Pinnotheres atrinicola, Roderic D. M. Page, 1983
|
publication ID |
https://doi.org/ 10.1080/03014223.1983.10423904 |
|
publication LSID |
lsid:zoobank.org:pub:84223D9F-7E5F-4AF3-87F5-10FF8426D814 |
|
DOI |
https://doi.org/10.5281/zenodo.5696926 |
|
persistent identifier |
https://treatment.plazi.org/id/A30440F2-1D7B-4D5B-B62C-656EA92FE48D |
|
taxon LSID |
lsid:zoobank.org:act:A30440F2-1D7B-4D5B-B62C-656EA92FE48D |
|
treatment provided by |
Plazi |
|
scientific name |
Pinnotheres atrinicola |
| status |
sp. nov. |
Pinnotheres atrinicola View in CoL n.sp. ( Fig. 21 View FIg.2 ,J, 3 View Fig.3 )
Pinnotheres novaezelandiae Filhol View in CoL . -*Chilton, 1911: 295 296. - Scott, 1961: 307 (part). -Bennett, 1964: 76-79 (part; not figs). - Takeda & Miyake, 1969: 18D-181.
Pinnotheres schauinslandi Lenz. -Bennett, 1964 View in CoL : 79-80 (part; not fig. 87 and 89-91).
*The original synonym is misspelt in this reference Pinnotheres View in CoL . Waite, 1909: 52.
Pinnotheres View in CoL "undescribed species". Gordon, 1936: 165.
Pinnotheres View in CoL sp. ("probably P. schauinslandi"). Wear, 1965: 16, 18.
Diagnosis. Hard-stage: chelae with a continuous dorsal row of setae on propodus; terminal segment of abdomen quadrate; male 1st pleopod slender, strongly curved in distal third, less setose than in P. novaezelandiae. Mature female: legs noticeably asymmetric; 2nd leg with dactylus subequal to carpus or longer, propodus longer than carpus. First 3 legs with long setae on carpus, propodus, and dactylus. First-stage zoe a larger than in P. novaezelandiae, and with a different chromatophore pattern.
Description. Similar to P. novaezelandiae, against which characters are compared.
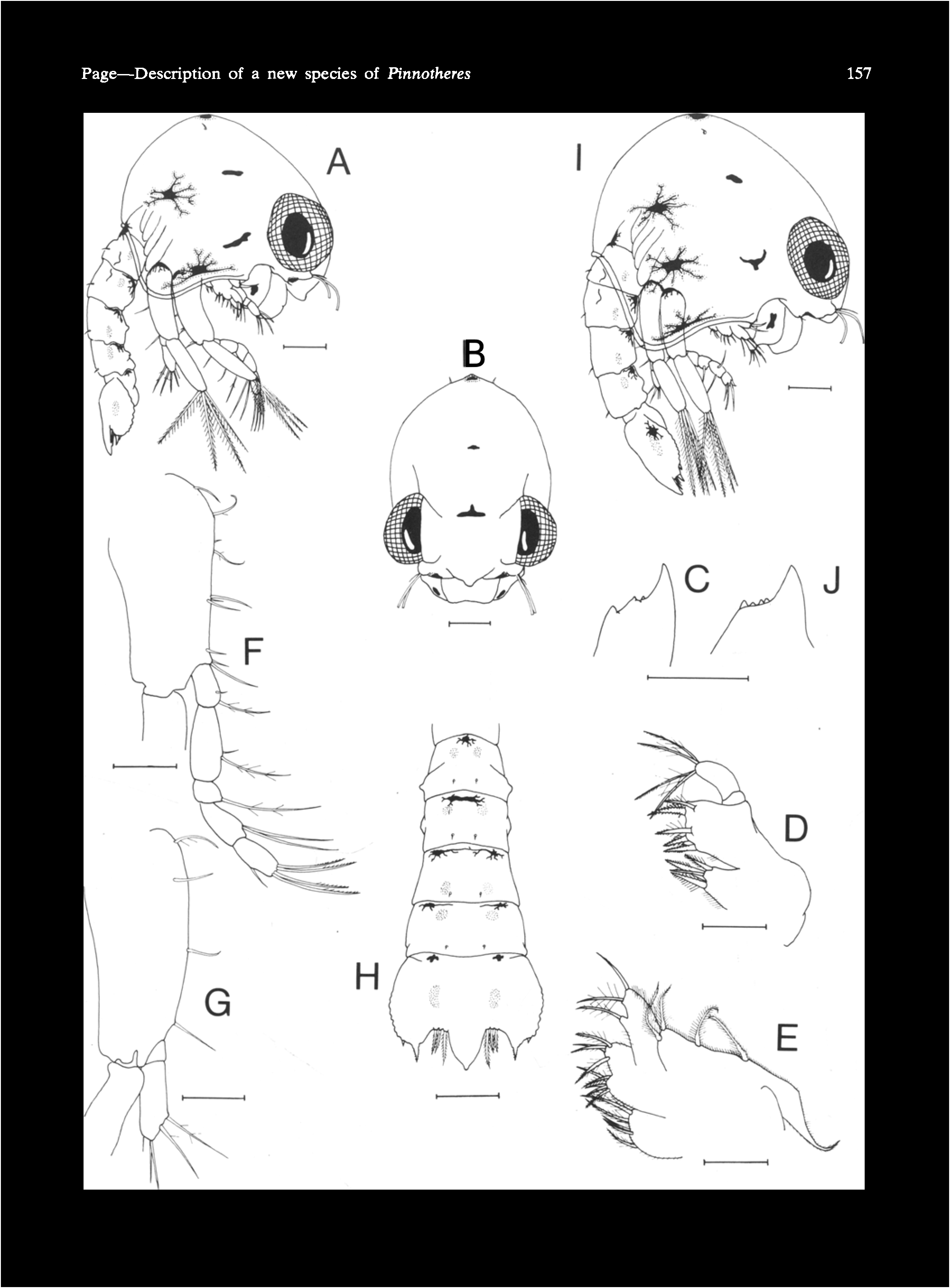
HARD-STAGE ( Fig. 3 View Fig.3 A-G). Carapace ( Fig. 3A View Fig.3 ) less dorsally inflated, less shouldered, 4.1--8.0 rom wide; front more protruding and convex.
Third maxilliped ( Fig. 3B View Fig.3 ) slightly narrower.
Chelae ( Fig. 3C,D View Fig.3 ) stouter, less inflated in male than female; row of setae on inner dorsal face of propodus extending from articulation with carpus to articulation with moving finger.
Legs with all setae usually more strongly developed and less reduced in large males.
Male abdomen ( Fig. 3E View Fig.3 ) with segments 1-3 broader, terminal segment quadrate. Female abdomen ( Fig. 3F View Fig.3 ) broad, convex ventrally, tapering strongly to quadrate terminal segment.
Male 1st pleopod ( Fig. 3G View Fig.3 ) less setose, slender, strongly curved laterally in distal third.
Colour pattern typically with a large, orange marking along midline of posterior half of carapace.
