Macrosiphoniella (Ramitrichophorus) janckei Börner, 1939
|
publication ID |
https://doi.org/ 10.11646/zootaxa.5183.1.23 |
|
publication LSID |
lsid:zoobank.org:pub:AD0BED04-C49A-4068-8D3A-22DE8B172744 |
|
DOI |
https://doi.org/10.5281/zenodo.7075536 |
|
persistent identifier |
https://treatment.plazi.org/id/03A58798-FFFC-FFD0-8CCA-8804FBE3AE7B |
|
treatment provided by |
Plazi |
|
scientific name |
Macrosiphoniella (Ramitrichophorus) janckei Börner, 1939 |
| status |
|
Macrosiphoniella (Ramitrichophorus) janckei Börner, 1939 View in CoL
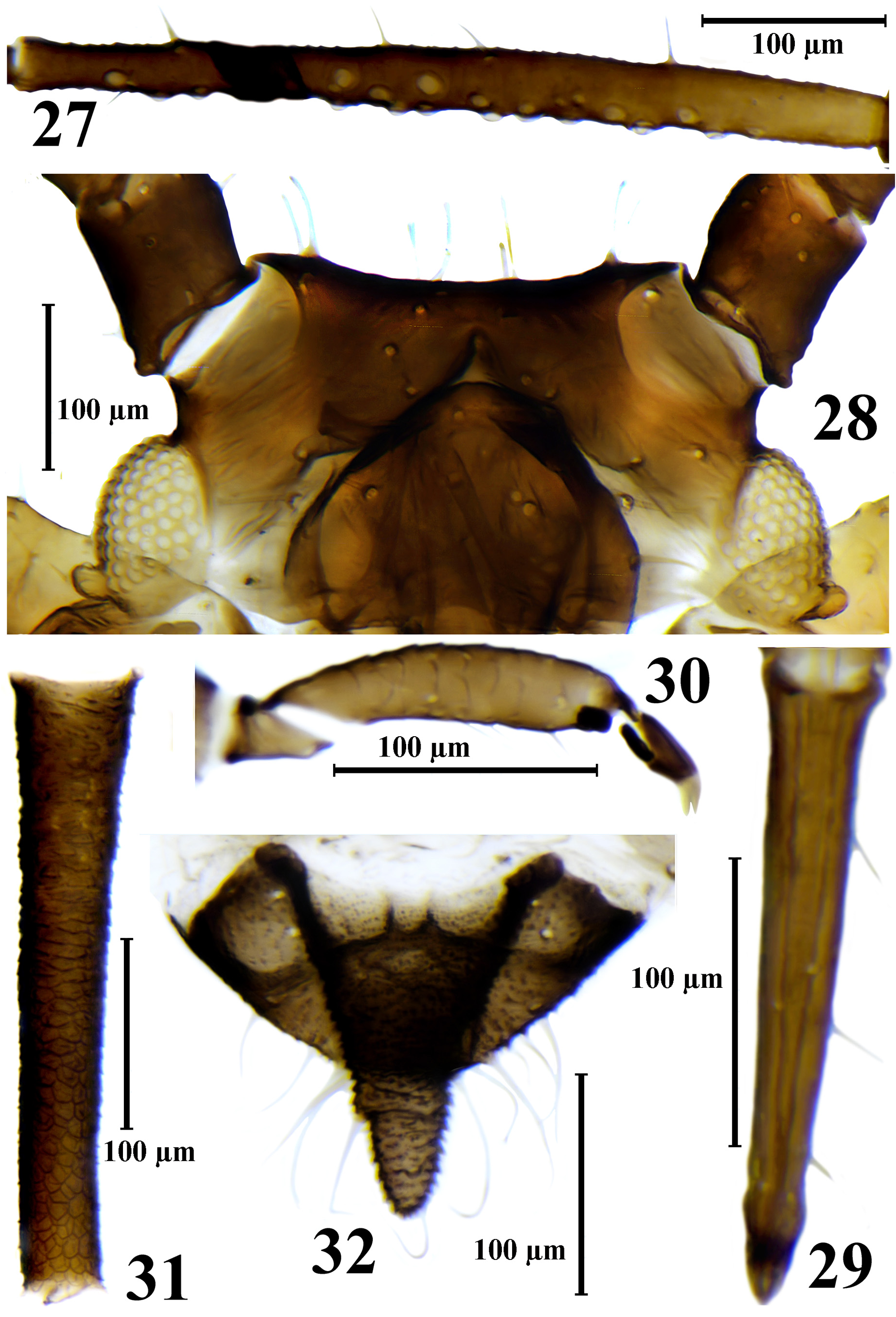
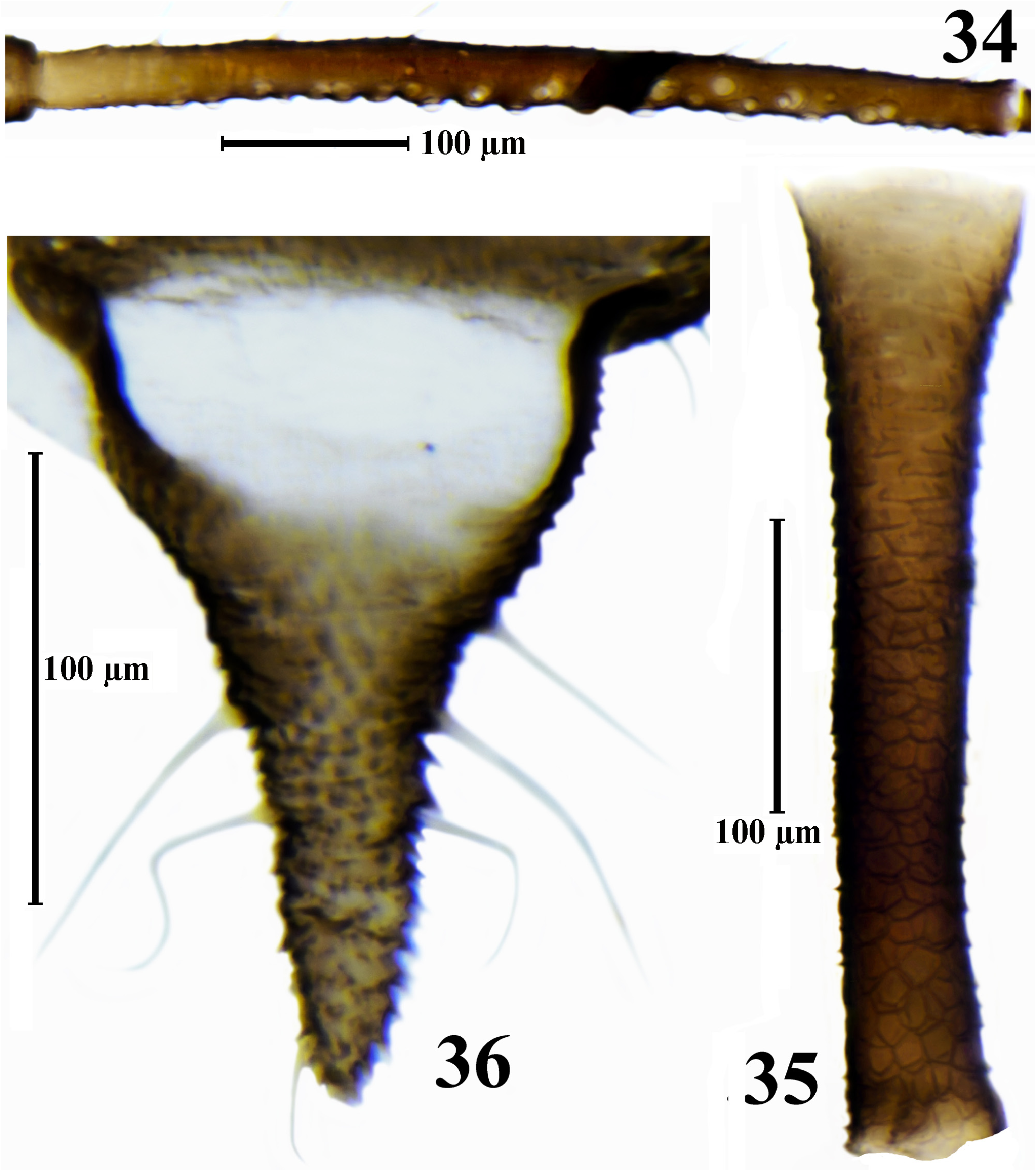
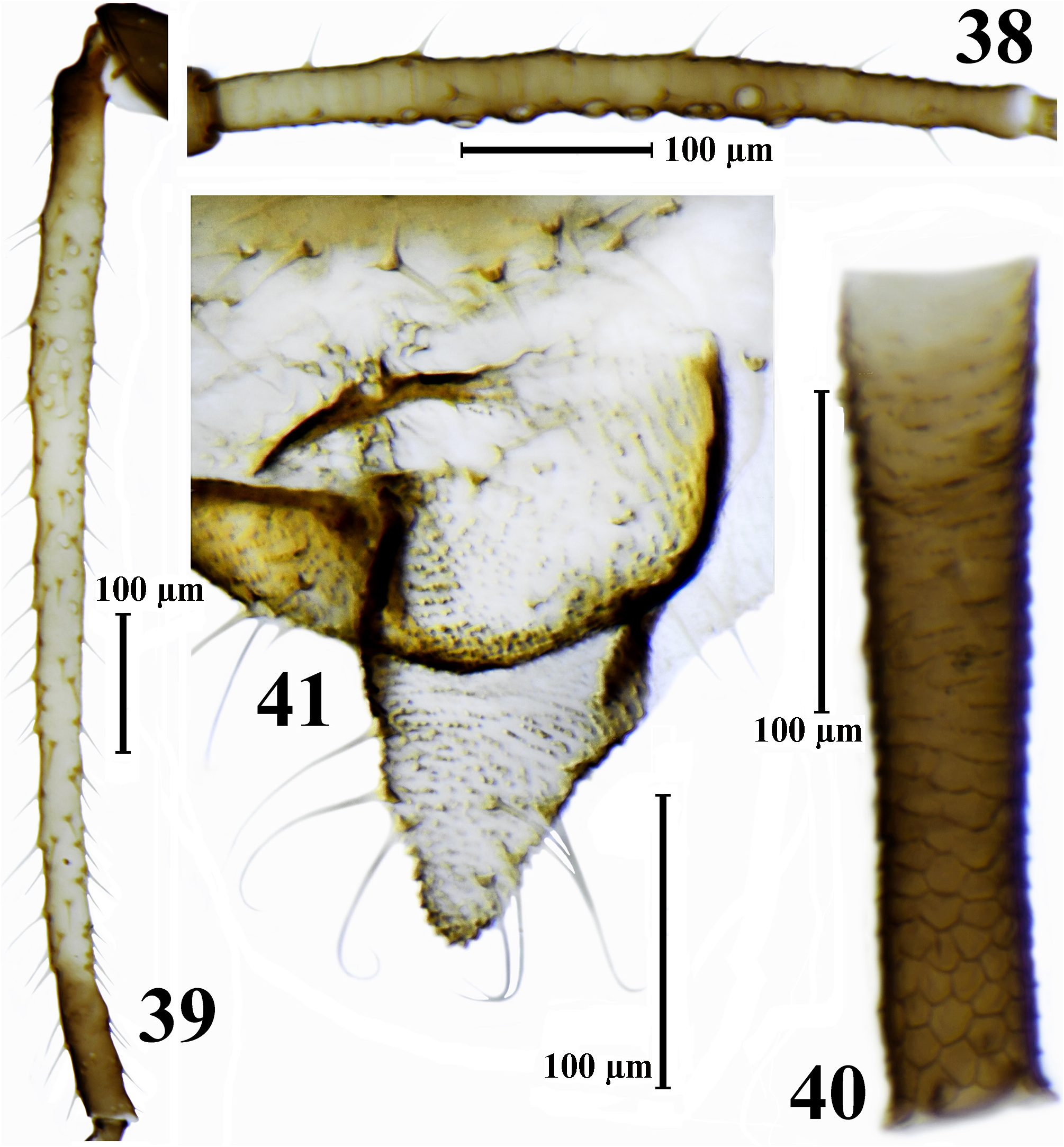
( Figs. 2 View FIGURES 1–3 , 25–41 View FIGURE 25 View FIGURE 26 View FIGURES 27–32 View FIGURE 33 View FIGURES 34–36 View FIGURE 37 View FIGURES 38–41 , Tabl. 3 View TABLE 3 )
