Dracoleuctra, Lee & Baumann, 2022
|
publication ID |
https://doi.org/ 10.11646/zootaxa.5115.2.4 |
|
publication LSID |
lsid:zoobank.org:pub:85A2F1F0-BC45-4ED0-AC0A-41757CCCB41B |
|
DOI |
https://doi.org/10.5281/zenodo.6354789 |
|
persistent identifier |
https://treatment.plazi.org/id/03B587C0-1C4E-A511-FF68-0EC6FD75AFA1 |
|
treatment provided by |
Plazi |
|
scientific name |
Dracoleuctra |
| status |
gen. nov. |
Dracoleuctra View in CoL , new genus
Type species:. Dracoleuctra siskiyou , new species, by monotypy.
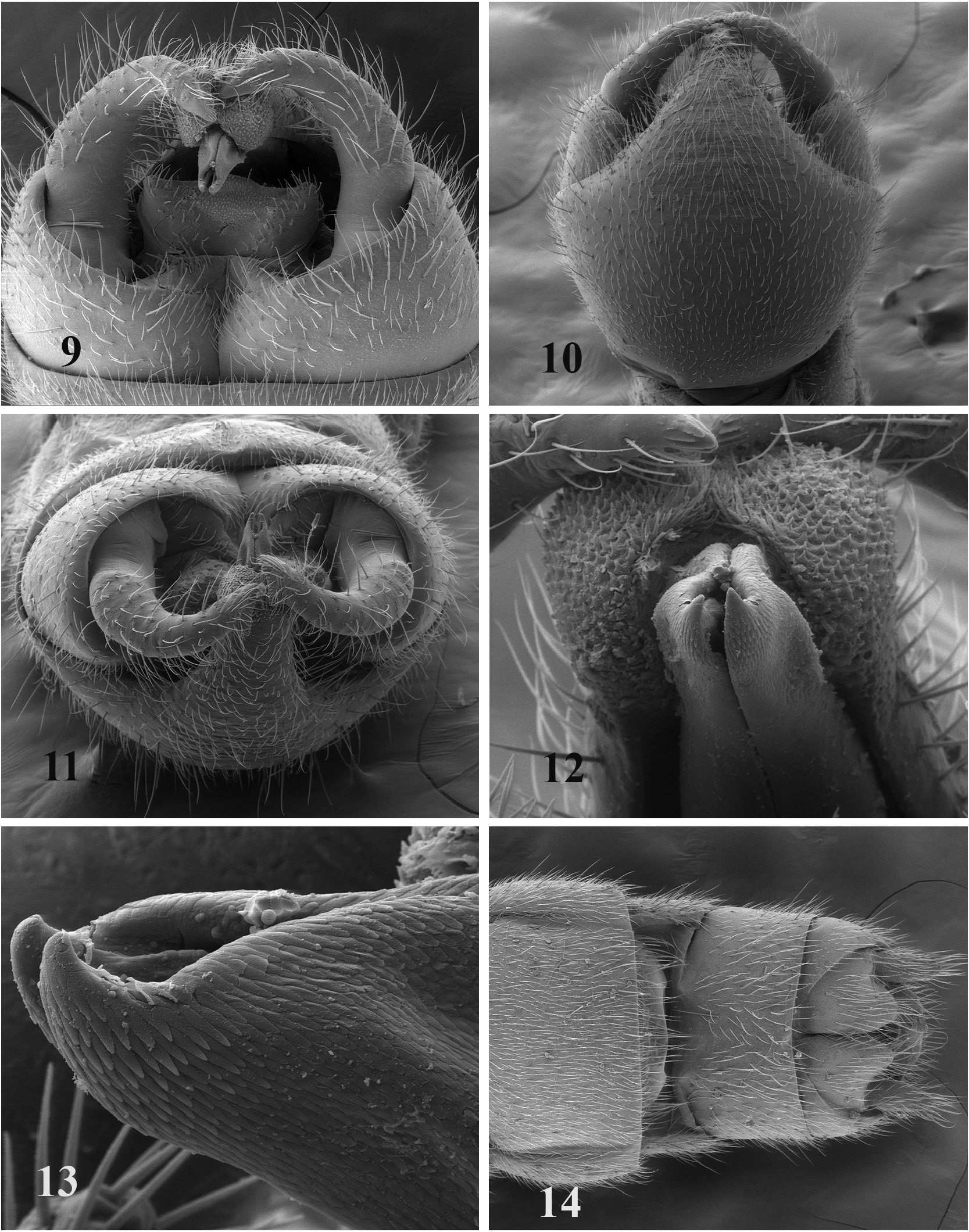
Morphology. Adults. Apterous ( Fig. 1 View FIGURES 1–4 ), head brown, lacking ocelli, antennae elongate, with at least 34 segments, body bearing numerous dark and light clothing hairs, sclerotized parts brown to dark brown, membranous areas pale.
Prothoracic tergum with large central pronotum wider than long, central disc brown with dark brown middorsal line, dark brown rugosities lateral to middorsal line ( Fig. 1 View FIGURES 1–4 ), light brown marginal groove ( Claassen, 1931) surrounding central disc; anterior and posterior margins with three shallow longitudinal furrows not reaching central disc, demarking four anterior and four posterior rectangular areas, larger in posterior row. Mesonotal anterior prescutum narrow, transverse, anteriorly curved, projecting slightly under pronotum, separated from scutum by only a shallow groove; scutum widest anteriorly projecting laterally forward of anepisternum, narrowing then widening slightly posteriorly, rugosities lateral to middorsal line; scutellum not apparent; postscutellum widest anteriorly, gradually narrowing posteriorly, middorsal suture line reaching scutum, demarked from scutum by transverse light brown line, suture not apparent. Metanotum similar to mesonotum but smaller and not as narrow posteriorly. Prothoracic basisternum and presternum fused, spade-shaped ( Fig. 2 View FIGURES 1–4 ), furcasternum narrow mesally, expanded laterally, somewhat bowtie-shaped, separate from basisternum; postfurcasternum large, transversely oval, midventral line membranous. Mesosternal spinasternum 1 with dark oval mesal depression, lateral sclerotized bands shorter than prothoracic furcasternal width; presternum heart-shaped, basisternum large, ray-shaped, “wings” extending laterally past the lateral margin of coxae, bearing numerous clothing hairs, with posteromesal depression where furcasternum is embedded, suture lines not apparent; furcasternum round-oval, furcasternal pits short, arms not apparent; spinasternum 2 dark, reaching metathoracic presternum, postfurcasterna oval, separate from spinasternum 2. Metasternal presternum transversely oval, basisternum similar in shape to mesobasisternum but truncate posteriorly where furcasternum is embedded.
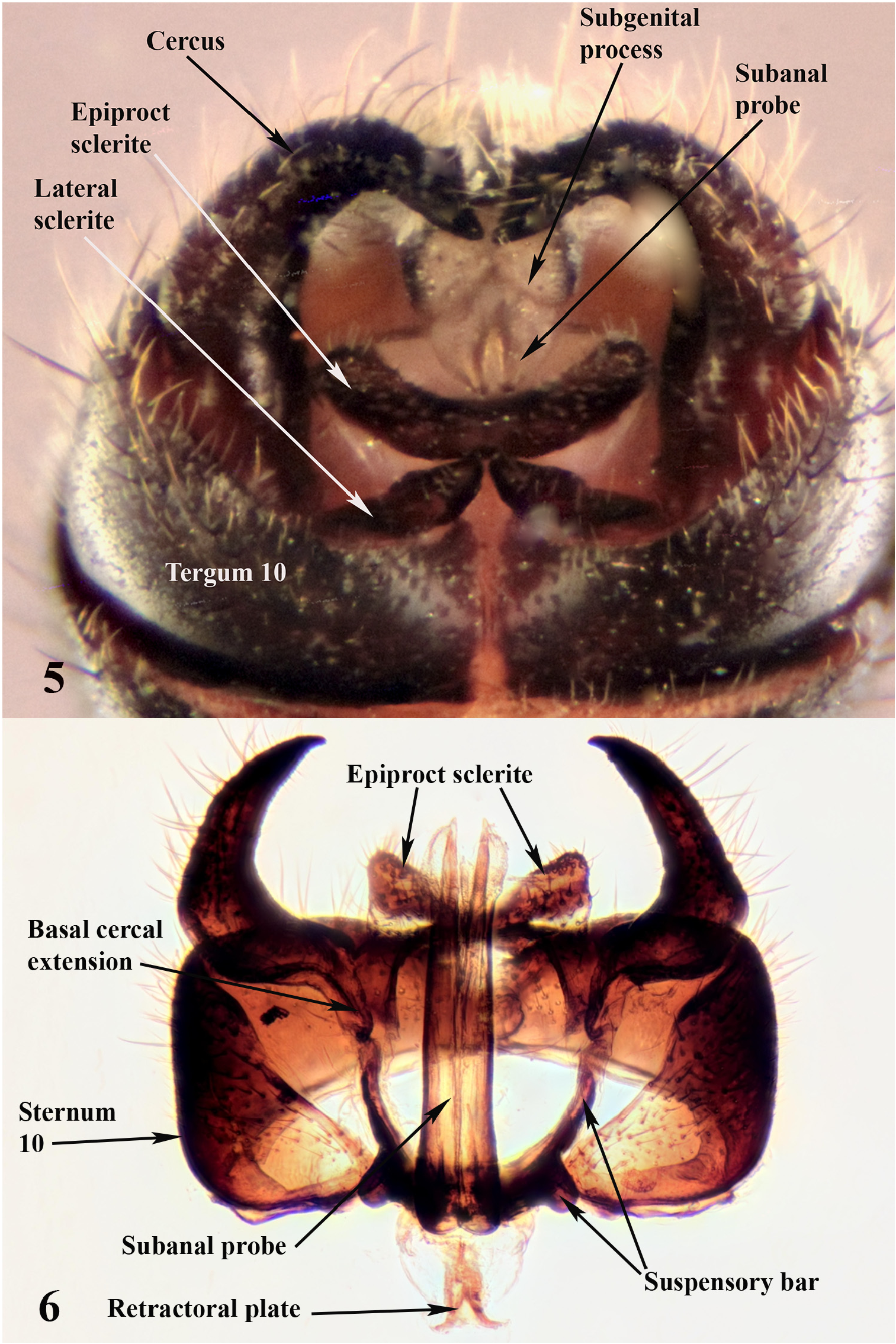
Male. Length 4.5–6.0 mm, mean length 5.3 mm (n = 8). Abdominal segments sclerotized, dark to light brown. Abdominal segment 9 lacking vesicle ( Fig. 10 View FIGURES 9–14 ), heavily sclerotized, uniformly dark brown, tergum narrow and widening laterally, sternum broad, cup-shaped then narrowing posteriorly to a dorsally produced subgenital process (we prefer this term to subgenital plate), process apically slightly emarginate; inner portion a membrane lined furrow where the subanal probe lies, membranous layer thick apically ( Fig. 5 View FIGURES 5–6 , 11 View FIGURES 9–14 ). Segment 10 forming sclerotized band under cerci and articulating with suspensory bars of paraprocts ( Fig. 6 View FIGURES 5–6 ), tergite with mesal longitudinal membranous line, mesoposterior border slightly produced posteriorly ( Figs. 5 View FIGURES 5–6 , 9 View FIGURES 9–14 ). Lateral sclerites narrowly contacting tergum 10 anteriorly, bases of cerci laterally, and epiproct sclerite posteromesally ( Fig. 7 View FIGURES 7–8 ); epiproct sclerites fused mesally, extending posterolaterally, dorsally projecting epiproct lacking ( Figs. 5 View FIGURES 5–6 , 7 View FIGURES 7–8 ); setose membranous pad present posterior to epiproct sclerite. Outer lobes of paraproct (suspensory bars) broad basally extending a short distance laterally then directed anteriorly as narrow sclerotized straps that articulate with posterior base of cerci ( Fig. 6 View FIGURES 5–6 ), lateral portion with large distoposterior knob that articulates with posteroventral portion of segment 10. Basal sclerotization extends mesally along subanal probe as paired sclerotized rods that appear to be unsclerotized on mesal margin, dorsoapical sclerotization acuminate ( Figs. 6 View FIGURES 5–6 , 8 View FIGURES 7–8 , 12–13 View FIGURES 9–14 ), ventral sclerotization ends subapically in a membranous structure ( Fig. 5 View FIGURES 5–6 ). Anterior to base of subanal probe is an interior membranous sac, at the mesoanterior end of sac lies the retractoral plate ( Figs. 6 View FIGURES 5–6 , 8 View FIGURES 7–8 ), a bluntly triangular sclerotized process (flat in lateral view), a pair of sclerotized straps (ducts?) extend from its base and are directed to the base of the subanal probe. Cerci large ( Figs. 5 View FIGURES 5–6 , 11 View FIGURES 9–14 ), heavily sclerotized, dark brown, horn shaped, directed posteriorly, curving mesally and slightly dorsally, apex acute with a subapical ventral tubercle, mesobasal margin with belt-like extension articulating with suspensory bars of paraproct ( Fig. 6 View FIGURES 5–6 ).
Female. Length 4–7 mm, mean length 5.7 mm (n = 19). Abdominal tergum 1 with large sclerite covering most of tergum; tergum 2 mostly membranous with large central sclerite and a few small sclerites laterally; terga 3–7 membranous with numerous small sclerites scattered over surface ( Fig. 3 View FIGURES 1–4 ), each sclerite bearing one large dark seta; some sclerites longitudinally elongate and bearing 2–3 dark setae, these appear to be coalesced smaller sclerites. Tergum 8 mostly sclerotized with submesal membranous area wide anteriorly, narrowing posteriorly, bearing few small sclerites; terga 9 and 10 sclerotized. Abdominal sterna 1–6 with mesal sclerite surrounded by membranous area, sclerites increase in size with sternal number; sternum 7 mostly sclerotized, membranous anteriorly at pleural fold; sternum 8 ( Figs. 4 View FIGURES 1–4 , 14 View FIGURES 9–14 ) with darkly sclerotized subgenital plate about two-thirds width and one-half length of sternite, posterior margin curving anteriorly in a symmetrical gentle arc or with a slight mesal posterior projection, posterior one-half of sternite membranous; sternum 9 sclerotized with a short anteromesal projection; sternum 10 membranous mesally; paraprocts roughly triangular, posterior margin blunt; cerci one segmented, setose.
Larva. Unknown.
Diagnosis. Morphology. Both males and females of Dracoleuctra can be recognized (even in the field) by the lack of wings and the one-segmented cerci. The following combination of characters are diagnostic for males: segment 9 heavily sclerotized, subgenital process produced dorsally with a membrane–lined interior furrow, inner apical portion with a thick membranous layer, vesicle lacking, heavily sclerotized horn-shaped cerci, lack of a dorsally projecting epiproct but with the lateral sclerites and epiproct sclerite together butterfly-shaped, and the membranous, bluntly cone shaped apex of the subanal probe pressed to inner membranous portion of subgenital process. Females can be recognized by the membranous abdominal tergites bearing numerous small, setose sclerites, and the size and shape of the subgenital plate.
Ricker and Ross (1969), Nelson and Hanson (1973), Nelson (1977), and Shepard and Baumann (1995) have proposed phylogenies for Leuctridae . Although we do not propose a possible Leuctridae phylogeny, we have studied these papers and examined leuctrid specimens we have in hand, including the western North American species of the following genera: Calileuctra Shepard & Baumann, 1995 , Despaxia Ricker, 1943 , Megaleuctra Neave, 1934 , Moselia Ricker, 1943 , Paraleuctra Hanson, 1941 , Perlomyia Banks, 1906 , Pomoleuctra Stark & Kyzar, 2001 , plus the apterous Leuctra aptera Kaćanski & Zwick, 1970 from Europe and Paraleuctra ambulans Shimizu, 2000 from Japan. With input from Charles Nelson (personal communication, 2020), we feel Dracoleuctra is closest to Paraleuctra and Pomoleuctra based on the general structure of the paraproct and paraproct lobes. Stark and Kyzar (2001) list nine shared features uniting species in Paraleuctra . Two of the features are wing vein characters, so obviously these do not pertain to Dracoleuctra . Additional unshared characters include basisternum and furcasternum of prothorax fused (not fused in Dracoleuctra ), epiproct reduced to a small, curved hook (dorsally projecting curved hook lacking in Dracoleuctra ), and female subgenital plate usually bilobed (not bilobed in Dracoleuctra and only onehalf length of sternum 8). These Dracoleuctra characters are also not shared with Pomoleuctra . Dracoleuctra also differs from Paraleuctra and Pomoleuctra in the lack of a vesicle, and from Paraleuctra , having a robust sternum 9 with a posteriorly and dorsally produced subgenital process harboring the subanal probe. The western North American Paraleuctra and Pomoleuctra we have examined also have furrows in sternum 9 demarcating the subgenital process, this feature is lacking in Dracoleuctra . Additionally, the female abdominal tergum, membranous but bearing numerous small setose sclerites, appears to be unique to Dracoleuctra . We have avoided using meso-metasternal characters for diagnosis since Zwick (2000) questions how variable flight ability affects use of thoracic sternal characters for generic correlation. Dracoleuctra does have meso-metasternal characters similar to the descriptions and figures of apterous Leuctra from the Caucasus, Georgia ( Teslenko et al., 2019). We are not sure if flight ability would affect prosternal sclerites, but we do include prosternal characters in our diagnosis.
Type specimens. Holotype male: Oregon, Josephine County, Happy Camp Road , Siskiyou Spring , 42.00162 -123.53785, ~ 1440 m elevation, Klamath-Siskiyou Mountains , 6 May 2020, J. Lee. To be deposited in the entomology collection at California Academy of Sciences, San Francisco, CA, USA ( CAS) GoogleMaps . Paratypes: 4 males, same data as holotype GoogleMaps ; 9 females, same data as holotype but 1 May 2020. To be deposited at CAS (2 males; 5 females) and the Brigham Young University Collection, Provo, Utah, USA ( BYUC) GoogleMaps 2 males; 4 females.Additional material: 3 males, 8 females (Jonathan J. Lee Collection); 3 males 8 females ( BYUC) .
Etymology. The prefix of the genus name Draco (Latin for lizard or dragon) was selected because the males had a reptilian quality running across the beating sheet. The suffix name was selected to indicate placement of the genus within the family Leuctridae . Gender of the name is neuter. The trivial name siskiyou was selected because the specimens were collected from Siskiyou Spring in the Siskiyou Mountains subregion of the Klamath Mountains.
Biology. Siskiyou Spring is located in the Siskiyou Mountains subregion of the Klamath Mountains and is a headwater tributary of Althouse Creek in the Rogue River drainage. The spring emerges ( Fig. 15 View FIGURES 15–20 ) at an elevation of about 1450 m on a steep hillside as small creeks and seeps creating a wetted area several meters wide. At least two small creeks flow within the wetted area; between them seeps are moss and forb covered ( Figs. 16–17 View FIGURES 15–20 ). The longitudinal distance between the springhead and road is <100 m. The entire wetted area is covered by a nearly impenetrable stand of Sitka Alder ( Alnus crispa ssp. sinuata ) ( Figs. 18–20 View FIGURES 15–20 ). Adult stoneflies collected with Dracoleuctra were Paraleuctra forcipata (Frison, 1937) , Pomoleuctra andersoni (Harper & Wildman, 1985) , females of an undescribed apterous Capniidae , Soyedina nevadensis (Claassen, 1923), S. producta (Claassen, 1923), and Zapada cordillera (Baumann & Gaufin, 1971). Adult Trichoptera collected were Goeracea genota (Ross, 1941) (Goeridae) and Moselyana comosa Denning, 1949 (Apataniidae). Plecoptera larvae collected were Moselia, Yoraperla Ricker, 1952 , Sasquaperla hoopa Stark & Baumann, 2001, Sweltsa pr. fidelis (Banks, 1920), Chernokrilus misnomus (Claassen, 1936), and Isoperla laucki Baumann & Lee, 2009. We have not visited the spring in late summer but presume it is perennial.
Initially, only females were caught by beating low vegetation next to running water. Some males were caught in low vegetation near the springhead, but it was later discovered that males were more easily caught by beating the vertical branches of Sitka Alder> 2 m high in the upper section of the spring creek. Anecdotally, a small female was found on the beating sheet with a male. We are guessing that the females higher up in the vegetation were virgins while many of the females caught nearer the ground were larger and probably gravid. We hypothesize that the males climb up the branches in search of virgin females.
Apterism in the Klamath Mountains Region. Relatively few apterous stoneflies are known from North America compared to other parts of the world, such as regions in Europe and Japan. Outside of the Klamath Mountains region, fully apterous North American Plecoptera include Allocapnia harperi Kirchner, 1980 and A. vivipara (Claassen, 1924) in the eastern United States; Capnia lacustra Jewett, 1963, from Lake Tahoe, California; Mesocapnia bergi (Ricker, 1965), known from the lower Yukon River, Alaska; Paracapnia disala (Jewett, 1962) , known from California and Oregon, P. ensicala (Jewett, 1962) , known from Oregon and Washington; and Utacapnia tahoensis (Nebeker & Gaufin, 1965), Lake Tahoe, California. Although the environmental processes leading to apterism are not well established in the literature, isolation in a stable environment over time has been suggested. Brinck (1949) states that “species with both sexes micropterous or apterous exist only in regions that have changed little during a long period of time.” Illies (1968) echoed Brinck. Loskutova & Zhiltzova (2016) also suggested that short wingedness is associated with populations being isolated over a long period of time. Stability of a region over a long period of time may explain the degree of apterism in the Klamath Mountains, however, it might not be a predictive cause in other regions. The Klamath Mountains region is known for its great biological diversity containing more than 3,500 plant species, more than 200 of which are endemic ( Sawyer 2007). Sawyer (2007) also notes that a large portion of the Klamath Mountains have been above sea level for 65 million years, with no records of volcanic deposits, and no glaciers were regionally extensive. This region is also isolated with poor access, particularly during winter months. Several species of apterous capniids are known from the Klamath Mountains of southern Oregon (OR) and northern California (CA), including Capnia fialai Nelson & Baumann, 1990, known from one perennial headwater creek in Humboldt Co., CA; Paracapnia disala , known from several headwater sites in southwestern OR and northwestern CA; the “ California Paracapnia ” (very probably a true phylogenetic lineage, David Murányi, unpublished), P. boris Stark & Baumann, 2004 , known from perennial and perhaps seasonal creeks in Colusa, Shasta and Trinity counties, CA, P. humboldta Baumann & Lee, 2007 , known from two seasonal creeks in Humboldt Co., CA, P. baumanni Kondratieff & Lee, 2010 , known from seasonal creeks in Shasta and Trinity counties, CA; and Mesocapnia aptera Lee & Baumann, 2011 , known from one seasonal creek in Mendocino Co., CA. The addition of Dracoleuctra and an undescribed apterous capniid (known only from females and early instar larvae that DNA sequence analysis places near C. fialai) suggests that the region is a relative hotbed for apterous stoneflies. Future collecting may continue to produce surprising finds.
Epilogue. The 2020 Slater Fire in northern California and southern Oregon burned through the entire area where Siskiyou Spring occurs. Hazardous tree removal has kept Happy Camp Road (the access road) closed at least into February 2022. Fire has probably burned through the area many times in the past and we expect that the Dracoleuctra population has survived this recent fire.
| CA |
Chicago Academy of Sciences |
| CAS |
California Academy of Sciences |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |