibular length, 114.1 (104.1–121.0— Lewis et al. 1968). Mean cranial measurements (mm; with range and n) of Egyptian adult C. a. lupaster were: condyloincisive length, 185.2 (173.5–196.0, 13); zygomatic width, 101.4 (93.5–111.3, 14); rostral width, 33.8 (31.2–37.8, 14); postorbital width, 34.8 (31.8–38.9, 13); basioccipital width at tempoparietal suture, 54.4 (51.8–59.1, 13); nasal length, 72.0 (65.9–84.8, 14); width across upper molars, 58.2 (54.2–63.0, 14); distance from anterior most surface of canine to posterior of 2nd molar, 80.3 (75.6–86.8, 14); skull height, 66.8 (62.0–74.0, 9—Osborn and Helmy 1980). In Bulgaria, mean cranial and tooth measurements (mm) for 92 males and 56 females, respectively, were: total skull length, 166, 161; neurocranium length, 89, 86.5; greatest length of nasals, 59.7, 57.9; greatest neurocranium breath, 52.5, 52.6; total length of mandible, 121.9, 118.6; height of vertebral ramus, 48.6, 46.7; length of P1 to P4, 33.9, 33.4; length of M1 to M3, 31.8, 31.0 ( Stoyanev 2012). Mean mandible length and depth at 2nd molar (mm) for 5 females and 3 males, respectively, was f: 102.8, 14.3; m: 108.9, 15.3 ( Palmqvist 2002). Condylobasal length (mm) from 10 males was 154, whereas it was 140 from 7+ female skulls ( Pocock 1938).
DISTRIBUTION
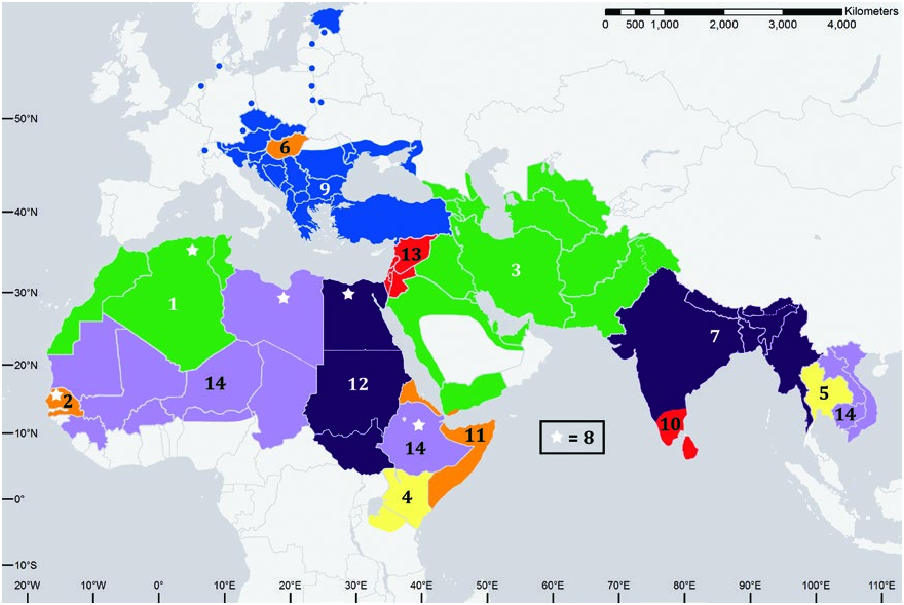
Canis aureus is widespread in North and East Africa, Europe, the Middle East, Central Asia, and Southeast Asia ( Fig. 3 View Fig ). They range from sea level in Eritrea to 3,500 m in the Bale Mountains of Ethiopia and 2,000 m in India ( Prater 1971; Admasu et al. 2004).
In Greece and Dalmatia, C. aureus is documented from the Holocene (Sommer and Benecke 2005; Malez 1984 in Rutkowski et al. 2015) and the ancient Mediterranean populations have persisted and merged with jackals coming from Asia ( Fabbri et al. 2014; Rutkowski et al. 2015). The expanding population of C. auerus in Europe is due to natural longdistance dispersal ( Arnold et al. 2012; Rutkowski et al. 2015). C. aureus expanded from southeastern Europe to Hungary in the late 1800s (Tóth et al. 2009). In the late 20th century, the expansion was rapid and is still ongoing ( Kryštufek et al. 1997; Arnold et al. 2012).
FOSSIL RECORD
The fossil record of Canis aureus is limited with many fossils that are controversial as to their specific attribution. A right lower carnassial from a Paleolithic Lebanon cave may be C. aureus ( Hooijer 1961) . Fossil remains attributed to C. aureus dating from the Upper Pleistocene were found in North Africa ( Savage 1977). Palestinian fossils attributed to this species have been reattributed to C. lupaster ( Kurten 1974) and hence might be a separate species ( Spassov 1989). Reliable fossils of C. aureus have not been found in the Pleistocene of Europe ( Spassov 1989). C. aureus appears to have arrived in sub-Saharan Africa after C. mesomelas and C. adustus , which have a fossil record in eastern and southern Africa from 500,000 to 2.5 million years ( Ewer 1956; Savage 1977).
FORM AND FUNCTION
The dental formula is i 3/3, c 1/1, p 4/4, m 2/3, total 42 ( Fig. 2 View Fig ). Of 31 Canis aureus skulls, 13% had broken teeth (van Valkenburgh 2009). Baculum length is 62.7 mm ( Dixson 1995), but 2 bacula of C. a. lupaster were 70 and 73 mm (Osborn and Helmy 1980).
Male and female skulls can be differentiated but cranial measures overlap ( Stoyanev 2012). Differentiation was not dependent on age or collection site. Only juveniles below the age of 6 months could be differentiated from adults. C. aureus from Bulgaria did not differ from European or Asian skulls and encompassed both African and Dalmatian jackals, but skulls from Canis aureus lupaster were larger, longer, and broader ( Stoyanev 2012). Croatian skulls were of a shorter length and broader ( Stoyanev 2012). In Bulgarian skulls, age could be assessed in 223 of 228 from craniometrics differences ( Stoyanev 2012). Across 7 populations, sexual dimorphism in skull length and canine diameter was significant for East African and northern Indian C. aureus but not north African, north central African, Middle Eastern, Pakistani, or southern Indian animals (Van Valkenburgh and Wayne 1994). In Bangladesh, mean organ weights (g, SD, n) for adult females and males, respectively, were: heart, 71.0 (7.35, 3), 79.2 (14.02, 4); liver, 297.7 (14.52, 3), 365.5 (72.06, 4); paired kidneys, 45.7 (8.73, 3), 64.0 (4.37, 4); spleen, 75.3 (21.1, 3), 43.0 (10.61, 3); lungs, 64.7 (11.47, 3), 85.7 (18.87, 3— Poché et al. 1987).
Adult C. aureus can be immobilized for handling up to 55 min with 130 mg ketamine hydrochloride and 15 mg promazine hydrochloride ( Fuller et al. 1989). For 3 female and 4 male C. aureus , doses of 15–80 mg /kg of ketamine hydrochloride generated recovery times of 32–300 min with no apparent side effects ( Poché et al. 1987). Time to immobilization was 2–3 min ( Poché et al. 1987). Trapped jackals were anesthetized with an intramuscular injection of a mixture of medetomide 0.09 mg /kg and ketamine 2.8 mg /kg and time to immobilization was an average of 5.0 min ( Admasu et al. 2004).
ONTOGENY AND REPRODUCTION
Ontogeny.— The estimated weight from a captive born litter of 2 female and 2 male Canis aureus neonates was 189 g (Moehlman and Hofer 1997). A male pup weight at day 8 was 249 g, day 13: 500 g, and at day 15: 590 g. A female pup at day 5 weighed 295 g, and at day 21 weighed 670 g ( Seitz 1959). Pups are blind at birth with eye opening at 8–11 days and tooth eruption at 11 days ( Seitz 1959; Wandry 1975). In captivity, canine teeth erupted at 28 days ( Seitz 1959) and pups emerged from the den box at approximately 4 weeks (Wandry 1975). At 13 months of age a female had her 1st litter and hence was reproductively mature at 11 months of age ( Seitz 1959).