Material. Syntype: Macrosiphoniella (Ramitrichophorus) janckei Börner, 1939 —3 apterous viviparous female, „ Helichrysum arenarium, Kolberg , Allemagne, Jancke leg., Macrosiphoniella janckei ” (from collections of MNHN and NHM). Additional materials: 3 apterous viviparous females, without date, Poland, West Pomeranian Voivodeship, Kołobrzeg County, Kołobrzeg (Kolberg), Helichrysum arenarium (from collections of IE BC CAS); 2 apterous viviparous females, 8.vi.1954, Russia, Stavropol Krai, Stepnovsky District, Irgakly, Helichrysum sp. , (from collections of NHM); 4 apterous viviparous females, 29.vi.1960, Germany, Hesse, Darmstadt, Frankfurt am Main, Helichrysum arenarium (from collections of NHM); 3 apterous viviparous females, 10.viii.1965, Poland, West Pomeranian Voivodeship, Kamień County, Międzyzdroje, Helichrysum arenarium (from collections of IE BC CAS); 15 apterous viviparous females, 03.vi.1979, Moldova,Autonomous Territorial Unit of Gagauzia, Vill. Bugeac, Helichrysum arenarium (from collections of MNHN); 15 apterous viviparous females, 07.vii.2004, Lithuania, Klaipėda County, Neringa, Vill. Pervalka, Helichrysum arenarium (from collections of IE BC CAS and ZIN RAS), 1 apterous viviparous female, 08.vii.2004, Lithuania, Klaipėda County, Neringa, Nida, Helichrysum arenarium (from collections of IE BC CAS); 1 apterous viviparous female, 17.vii.1996, Russia, Pskov Province, Nevel` District, near Dubokray Vill., Helichrysum arenarium (from collections of BSU); 1 apterous viviparous female, 14.vii.2014, Belarus, Minsk Region, Maladzyechna District, near Udranka Vill., Helichrysum arenarium (from collections of BSU); 38 apterous viviparous females, 1 alate viviparous female, 16.vii.2015, Belarus, Vitebsk Province, Gorodock District, near Zadrach`e Vill., Helichrysum arenarium (from collections of BSU); 57 apterous viviparous females, 1 oviparous female, 28.viii.2016, Russia, Pskov Province, Nevel` District, near Dubokray Vill., Helichrysum arenarium (from collections of BSU); 18 apterous viviparous females, 30.viii.2016, Russia, Pskov Province, Nevel` District, near Dubokray Vill., Helichrysum arenarium (from collections of BSU). Also used data from Börner (1939), Bozhko (1976), and Heie (1975).
Etymology. The species was named in honor of the famous German biologist Oldwig Jancke, who first collected the apterous viviparous females of this species.
