Tosana dampieriensis, Gill & Pogonoski & Johnson & Tea, 2021
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4996.1.2 |
|
publication LSID |
lsid:zoobank.org:pub:9233EEEF-BBDF-4D43-BE77-1931B26BCB9B |
|
persistent identifier |
https://treatment.plazi.org/id/03AAEB28-384F-655F-FF7E-6352FB5FF8AC |
|
treatment provided by |
Plazi |
|
scientific name |
Tosana dampieriensis |
| status |
sp. nov. |
Tosana dampieriensis n. sp.
Figures 8–10 View FIGURE 8 View FIGURE 9 View FIGURE 10 ; Tables 1, 4–8
English common name: Dampierian Threadtail Anthias urn:lsid:zoobank.org:act:1E4DDCAF-936D-4ABF-8065-F92B5D0C9147
Holotype. NMV A.29686-007, 46.2 mm SL, Australia, Western Australia, from 18°25.52′S, 120°05.92′E to 18°26.20′S, 120°06.50′E, 103–105 m, beam trawl, M.F. Gomon, RV Southern Surveyor , 19 Jun 2007 ( Fig. 9A View FIGURE 9 ). GoogleMaps
Paratypes (n = 2). CSIRO H 6454-05 View Materials , 47.5 mm SL, Australia, Western Australia, west of Exmouth Gulf , from 21°58.70′S, 113°49.20′E to 21°59.05′S, 113°49.20′E, 170–177 m, beam trawl GoogleMaps , CSIRO personnel, FRV Southern Surveyor , 10 Dec 2005 ( Fig. 9B View FIGURE 9 ) (COI: BOLD FOAO1480-18 ) ; CSIRO H 8279-02 View Materials , 44.6 mm SL, Australia, Western Australia, north of Dampier Archipelago , from 19°56.74′S, 116°39.40′E to 19°56.92′S, 116°39.23′E, 66 m, benthic sled, A. Graham & J. Pogonoski GoogleMaps , RV Investigator , 26 Oct 2017 (COI: BOLD FOAO1570-18 ) .
Diagnosis. Tosana dampieriensis is distinguished from congeners in having a combination of 24 circumpeduncular scales, and a pectoral-fin length of 28.4–28.9% SL.
Description. Frequency distributions for selected meristic characters are provided in Table 5. Dorsal rays X,14, all but first segmented rays branched (all branched in paratypes); anal rays III,7, all segmented rays branched; pectoral rays 16/16 (16–17), all rays branched except upper 2/2 and lower 9/9 (variable in paratypes, see Table 4); pelvic rays I,5; principal caudal rays 9+8; branched caudal rays 7+6; upper procurrent caudal rays 9; lower procurrent caudal rays 9 (9–10); total caudal rays 35 (35–36); lateral-line scales 36/36 (38–39); scales above lateral line to origin of dorsal fin 5/4 (5); scales above lateral line to base of fifth dorsal spine 3/3 (3–4); scales below lateral line to origin of anal fin 16/17 (16–17); circumpeduncular scales 24; gill rakers 10+25 (10+23–25); branchiostegal rays 7; pseudobranch filaments 16 (15).
Morphometric values are presented in Table 6.
Vertebrae 10+16; supraneurals 2; predorsal formula 0/0/2/1+1; main shaft (proximal component) of first dorsal pterygiophore almost vertical, the distal end slightly posterior to vertical through proximal tip; dorsal pterygiophores in interneural spaces 9–13 1/1/1+1/1/1+1; terminal dorsal pterygiophore in interneural space 18; no trisegmental pterygiophores associated with dorsal fin; proximal tip of first anal pterygiophore between parapophyses and haemal spine on vertebra 11; terminal anal pterygiophore in interhaemal space 5; no trisegmental pterygiophores associated with anal fin; ribs present on vertebrae 3 through 10; epineurals present on vertebrae 1 through 12; paired parapophyses present on first caudal vertebra; parhypural and hypurals autogenous; well-developed hypurapophysis on parhypural; epurals 3; single uroneural (posterior uroneural absent); ventral tip of cleithrum with well-developed posteroventral process ( Fig. 10 View FIGURE 10 ).
