Nitidiscala tinctum, (CARPENTER 1864)
|
publication ID |
https://doi.org/ 10.1046/j.1096-3642.2003.00058.x |
|
persistent identifier |
https://treatment.plazi.org/id/03B6B923-EE2C-FFFE-8C21-64CC38CA6C6F |
|
treatment provided by |
Carolina |
|
scientific name |
Nitidiscala tinctum |
| status |
|
NITIDISCALA TINCTUM (CARPENTER 1864) View in CoL
Material examined
Venice, California ( USNM 890942).
External anatomy and mantle cavity
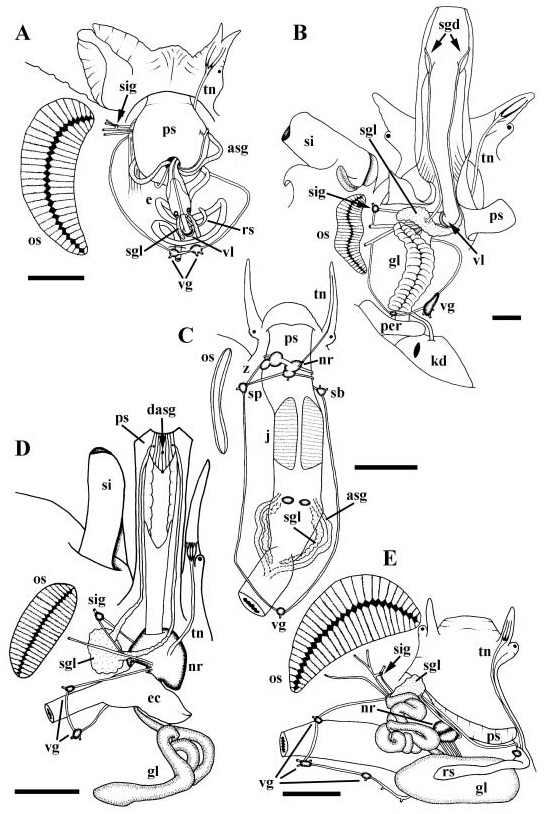
Mantle edge smooth. Hypobranchial gland thick, bright orange/brown in colour, producing characteristic purple secretion when disturbed. Osphradium ( Fig. 7C View Figure 7 , os) long, extending length of ctenidium, consisting of broad central axis bordered by two, densely ciliated ridges. Anterior pedal gland composed of basophilic cells, opening to deep cleft along anterior edge of propodium. Mesopodial pedal gland composed of basophilic and acidophilic cells, penetrated by numerous small ciliated ducts coalescing and opening to groove in foot sole via single, broad duct. Groove mid-ventral, extending approximately two thirds length of foot from posterior tip.
Reproductive system
Protandrous hermaphrodites. Gonad dorsally enclosing digestive gland on right. Vas deferens emerging from testis, rapidly enlarging to form seminal vesicle storing spermatozeugmata. Vas deferens narrowing anteriorly, opening to pallial gonoduct at base of mantle cavity. Prostate comprising paired, open laminae with shallow intervening gonoductal groove, densely ciliated anteriorly. Penis absent.
Gonopericardial canal absent. Oviduct opening to glandular pallial gonoduct short distance from its base. Pallial gonoduct comprising paired laminae, open along anterior end of albumen gland and length of capsule gland. Enclosed base of albumen gland bearing ciliated ridges directing sperm back and dorsally to opening of seminal receptacle. Small, ovate receptacle attached to dorsal wall of albumen gland, storing orientated sperm. In many females, degenerating spermatozeugmata visible adhering to floor of pallial cavity adjacent to albumen gland.
Alimentary System
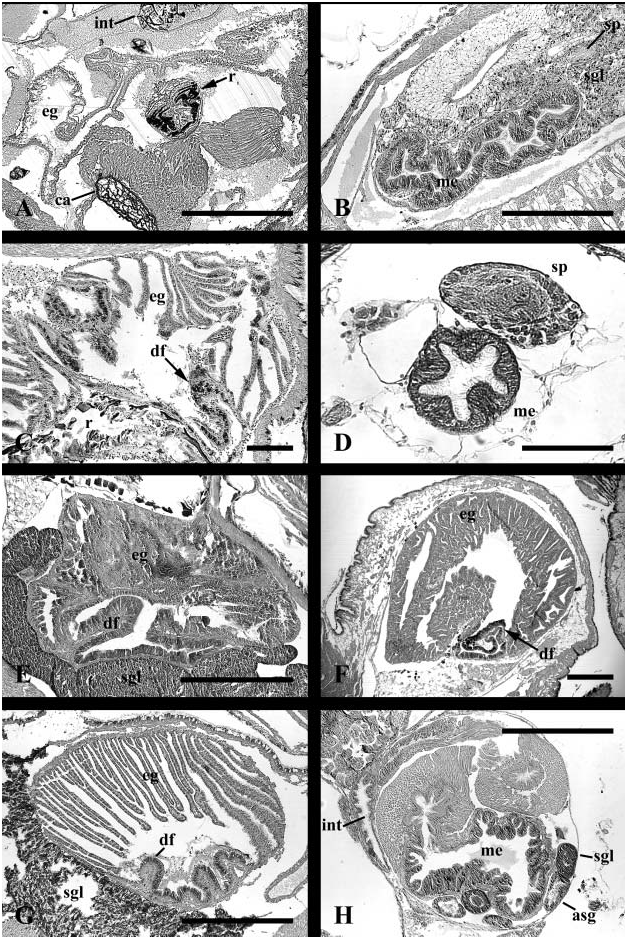
Foregut. Foregut modified into acrembolic proboscis ( Fig. 7C View Figure 7 , ps). Numerous long, thin branching protractor and retractor muscles connecting proboscis to walls and floor of cephalic haemocoel. Proboscis comprising long proboscis sheath opening to elongate buccal mass. Paired jaws (j) present, consisting of long sheets of cuticle comprised of rods with laterally overlapping, dark-staining homogeneous layer. Jaw supported by muscular, compartmentalized tissue and posteriorly flanking odontophore. Paired, thin, flexible cartilages supporting medially incised odontophore. Cartilages posteriorly L-shaped in cross section, becoming U-shaped anteriorly. Lateral arms of cartilages invading walls of buccal mass, supporting jaw, extending between cuticle and compartmentalized tissue beneath. Ptenoglossate radula extending completely over odontophore into deep sublingual cavity; subradular organ absent. Dorsal folds indistinct. Anterior oesophagus highly folded and uniformly glandular ( Fig. 10B View Figure 10 ). Discrete ventral folds and ventro-lateral folds absent. Dorsal pair of salivary glands ( Fig. 7C View Figure 7 , sgl) and ventral pair of accessory salivary glands (asg) present. Distal tips of salivary glands and fused distal tip of accessory glands attaching to ventral surface of oesophagus. Ducts of foregut glands extending anteriorly embedded within buccal cavity walls. Dorsal glands opening laterally beside odontophore. Ventral accessory glands, opening labially at base of proboscis sheath, terminating in cuticular stylets. Mid-oesophagus short and broad, with folded glandular walls; septate oesophageal gland absent ( Fig. 11H View Figure 11 , me). Proximal posterior oesophagus highly muscularized. Distal posterior oesophagus glandular, thin-walled, opening to midgut.
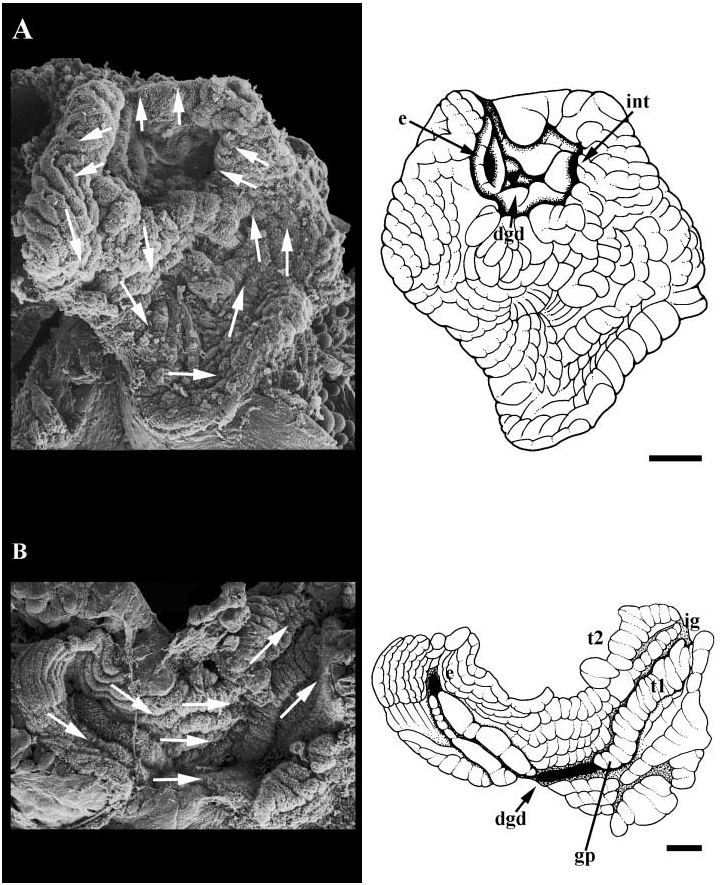
Midgut. Midgut comprising small anterior chamber, and elongate, highly folded, sacculate gastric chamber ( Fig. 17A View Figure 17 ). Oesophagus (e) entering left side of small anterior chamber, bounded by muscular valves. Anterior chamber also receiving single, large digestive gland duct (dgd). Gastric chamber walls densely ciliated; ciliary currents circulating particles posteriorly along left wall and anteriorly along right wall. Style sac region characterized by presence of single (major) typhlosole and longitudinally folded epithelium bearing uniform cilia.
Hindgut. Style sac region emerging from right, anterior end of caecum opposite oesophageal aperture. Intestine straight.
Reno-pericardial system
Kidney small, surrounding pericardium on right. Afferent renal vessel entering kidney floor just behind base of mantle cavity. Afferent renal vessel extending dorsally, branching into two large vessels that ramify to supply excretory tissue. Nephridial gland present.
Nervous system and sensory structures
Nervous system epiathroid, left zygoneurous ( Fig. 24E View Figure 24 ). Circum-oesophageal nerve ring ( Fig. 7C View Figure 7 , nr) lying far anterior to buccal mass, surrounding elongate proboscis sheath (ps). Long buccal connectives extending posteriorly to buccal ganglia lying ventrally at back of buccal mass, flanking short radular sac. Tentacular nerve (tn) single with small tentacular ganglion near base of nerve. Long pedal connectives extending around proboscis sheath to small pedal ganglia ( Fig. 24E View Figure 24 , pdg) partially embedded in mesopodial mucus gland. Supra-oesophageal ( Fig. 7C View Figure 7 , sp.) and sub-oesophageal (sb) connectives long. Pallial nerve from left pleural ganglion producing two small nerves before merging with supra-oesophageal ganglion. Single visceral ganglion (vg) under pericardium within cephalopedal sinus. Statocysts with single statoliths ( Fig. 24E View Figure 24 , sc) present on postero-dorsal surfaces of pedal ganglia.
Discussion
The Epitoniidae View in CoL is currently classified in the Superfamily Janthinoidea with the Aclididae View in CoL and Janthinidae. The anatomy of the family Epitoniidae View in CoL , as well as of the superfamily to which it belongs, has been insufficiently investigated. Of the three families, the Aclididae View in CoL is the least known; little more is established than the fact that they possess an acrembolic proboscis with a ptenoglossate radula, the males are aphallate and there are paired pedal folds that cover the base of the shell when the foot is extended ( Fretter & Graham, 1982; Bouchet & Warén, 1986).
Considerably more is known about the two remaining families. The foot of Janthina janthina View in CoL is modified, reflecting its pelagic life habit with a so-called funnel into which numerous glands empty ( Simroth, 1896 – 1907). But like Nitidiscala tinctum View in CoL , other epitoniids possess a foot that is indented posteriorly and bears a central groove ( Fretter & Graham, 1982) into which a mesopodial pedal gland opens (in both males and females) and secretes the mucus thread that connects egg capsules ( Robertson, 1983).
Pallial cavity anatomy is fairly conservative within the Janthinoidea , consisting of an elongate ctenidium, a thick hypobranchial gland that releases a characteristic purple secretion, and a long osphradium ( Bouvier, 1886; Thiele, 1928b; Clench & Turner, 1950; Taki, 1956, 1957; Robertson, 1963; Warén, 1980). Robertson (1983) suggested that the purple dye is repugnatorial. The description of Bouvier (1886) for Janthina globosa suggested a regionation of the osphradium as described here.
There has been some controversy surrounding the reproductive mode of janthinoideans (see Fretter & Graham, 1962, 1982 and Robertson, 1981a for discussion and review). However, it seems clear that most are protandrous hermaphrodites with only a single sex change ( Ankel, 1930; Graham, 1954; Breyer, 1980; Robertson, 1981a,b, 1983; Fretter & Graham, 1982; Smith & Breyer, 1983), but sex reversal may occur in some species ( Ankel, 1936). Due to the absence of copulatory organs in the Janthinoidea , sperm are transferred via spermatozeugmata which are stored in the seminal vesicle and distal end of the male genital tract ( Ankel, 1930; Graham, 1954; Bulnheim, 1968; Robertson, 1981a, 1983; Fretter & Graham, 1982). While abundant in the seminal vesicle, no spermatozeugmata were found in the pallial gonoduct in Nitidiscala tinctum . However, spermatozeugmata were routinely found at the base of the pallial cavity alongside the proximal pallial oviduct. In the absence of a bursa copulatrix, this most likely represents the site of spermatozeugmata degeneration whereupon sperm are passed to the seminal receptacle.
Available descriptions of epitoniids reveal that both open ( Fretter & Graham, 1982) and closed ( Taki, 1956, 1957) gonoducts are present. Janthina janthina ( Graham, 1954) possesses a closed pallial gonoduct that functions as a prostate during the male phase; posterior to the prostate is a blind sac with tubular glands that stores spermatozeugmata (Graham). During the female phase in J. janthina , the proximal portion of the pallial oviduct bears a highly villous appearance, similar to the glandular sac found in the same position during the male phase (Graham). No tubular glands were evident in female Nitidiscala tinctum . Janthina janthina (Graham) and other epitoniids ( Taki, 1956, 1957) apparently lack a seminal receptacle.
Epitoniids possess a long acrembolic proboscis ( Thiele, 1928b; Taki, 1956, 1957; Fretter & Graham, 1962, 1982; Warén, 1980; Robertson, 1981b, 1983) with two pairs of salivary glands ( Bouvier, 1886; Thiele, 1928b; Taki, 1956, 1957; Fretter & Graham, 1962). Janthinids possess only a short extensible snout ( Graham, 1965) equipped with two ( Bouvier, 1886; Simroth, 1902; Risbec, 1953; Graham, 1965) or three pairs of glands ( Thiele, 1928b; Marcus, 1956). All janthinoideans have an odontophore supported by Ushaped cartilages that extend into the buccal walls and support the jaw ( Thiele, 1928b; Marcus, 1956; Graham, 1965). In some, the jaw is massive ( Taki, 1956, 1957). However, most janthinoideans lack the thick, muscular, compartmentalized tissue supporting the jaw ( Thiele, 1928b; Marcus, 1956; Graham, 1965). Other species possess only small cuticular plates at the anterior end of the buccal cavity, with glands in the buccal cavity walls that may correspond to the compartmentalized tissue described here (e.g. Clathrus clathrus ; Fretter & Graham, 1962). Terminal stylets on the ventral salivary gland ducts may be present ( Simroth, 1902; Thiele, 1928b; Ankel, 1936, 1949; Fretter & Graham, 1962, 1982; Robertson, 1963; Warén, 1980) or absent ( Thiele, 1928b); the presence of stylets was neither confirmed nor denied by Taki (1956, 1957).
Janthinoideans possess a simple midgut ( Taki, 1956, 1957; Fretter & Graham, 1982), but there is some minor variability in the posterior oesophagus and in the position of intestinal, oesophageal and digestive gland apertures. The distal, glandular portion of the posterior oesophagus of Nitidiscala tinctum corresponds to the so-called cardiac chamber of other epitoniids ( Taki, 1956, 1957); the chamber varies from small and poorly developed, to large and muscular (Taki). In addition, the cardiac chamber may open separately to the gastric chamber (Taki), rather than to the caecum as in N. tinctum . Digestive gland ducts may be paired or single (Taki).
Similar to Nitidiscala tinctum , Janthina is epiathroid and left zygoneurous ( Bouvier, 1886; Thiele, 1928b). Bouvier also described the nervous system of Scala communis (as Scalaria communis ) as epiathroid but lacking secondary connections between ganglia. However, his preparation was apparently compromised by the purple secretions of the hypobranchial gland ( Thiele, 1928b). Statocysts with single statoliths are present between the pedal ganglia ( Bouvier, 1886; Marcus, 1956).
| USNM |
Smithsonian Institution, National Museum of Natural History |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Family |
|
|
Genus |
Nitidiscala tinctum
| Strong, Ellen E. 2003 |
Janthina janthina
| janthina (Graham 1954 |
Epitoniidae
| Berry 1910 |
Epitoniidae
| Berry 1910 |
Aclididae
| G.O.Sars 1878 |
Aclididae
| G.O.Sars 1878 |
Janthinoidea
| Lamarck 1810 |