Cypraea robertsi, Hidalgo, 1906
|
publication ID |
https://doi.org/ 10.1046/j.1096-3642.2003.00058.x |
|
persistent identifier |
https://treatment.plazi.org/id/03B6B923-EE2E-FFF8-8CD9-64CD3F7B6BE3 |
|
treatment provided by |
Carolina |
|
scientific name |
Cypraea robertsi |
| status |
|
CYPRAEA ROBERTSI View in CoL HIDALGO 1906
Material examined
Playa Bique, Panama ( USNM 890940).
External anatomy and mantle cavity
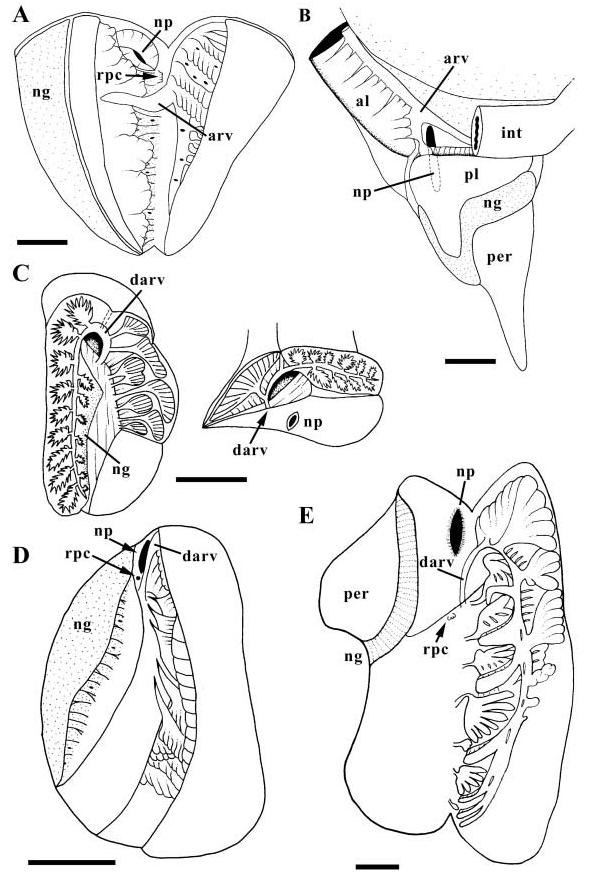
Anterior pedal gland opening to deep propodial groove. Mantle surface pustulose with conical papillae; short siphonal fringe. Anterior kidney chamber extending into pallial roof. Osphradium triradiate. Hypobranchial gland extensive, comprising large, pendulous folds. Operculum absent.
Reproductive system
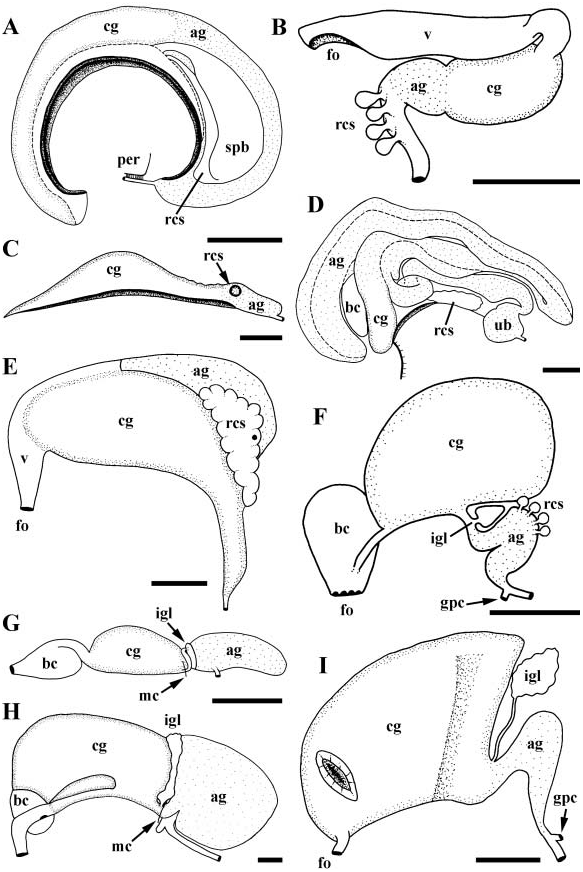
Gonad occupying terminal whorls, anteriorly bounding digestive gland and midgut on right. Gonopericardial canal absent. Vestigial gonopericardial canal present, forming small, blind pericardial diverticulum. Oviduct entering capsule gland near base of mantle cavity. Capsule gland ( Fig. 2E View Figure 2 , cg) long and crescentic, narrow posteriorly, expanding anteriorly. Albumen gland (dg) lying along dorsal margin of capsule gland, opening to capsule gland anteriorly via narrow duct. Large, lobate seminal receptacle (rcs) opening to blind posterior end of albumen gland. Finely grooved tract extending anteriorly from aperture of albumen gland, expanding at distal end of capsule gland, continuous with narrow vagina (v). Vagina terminating at small female opening (fo).
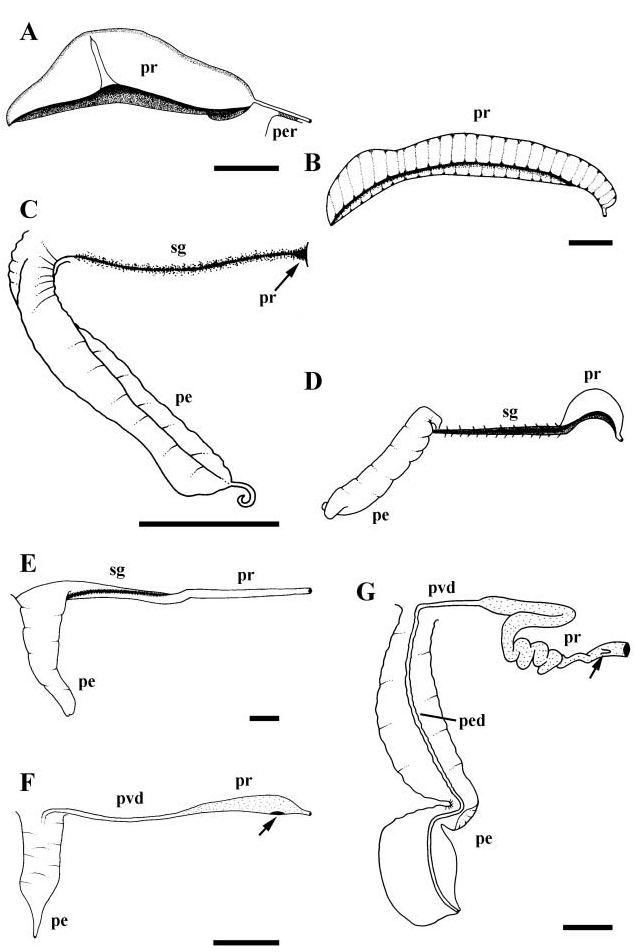
Vas deferens forming large and complexly coiled seminal vesicle. Seminal vesicle narrowing anteriorly and opening to closed, narrow, glandular, basophilic prostate at base of mantle cavity ( Fig. 3E View Figure 3 , pr). Vas deferens emerging from pallial roof, opening to large pocket and open seminal groove lined with acidophilic cells. Seminal groove extending across neck and along ventral surface of long, simple, narrowly triangular penis (pe).
Alimentary system
Foregut. Radula taenioglossate. Foregut modified into muscular, acrembolic proboscis ( Fig. 6C View Figure 6 , ps). Jaw absent; buccal cavity uncuticularized. Subradular membrane completely covering ventral surface of odontophore; subradular organ absent ( Fig. 4G View Figure 4 ). Short mid-ventral fold ( Fig. 8G View Figure 8 ) present forming dorsal lip of radular sac. Paired muscular, non-glandular pouches present opening to anterior oesophagus. Shallow ridge running transversely across floor of oesophagus between pouches; ventro-lateral folds absent. Salivary gland ducts passing through broad nerve ring, under long pedal connectives. Salivary gland massive, filling left side of cephalic haemocoel and overlying oesophageal gland. Radular sac long and coiled. Midoesophagus with well-developed, septate oesophageal gland and open dorsal food groove.
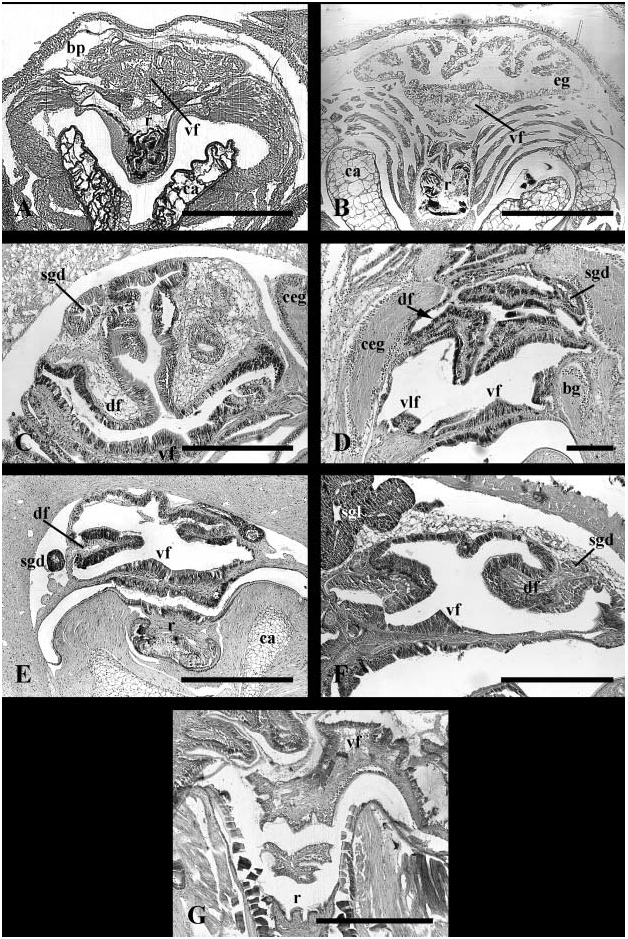
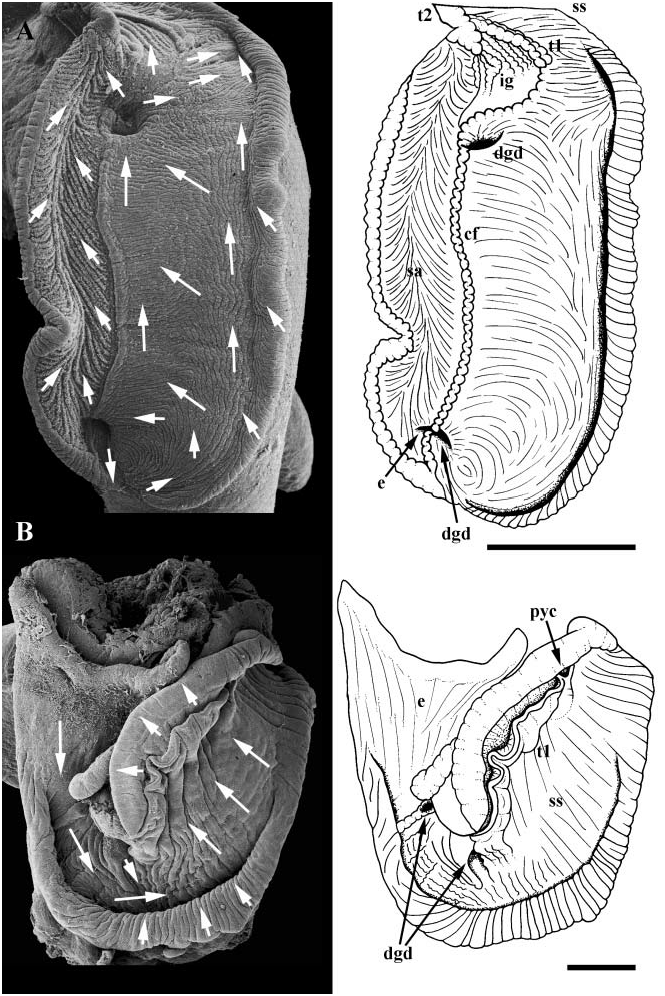
Midgut. Oesophagus opening to left limb of large, Ushaped midgut ( Fig. 16B, e View Figure 16 ). Gastric shield absent. One digestive gland duct (dgd) opening near oesophagus, draining small posterior lobe of digestive gland. Second digestive gland duct draining large anterior lobe of digestive gland, forming caecum. Broad mouth of caecum opening ventrally, along anterior wall of midgut. Anterior extension of major typhlosole (t1) bounding caecum on right. Style sac region characterized by single (major) typhlosole and transversely folded epithelium bearing uniform cilia; minor typhlosole absent. Ciliated strip on major typhlosole absent. Ciliary currents flowing clockwise within style sac region. Major typhlosole turning to right at distal end of style sac. Transverse lip of major typhlosole bounding deep pyloric caecum (pyc), extending posteriorly under style sac. Minor typhlosole beginning within pyloric caecum, paralleling major typhlosole straight out of caecum. Posterior digestive gland lobe occupying terminal visceral whorls; anterior lobe extending into pallial cavity.
Hindgut. Major and minor typhlosoles continuing only short distance within proximal intestine. Intestine straight.
Reno-pericardial system
Kidney large, differentiated into posterior and anterior, histologically distinct lobes ( Fig. 23B View Figure 23 ). Posterior lobe (pl) overlying visceral kidney lumen. Anterior lobe (al) long and narrow, extending within pallial roof under intestine (int), along digestive gland and pallial gonoduct. Nephridial gland present (ng). Afferent renal vessel entering kidney posteriorly, extending anteriorly along floor, producing numerous small branches supplying lamellae along ventral surface of digestive gland. Afferent renal vessel splitting near nephropore (np) into two large branches of equal size. Ventral branch supplying anterior lobe of kidney. Dorsal branch supplying anterior and posterior lobes.
Nervous system and sensory structures
Nervous system epiathroid, left zygoneurous, possibly right dialyneurous ( Fig. 24D View Figure 24 ). Circum-oesophageal nerve ring ( Fig. 6C View Figure 6 , nr) lying obliquely within cephalic haemocoel, with concentrated cerebro-pleural complex near left cephalic tentacle. Buccal ganglia present just anterior to buccal pouches (bp). Tentacular nerve (tn) dividing into nerve plexus upon entering tentacle. Long pedal connectives passing over anterior oesophagus. Pedal ganglia ( Fig. 24D View Figure 24 , pdg) large with pedal cords (pc) forming numerous cross connections. Supraoesophageal (sp) connective very short; suboesophageal (sb) connective long. Single large visceral ganglion ( Fig. 6C View Figure 6 , vg) lying within cephalopedal sinus. Four smaller accessory visceral ganglia (vg) present straddling posterior oesophagus and along left side of cephalic haemocoel. Statocysts with single statolith ( Fig. 24D View Figure 24 , sc) present on dorsal surface of pedal ganglia.
Discussion
The foregut of cypraeoideans is modified to form a muscular, acrembolic proboscis ( Vayssière, 1923; Kay, 1960). The position, number and glandular development of pouches within the anterior oesophagus is variable. They may be paired ( Kay, 1960; present study) or unpaired and thin-walled or glandular ( Amaudrut, 1898; Vayssière, 1923; Thiele, 1929; Rau, 1934; Fretter, 1951; Fretter & Graham, 1962). Such unpaired mucus diverticulae have been described as opening to the oesophagus at the origin of the oesophageal gland, slightly posterior to that described here. It seems likely that these glandular and nonglandular diverticulae are homologues (cf. Kay, 1960) despite slight differences in position within the anterior oesophagus. In fact, given that twisting of the anterior oesophagus brings the left pouch into a hidden ventral position, it suggests that many of the described single diverticulae may be paired with asymmetrical development of glandular tissue.
Cypraeoideans display an extraordinary conservatism of midgut morphologies. The structure and development of such features as the digestive caecum, the major typhlosole, and the intestinal groove are remarkably similar in many species ( Haller, 1890; Rau, 1934; Kay, 1960). Cypraea robertsi differs in that it lacks both the gastric shield and the ciliated strip on the major typhlosole, which might otherwise confound attempts to interpret midgut homologies. However, the ovulid Jenneria pustulata has a midgut that is similar in overall organization, and also possesses both the shield and the ciliated strip (pers. obs.). Owing to this basic conservatism, midgut morphology of J. pustulata permits an interpretation of cypraeoidean midgut homologies and a reinterpretation of the hypotheses offered by Kay (1960), even in those taxa lacking structures such as the gastric shield.
As stated above, Jenneria pustulata possesses a gastric shield and a ciliated strip on the major typhlosole. The gastric shield and a prominent horizontal fold distal to the shield (= lip of style sac) in J. pustulata are similar to style-bearing caenogastropods in their position and connection to other landmarks. This suggests that the portion of the midgut along the major typhlosole referred to as the ‘cuticular area’ by Kay (1960), represents the style sac region of other caenogastropods. Kay suggested instead that the region of the gut distal to the transverse segment of the major typhlosole is comparable to the style sac region. This reinterpretation indicates that the transverse segment of the major typhlosole lies at the distal end of the style sac, not at the proximal end as suggested by Kay, confirming the identification of a pyloric caecum at the distal end of the style sac in C. robertsi . Kay did not describe a pyloric caecum under the transverse segment of the major typhlosole. However, a depression appears to be visible in the appropriate position. Cypraea robertsi is unique in lacking a minor typhlosole in the style sac region.
As in naticids, the excretory tissue of cypraeids is subdivided into two lobes that are histologically and macroscopically distinct ( Haller, 1890; Simroth, 1896 – 1907). Haller’s detailed description of the kidney in C. testudinaria also clearly documented the branching of the afferent renal vessel to supply the anterior and posterior lobes. Rau (1934) provided a largely erroneous interpretation of the reno-pericardial system of Cypraea arabica , referring to the anterior kidney lobe as the right kidney, complete with a rudimentary nephropore near the anus.
Cypraeoidean nervous systems are displaced to the left of the buccal mass, with highly concentrated cerebral and pleural ganglia; the supra-oesophageal and left pleural ganglia are in close proximity, and the suboesophageal ganglion is widely separated from the others ( Haller, 1890; Amaudrut, 1898; Vayssière, 1923; Riese, 1931). Riese’s (1931) detailed analysis of the nervous system of Cypraea moneta described at least two anastomoses of the sub-oesophageal and supra-oesophageal ganglia that pass through the mantle roof; presence or absence of these dialyneuries could not be confirmed in sections of the highly glandular mantle roof of C. robertsi . Pedal cords with numerous cross connections are present ( Haller, 1890; Shaw, 1909; Riese, 1931). In contrast to the present findings, the tentacular nerve typically is shown as a simple, unbranched nerve ( Haller, 1890; Vayssière, 1923). The number of visceral ganglia is somewhat inconsistent among existing descriptions. For example, Haller (1890) depicted a single main ganglion with only a single accessory ganglion, while Riese (1931) described one main and three accessory ganglia. The position of the accessory ganglia is also variable. This inconsistency may be due to differing interpretations of the swellings along the visceral loop, several of which were only visible in C. robertsi with the aid of histological sections; these are highly variable in size and not all produce nerves.
| USNM |
Smithsonian Institution, National Museum of Natural History |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.