Dorsal-fin origin slightly in front of vertical through posterior edge of operculum; third or fourth dorsal spine longest; distal tips of dorsal spines with fleshy tabs; dorsal fin not incised before first segmented ray; soft dorsal fin pointed, 10 th to 13 th ray longest; second or third anal spine longest, second spine stoutest; anal fin pointed, with third or fourth segmented ray longest; caudal fin lunate with filamentous tips; pectoral bluntly pointed, reaching area ranging from anus to anal-fin origin; second pelvic ray longest, reaching anus.
Mouth large, oblique, posterior margin of maxilla reaching vertical through posterior edge of pupil; mouth terminal, lower jaw projecting slightly; premaxilla with pair of enlarged canines anterolaterally, a band of small conical teeth about 3 or 4 rows wide at symphysis reducing to 1 or 2 rows on sides of jaw, with the outer row of teeth much larger, those of posterior half of jaw curved anteriorly; dentary with 1 or 2 laterally curved, enlarged canines at front of jaw, followed by band of small conical teeth about 3 or 4 rows wide reducing to 1 or 2 rows posteriorly, the outer-row teeth much larger, those of posterior part of jaw curved anteriorly; anterior third of dentary with 1 or 2 enlarged, posteriorly curved canines outside band of teeth; vomer with small triangular patch of small conical teeth, about 3 rows wide in midline; palatine with a narrow band of small conical teeth, 2 or 3 rows wide at widest point; ectopterygoid, mesopterygoid and tongue edentate.
Opercle with 3 flat spines, middle spine longest; preopercle with 26/27 (22–23) fine serrations, those largest near preopercle angle, weakly developed or absent on ventral edge of bone; interopercle with 1 or 2 weak serrations; subopercle with no or 1 serration; posttemporal without serrations. Anterior nostril positioned at middle of snout, tubular with small flap on posterior rim, flap reaching about 1/2 distance to posterior nostril when laid back; posterior nostril at mid-upper, anterior border of orbit, covered by thin, narrow membrane anteriorly, membrane bearing small flap. No papillae on posterior rim of orbit. Snout not hypertrophied with papillae at symphysis.
Scales ctenoid with peripheral cteni ( Roberts, 1993); lateral line broadly arched over pectoral fin following body contour to caudal-fin base; head scaly, including mandibles, maxilla, snout, and infraorbitals, no scales on lips; no auxiliary scales on body, a few auxiliary scales on cheeks and operculum; low scaly sheath on soft dorsal and anal fins, with indistinct small scales present between segmented rays; caudal fin with scaly basal sheath, with small scales extending over almost all of fin, except for fin tips and posterior part of membranes of middle rays; pectoral fins with basal, wedge-shaped sheath of small scales.
Colour of holotype when fresh (based on photograph when freshly dead; Fig. 9A View FIGURE 9 ): Head orange-brown with yellow stripe behind eye to upper part of opercle, connecting ventrally with indistinct yellow bar on cheek and preopercle; subopercle and area immediately above yellow stripe pinkish purple; iris bright yellow, dusky grey to black dorsally and ventrally, with iridescent blue-black crescent in front of pupil; body orange, with yellow stripe from behind eye continuing mid-laterally to mid-side; yellow stripe narrowly edged dorsally with pinkish purple; breast and sides above anal fin pinkish purple; scales of upper part of caudal peduncle and body beneath dorsal fin each with indistinct orange-brown crescent; spinous part of dorsal fin pink, yellow on distal half of fin, with yellowbrown spot in front of base of each fin spine; soft part of dorsal fin pink, with 2–3 rows of yellow spots; anal fin hyaline; caudal-fin base with indistinct pale pink blotch dorsally and ventrally, remainder of fin bright yellow, paler on dorsal and ventral margins and on distal half of concave, central part of fin; pelvic fins whitish hyaline; pectoral fins hyaline. Paratypes when fresh (based on photographs taken between 7 months and 13 years after being frozen; Fig. 9B View FIGURE 9 ): Head pale tan, paler ventrally, with yellow stripe from snout tip to anterior edge of eye, continuing behind eye to upper part of opercle; iris dark grey, bright yellow posteriorly where adjacent to yellow stripe; body pale tan, pale yellow to white ventrally and on breast; yellow stripe from behind eye continuing mid-laterally to caudal-fin base, indistinct posteriorly; body with pinkish purple stripe immediately beneath yellow stripe, becoming indistinct on caudal peduncle; scales of upper part of caudal peduncle and body beneath dorsal fin each with grey-brown crescent; fins similar to holotype, but paler.