In the Serengeti, Tanzania, the pups are born and remain in underground dens until they are 3 weeks old during which time their mother spends most of her time with them ( Moehlman 1983). Pups emerge at approximately 3 weeks of age and in addition to nursing, eat regurgitated food ( Moehlman 1986). Some families have helpers which are offspring from the previous year’s litter. During pregnancy, the female is provisioned by her mate and while she is lactating she is provisioned by her mate and the “helpers” which are her older offspring (Moehlman and Hofer 1997). One of us (PDM) has observed that helpers also provision the pups which are their full siblings. They are weaned at 8–10 weeks of age and stay within several hundred meters of the den until they are 10 weeks old. At 14 weeks of age they are well coordinated and start to forage with the adults. They will continue to receive some food from their parents until they are 8 months old. Even when they are helpers (11–18 months) they may infrequently receive food from their parents ( Moehlman 1986).
Adults can be distinguished from juveniles by frontoparietal ridges that are fused posteriorly to the frontoparietal suture creating a high sagittal ridge (Osborn and Helmy 1980). Tooth wear can be used to age C. aureus ( Raichev 2011) .
Reproduction. —In the Serengeti mating typically occurs from October to December ( Moehlman 1983, 1986) and birth from December to March ( Moehlman 1983). In Egypt, litters have been observed in March, April, and May ( Flower 1932). In India reproductive behavior begins in February–March (Jhala and Moehlman 2004) and in Israel between October and March (Golani and Keller 1975; Ginsberg and Macdonald 1990). In Bangladesh, pups are present year-round ( Poché et al. 1987). Gestation is about 63 days ( Sheldon 1992). Litter size ranges from 1 to 9, average 5.7 (Moehlman and Hofer 1997). In Tanzania, an average of 2 pups emerged from the natal den at 3 weeks (Wyman 1967). Lactation lasts for 8–10 weeks ( Moehlman 1986). Six females captured in Kenya in June to August were not lactating ( Fuller et al. 1989).
ECOLOGY
Population characteristics. —In Tanzania, densities up to 2 adults per km 2 may occur ( Moehlman 1983, 1986, 1989). In northern Dalmatia, Croatia, territory density was 0.61–1.15 groups per 10 km 2 ( Krofel 2008), whereas in southern Dalmatia density was 6–7 groups per 10 km 2 ( Krofel 2007). In Kenya, juveniles comprised 47% of captured Canis aureus ( Fuller et al. 1989) . In the Serengeti, maximum longevity is about 14 years (Moehlman and Hofer 1997). In captivity, individuals have lived up to 18 years 10 months (Weigl 2005). Sex ratio at approximately 3 weeks when pups emerge from the den is 1:0.8 (n = 33), surviving at age 14 weeks (1:0.8, n = 44), helping as juveniles at approximately 52 weeks (1:1, n = 16), and reproductive adults (1:1, n = 46— Moehlman 1986). The sex ratio is equal for all age groups and sex ratio is equal for helpers.
Space use. —Tolerance of dry habitats and an omnivorous diet allow Canis aureus access to a wide variety of habitats (Jhala and Moehlman 2008). They normally live in open grassland habitat but also occur in deserts, woodlands, mangroves, and agricultural and rural habitats in India and Bangladesh (Jhala and Moehlman 2008)
In the Serengeti, C. aureus maintains year-round exclusive territories of 0.5–7.0 km 2 ( Moehlman 1983) and 2–5 km 2 in Ngorongoro (van Lawick and van Lawick-Goodall 1970), but will make excursions beyond these territorial boundaries to gain access to fresh carcasses. In Algeria, seasonal territories as may be as small as 0.39 km 2 ( Khidas 1990). Territories are within larger home ranges. Home range size of radiocollared individuals in some locales can vary according to habitat and age. An adult pair in Acacia woodland in Kenya had a home range of 2.4 km 2 and 2 juvenile females had home ranges of 5.6 and 21.7 km 2 ( Fuller et al. 1989), although range size over a 16-month period in the Bale Mountains of Ethiopia varied from 7.9 to 48.2 km 2 for adults and from 24.2 to 64.8 km 2 for subadults ( Admasu et al. 2004). Subadults may have larger home ranges as they explore and search for a mate and a territory. Home ranges of individuals within a social group tend to overlap (van Lawick and van Lawick-Goodall 1970; Khidas 1990; Admasu et al. 2004). In Bangladesh, breeding pairs defend an average of 37.3 ha of cover but not the peripheral foraging areas ( Jaeger et al. 1996, 2007) and a male and female had home ranges of 1.1 and 0.6 km 2, respectively ( Poché et al. 1987). In Israel, home and core ranges (km 2) of C. aureus near villages were about one-third of those in more natural areas (mean, SD): home, 6.6 (4.5) versus 21.2 (9.3); core, 1.2 (0.9) versus 3.5 (1.6— Rotem et al. 2011).
In the Serengeti, territories are maintained for up to 8 years albeit with changes in boundaries and size through time. Both members of the pair mark and defend the territory ( Moehlman 1986).
Diet. —In Bangladesh, rodents are the most common food ( Jaeger et al. 2007). Similarly, in Bulgaria, rodents comprise 59.3% of the diet with brown hares ( Lepus europaeus , 20.1%) and plants (primarily fruit, 19.7%) as the next most common components (Markov and Lanszki 2012). In Rajasthan, India, rodents make up 45% of the diet ( Mukherjee et al. 2004). In Algeria, mammals comprise about a one-third of the diet with 17–24% poultry and 12–27% insects depending on location ( Amroun et al. 2006). In Israel, domestic carrion may form a significant component of the diet along with fruit, birds, small mammals, and invertebrates ( Borkowski et al. 2011). In Bangladesh, 12 stomachs contained primarily carrion, poultry, and vegetation but no rodents ( Poché et al. 1987).
Single C. aureus typically hunt smaller prey like rodents and birds, using their hearing to locate rodents in the grass and then pouncing on them by leaping through the air, or digging out rodents from their burrows. However, single animals will also hunt Thomson’s gazelle ( Eudorcas tomsonii ) fawns. They have been observed to hunt young, old, and infirm ungulates that are sometimes 4–5 times their body weight (van Lawick and van Lawick-Goodall 1970; Eisenberg and Lockhart 1972). C. aureus in Bale, Ethiopia, may be more solitary than elsewhere in the range, with individuals having been observed foraging alone on 87% of occasions ( Admasu et al. 2004). This is likely because food resources were widely dispersed and rarely concentrated enough for jackals to forage in groups ( Admasu et al. 2004).
In the Serengeti, mated pairs will hunt cooperatively and regularly kill Thomson Gazelle fawns and occasionally adults; pairs have a higher kill rate than individuals (Wyman 1967; Kruuk 1972; Rosevear 1974). Cooperative hunting permits them to harvest much larger prey in areas where they are available Temu et al. 2017. In the Ngorongoro Crater, C. aureus pairs cooperatively killed Thomson’s gazelle fawns and occasionally killed adult Thomson’s gazelle. Individuals successfully hunted Abdim’s storks ( Ciconia abdimii ) and during Wildebeest calving season consumed the abundantly available placentas. There was one observation of C. aureus cooperatively killing and eating a bat-eared fox ( Otocyon megalotis ) (Temu et al. 2017). Golden jackals consumed significantly more grams of food per hour of foraging when compared with sympatric Silver-backed jackals (Temu et al. 2016). In some areas, particularly where food resources are clumped, aggregations of C. aureus may occur, such as 14 jackals on a carcass in Ngorongoro (van Lawick and van Lawick-Goodall 1970) and aggregations of 5–18 jackals scavenging on carcasses of large ungulates in India (Y. V. Jhala, in litt.). C. aureus will cache excess food (van Lawick and van Lawick-Goodall 1970). In India, they will kill poultry as well as young and weak sheep and goats (Jhala and Moehlman 2004), and will venture into human habitation at night to feed on garbage ( Poché et al. 1987). In the Golan Heights, 1.5–1.9% of domestic calves are killed by predators, primarily C. aureus (Yom-Tov et al. 1995) .
In the Serengeti, C. aureus scavenges the kills of spotted hyenas ( Crocuta crocuta ) and lions ( Panthera leo ) and leopards ( Panthera pardus —van Lawick and van Lawick-Goodall 1970; Moehlman 1986). In the Serengeti during the dry season, C. aureus does not have access to free standing water and can tolerate dry habitats (Moehlman and Jhala 2013).
Diseases and parasites. —In the Serengeti, blood of 2 Canis aureus males was negative for leptospirosis, canine distemper, canine brucellosis, rabies, rinderpest, African horse sickness, and rift valley fever. Both individuals had titers for canine adenovirus and canine coronavirus. The older male was positive for canine parvovirus (Karesh 1990 in litt.). In 1994 during an outbreak of distemper, one C. aureus tested positive for canine distemper virus (Roelke- Parker et al. 1996). In 2011, samples collected from C. aureus pups were positive for canine distemper virus, but only 1 death was observed (Olarte-Castillo et al. 2015). In Kenya (1987–1988) testing for canine parvovirus (CPV-2), canine distemper virus, Ehrlichia canis , and rabies indicated that 9 of 16 individuals were positive for CPV-2; all were negative for canine distemper virus, E. canis , and rabies ( Alexander et al. 1994).
In Israel, a serological survey indicated the presence of canine distemper virus, E. canis , and Leshimania infantum. Blood smears in one individual revealed Hepatazoon canis ( Shamir et al. 2001) . Leshimania tropica is also known from Israeli jackals (Talmi-Frank et al. 2010). Six of 8 C. aureus were positive for Toxoplasmosis gondii in the United Arab Emirates ( Dubey et al. 2010).
Canis aureus in Serbia had an infection rate of 46 out of 447 jackals (10.3%) of at least one helminth species and in total 12 species were identified: 2 trematodes, Alaria alata and Pseudamphistomum truncatum , 3 nematodes, Toxocara canis , Ancylostoma caninum , and Gongylonema , and 7 cestodes, Taenia pisiformis , Taenia hydatigena , Multiceps multiceps , Multiceps serialis , Mesocestoides lineatus, Macrocanthorynchus hirudinaceus, and Onicola canis ( Ćirović et al. 2015) . In Israel, fecal samples had A. caninum and Dipylidium caninum ( Shamir et al. 2001) . In Tanzania, fecal samples from C. aureus had Coccida, Ancyclostoma , Psoroptes , and T. canis (W. B. Karesh, in litt.). In Rumania, the roundworm, Trichinella britovi occurs in C. aureus ( Blaga et al. 2008) . In Uzbekistan and Turkmenistan, the Ministries of Health ( Berdyeva 1998; L. M. Isaev Institute of Medical Parasitology 1998) reported that C. aureus may be infected with the guinea worm, Dracunculiasis medinensis; thus, C. aureus potentially can infect water sources. In Italy, a subadult male had 2 ectoparasitic species, Dermacentor reticulatus , Isodes ricinus , and 1 endoparasitic species, Metagonimus yokogawai ( Lapini et al. 2009) . One C. aureus from Tunisia had 72 tapeworms, Echinococcus granulosus while 4 others had fecal evidence of the species ( Lahmar et al. 2009). The protozoan, Babesia gibsoni , may use C. aureus as a host ( Patton 1910).
Interspecific interactions. —In East Africa, Canis aureus is sympatric with C. mesomelas and C. adustus . Skull characters indicate that C. aureus are more carnivorous than C. adustus and less carnivorous than C. mesomelas (van Valkenburgh and Wayne 1994) . In Hungary, dietary overlap between sympatric C. aureus and red foxes ( Vulpes vulpes ) is high at 73% as both primarily eat small mammals <50 g ( Lanszki et al. 2006).
In East Africa, spotted hyenas will kill and feed on C. aureus ( Kruuk 1972; Kingdon 1977). In the Serengeti, C. aureus give a “warning yowl” when spotted hyenas approach their dens. The adult jackals then chase the hyenas and bite them on the rump or genitals. Honey badgers ( Mellivora capensis ) have been observed near C. aureus dens, but have always been chased away by the parents and helpers. In India, striped hyenas ( Hyaena hyaena ) may prey on C. aureus (Y. V. Jhala, in litt.) and pythons ( Python molurus ) can be major predators ( Singh 1983). In India, when C. aureus approaches human habitation it may be killed by feral domestic dogs and death due to road kills increased during its breeding season (Y. V. Jhala, in litt.). C. aureus may be killed as a side effect of predator control programs when it scavenges poisoned carcasses (Moehlman and Jhala 2013).
Miscellaneous. —Capture methods include rubber-padded, steel, foot-hold traps, cage-type live traps, large box traps, and Victor steel traps with off-set jaws ( Poché et al. 1987; Fuller et al. 1989; Admasu et al. 2004; Rotem et al. 2011). Differences in white markings on throat and chest may allow individual identification ( Macdonald 1979). Unique facial scars and naturally notched ears can be used to identify individuals ( Moehlman 1983, 1986).
HUSBANDRY
As of March 2000, approximately 193 Canis aureus were in Indian zoos (Jhala and Moehlman 2004). C. aureus have been raised in captivity for behavioral studies ( Seitz 1959; Wandry 1975). Individuals were housed in a kennel of 225 m 2 which included 5 separate and interconnected areas/cages. Each cage had a large box with a tunnel-like entrance (Wandry 1975).
BEHAVIOR
Grouping behavior. — Canis aureus are typically monogamous but can potentially be socially flexible depending on demography and food resources ( Macdonald 1979; Moehlman
1983, 1986, 1989; Fuller et al. 1989; Moehlman and Hofer 1997; Admasu et al. 2004). The basic social unit is the breeding pair and some offspring from the previous year’s litter may remain with the parents and help to raise the current littler of pups ( Moehlman 1983, 1986, 1989). In Tanzania, C. aureus usually form long-term pair bonds which can last for up to 6 years. The bonded pair is territorial and both male and female mark within and on the boundaries of their territory and defend their territories against intruders. Same sex aggression is the norm with the territorial male attacking intruding males and females attacking intruding females. The pair hunt together, share food, and cooperatively rear their young ( Moehlman 1983, 1986, 1989). Moehlman and Hofer (1997) give average group size as 2.5 (Serengeti, Tanzania), similar to average group size (3; n = 7) in Velavadar N. P., India (Y. V. Jhala, in litt.).
Reproductive behavior. — Canis aureus has a strong pair bond which normally lasts for its lifetime ( Moehlman 1983, 1986, 1989). Mating involves a copulatory tie that lasts for several minutes (Golani and Mendelssohn 1971; Golani and Keller 1975). Pups are born in an underground den and normally emerge at about 3 weeks of age. Once pups emerge from the den they are fed solid food by adults regurgitating to them in addition to nursing. The origin of the dens can vary. Some authors (e.g., Rosevear 1974) report the use of existing aardvark Oryceropus afer or warthog Phacochoerus burrows, but in the Serengeti this is unlikely as C. aureus den openings are too small for either of these species (Jhala and Moehlman 2004). Earthen dens may have 1–3 openings and are typically about 2–3 m long and 0.5–1 m deep (Jhala and Moehlman 2004). Pups may be moved to different dens 2–4 times during their first 14 weeks of development ( Moehlman 1983, 1986, 1989).
In Tanzania, C. aureus typically produces litters once a year during the wet season (December to February) when the large migratory herds of wildebeest ( Connochaetes taurinus ), zebra ( Equus burchelli ), and Thomson’s gazelles are present on the short-grass plains ( Maddock 1979). Thus, when C. aureus has pups, food items ranging in size from dung beetles to Thomson’s gazelle fawns are abundant. C. aureus also scavenges at carcasses of wildebeest and zebra that are killed by larger predators, mainly spotted hyenas (van Lawick and van Lawick-Goodall 1970; Moehlman 1983, 1986).
In the Serengeti since mated pairs are monogamous with long-term bonds, sequential litters are full siblings as confirmed by genetic analysis of microsatellite DNA (P. D. Moehlman, in litt.). The male provisions the female while she is pregnant and both the male and the helpers (the previous years pups) provide the female with food while she is lactating ( Moehlman 1983, 1986, 1989; Moehlman and Hofer 1997). The presence of helpers correlates significantly with a higher pup survival (Macdonald and Moehlman 1983; Moehlman 1986, 1989).
Communication. —Territories are maintained indirectly by scent marking and vocalizations. Both members of a pair use a raised-leg urination posture to scent mark grass tufts, bushes, and trees at nose height. Urination with a raised rear leg instead of squatting is considered to be a ‘marking posture’ and indicates that the individual is a territory holder and on its territory. Marking, for example “raised-leg” urinations indicate that an individual is a territory holder and on its territory. When a territorial male or female leaves their area they do not “mark.” Both the boundary and the area within a territory are “marked.” When foraging with their mate, territorial individuals mark at twice the frequency as when they are foraging alone. When travelling together they mark the same spot in tandem ( Moehlman 1983, 1986). The presence of scent from both members of the pair may advertise to potential intruders that they are both actively in residence ( Moehlman 1983). Such scent marks are considered to play an important role in territorial defense ( Rosevear 1974).
Affiliative behaviors like greeting ceremonies, grooming, and group vocalizations are common in C. aureus social interactions (Van Lawick and van Lawick-Goodall 1970; Golani and Keller 1975). Vocalization consists of a complex howl repertoire beginning with 2–3 simple low-pitch howls and culminating in a high-pitched staccato of calls. In the Serengeti, individuals give reciprocal howls to locate their mates and family members. Families also occasionally group howl in response to neighboring family howls ( Moehlman 1983). Similar functions accrue to howling in Bangladesh, for territory maintenance and advertisement of dominance ( Jaeger et al. 1996). C. aureus has a “rumble growl” and “predator bark” when potential predators like spotted hyenas approach a den with pups. Pups respond to these vocalizations by running into the den ( Moehlman 1983).
Miscellaneous behavior. —In Kenya, Canis aureus was active during the day, as well as at dawn and dusk but not in the middle of the night ( Fuller et al. 1989). In Bangladesh, it preferred the cover of sugarcane during the day ( Jaeger et al. 2007).
GENETICS
The diploid chromosome number (2N) of Canis aureus is 78 (Wurster and Benirschke 1968). A total of 12–24 microsatellite loci from Israeli C. aureus generated expected levels of heterozygosity ( Cohen et al. 2013). In Serbian C. aureus , mitochondrial DNA has no variation and nuclear variability is low (Zachos et al. 2009). The mitochondrial DNA control regions are variable across C. aureus from Bulgaria, Serbia, Croatia (Dalmatia and Slavonia), and Italy and most genetically distinct in the population from Dalmatia ( Fabbri et al. 2014).
A melanistic individual occurred in Turkey (Ambarli and Bilgin 2012). Other melanistic and piebald forms are known (Muller-Using 1975; Jerdon 1984). Hybridization with domestic dogs may occur in Croatia ( Galov et al. 2015) but not in India (Yumnam et al. 2015).
Genetic data suggest that C. a. lupaster represents an ancient wolf lineage that most likely colonized Africa prior to the northern hemisphere radiation ( Rueness et al. 2011). A combined analysis of nuclear and mitochondrial DNA ( Bardeleben et al. 2005) indicates that C. aureus is in a wolf-like clade that includes Canis adustus , C. mesomelas , C. familiaris , C. lupus , C. latrans , Cuon alpinus , and Lycaon pictus . A phylogeny of living canids based on 22 nuclear and 3 mitochondrial loci produced a similar wolf-like clade ( Perini et al. 2010).
CONSERVATION
Canis aureus is fairly common throughout its range and is listed on the International Union for Conservation of Nature and Natural Resources Red List as “Least Concern” with at least 80,000 individuals on the Indian continent and an unknown number in Africa (Jhala and Moehlman 2008). This species is not listed in CITES. In the European Union, C. aureus is listed as an Annex V species and should be legally protected ( Rutkowski et al. 2015; Trouwborst et al. 2015). As the expansion of C. aureus is a natural long-range dispersal, it should not be considered an alien invasive species ( Rutkowski et al. 2015).
In the Serengeti–Ngorongoro ecosystem the population may be in decline ( Durant et al. 2011) but it is still at a relatively high density (0.5 individuals per km 2). In Greece, the population has been declining from at least 1975 ( Giannatos et al. 2005). In areas where C. aureus is perceived as a threat to livestock, it is often killed in indiscriminate predator control programs (Moehlman and Jhala 2013).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Canis aureus Linnaeus, 1758
| Moehlman, Patricia D. & Hayssen, Virginia 2018 |
Canis lupaster doederleini:
| Saleh and Basuony 2014: 49 |
Canis aureus qattarensis
| Saleh and Basuony 2014: 49 |
Thos aureus ecsedensis
| Kretzoi 1947: 287 |
Thos aureus algirensis:
| Allen 1939: 194 |
Thos aureus anthus:
| Allen 1939: 194 |
Thos aureus lupaster:
| Allen 1939: 194 |
Thos aureus maroccanus:
| Allen 1939: 195 |
Thos aureus soudanicus:
| Allen 1939: 195 |
Thos gallaensis:
| Allen 1939: 195 |
Thos lamperti:
| Allen 1939: 195 |
Canis aureus hungaricus Éhik, 1938:11
| Ehik 1938: 11 |
Canis aureus minor: Éhik, 1938:13
| Ehik 1938: 13 |
Thos lupaster maroccanus
| Cabrera 1921: 263 |
Canis indicus kola
| Wroughton 1916: 651 |
Canis naria
| Wroughton 1916: 651 |
Canis lanka
| Wroughton 1916: 652 |
naria
| Wroughton 1916 |
lanka
| Wroughton 1916 |
Thos aureus bea
| Heller 1914: 5 |
Canis somalicus
| Lorenz 1906: 306 |
Canis gallaënsis
| Lorenz 1906: 307 |
Canis doederleini
| Hilzheimer 1906: 116 |
Canis thooides
| Hilzheimer 1906: 364 |
Canis lupaster grayi
| Hilzheimer 1906: 367 |
Canis studeri
| Hilzheimer 1906: 368 |
lamperti
| Hilzheimer 1906: 371 |
gallaënsis
| Lorenz 1906 |
lamperti
| Hilzheimer 1906 |
somalicus
| Lorenz 1906 |
doederleini
| Hilzheimer 1906 |
thooides
| Hilzheimer 1906 |
Canis anthus soudanicus
| Thomas 1903: 295 |
Canis cruesemanni
| Matschie 1900: 145 |
cruesemanni
| Matschie 1900 |
Canis mengesi
| Noack 1897: 518 |
Canis lupus minor
| Mojsisovico 1897: 202 |
mengesi
| Noack 1897 |
mengesi
| Noack 1897 |
Canis hadramaticus
| Noack 1896: 356 |
Canis aureus balcanicus
| Brusina 1892: 317 |
Canis aureus typicus
| Kolenati 1858: 96 |
Canis aureus typicus var caucasica
| Kolenati 1858: 96 |
Canis aureus var syriaco
| Kolenati 1858: 96 |
Canis aureus var indica
| Kolenati 1858: 96 |
Canis aureus var nubica
| Kolenati 1858: 96 |
Canis aureus var algira
| Kolenati 1858: 96 |
Canis aureus var senegalensis:
| Kolenati 1858: 96 |
Canis aureus var barbarus:
| Kolenati 1858: 96 |
Canis graecus
| Wagner 1841: 383 |
Canis aureus algirensis
| Wagner 1841: 384 |
graecus
| Wagner 1841 |
Thous anthus:
| Hamilton Smith 1839: 195 |
Thous variegatus:
| Hamilton Smith 1839: 198 |
Thous sengalensi
| Hamilton Smith 1839: 201 |
Sacalius aureus:
| Hamilton Smith 1839: 214 |
Sacalius barbarus
| Hamilton Smith 1839: 218 |
Sacalius indicus:
| Hamilton Smith 1839: 219 |
Canis aureus var. moreotica
| I. Geoffroy Saint-Hilaire 1835 |
C. aureus moreotica
| I. Geoffroy Saint-Hilaire 1835 |
Canis lupaster
| Hemprich and Ehrenberg 1833 |
Canis sacer
| Hemprich and Ehrenberg 1833 |
Canis riparius
| Hemprich and Ehrenberg 1833 |
Canis aureus indicus
| Hodgson 1833: 237 |
indicus
| Hodgson 1833 |
indicus
| Hodgson 1833 |
lupaster
| Hemprich and Ehrenberg 1833 |
sacer
| Hemprich and Ehrenberg 1833 |
riparius
| Hemprich and Ehrenberg 1833 |
Canis syriacus
| Hemprich and Ehrenberg 1830 |
syriacus
| Hemprich and Ehrenberg 1830 |
syriacus
| Hemprich and Ehrenberg 1830 |
Canis spelaeus minor
| Wagner 1829 |
Canis variegatus
| Cretzschmar 1826: 31 |
Canis variegatus
| Cretzschmar 1826 |
variegatus
| Cretzschmar 1826 |
variegatus
| Cretzschmar 1826 |
Canis]
| F. Cuvier 1820 |
anthus
| F. Cuvier 1820 |
Canis barbarus
| Shaw 1800: 311 |
Canis aureus
| Linnaeus 1758: 40 |
aureus
| Linnaeus 1758: 40 |
Vulpes indiae orientalis
| Linnaeus 1758: 41 |
Canis aureus
| Linnaeus 1758 |
aureus
| Linnaeus 1758 |
aureus
| Linnaeus 1758 |
Canis aureus
| Linnaeus 1758 |
aureus
| Linnaeus 1758 |