TABLE 3 (Continued)
TABLE 3 (Continued)
Description. Apterous viviparous female. Body elliptical or elongate elliptical, 1.5–2.0 times as long as wide. The living specimens from pale gray-olive to reddish brown or black, with green spots at siphuncular bases; waxpowdered except in the middle of the abdominal dorsum and along body margins, in wax-powder pale ash gray. Cleared specimens with dark brown antennae (except base of 3rd antennal segment), distal half of the rostrum, legs (except base of femora) and siphunculi, with brown head, base of 3rd antennal segment, sclerites at the base of the coxae, peritremes, anal and subgenital plates and cauda, with light-brown or rarely brown bands and scleroites on thorax and abdomen. Thorax with more or less wide bands on pro- and mesonotum and row of sclerites on metanotum; abdomen with rows of spinal sclerites, with small marginal sclerites and peritrems on I–VI segments, with antesiphuncular sclerites and with bands on VII–VIII tergites; sclerites on the thorax and abdomen are often small, pale and rare or, especially marginal sclerites, absent; sclerites on VII tergite almost always fused to form a short band. Surface of head, thoracic dorsum and abdominal tergites I–V smooth, sometimes weakly wrinkled, on VI–VII abdominal tergites with rows of small pointed spinules, which on VIII abdominal tergite partially fuse to form scales; thoracic venter smooth, ventral abdomen with long rows of small hardly visible spinules, sometimes forming strongly elongate cells. Setae on head, dorsal side of thorax and abdomen relatively long, with bifurcate or flabellate apices, on ventral side finely pointed. Third antennal segment with 10–30 secondary rhinaria, fourth segment with 0–4, fifth with 0–1 rhinaria. Rhinaria with internal diameter 6–30 μm, with external diameter 1.7–8.2 times as long as high of rhinaria. Setae on antennae blunt, bifurcate or apically flabellate. Rostrum is often very long, reaching abdominal segment III–V. Ultimate rostral segment 4.6–6.7 times as long as its basal width. Setae on legs blunt or bifurcate, rarely pointed, finely pointed or apically flabellate. Peritremes on abdominal segments I and II separated by a distance equal to or less than diameter of peritreme. Siphunculi with polygonal reticulation on distal 0.33–0.55 of length (largest transverse row in reticulate part of siphunculi consisting of 4–8 cells), distinctly imbricated, with short rows of pointed spinules at the base that fused into large scales in front of the reticular part. Subgenital and anal plate with finely pointed, bifurcate or weakly flabellate apically setae. Cauda nearly triangular, with some constriction in the middle, which weakly divides it into broad base and conical and rounded on apex distal part; setae on cauda long, finely pointed, bifurcate or weakly flabellate apically.
Alate viviparous female. Body elongate elliptical, 2.2 times as long as wide. Colour of living specimen unknown. Cleared specimens with dark brown thorax; abdomen with distinct brown marginal sclerites on segments I–VII, spinal sclerites on abdominal tergites I–VI and antesiphuncular sclerites absent. Third antennal segment with 29 secondary rhinaria, 4th segment with 7–8 and 5th segment with 1 secondary rhinaria. Rostrum reaching abdominal segment I. Peritremes on abdominal segments I and II separated by a distance less than diameter of peritreme or fused.
Male. Apterous ( Müller 1975).
Oviparous female. Body 1.8 times as long as wide. Colour of living specimen unknown. Tibiae of all legs dark brown only on the apices and brown or light-brown in the middle. Spinal sclerites on abdominal tergites I–VI, marginal sclerites on all abdominal segments, antesiphuncular sclerites, and band on abdominal tergite VII absent. Setae on head, dorsal side of thorax and abdomen not only with bifurcate or flabellate, but also with blunt or weakly capitate apices; setae on abdominal tergite VIII with blunt, bifurcate or weakly capitate apices. Third antennal segment with 11–12 secondary rhinaria, fourth and fifth segments with 0 rhinaria. Setae on legs blunt, pointed or finely pointed. Subgenital and anal plate with finely pointed or bifurcate setae. Cauda triangular, without constriction in the middle. Hind tibiae weakly swollen on basal half, with 23–27 round or oval pheromone plates, more of them located on basal half.
Systematic relationships. The species is very close to Macrosiphoniella (Ramitrichophorus) medvedevi ( Bozhko, 1950) . The differences between them are given in the keys.
Distribution. Germany (Hesse, Darmstadt, Frankfurt am Main; Mecklenburg-Vorpommern: Mecklenburgische Seenplatte and Rostock, Laage) ( Müller 1964, 1975; Szelegiewicz 1968), Poland (West Pomeranian Voivodeship: Kołobrzeg County, Kołobrzeg [Kolberg] and Kamień County, Międzyzdroje; Kuyavian-Pomeranian Voivodeship, Bydgoszcz) ( Börner 1939, 1952; Szelegiewicz 1968, 1978; Nast et al. 1990; Węgierek & Wojciechowski 2004; Wrzesińska & Sawilska 2009; Osiadacz & Hałaj 2010), Lithuania (Klaipėda County, Neringa, Vill. Pervalka and Neringa, Nida)( Rakauskas et al. 2008, as Macrosiphoniella (Ramitrichophorus) janckei and M. (R.) hillerislambersi ), Belarus (Minsk Region, Maladzyechna District, near Udranka Vill. ( Buga & Stekolshchikov 2012), Moldova (Autonomous Territorial Unit of Gagauzia, Vill. Bugeac) ( Vereshchagin et al. 1985; Andreev & Vereshchagin 1993 as Macrosiphoniella medvedevi ), Ukraine (forest-steppe and steppe zones) ( Bozhko 1963, 1976), Russia (Pskov Province, Nevel` District, near Dubokray Vill.; Stavropol Krai, Stepnovsky District, Irgakly), Kazakhstan (West Kazakhstan Region, Terekti District, east of Uralsk; Atyrau Region, Makhambet District, near Makhambet Vill. and Isatay District, near Isatai Vill.; Aktobe Region, Shalkar District, near Shalkar; Akmola Region, Birzhan sal District, east of Stepnyak; Karaganda Region, Ulytau District, south-west of Ulytau Vill.; Almaty Region, right bank of the river Ili) ( Kadyrbekov 2003, 2017).
Ivanovskaya (1977) records this species from the Altai Republic ( Russia) on the basis of nine immature of the third instar collected on an unknown Asteraceae . On the basis of these data, many researchers in their later publications indicate Western Siberia as part of the area of this species. However, any reliable identification based on such material seems very doubtful and, therefore, there are no reliable data on the finding of this species in Western Siberia and Altai (see Stekolshchikov & Novgorodova 2015).
Biology. The species lives on stems, inflorescences and the lower surface of leaves of Helichrysum arenarium (L.) Moench, actively attended by ants ( Szelegiewicz 1968, Bozhko 1976, Kadyrbekov 2003, Buga & Stekolshchikov 2012). This species has never been observed on other species of the genus Helichrysum or plants of other genera. Colonies of these aphids in Belarus and the Pskov Province of Russia were always collected from beneath the inflorescences and from stems of H. arenarium . All these colonies were attended by ants Formica cinerea Mayr. Apterous males and oviparous females of M. (R.) janckei were found in Germany (Rostock, Laage) by Müller (1975) on 28.ix.1970, and a single oviparous female only was found together with apterous viviparous females in Russia (Pskov Province, Nevel` District, near Dubokray Vill.) in August 2016.
TABLE 3. Biometric data for apterous and alate viviparous females and oviparous females of Macrosiphoniella
| Apterous viviparous females | Alate viviparous females | Oviparous female | |
|---|---|---|---|
| Number of samples / specimens | 12/154 + data of Börner (1939), Bozhko (1976), and ( Heie, 1995) | 1/1 | 1/1 |
| Length of body | 1492–2157 | 2020 | 1782 |
| Length of antenna | 1164–2094 | 2085 | 1735 |
| Length of antenna / length of body | 0.68–1.24 | 1.03 | 0.97 |
| length | 447–624 | 588 | 518–523 |
| HFem length / length of body | 0.27–0.37 | 0.29 | 0.29 |
| length / W Head | 1.10–1.43 | 1.44 | 1.14–1.15 |
| length HTib length / length of body | 761–1066 | 1010–1015 | 853 |
| 0.44–0.61 | 0.50 | 0.48 | |
| W Head | 369–488 | 408 | 455 |
| Depth of frontal sinus / width between antennal bases | 0.02–0.12 | 0.12 | 0.06 |
......Continued on the next page
| MNHN |
Museum National d'Histoire Naturelle |
| NHM |
University of Nottingham |
| IE |
Cepario de Hongos del Instituto de Ecologia |
| CAS |
California Academy of Sciences |
| ZIN |
Russian Academy of Sciences, Zoological Institute, Zoological Museum |
| RAS |
Union of Burma Applied Research Institute |
| BSU |
Belgorod State University |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |