Paraphasma umbretta ( Lichtenstein, 1796 )
|
publication ID |
https://doi.org/10.11646/zootaxa.5122.1.1 |
|
publication LSID |
lsid:zoobank.org:pub:EC13A69D-D6FA-4926-AC59-648A5626C9B9 |
|
DOI |
https://doi.org/10.5281/zenodo.10718251 |
|
persistent identifier |
https://treatment.plazi.org/id/03B587AA-FFEE-FF89-FF2A-FF2DFAB1F198 |
|
treatment provided by |
Plazi |
|
scientific name |
Paraphasma umbretta ( Lichtenstein, 1796 ) |
| status |
|
Paraphasma umbretta ( Lichtenstein, 1796) View in CoL
Figs 43–49 View FIGURE 43 View FIGURE 44 View FIGURE 45 View FIGURE 46 View FIGURE 47 View FIGURE 48 View FIGURE 49 , Table 10 View TABLE 10 .
Phasma umbretta Lichtenstein, 1796: 78 [name for the species “Le Double-Epine Brun” of Stoll (1787: 24, fig. 27)]; Lichtenstein, 1802: 14; Gray, 1835: 26 [= Phasma lineare Stoll, 1813 ]; De Haan, 1842: 123; Redtenbacher, 1906: 116 [= Paraphasma maculatum ( Gray, 1835) View in CoL ]; Kerzhner, 1994: 111–112; ICZN, 1995: 283–284 [the name Phasma umbretta Lichtenstein, 1796 is available despite having been published in a work suppressed for nomenclatural purposes]; Otte & Brock, 2005: 251 [= Paraphasma maculatum ( Gray, 1835) View in CoL ]; Conle et al., 2011: 33 [= Paraphasma maculatum ( Gray, 1835) View in CoL ]; Conle et al., 2020: 126 [= Paraphasma maculatum ( Gray, 1835) View in CoL ]. Unspecified type (s): ♀, Suriname [probably lost].
Necroscia umbretta, Westwood, 1859: 147 ; Redtenbacher, 1906: 116 [= Paraphasma maculatum ( Gray, 1835) View in CoL ].
Prexaspes umbretta, Kirby, 1904a: 414 View in CoL .
Paraphasma umbretta, Brock et al., 2022 View in CoL [database Phasmida Species File Online].
Phasma linearis Stoll, 1813: 77 [name for the species “Le Double-Epine Brun” of Stoll (1787: 24, fig. 27)]; Westwood, 1859: 147 [= Necroscia umbretta ( Gray, 1835) ]; Kirby, 1904a: 415 [= Prexaspes umbretta ( Gray, 1835) View in CoL ]; Redtenbacher, 1906: 116 [= Paraphasma maculatum ( Gray, 1835) View in CoL ]; Otte & Brock, 2005: 251 [= Paraphasma maculatum ( Gray, 1835) View in CoL ]; Conle et al., 2011: 33 [= Paraphasma maculatum ( Gray, 1835) View in CoL ]; Conle et al., 2020: 126 [= Paraphasma maculatum ( Gray, 1835) View in CoL ].
Phasma lineare, Gray, 1835: 26 ; De Haan, 1842: 123.
[Non Mantis linearis Fabricius, 1787 , valid name: Necroscia manicata ( Lichtenstein, 1802) ]
Phasma acanthophora Stoll, 1813: 62 , 77, fig. 78 [“Le Spectre Moucheté”, named Phasma acanthophora in the appendix (p. 77)]; Otte & Brock, 2005: 251 [= Paraphasma maculatum ( Gray, 1835) View in CoL ]; Conle et al., 2011: 33 [= Paraphasma maculatum ( Gray, 1835) View in CoL ]; Conle et al., 2020: 126 [= Paraphasma maculatum ( Gray, 1835) View in CoL ]. Unspecified type (s): ♀, Ambon Island [probably lost; locality probably wrong].
Phasma acanthopterum, Gray, 1835: 23 [misspelling of Phasma acanthophora Stoll, 1813 reproduced by subsequent authors]; Burmeister, 1838: 586; Serville, 1839: 268–269; De Haan, 1842: 123; Redtenbacher, 1906: 116 [= Paraphasma maculatum ( Gray, 1835) View in CoL ]; Conle et al., 2020: 126 [= Paraphasma maculatum ( Gray, 1835) View in CoL ].
Necroscia acanthoptera, Westwood, 1859: 147 ; Redtenbacher, 1906: 116 [= Paraphasma maculatum ( Gray, 1835) View in CoL ].
Trigonophasma acanthophora, Kirby, 1904a: 373 .
Phasma maculatum Gray, 1835: 26 ; Serville, 1839: 269; De Haan, 1842: 123; Westwood, 1859: 122; Bates, 1865: 350. Holotype: ♂, Brazil, “Amazon” (NHM 012502909) ( Fig. 43 View FIGURE 43 ).
Prexaspes maculatus, Kirby, 1904a: 414 .
Paraphasma maculatum, Redtenbacher, 1906: 116 View in CoL , pl. IV, fig. 17; Handlirsch, 1930: 760, fig. 804; Zompro, 2004: 159; Otte & Brock, 2005: 251; Conle et al., 2011: 33; Brock et al., 2016: 181 View Cited Treatment ; Conle et al., 2020: 70, 126; Chiquetto-Machado & Cancello, 2021: 4, 26, figs 4, 15B, 16B, 17A, 19A, 23B, 24B, 25, 26.
Prexaspes janus Kirby, 1904a: 415 View in CoL [new name for Phasma acanthopterum Gray, 1835 (incorrectly attributed to Serville, 1839)]; Conle et al., 2020: 126 [= Paraphasma maculatum ( Gray, 1835) View in CoL ].
Prexaspes (Elasia) janus, Otte & Brock, 2005: 281 View in CoL .
Diagnosis. Easily distinguishable from other species of Paraphasma by the following characters of external morphology: anterior region of the male subgenital plate with a pair of small, approximately styliform lateral projections entirely covered by tergite IX ( Fig. 45E View FIGURE 45 : arrowhead); posterior margin of the male subgenital plate truncate in lateral view ( Fig. 45B View FIGURE 45 ) and V-shaped in caudal view ( Fig. 45D View FIGURE 45 ); female sternite VII with straight posterior margin, lacking a rounded indentation; body distinctly stained in black or brown (especially on the head, pronotum, mesonotum and costal region of the hindwings) and lacking the pair of lateral light stripes present in most species of Paraphasma ( Figs 43 View FIGURE 43 , 44 View FIGURE 44 , 47 View FIGURE 47 , 49 View FIGURE 49 ). Other features that may be useful for the identification of P. umbretta are the tegmina shoulder pads developed into sharp spines ( Figs 43B View FIGURE 43 , 44D View FIGURE 44 ), the approximately pyriform male cerci with rounded apex ( Fig. 45 View FIGURE 45 ), and females with large body length for the genus (> 66 mm, sometimes surpassing 70 mm).
Redescription of male. Color ( Figs 43–45 View FIGURE 43 View FIGURE 44 View FIGURE 45 , 49 View FIGURE 49 ): Body mostly yellowish-brown, but with many black or dark brown areas. Head, pro- and mesonotum strongly black-stained, with distinct light brown dorsomedian line.Antennae dark brown or black, sometimes with sparse light brown regions. Legs mostly dark brown or black, with irregular lighter areas; femora-tibiae joints light brown. Tegmina dark brown or black with conspicuous light brown venation. Costal region of hindwings yellowish or grayish-brown, with irregular dark brown stains. Body ventrally brown; abdominal sternite VIII and subgenital plate black-stained. Live specimens fairly bright-colored ( Fig. 49 View FIGURE 49 ), with head, thorax and legs shiny black with reddish-brown areas; hindwings with yellowish-gray costal region and orange anal region (orange color completely fades in dried specimens). Head ( Figs 43 View FIGURE 43 , 44 View FIGURE 44 ): As in Paraphasma conspersum , but vertex almost flat and compound eye slightly elongate in lateral view. Thorax and legs ( Figs 43 View FIGURE 43 , 44 View FIGURE 44 ): As in Paraphasma conspersum . Wings ( Figs 43 View FIGURE 43 , 44 View FIGURE 44 ): Tegmina short, not reaching median region of metanotum; in dorsal view about 2.5–2.8x longer than wide; posterior and apical margins rounded; shoulder pad developed into a sharp spine; anal region with conspicuous reticulate venation. Hindwing usually reaching anterior region of abdominal tergite VII. Abdomen ( Figs 44A–C View FIGURE 44 , 45 View FIGURE 45 ): About 1.8x longer than the combined length of head, thorax and median segment; dorsally and ventrally smooth, but tergite X sometimes slightly carinate on posterior region. Segments gradually shortening from II to VIII. Tergites VIII–X ( Fig. 45A,B,E View FIGURE 45 ) very short and distinctly wider than II–VII; tergite IX about as long as VIII and slightly longer than X. Tergite X strongly convex; narrower than VIII and IX; wider than long, narrowing towards the posterior region; posterior margin slightly emarginate in dorsal view. Thorn pads weakly developed, formed by 5–10 minute teeth grouped on a slightly swollen region on each side of posterior margin of tergite X. Cerci ( Fig. 45 View FIGURE 45 ) about as long as tergite X, circular in cross-section; somewhat pyriform, thicker basally and tapering towards apex; apex rounded, slightly incurved. Vomer ( Fig. 45F View FIGURE 45 ) subtriangular, approximately symmetric, wider than long, gradually narrowing from base to sharp apex; anterior margin distinctly concave; basal region formed by two narrow lateral projections. Sternite VIII ( Fig. 45B,C View FIGURE 45 ) distinctly shorter than corresponding tergite. Subgenital plate 2.5–3x longer than sternite VIII; distinctly divided into anterior and posterior region ( Fig. 45B–E View FIGURE 45 ). Anterior region of subgenital plate originating a symmetric pair of small lateral projections ( Fig. 45E View FIGURE 45 : arrowhead), directed posteriorly, covered by tergite IX and not reaching its posterior margin; lateral projections approximately styliform, slightly compressed laterally, with rounded apex. Posterior region of subgenital plate strongly convex; longer than anterior region; posterior margin truncate in lateral view, V-shaped in caudal view. Cerci, posterior margin of tergite X and subgenital plate densely pilose. Phallic organ ( Fig. 46 View FIGURE 46 ): Dorsal sclerite ( Fig. 46 View FIGURE 46 , in red) distinctly displaced to right side of phallic organ, about as wide as long, roughly triangular in dorsal view; distal process ( Fig. 46 View FIGURE 46 : dp) narrow and elongate, directed posteriorly and to the left, approximately in angle of 45º with longitudinal axis of organ. Dorsal and ventral lobes almost completely fused on left side. Dorsal lobe undivided ( Fig. 46 View FIGURE 46 : dl). Sclerite of the ventral lobe of “small-simple type ” ( Fig. 46 View FIGURE 46 , in green), covering only central region of inner face of ventral lobe; roughly oval in dorsal view, broader on right region; with two protuberances partially linked by a carina, the left one broader and rounded, the right one smaller, approximately lamellate. Base apodemes ( Fig. 46 View FIGURE 46 , in blue) forming two spatulate expansions, one directed anteriorly and the other posteriorly, projecting into dorsal lobe.
(NHM 012502909). A. Habitus, dorsal view. B. Anterior region of the body, dorsal view. C. Labels. Scale bars = 10 mm.
Redescription of female. Color ( Fig. 47 View FIGURE 47 ): As in male, but posterior region of subgenital plate light brown and some specimens with costal region of hindwings more strongly stained in dark brown. Head and thorax ( Fig. 47A–D View FIGURE 47 ): As in male. Legs ( Fig. 47A–C View FIGURE 47 ): As in male, but slightly shorter in relation to body and anterodorsal carina of profemur distinctly raised medially. Wings ( Fig. 47A–D View FIGURE 47 ): As in male, but hindwing reaching abdominal tergite VIII. Abdomen ( Fig. 47A–C,E–G View FIGURE 47 ): As in Paraphasma conspersum , but sternite VII with straight posterior margin (lacking rounded indentation).
Description of egg ( Fig. 48 View FIGURE 48 ). Capsule somewhat elongate, approximately cylindrical, about as high as wide or slightly higher than wide; slightly narrowing towards polar area, the latter almost flat; capsule densely covered with fairly thick bristles, longer around operculum and on polar area. Operculum approximately oval, slightly higher than wide, perpendicular in relation to longitudinal axis of capsule; covered with long bristles except for a small central area. Micropylar plate oval, somewhat elongate, positioned medially on capsule; smooth except for a slight longitudinal ridge which may bear 1–3 short bristles. Median line short and inconspicuous ( Fig. 48A View FIGURE 48 : arrow). Internal micropylar plate not examined. Capsule and operculum uniformly light brown; micropylar plate and median line in the same color or slightly darker. Measurements (mm, n = 4): capsule length, 2.7–2.8; capsule width, 1.6; capsule height, 1.6–1.7; operculum width, 1.1–1.2; operculum height, 1.3–1.4; micropylar plate length, 0.6–0.7; micropylar plate width, 0.4.
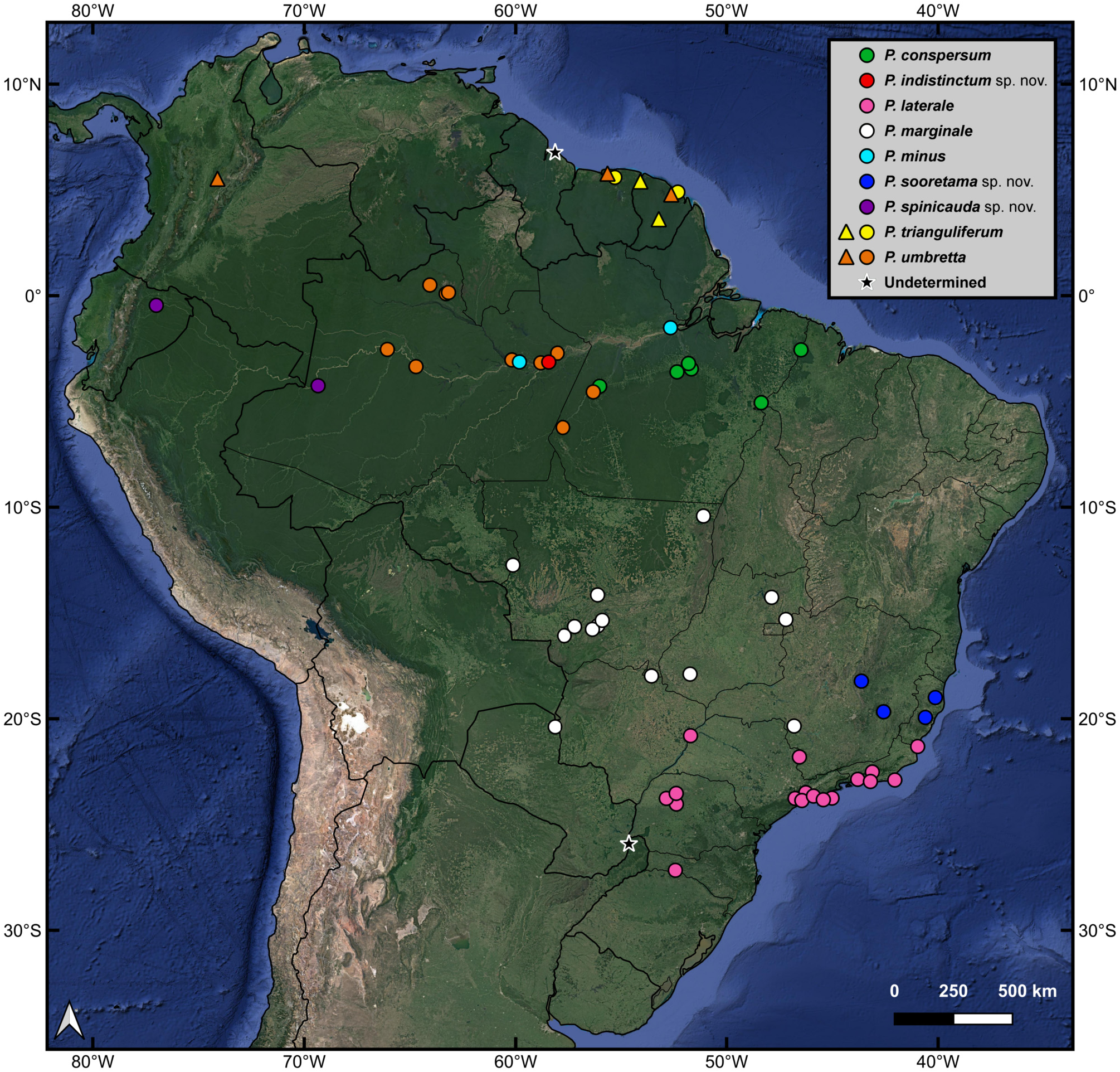
Distribution ( Fig. 1 View FIGURE 1 : orange circles, orange triangles). Paraphasma umbretta is distributed over a large part of the Amazon Forest. It is known from the Brazilian states of Amazonas and western Pará and is also recorded from Suriname (type locality), French Guiana ( Serville 1839) and north-central Colombia ( Conle et al. 2011).
Remarks. Fabricius (1787) described Mantis linearis , from India, which was later transferred to Phasma in a subsequent work of the same author ( Fabricius 1798) [this species is currently under the valid name Necroscia manicata ( Lichtenstein, 1802) due to being a junior primary homonym of Mantis linearis Drury, 1770 ]. Lichtenstein (1796) proposed the binomen Phasma umbretta for Stoll’s species ‘Double-Epine Brun’, from Suriname ( Stoll 1787: pp. 24–25, fig. 27), and also suggested that this species might be the male of Mantis linearis Fabricius [a clearly wrong interpretation, considering the very different collecting locality and that the specimen illustrated by Stoll (1787) is a female]. Later, Stoll (1813) himself proposed the name Phasma linearis for his species ‘DoubleEpine Brun’, simultaneously creating an objective synonym of Phasma umbretta Lichtenstein [as indicated by Westwood (1859)] and a primary homonym of Phasma linearis (Fabricius) .
Stoll (1813) mentioned Ambon Island as the place of origin of Phasma acanthophora Stoll, 1813 (= ‘Spectre Moucheté’: p. 62, fig. 78). However, the specimen illustrated is similar to Paraphasma umbretta and this type locality is likely to be wrong—the specimen is possibly from Suriname, like the type of Paraphasma umbretta . Therefore, we maintain the synonymy between these species, which was first pointed out by Redtenbacher (1906). The types of both Paraphasma umbretta and Phasma acanthophora are probably lost.
Gray (1835) described Phasma maculatum based on a male from the Brazilian Amazon. Redtenbacher (1906) transferred this species to Paraphasma and treated Paraphasma maculatum (Gray) as the valid name for Phasma umbretta Lichtenstein , Phasma linearis Stoll and Phasma acanthophora Stoll [in the case of P. acanthophora , the author reproduced the incorrect spelling of Gray (1835), “ Phasma acanthopterum ”].
Despite the suppression of the publication by Lichtenstein (1796) for nomenclatural purposes, Phasma umbretta and 18 other specific names published in that work were conserved [see Kerzhner (1994) and the ruling of the International Commission on Zoological Nomenclature ( ICZN 1995)]. Paraphasma umbretta ( Lichtenstein, 1796) is thus the valid name of this species, notwithstanding that its junior synonym Paraphasma maculatum (Gray) has been more commonly used in the literature (see e.g. Handlirsch 1930; Zompro 2004; Conle et al. 2011, 2020; Brock et al. 2016; Chiquetto-Machado & Cancello 2021), possibly as a consequence of Redtenbacher (1906) having used it as a valid name.
Additional material examined. BRAZIL. Amazonas: 1♂, 1♀, Barcelos, Rio Aracá, Boca Rio Curuduri , 00º05’50.2”N, 63º17’22.3”W, 15–19.vi.2010, Varredura, S. S. Oliveira, J. T. Câmara, J. A. Rafael, V. Linard ( INPA *) GoogleMaps ; 1♀, Barcelos, Rio Aracá, Comunidade Bacuquara , 00º09’09”N, 63º10’38”W, 11.viii.2007, Coleta manual, T. K. Krolow ( INPA) GoogleMaps ; 1♀, Barcelos, Rio Padauri, Parada Ararão , 23.ix.2006, Nogueira & Braga-Neto ( MZUSP 0337 View Materials ) ; 1♂, Fonte Boa, Estr. Mamopina , 023227S , 660408W , 21–24.ix.2005, Coleta manual, J. A. Rafael & F. F. Xavier Fº ( INPA *) ; 1♀, Itacoatiara , xii.1957 ( MZUSP 0359 View Materials ) ; 1♀, Itapiranga, Rod. AM 363, km 111 , Ramal Madubá , 02º42’57”S, 58º00’46”W, 8–9.x.2010, A. Agudelo ( INPA) GoogleMaps ; 1♂, Manaus, Cj. Tiradentes , 7.iv.2004, Rede entomológica, Ogawa, G. M. ( INPA) ; 1♂, Manaus, Instituto Nacional de Pesquisas da Amazônia—Campus I , 28.iv.2002, Coleta manual, Coletto-Silva, A. ( INPA 3619 View Materials *) ; 1♀, Tefé , ix.1958 ( MZUSP 0151 View Materials ) ; 1♂, Tefé , x.1958, Dirings ( MZUSP 0152 View Materials *) ; 3♀, Tefé , xii.1958 ( MZUSP 0354 View Materials , 0355 View Materials , 0358 View Materials ) ; 1♀, Tefé , vii.1959, Dirings ( MZUSP 0150 View Materials ) ; 1♀, Tefé , v.1962 ( MZUSP 0357 View Materials ) ; 1♀, Tefé ( MZUSP 0356 View Materials ) ; 4 eggs from females with locality “Amazonas” (NHMW). Pará: 1♂, Jacareacanga , x.1959, M. Alvarenga ( DZUP 379996 View Materials *) ; 1♂, Parque Nacional da Amazônia, Base do Uruá , 04º32’58”S, 56º18’14”W, 5.i.2016, P. I. Chiquetto-Machado, A. Z. Ramin ( MZUSP 0546 View Materials *) GoogleMaps .
| INPA |
Instituto Nacional de Pesquisas da Amazonia |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Paraphasma umbretta ( Lichtenstein, 1796 )
| Chiquetto-Machado, Pedro I., Morales, Adriana C. & Cancello, Eliana M. 2022 |
Prexaspes (Elasia) janus
| Otte, D. & Brock, P. D. 2005: 281 |
Paraphasma maculatum
| Chiquetto-Machado, P. I. & Cancello, E. M. 2021: 4 |
| Conle, O. V. & Hennemann, F. H. & Bellanger, Y. & Lelong, P. & Jourdan, T. & Valero, P. 2020: 70 |
| Brock, P. D. & Marshall, J. A. & Beccaloni, G. W. & Harman, A. J. E. 2016: 181 |
| Conle, O. V. & Hennemann, F. H. & Gutierrez, Y. 2011: 33 |
| Otte, D. & Brock, P. D. 2005: 251 |
| Zompro, O. 2004: 159 |
| Handlirsch, A. 1930: 760 |
| Redtenbacher, J. 1906: 116 |
Prexaspes umbretta
| Kirby, W. F. 1904: 414 |
Trigonophasma acanthophora
| Kirby, W. F. 1904: 373 |
Prexaspes maculatus
| Kirby, W. F. 1904: 414 |
Prexaspes janus
| Conle, O. V. & Hennemann, F. H. & Bellanger, Y. & Lelong, P. & Jourdan, T. & Valero, P. 2020: 126 |
| Kirby, W. F. 1904: 415 |
Necroscia umbretta
| Redtenbacher, J. 1906: 116 |
| Westwood, J. O. 1859: 147 |
Necroscia acanthoptera
| Redtenbacher, J. 1906: 116 |
| Westwood, J. O. 1859: 147 |
Phasma lineare
| De Haan, W. 1842: 123 |
| Gray, G. R. 1835: 26 |
Phasma acanthopterum, Gray, 1835: 23
| Conle, O. V. & Hennemann, F. H. & Bellanger, Y. & Lelong, P. & Jourdan, T. & Valero, P. 2020: 126 |
| Redtenbacher, J. 1906: 116 |
| De Haan, W. 1842: 123 |
| Serville, J. G. A. 1839: 268 |
| Burmeister, H. 1838: 586 |
| Gray, G. R. 1835: 23 |
Phasma maculatum
| Bates, H. W. 1865: 350 |
| Westwood, J. O. 1859: 122 |
| De Haan, W. 1842: 123 |
| Serville, J. G. A. 1839: 269 |
| Gray, G. R. 1835: 26 |
Phasma umbretta
| Conle, O. V. & Hennemann, F. H. & Bellanger, Y. & Lelong, P. & Jourdan, T. & Valero, P. 2020: 126 |
| Conle, O. V. & Hennemann, F. H. & Gutierrez, Y. 2011: 33 |
| Otte, D. & Brock, P. D. 2005: 251 |
| ICZN 1995: 283 |
| Kerzhner, I. M. 1994: 111 |
| Redtenbacher, J. 1906: 116 |
| De Haan, W. 1842: 123 |
| Gray, G. R. 1835: 26 |
| Lichtenstein, A. A. H. 1802: 14 |
| Lichtenstein, A. A. H. 1796: 78 |
| Stoll, C. & Qui se trouvent dans les quatre parties du monde, L'Europe 1787: 24 |
Phasma linearis
| Conle, O. V. & Hennemann, F. H. & Bellanger, Y. & Lelong, P. & Jourdan, T. & Valero, P. 2020: 126 |
| Conle, O. V. & Hennemann, F. H. & Gutierrez, Y. 2011: 33 |
| Otte, D. & Brock, P. D. 2005: 251 |
| Redtenbacher, J. 1906: 116 |
| Kirby, W. F. 1904: 415 |
| Westwood, J. O. 1859: 147 |
| Stoll, C. & Qui se trouvent dans les quatre parties du monde, L'Europe 1787: 24 |