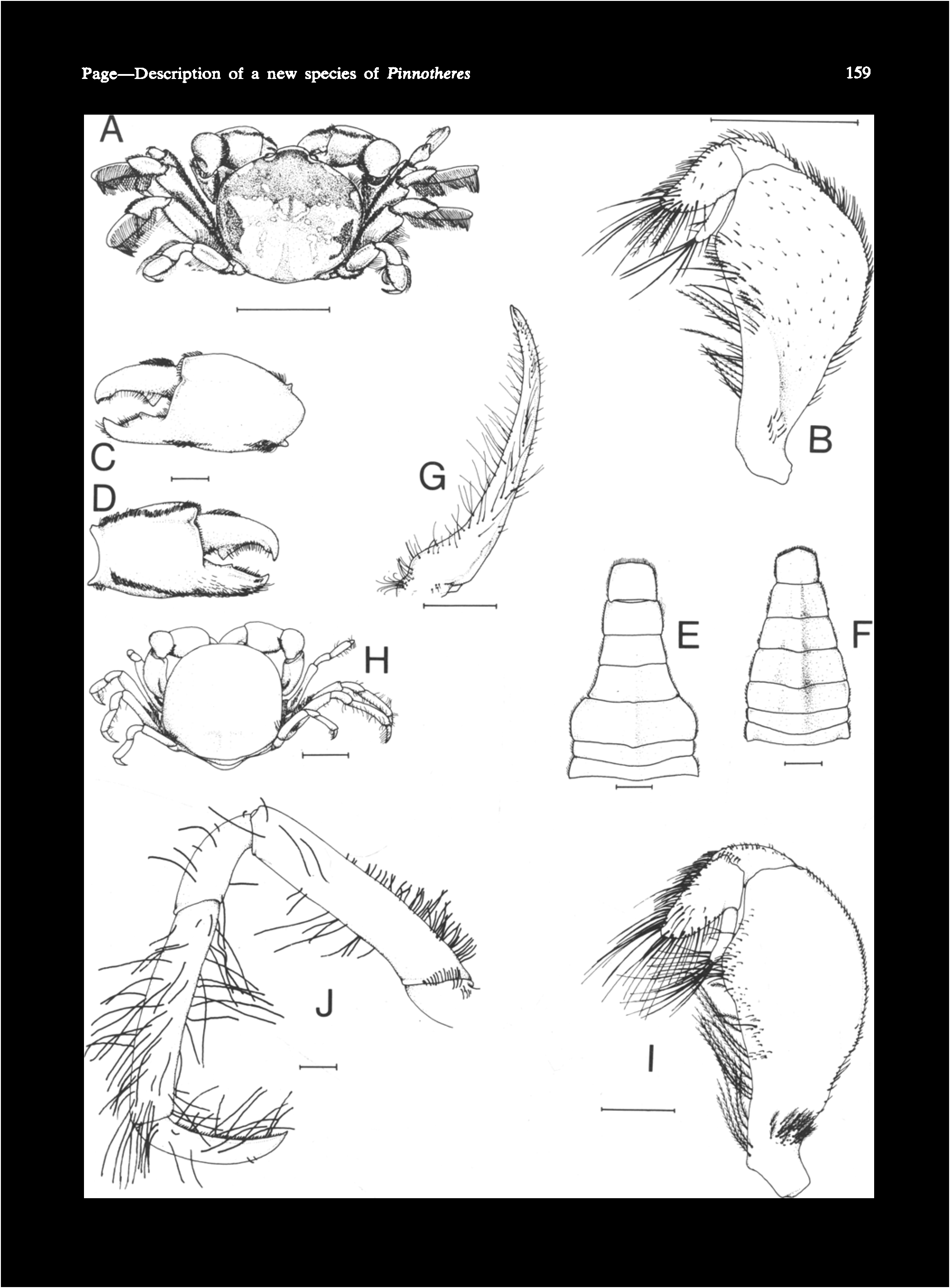
MATURE FEMALE ( Fig. 3 View Fig.3 H-J). Carapace ( Fig. 3H View Fig.3 ) more laterally inflated and more rounded, less arched longitudinally, less convex dorsally, 10.0 19.9 rom wide.
Third maxilliped (Fig. 31) slightly narrower than in hard-stage,
Cheliped merus with dorsal setae reduced or absent, ventral row absent; chelae stout, inflated.
Legs slender, subcylindrical, the 2nd longest, the 3rd subequal in length to it, the 4th shortest. Second leg ( Fig. 3J View Fig.3 ) with propodus longer than carpus (ratio 1:1.48 ± 0.12 SD; n = 24), dactylus subequal to carpus or longer. Legs noticeably asymmetric (ratio of sum of lengths of last 3 segments of 2nd leg, shortest to longest, 1:1.7 ± 0.07 SD; n = 22); legs on 'shorter' side less robust and setose than those on 'longer' side. Merus with dorsal row of setae extending only halfway along margin. First 3 legs with scattered setae on lower half of merus and carpus, long setae on carpus, propodus, and dactylus. Last leg with scattered setae on dactylus and distal ventral margin of propodus.
Abdomen as in P. novaezelandiae.
Usually unpigmented.
FIRST-STAGE ZOEA (Fig. 21) larger - mean carapace length 0.56 mm (range 0.53-0.63 mm), mean width 0.39 mm (range 0.37-0.43 mm). Rostrum usually shorter.
Mandible with incisor process variable, usually bearing 1 major tooth and 4 smaller accessory teeth ( Fig. 2J View FIg.2 ).
Chromatophore pattern (Fig. 21; Table 1 View Table 1 ): lateral carapacial chromatophore with 2, sometimes 3 centres; abdomen with paired black and yellow chromatophores; black subintestinal chromatophores of telson immediately ventral to yellow lateral intestinal chromatophores.
Type data. Holotype'; (hard-stage): NEW ZEALAND, Whangarei Harbour between High Island and mainland , 0-1 m, from Atrina zelandica , 13 May 1982, B. Dobson, G. Miles, C. Turbott, and C. Worthington ( National Museum of New Zealand, Cr. 3021 ). Paratypes (National Museum of New Zealand). Bay of Plenty, B. L. Godfriaux, 3 S?, 7'; (sample no. 39A; Cr. 2529). B.S. 488, 40 009.5'S, 174°36'E, c. 18 miles S of Waitotara R. mouth, in 82 m, 2 Mar 1976, LV. Acheron , 1 S? (Cr. 2521). Evans Bay powerhouse intake, 13 Oct 1954, R. K. Dell, 1 S? ( Pinnotheres novaezelandiae det. M. Scott, 1959; CL 952). Lyall Bay, Wellington, from A. zelandica, Sep 1949, R. K. Dell, 1 S? (P. novaezelandiae det. M. Scott, 1959; Cr. 955). B.S. 528, 40 036.5'S, 173°oo.5'E, off shelf flats inside Farewell Spit (Tasman Bank), in 24-26 m, 9 Mar 1976, r.v. Acheron , 2 <;? (CL 2519, 2520). B.S. 527, 40 037'S, 172°48'E, c. 5 miles off Pakawau Beach, Golden Bay, in 24 m, 9 Mar 1976, r.v, Acheron , 19<;? (P. novaezelandiae det. G. R. F. Hicks, in A. zelandica; Cr. 2524). 40OJ3'S, 173OZ7'E, in 73 m, from Modiolus areolatus, 10 Apr 1964, r.v. Constantia , 1 <;? (pres. N.Z. Marine Department; Cr. 2528). B.S. 431, Orchard Bay, Marlborough Sounds, from head to entrance, 16 fm, 30 Aug 1975, LV. Acheron , 1 <;? (Cr. 2522). B.S. 515 (41OOO.5'S, 174°oo'E), W side of Forsyth Bay, Marlborough Sounds, in 9-18 m, 5 Mar 1976, r.v, Acheron , 1 <;?, 3gS? (Cr. 2518). Off Cape Campbell, 40 fm, 5 Dec 1956, F. Abernethy, 1 <;? (P. novaezelandiae det. M. Scott, 1959; Cr. 956). Off Cape Campbell, 40 fm, from A. zelandica, Mar 1957, F. Abernethy, 1 <;? (P.
novaezelandiae det. M. Scott, 1959; Z. Cr. 687). Off Otago Harbour, from A. zelandica, 9 Mar 1952, vessel Taiaroa, J. C. Yaldwyn , IgS? (Cr. 941).
Other material examined. AUTHOR'S PERSONAL COLLEC TION. Whangarei Harbour: same data as holotype, from 86 A. zelandica, 48 2, 28g 2, 50; MacDonald Bank, from A. zelandica, 10 May 1982, 52. Ngataringa Bay, Waitemata Harbour, from A. zelandica, 12 Dec 1981, 42, 2g2, 14 Dec 1981, 112, 4g2, 20, 13 Jan 1982, 92, 2g2, 27 Feb 1982,12,lg2.
UNIVERSITY OF AUCKLAND DEPARTMENT OF ZOOLOGY. Okahu Bay, Waitemata Harbour, from Chione stutchburyi, 15 Aug 1981, A. Grimm, 10.
AUCKLAND INSTITUTE AND MUSEUM. Ruakaka, Marsden Pt, 2 Jan 1969, W. Farley, 12 (AIM. 3929).
J. B. JONES PERSONAL COLLECTION. Seatoun beach, from A. zelandica, 1 2.
CANTERBURY MUSEUM. Takapuna Beach, from A. zelandica, 29 Sep 1914, 22 (P. novaezelandiae del. E. W. Bennett, 18 Nov 1930; AQ 2369). Cheltenham Beach, from A. zelandica (ex Chilton Coli., no. 489), 22 (P. schauinslandi det. E. W. Bennett, 18 Nov 1930), 10 (P. novaezelandiae det. E. W. Bennett, 18 Nov 1930; AQ 2383). Auckland (dredged), Capt. Bollons, 22 (P. novaezelandiae det. E. W. Bennett, 18 Nov 1930; AQ 2396). OffLyttelton heads, trawled sand-mud, 25-28 fm, 7 Mar 1967, vessel Golden Light, Mr Smith, 12 (AQ 2066). Nora Niven Expdn 1907, Stations 20 and 44, 52 (AQ 2242).
Distribution. North and South islands. Intertidal to 200 m. Endemic.
Hosts. The principal host is Atrina zelandica . There are single records of hard-stages from Modiolus areolatus (Gould) and Chione stutchburyi. Waite (1909, p. 52) found that "almost every adult Pinna [= Atrina ] taken had its crustacean commensual, Pinnotheres ", as did Stead (1971): in a sample of 87 A. zelandica taken from Whangarei Harbour on 13 May 1982, 83 (95.4%) contained a specimen of P. atrinicola .
Remarks. Differences between P. novaezelandiae and Pinnotheres from Atrina zelandica were first noted by Bennett (1964, p. 76), who observed that mature females of "P. novaezelandiae" from A. zelandica were usually larger and had relatively longer legs. Wear (1965) reported zoea larvae of 2 species of Pinnotheres in Wellington Harbour plankton: one he could not distinguish from P. novaezelandiae as described by Bennett (1964, p. 78-79, fig. 92 and 93); the other, he suggested, was probably P. schauinslandi, "as this is the only other adult of the genus Pinnotheres recorded from New Zealand (Bennett, 1964)". Apart from figuring the telson of P. novaezelandiae, Wear did not describe his specimens. Jones (1977) has confirmed that they belong to different species.
Using scanning electron microscopy, Jones (1975) compared first-stage zoeae of P. novaezelandiae and Pinnotheres from A. zelandica, noting small differences in the morphology of the rostrum, labrum, and mandibles which led him to suggest that the latter was a different species (Jones 1975, 1978).
P. atrinicola is obviously very similar morphologically to P. novaezelandiae. The latter is more common in mytilids, especially Perna canaliculus, while with 2 exceptions P. atrinicola is known only from A. zelandica. Chione stutchburyi is the only host the 2 species have in common. This relative lack of overlap in hosts raises the possibility that the crabs I have recognised here as distinct species are in reality morphs of a single, polymorphic species in which post-planktonic morphology is determined by the identity of the host occupied. However, the morphology of both species is relatively constant, and both P. novaezelandiae and P. atrinicola show no patent morphological changes when found in bivalves other than their respective principal hosts, Perna canaliculus and Atrina zelandica. Further work, such as an electrophoretic study of allele frequencies in pea crabs from these 2 hosts, would help remove any remaining doubt about the status of P. atrinicola .
The specific epithet is constructed from Atrina , genus of the type host, and -cola (L.), a suffix denoting one who inhabits.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |