Minagrion caldense Santos, 1965
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4786.2.2 |
|
publication LSID |
lsid:zoobank.org:pub:9EEAC300-4179-41B9-B51F-FDB4131CD991 |
|
persistent identifier |
https://treatment.plazi.org/id/03B61158-FFDA-FFC7-FF44-A56EFAE9BB9A |
|
treatment provided by |
Plazi |
|
scientific name |
Minagrion caldense Santos, 1965 |
| status |
|
Minagrion caldense Santos, 1965 View in CoL
Figs. 1 View FIGURE 1 a–b (♂ hab), 2a–d (♂ and ♀ tub), 3a–h (♂ app), 4a−b (♂ lig), 5a–g (♀ hab), 6a–b (♀ lob), 7a (ptx), 8a–b (♂ app, lost holotype), 9a–c (♀ lost allotype), 10a–c (alive ♂ and ♀♀, respectively)
Minagrion caldense, Santos (1965b) View in CoL : 8−12 (original description ♂ and ♀);— Lencioni (2006): 169 (illustrated guide; distribu- tion);—Garrison e t al. (2010): 287 (synonymic list);— Machado & Bedê (2015): 290−291, 294 (distr., illustr., comparison with Minagrion franciscoi View in CoL ).
Minagrion franciscoi Machado & Bedê (2015) View in CoL : 288 −291 (original description ♂). New synonymy.
Holotype ♂ and allotype ♀: BRAZIL, Minas Gerais State, Poços de Caldas, Campo do Aterrado , 05.xii.1964, N.D. Santos & J. Machado leg., in MNRJ [examined]
Specimens examined. Total: 30♂♂, 20♀♀. 6♂♂, 4♀♀ BRAZIL, Minas Gerais State, Poços de Caldas, Campo do Aterrado , 23.x.1964, O. Roppa & O. Leoncini leg. , 1♂, 2♀♀ in ABMM, 1♂ in FAAL and 4♂♂, 2♀♀ in MNRJ; Minas Gerais State, São Roque de Minas, Serra da Canastra National Park (20°14’37” S, 46°26’47” W) in different dates and collections as follows: 3♂♂, 09.x.2014 (including the holotype and paratypes of M. franciscoi ), L.C. Bedê leg. GoogleMaps , in ABMM; 1♂, 25.ii.2015, L.C. Bedê leg. , in ABMM; 4♂♂, 9♀♀, 08.iii.2018, R. Guillermo−Ferreira leg. (of those, 2♂♂, 2♀♀ in LESTES, 2♂♂, 7♀♀ in ECOEVO) ; 3♂♂, 1♀, 19.x.2017, R. Guillermo−Ferreira leg. , in LESTES; 3♂♂, 20.x.2017, R. Guillermo−Ferreira leg. , in LESTES; 2♂♂, 21.x.2017, R. Guillermo−Ferreira leg. , in LESTES; 2♂♂, 2♀♀, Minas Gerais State, São Roque de Minas, Serra da Canastra National Park, Ponte 1 (20°14’25”, 46°35’15”), 21.x.2017, D.S. Vilela leg.
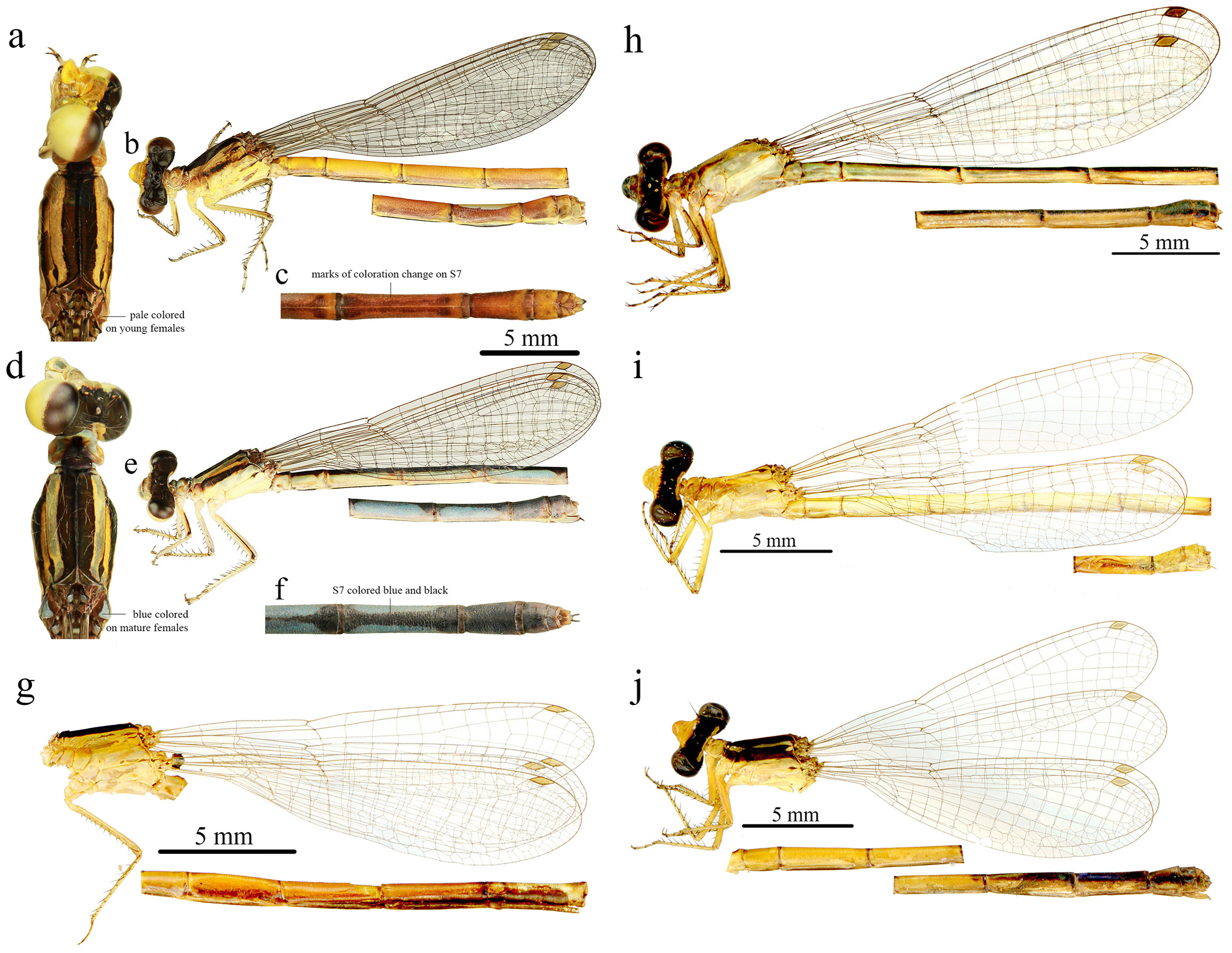
Additions to the original female description. Head ( Fig. 5 View FIGURE 5 a–b, d–e). Dorsally black except for the occipital bar: yellow in juvenile females, pale/white in mature females; from labrum to postfrons: yellow in juvenile females, pale/blue in mature females.
Thorax ( Figs. 5 View FIGURE 5 a–g, 6a–b, 7a, 9a). With a thick black humeral stripe extending from prothorax to posterior margin of middorsal carina; some females with lateral black stripe on the upper half of mesepimeron; remainder areas yellow in juvenile females and blue/yellow in mature females, remainder of mesepisternum, pale/white; posterior lobe of prothorax slightly convex, directed posteriorly; posterior margin of prothorax slightly widens towards the base in lateral view.
Legs ( Fig. 5b, e View FIGURE 5 ). Pale yellow with darker areas on femoral armature in juvenile females; legs white with a black line on femoral armature in mature females.
Wings ( Fig. 5b, e, g View FIGURE 5 ). Hyaline with a light brown pterostigma, occupying one cell; 8−9 Px in HW, 9 Px in FW; CuA extending for 8 cells distal to vein descending from subnodus in FW and 7 in HW; MP not reaching wing posterior margin and merged to MP&AA in FW and HW.
Abdomen ( Figs. 5 View FIGURE 5 b–c, e–f, g, 9b–c). Juvenile females: S1−5 yellow with slightly darker areas laterally; S6−8 al- most orange with darker areas laterally; S9−10 yellow/orange, cercus yellow. Mature females: S1−3 white laterally, black dorsally; S4 light blue laterally, black dorsally; S5−7 blue, interrupted by thin black dorsal stripes and black basal rings; S8−10 black, with white pruinosity on older females; cercus white. On all females, notopleural suture a smoothly curved line; anterior portion of tubercle ventrally pronounced, apex slightly excavated; genital valves surpassing S10 margin for a distance as long as the segment.
Measurements. Total 28−32; abdomen 22−26; FW 18−20; HW 14−17.
Diagnosis. Males of M. caldense can easily be separated from congeners by the almost unique excavated morphology of tubercle ( Fig. 2 View FIGURE 2 a–b). Females can be separated from females of other species by the following character combination: posterior lobe of prothorax with no separated lobes, middle lobe convex (concave in M. canaanense ) ( Fig. 6 View FIGURE 6 a–b); tubercle with a minor excavation, anterior portion ventrally pronounced ( Fig. 2 View FIGURE 2 c–d).
Molecular analysis. The target COI sequences of all samples were amplified and sequenced successfully. Sequences of old specimens of M. caldense were also analyzed, however, they were highly fragmented and therefore could not be used in this study. The analyzed males and females of M. caldense formed a monophyletic group with strong support with the six females analyzed (Bayesian posterior probability, BPP = 1.00) and formed a unique clade with no further divided branches (full tree available in Appendix A). The K2P distance of COI sequences among the M. caldense female types was 0.00% − 0.20%, while for the other species this value ranged between 13.20% and 20.10% (see Appendix B). These evidences implied that the males and females analyzed here belong to the same species.
Ontogenetic color change. The dissections made in both female stages of M. franciscoi lead us to the conclusion that females of this species undergo a complete ontogenetic coloration change, therefore representing juvenile (yellow females) and mature (blue females) instead of two different morphs. Juvenile females only presented undeveloped egg masses and all examined mature females presented fully developed eggs with an oval shape and resilient chorion ( Waage 1980, Higashi & Watanabe 1993).
Remarks. Upon the examination of holotypes and paratypes of M. caldense of M. franciscoi , we found no substantial characters in morphology to separate them, although some traits such as cercus ( Figs. 3 View FIGURE 3 a–h, 8a–b) and genital ligula ( Fig. 4 View FIGURE 4 a–b) present subtle differences, which can be interpreted as intraspecific variation. Machado & Bedê (2015: 291) state that the sclerotized lateral process in genital ligula is separated, forming two structures on each side in M. franciscoi . We noticed a subtle variation (size, shape) of this trait on the examined males, but we saw no such separation among material (including paratypes) identified as this species. The sclerotized lateral processes in all were entire (as in Fig. 4 View FIGURE 4 a–b). Excluding this character, coloration was the diagnostic means with which to separate M. caldense from M. franciscoi by Machado & Bedê (2015). However, this can be problematic since coloration has proven not always to be a reliable source of taxonomic diagnosis in Coenagrionids (Vilela et al. 2019). In both ‘species’, the posterior lobe of female prothorax is slightly convex, posteriorly oriented, and the lateral margins of prothorax slightly widening towards the base, in lateral view. Females of M. caldense are also morphologically identical to females that we associated with males of M. franciscoi . As in males, coloration accounted for the main differences between these females which cannot be used as a specific differential since females undergo ontogenetic changes in coloration. Therefore we suggest that M. franciscoi be treated as a junior synonym of M. caldense .
Distribution. This species remains one of the rarest in the genus. The only two known localities of occurrence are in Poços de Caldas (type locality), and the National Park of Serra da Canastra both in Minas Gerais State.
| MNRJ |
Museu Nacional/Universidade Federal de Rio de Janeiro |
| R |
Departamento de Geologia, Universidad de Chile |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Minagrion caldense Santos, 1965
| Vilela, Diogo Silva, Anjos-Santos, Danielle, Koroiva, Ricardo, Cordero-Rivera, Adolfo & Guillermo-Ferreira, Rhainer 2020 |
Minagrion franciscoi Machado & Bedê (2015)
| Machado, A. B. M. & Bede, L. C. 2015: 288 |