Colour in preservative: Head and body pale tan; brown crescents on upper body and caudal peduncle remain, becoming dusky brown; fins pale tan to hyaline, without markings.
Habitat and distribution. Known only from three specimens trawled off Western Australia between 18°25′S and 22°S in 66–177 m ( Fig. 8 View FIGURE 8 ). Specimens were collected by benthic sled and beam trawls .
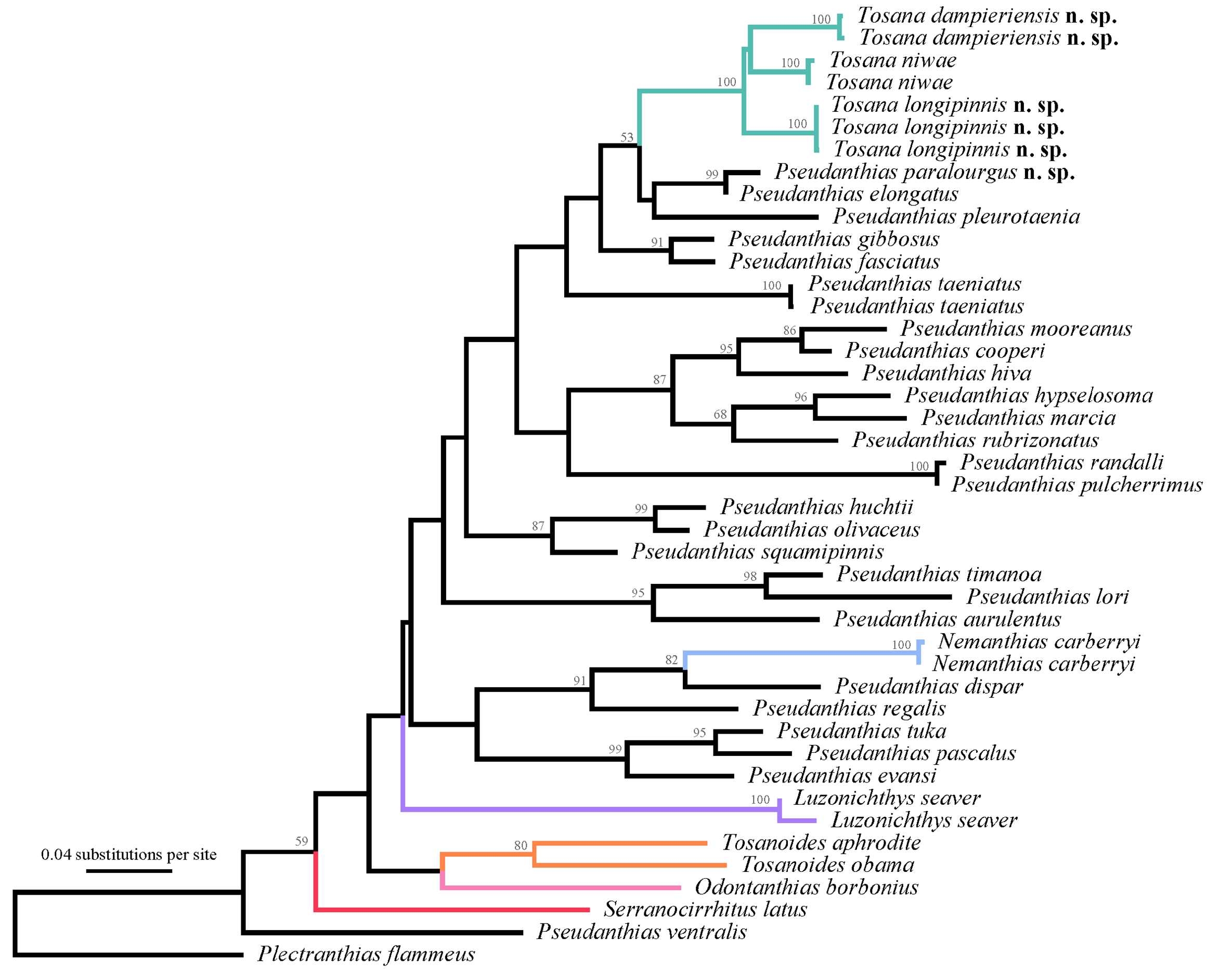
Comparisons. Analysis of mitochondrial COI sequence data retrieved Tosana as monophyletic, with T. niwae and T. dampieriensis forming the sister group of T. longipinnis ( Fig. 1 View FIGURE 1 ). Uncorrected pairwise distances for T. niwae to T. longipinnis and T. dampieriensis were 6.5–6.9% and 6.6–7.4%, respectively. The pairwise distance for T. longipinnis and T. dampieriensis was 7.4–7.6%.
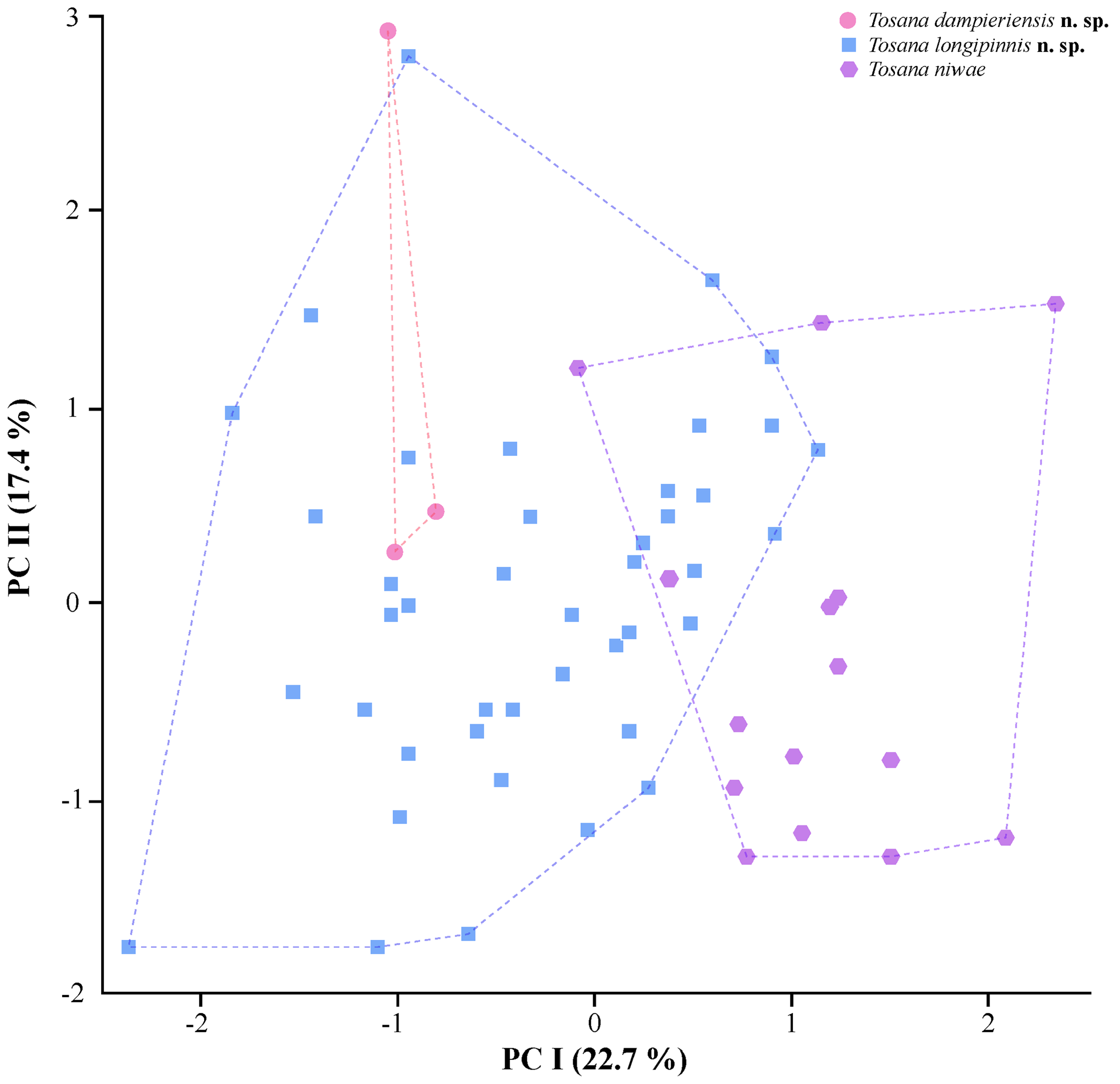
The three species of Tosana are very similar in morphology ( Tables 4–6). There is possibly little difference in live coloration, although we are unable to adequately assess this owing to our relatively poor understanding of fresh coloration in the two new species. Comparisons between the three species are also hampered by the small number and relatively small size of available specimens of T. dampieriensis , and we therefore appeal partly to the observed genetic differences as justification for our taxonomic decisions. Principal component analyses of meristic characters failed to fully distinguish the three species ( Fig. 11 View FIGURE 11 ). Although there was no overlap in PC I scores for T. dampieriensis and T. niwae , T. longipinnis overlapped with both species. Nonetheless, most specimens of T. longipinnis were distinguished from T. niwae on PC I. Eigenvalues for PC I and PC II for each character are provided in Table 7. PC I accounted for 22.7% of variance, whereas PC II accounted for 17.4% SL (cumulative variance explained 40.1%). Three characters had high loadings on PC I: lateral-line scales; scales below lateral line to origin of anal fin; circumpeduncular scales. Frequency distributions for these and other meristic characters are given in Table 5. Tosana niwae tends to have lower numbers of lateral-line scales: 78% of specimens had 36 or fewer lateral-line scales, whereas T. dampieriensis had 36–39 scales, and 93% of T. longipinnis specimens had 37 or more scales. Tosana niwae differed from T. dampieriensis in having fewer scales below the lateral line to the origin of the anal fin (12–16, of which 92% had 15 or fewer scales, versus 16–17 in T. dampieriensis ) and fewer circumpeduncular scales (21–23 versus 24).
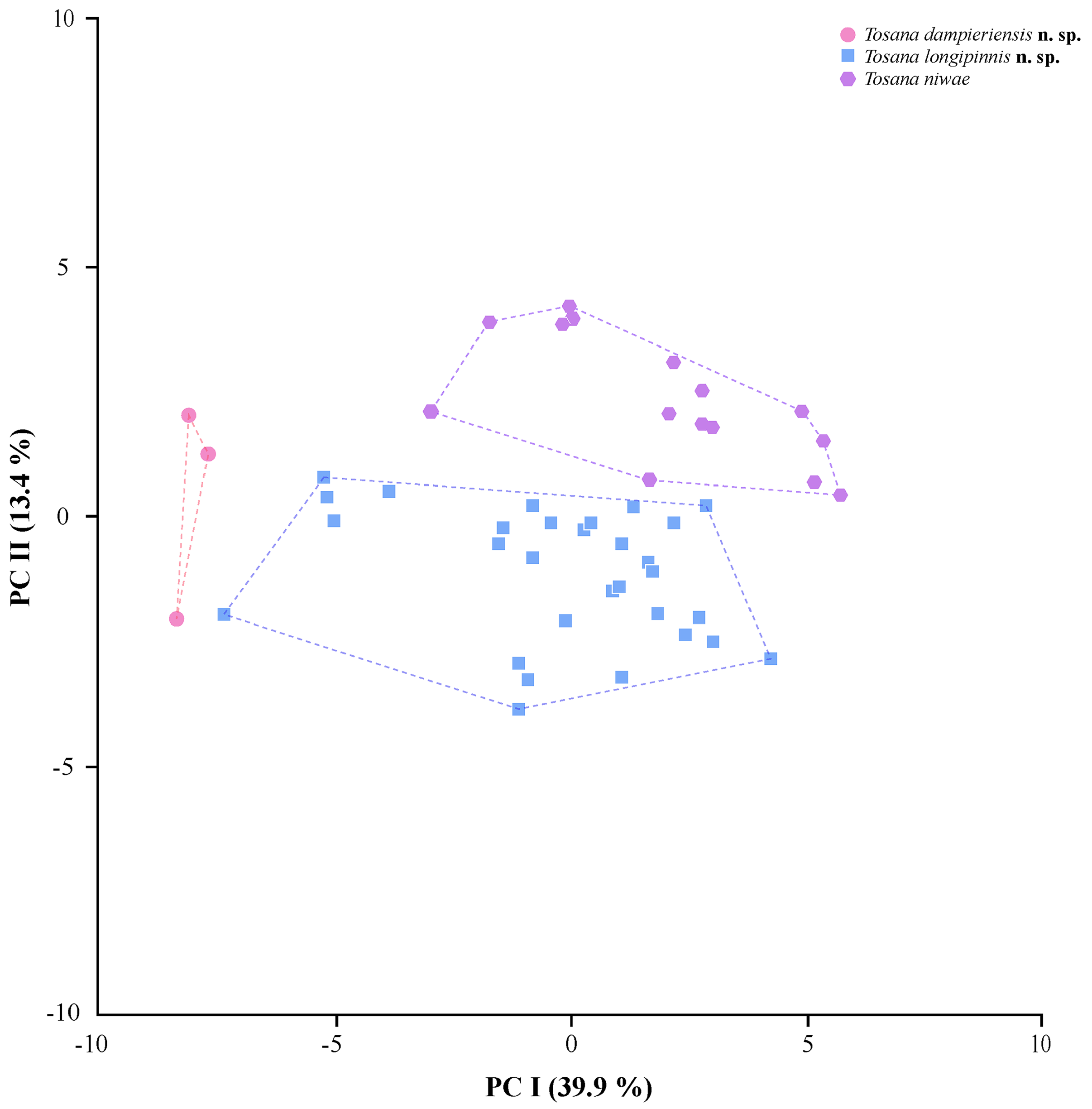
The morphometric PCA distinguished all three species, with T. dampieriensis distinguished from the other two species on the first principal component, and T. longipinnis and T. niwae distinguished from each other on the second principal component ( Fig. 12 View FIGURE 12 ). Eigenvalues for each character on the first two principal components are provided in Table 8. PC I accounted for 39.9% of variance, whereas PC II accounted for 13.4% (cumulative variance explained 53.3%). Of the characters loading highest on PC I, two were diagnostic for T. dampieriensis : length of the longest dorsal ray (16.4–18.3% SL for T. dampieriensis , versus 24.4–34.9% SL for T. niwae , and 17.8–38.9% SL for T. longipinnis ) and length of longest anal ray (21.1–22.0% SL for T. dampieriensis , versus 22.1–56.6% SL for T. niwae and 23.8–52.2% SL for T. longipinnis ). Of characters loading highest on PC II, three were apparently useful in diagnosing Tosana species : head length shorter in T. niwae (28.2–30.1% SL) than T. dampieriensis (32.0– 33.3% SL), though more variable in T. longipinnis (28.8–32.4% SL); upper-jaw length generally shorter in T. niwae (13.2–14.6%) than T. dampieriensis (14.9–15.8% SL) and T. longipinnis (14.1–16.0% SL); pectoral fin relatively short in T. niwae (22.3–27.7% SL), intermediate in T. dampieriensis (28.4–28.9% SL), and long in T. longipinnis (28.9–35.3% SL). However, the diagnostic reality of each of these characters must be tempered by the possibility that they may reflect allometric growth and the small size of the three specimens of T. dampieriensis (44.6–47.5 mm SL versus 54.9–118.1 mm SL and 68.5–127.8 mm SL, respectively, for T. longipinnis and T. niwae ).
Etymology. The specific epithet refers to this species’ distribution, which lies within the Australian marine biogeographical Dampierian Region. Named for the English explorer William Dampier, the area was originally proposed by malacologist Charles Hedley “for the marine fauna which extends from Torres Straits to Houtman’s Abrolhos” ( Hedley, 1904: 880), and is still recognised in this sense by historical biogeographers ( Ebach et al., 2013; Gill & Mooi, 2017).
Remarks. Although there are fresh colour photos of all three specimens, none are satisfactory. The holotype photo ( Fig. 9A View FIGURE 9 ) is heavily saturated, even after attempts to correct for saturation. The photos of the paratypes were taken after the specimens had been frozen for between 7 months and 13 years and had faded.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |