Channa pseudomarulius, Günther, 1861
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4299.4.4 |
|
publication LSID |
lsid:zoobank.org:pub:3437A45D-7D3E-4B21-BBCB-BD0B5EDAD44E |
|
DOI |
https://doi.org/10.5281/zenodo.6045575 |
|
persistent identifier |
https://treatment.plazi.org/id/03C27A4F-8861-FF96-FF04-CCDAFC83FEA9 |
|
treatment provided by |
Plazi |
|
scientific name |
Channa pseudomarulius |
| status |
|
Channa pseudomarulius View in CoL
( Figures 2 View FIGURE 2 , 3 View FIGURE 3 )
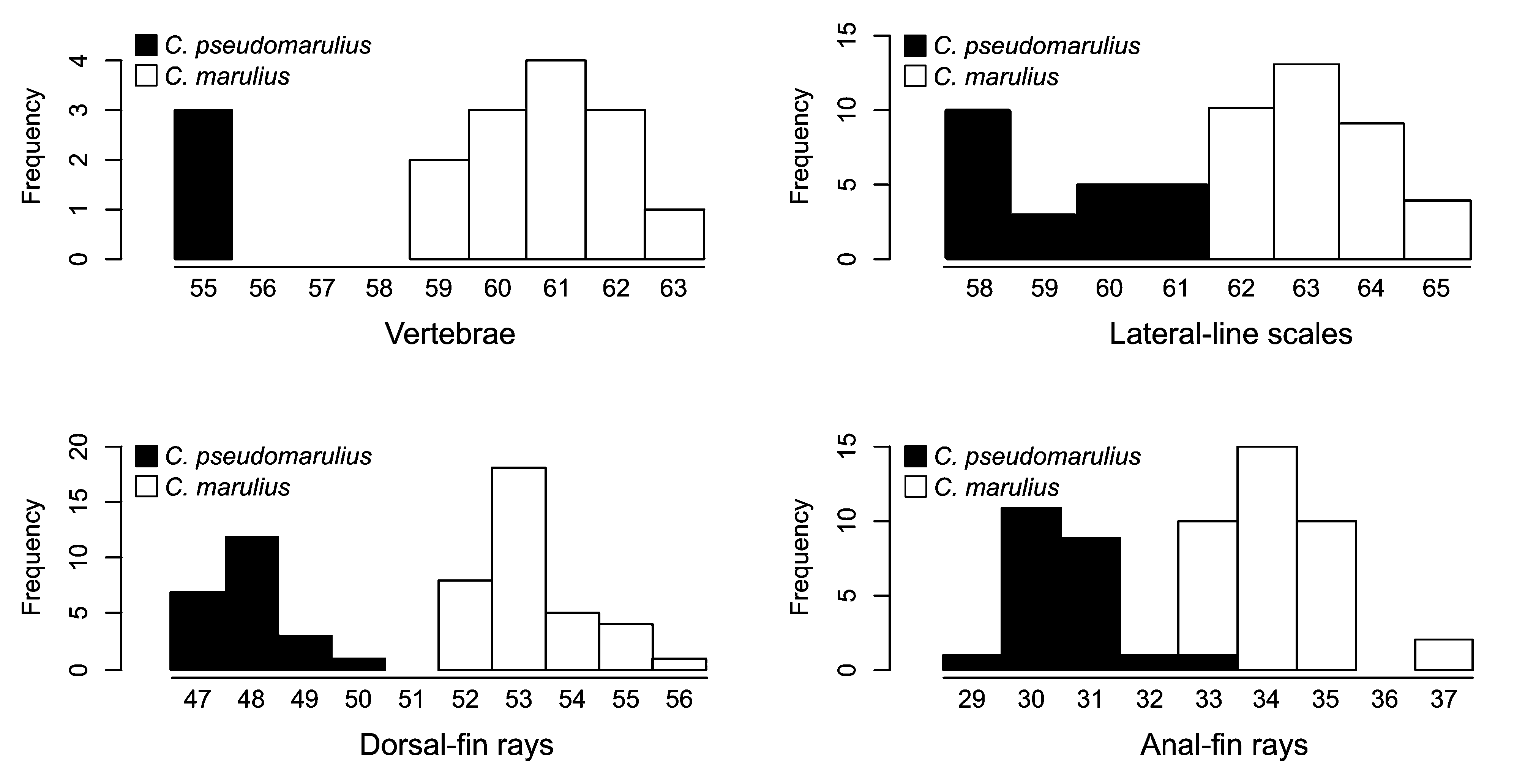
Diagnosis. Channa pseudomarulius can be distinguished from C. marulius by fewer lateral line scales (59–61 vs 63–65), fewer dorsal- (47–50 vs 52–56) and anal-fin (29–33 vs 33–37) rays and fewer vertebrae (55 vs 59–63) ( Table 2, Fig. 7 View FIGURE 7 ), and by a shorter body/longer head especially in specimens over 200 mm SL (see below).
Description. For general appearance, see Figures 2 View FIGURE 2 and 3 View FIGURE 3 , for morphometric information Table 1 and for meristic data Table 2. Dorsal profile rising in straight line from tip of snout to anterior base of dorsal fin, continuing in horizontal line to middle of dorsal fin, sloping ventrally from there to posterior dorsal-fin base. Ventral profile falling in straight line to vertical through base of pectoral fin, continuing in horizontal line parallel to dorsal profile up to posterior third of anal fin, rising from there to caudal-fin base. Dorsal fin with long base, 2/3 of SL, originating at vertical through pelvic-fin base and extending to vertical through posterior base of anal fin. Anal fin long, 2/5 of SL, originating behind vertical through distal tip of adpressed pectoral fin and ending at vertical through posterior dorsal-fin base. Head length about 1/3 of SL (Fig. 2,3). Eye located in anterior 1/3 of head. Lips prominent; mouth large, with posterior angle of gape behind vertical through posterior rim of orbit. Upper jaw with several rows of villiform teeth along its length, few canines near symphysis. Lower jaw with several rows of small recurved teeth at front, continuing posteriorly as single row, with several prominent canines interspersed. Vomer and palatine with several rows of small, recurved, conical teeth.
Lateral-line scales 58–61 in total; 15–18 scales pre-drop, 2–4 forming drop, and 39–42 scales postdrop. Dorsal-fin rays 47–50. Anal-fin rays 29–33. Pectoral fin with 16–18 rays almost reaching vertical through vent when adpressed. Pelvic-fin rays 6. Caudal fin rounded, with 14 principal rays.
Colouration. In preservative ( Fig. 2 View FIGURE 2 ), small juveniles of 50–60 mm SL with dorsum dark brown from snout to end of dorsal-fin base, followed ventrally by conspicuous light-brown longitudinal band extending from eye to base of caudal fin, limited ventrally by slightly wider, dark-brown to black mid-lateral band; ventral area of head and body greyish white. Fins cream, with ventral half of caudal fin darker at base; caudal-fin ocellus absent. Juveniles of about 100 mm SL with dark brown colour on top and sides of head ventrally to gular area ( Fig. 2 View FIGURE 2 B). Ventral side of head and throat whitish cream. Body dorsally dark brown, set off from midlateral series of dark brown blotches by narrow lighter line (a remnant of the small juvenile light stripe) running just below anterior part of lateral line before lateral-line drop and above it after drop. Series of up to seven large dark brown blotches midlaterally (a remnant of the dark brown longitudinal stripe of smaller juveniles) alternating with lighter narrow bars forming as dorsal extensions from lighter ventral side of abdominal and caudal areas of body. Pectoral and pelvic fins cream. Dorsal-fin membrane brownish with pattern of white, sometimes connected, lines. Anal fin whitish cream with few irregular grey spots in posterior part of fin. Caudal fin with dark brown blotch at base, a dark blotch with conspicuous white rim forming ocellus on dorsal part of fin and four cream to white vertical lines separating darker stripes from each other.
In adults of around 30 cm, dorsal and lateral area of head and body dark brown reaching ventrally to about level of lateral line ( Fig. 2 View FIGURE 2 C). Lower third of head yellowish beige, body ventral to lateral line light yellowish brown, surrounding series of four or five dark brown to black blotches. Blotches extending dorsally to or just beyond lateral line. Pectoral, dorsal, caudal, and anal fins brown, but pectoral fin with dark brown vertical mark at base. Pelvic fin yellowish beige. Caudal-fin ocellus indistinct.
In life ( Fig. 3 View FIGURE 3 ), Small juveniles of 50–60 mm SL with bright yellowish orange longitudinal band from eye (including iris) to caudal-fin base. Cheek and side of abdomen with golden iridescence. Middle and lower half of body with dark brown longitudinal band originating on opercle and extending onto base of caudal fin. Otherwise fin membranes hyaline.
Larger juveniles of ca. 80 mm SL light brown on top and sides with darker marbling dorsally from head to caudal peduncle ( Fig. 3 View FIGURE 3 A). Light stripe running from eye to end of caudal peduncle separating darker dorsal side from series of light brown mid-lateral blotches. Blotches less conspicuous anteriorly in series, separated from each other by orange golden blotches. Ventral side of head and body whitish cream. Pectoral and pelvic fins hyaline. Dorsal- fin membrane with irregular pattern of spots often rimmed by lighter yellowish lines, otherwise hyaline. Anal fin hyaline. Caudal with dark brown base and very conspicuous ocellus on dorsal part of fin formed by large black spherical spot rimmed by narrow yellow line. Additional dorsoventrally running oblique yellowish lines on otherwise brown caudal fin.
Subadults (164 mm SL) similar to juveniles in colouration of dorsal side of head and body, but lateral series of dark brown mid-lateral blotches more conspicuous and separated by bright orange blotches ( Fig. 3 View FIGURE 3 B). Mid-lateral dark blotches also with numerous white spots, more numerous in lower half of blotch, but also present above blotches in posterior dorsal half of caudal area. Pectoral and pelvic fins cream hyaline. Dorsal fin with series of dark spots at base and numerous white spots arranged in roughly three irregular longitudinal lines. Anal fin cream with series of white spots arranged in lines. Caudal fin brownish with several irregular vertical lines of white markings. Ocellus less conspicuous than in juveniles and appearing slightly faded.
TABLE]. Morphometric information for Channa pseudomarulius (n=11), C. marulius , the lectotype of Ophiocephalus grandinosus (MNHN A1959) anđ the holotype of O. theophrasti (MNHN A 668).
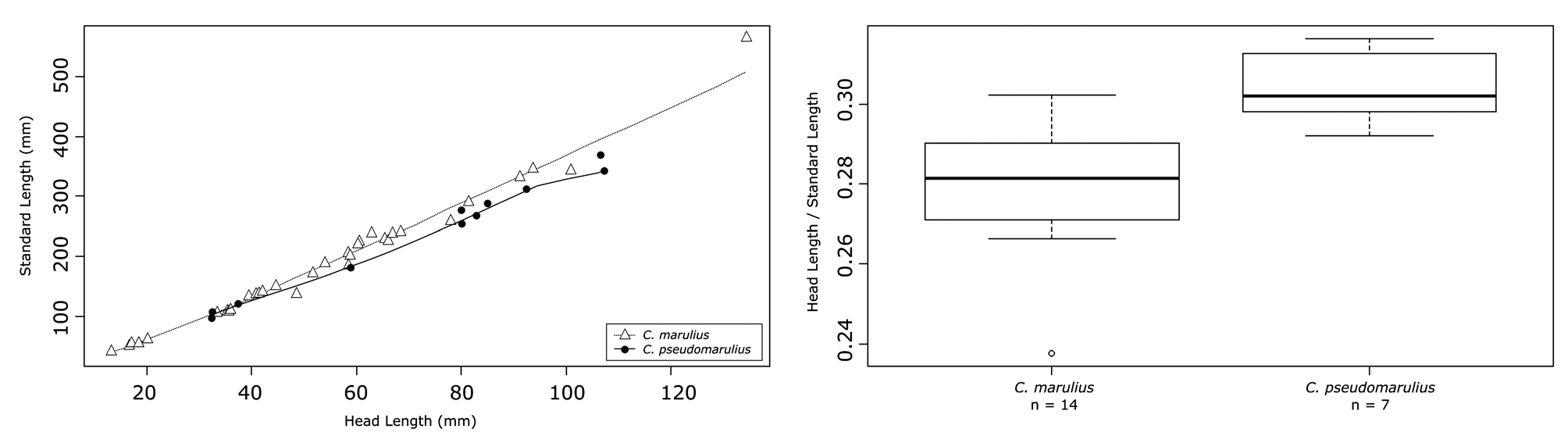
Comparative morphometry. In general appearance, Channa pseudomarulius is shorter bodied than C. marulius . This is reflected by comparatively lower number of vertebrae and lateral-line scales ( Fig 7 View FIGURE 7 .). Channa pseudomarulius also has fewer dorsal- and anal-fin rays, although the holotype of C. pseudomarulius specimen has 33 anal rays, overlapping with the lowest counts for C. marulius specimens ( Fig 7 View FIGURE 7 .). This overall shorter body length means that its head appears comparatively “larger”, as can be seen in Fig. 8 View FIGURE 8 , this difference expressed as the ratio of HL/SL is significant among larger specimens (> 200mm) (t = -4.482, df = 18.102, p = <0.0003), although not significant when available data from smaller fishes are included (t = -1.6489, df = 35.122, p = 0.1081).
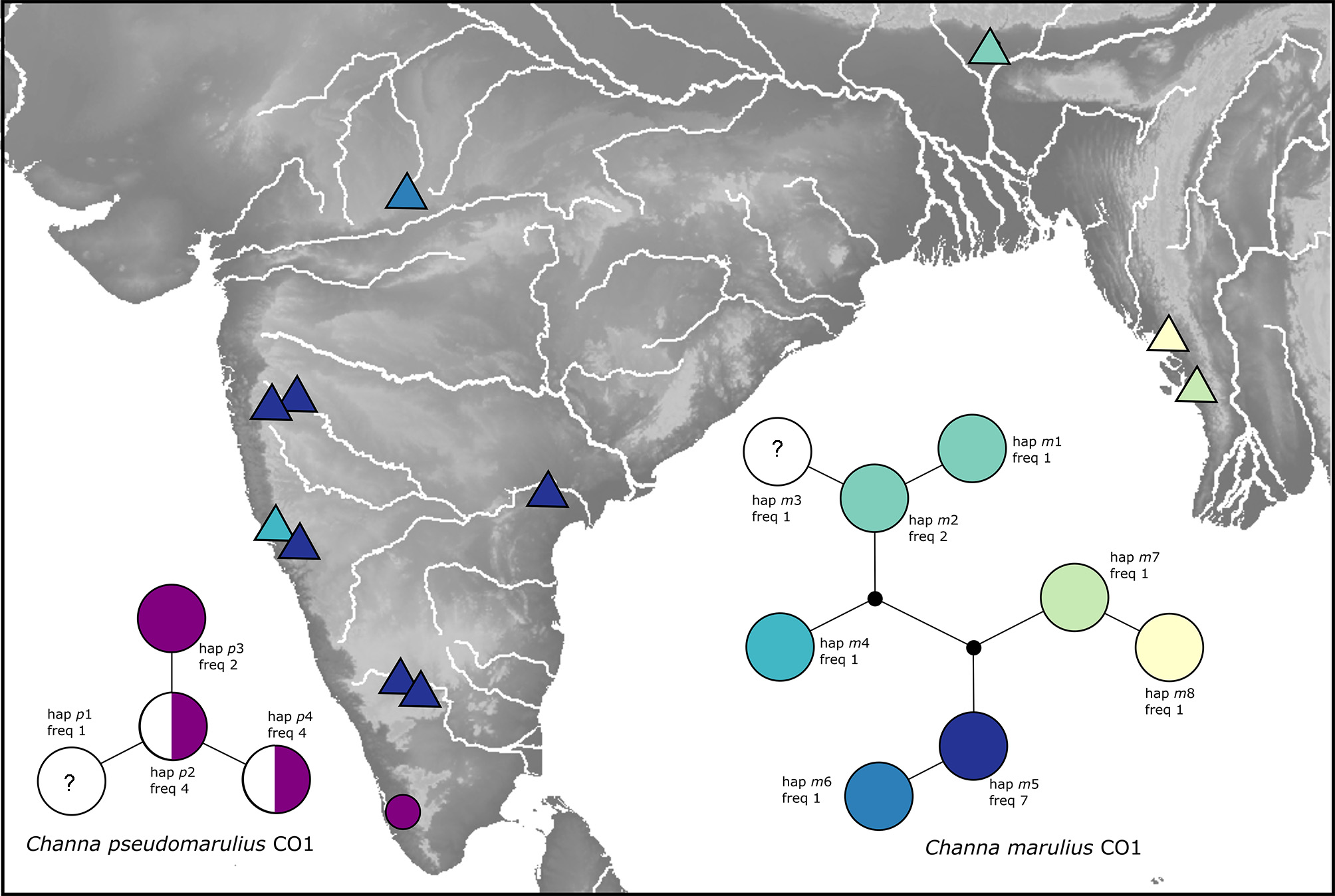
Molecular results. CO1 sequence data revealed two genetic groups; 1) Channa pseudomarulius (n = 11, 4 haplotypes p 1– p 4) collected exclusively in south west India, and; 2) C. marulius (n =15, 8 haplotypes m 1– m 8) collected throughout central and north east India and Western Myanmar (see Fig. 4 View FIGURE 4 ). Within each group genetic divergence was low (maximum distances of 0.003 and 0.008 for C. pseudomarulius and C. marulius respectively), while divergence between groups was an order of magnitude higher (net divergence 0.082, mean divergence 0.084), equating to a minimum of 8.1% uncorrected divergence (50 mutations). Nucleotide diversity was generally low (π = 1.09, 2.34 for C. pseudomarulius and C. marulius respectively) and Fu’s F S returned non-significant results when each species was analysed as a single ‘population’ (p values>0.05) providing no evidence for genetic signatures of demographic changes in recent evolutionary time.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |