Ceropsylla, Rileyi, 1885
|
publication ID |
https://doi.org/10.1080/00222933.2019.1676931 |
|
DOI |
https://doi.org/10.5281/zenodo.3664907 |
|
persistent identifier |
https://treatment.plazi.org/id/0399EA14-FFC0-BE25-CC60-FF03FE4E8E14 |
|
treatment provided by |
Valdenar |
|
scientific name |
Ceropsylla |
| status |
|
Ceropsylla View in CoL View at ENA sp.; Calácio 2016 ı p. 15; Naves 2017 ı p. 9.
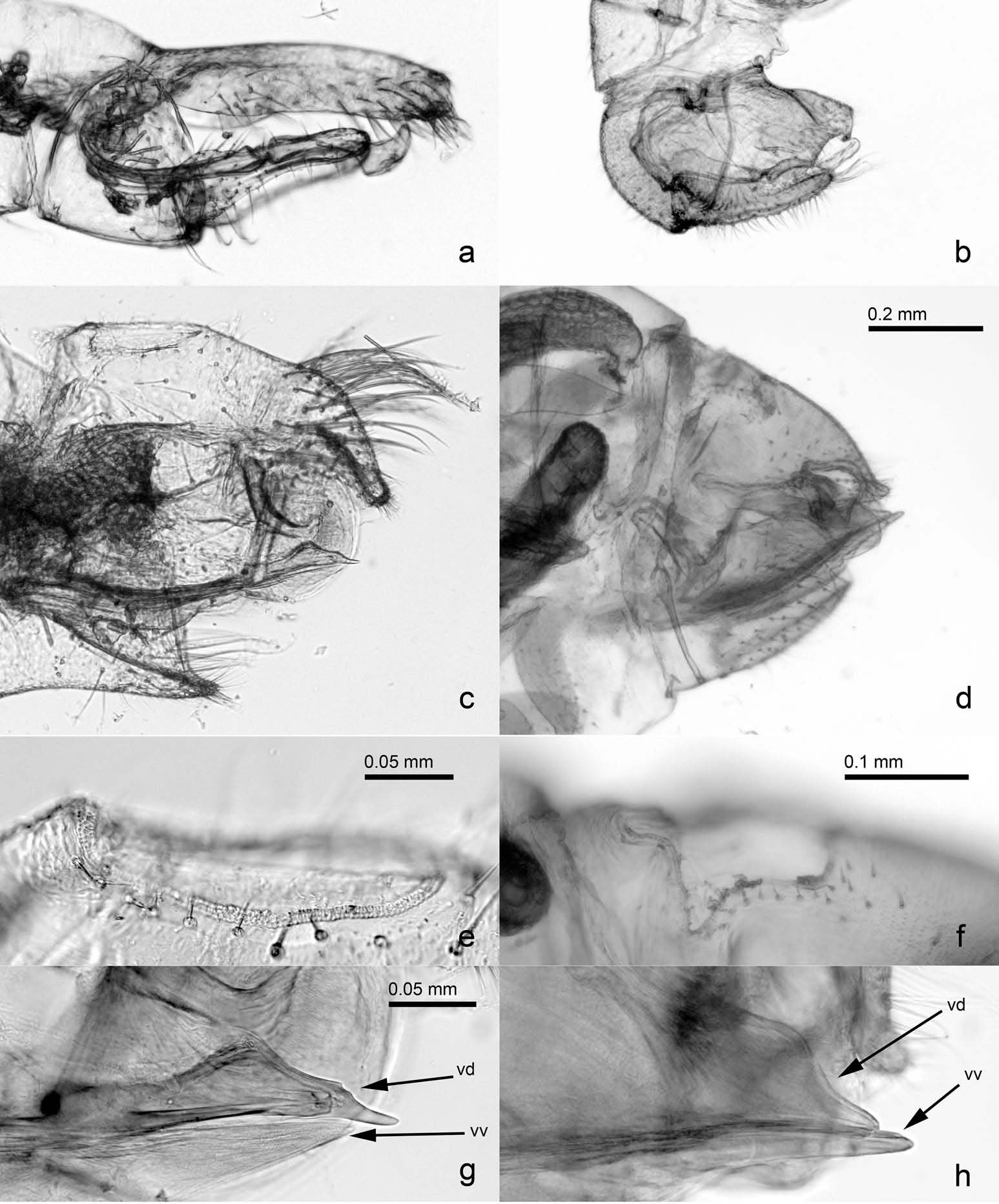
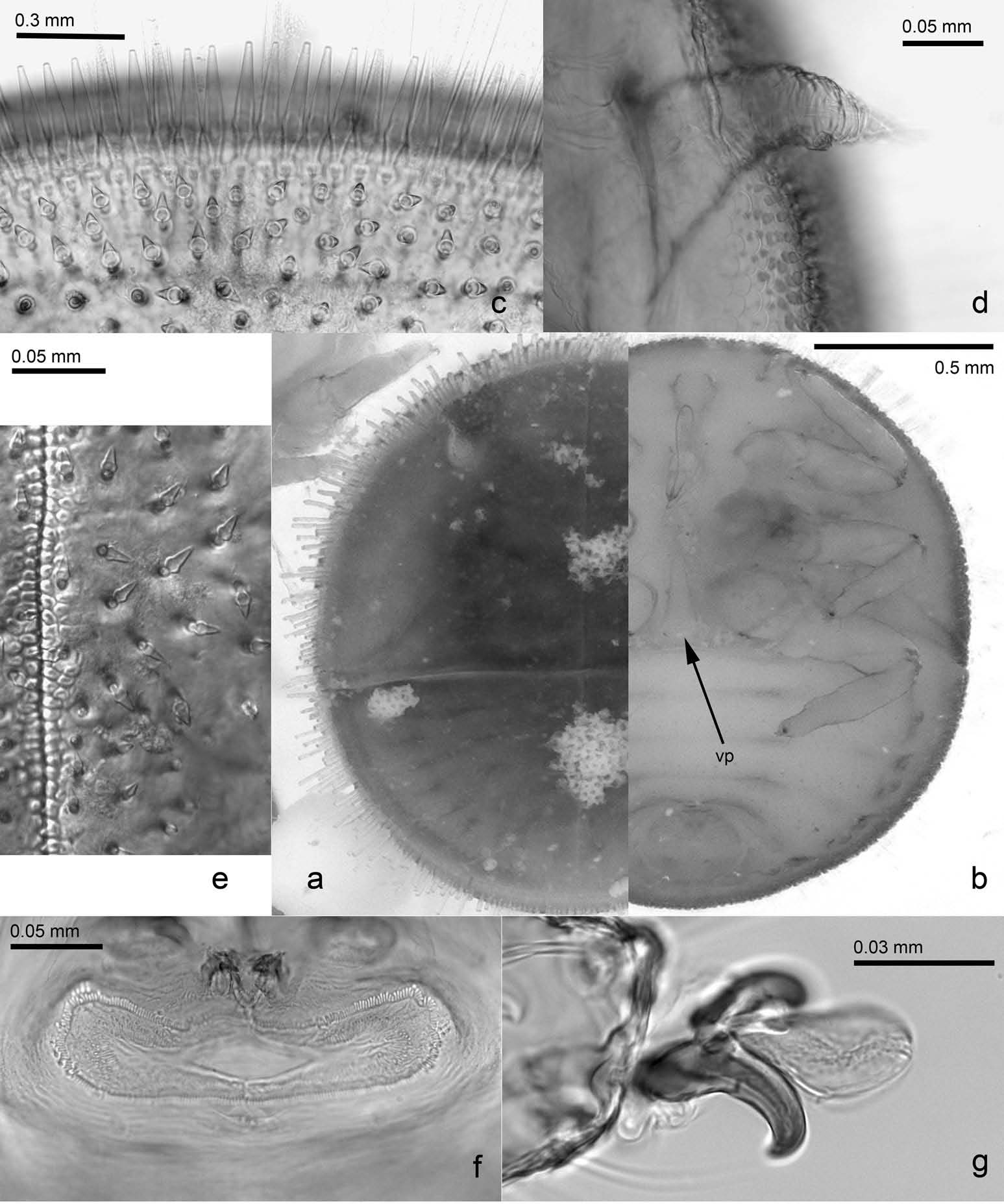
( Figures 2 View Figure 2 (aıcıe – g); 3(aıcıeıg); 4(aıcıf); and 5)
Material
Holotype maleı BRAZIL: Minas Geraisı Uberlândiaı Clube Caça e Pesca Itororoı −18.9932 −48.3065 ı 830 mı 17.ix. 2018ı Pouteria ramiflora (D.C. Oliveira) (UFPRı dry mounted) GoogleMaps . – Paratypes. BRAZIL: 8 malesı 9 femalesı same data as holotype but (NHMBı dry mounted and in 70% ethanol); 2 malesı 3 femalesı same but 20.vi.2015 (NHMBı UFPRı dry mounted); 3 malesı 3 femalesı 22 immaturesı same but 2.ix.2014 (NHMBı slide mounted and in 70% ethanol GoogleMaps ).
Material not included in type series. BRAZIL: 15 gallsı some with immaturesı same data as holotype but 2.ix.2014 (NHMBı in 70% ethanol) .
Diagnosis
Adult. Body conspicuously bicolouredı almost black dorsallyı straw-coloured ventrally. Flagellar segments of antenna conspicuously thickened. Forewing narrowly rounded apically. Male proctiger longı slender; paramere digitiform; apex of distal portion of aedeagus hook-shaped. Female proctiger strongly curved in apical thirdı apex down-curvedı blunt; bearing long setae in apical third; circumanal ring oval; subgenital plate pointed apically; ventral margin concave. – Fifth instar immature: Body with longı denseı subacute marginal sectasetae and smallı dorsal sectasetae; strongly bulbous ventrally. Tarsal arolium ovalı bearing short pedicel and unguitractor; slightly longer than well-developed claws. Very long membranous process present at edge of thorax and abdomen medially.
Description
Adult. Colouration. Headı including clypeus and antennaeı and prothorax completely blackı compound eyes dull reddishı ocelli orange. Remainder of body black dorsally and yellow ventrally. Fore and mid legs mostly blackı procoxa partlyı mesocoxa entirely and hind leg straw-coloured. Forewing veins almost black in anterior half and brown in posterior half of wing; membrane transparent and colourless apart from wing base and a narrow stripe in cell c+ sc along vein R + M+ Cu which are brown. Hindwing colourless and transparentı costal vein light brown. Terminalia straw-coloured with almost black proctiger in both sexes. Younger specimens with more expanded light colour.
Structure. Body length male 3.9 – 4.1 mm; female 4.1 – 4.3 mm (2 malesı 3 females). Body integument almost glabrous; head and thorax bearing longı fineı sparse setae dorsally ( Figure 2 View Figure 2 (c)). Genal processes separate in the middleı axes strongly converging towards apex ( Figure 2 View Figure 2 (c)). Antenna 1.3 – 1.4 times as long as head width; flagellar segments conspicuously thickened ( Figure 2 View Figure 2 (e))ı relative length of flagellar segments as 1.0: 0.5: 0.4: 0.5: 0.4: 0.5: 0.5: 0.5; terminal setae 0.6 and 0.4 times as long as segment 10. Metatibia 0.8 times as long as head width; with a group of smallı inconspicuous genual spines ( Figure 2 View Figure 2 (g)). Forewing 3.9 – 4.1 times as long as head widthı 2.6 – 2.8 times as long as wide; narrowly rounded apically ( Figure 2 View Figure 2 (f)). Terminalia as in Figures 3 View Figure 3 (aıcıeıg) and 4(aıcıf). Male proctiger long and slenderı 0.7 times as long as head width; paramere digitiformı sparsely beset with moderately long and a few long setae on inner face; distal portion of aedeagus hook-shaped apically. Female proctiger 0.8 times as long as head widthı strongly curved in apical thirdı apex downcurvedı blunt; bearing long setae in basal half laterally and in apical third; circumanal ring 0.3 times as long as proctigerı ovalı consisting of several rows of pores basally and two rows of unequal pores laterally and caudally. Subgenital plate pointed apicallyı beset with long setae; ventral margin concave. Valvula lateralis broadly rounded caudally.
Measurements (in mm; 1 maleı 1 female). Head width 0.78 – 0.80; antennal length 1.04 – 1.05; forewing length 3.11 – 3.16; proctiger length male 0.53ı female 0.63; paramere length 0.34; length of distal segment of aedeagus 0.25.
Fifth istar immature. Colouration. Dorsal side with head and wing pads ochreousı thorax and abdomen brownı the latter with ochreous margin. Eyes yellowish. Ventral side dirty whitishı caudal plate yellowish. Tip of rostrum and claws dark.
Structure. Body ( Figure 5 View Figure 5 (aıb)) ovalı 1.2 – 1.3 times as long as wide; flatı strongly sclerotised dorsally ( Figure 5 View Figure 5 (a))ı strongly bulbousı little sclerotised ventrally ( Figure 5 View Figure 5 (b)). Tarsal arolium ( Figure 5 View Figure 5 (g)) ovalı bearing short pedicel and unguitractor; slightly longer than well-developed claws. Forewing pad 6.3 – 6.6 times as long as antenna. Very long membranous processes present at limit of thorax and abdomen medially ( Figure 5 View Figure 5 (b): vp). Caudal plate 0.5 – 0.6 times as long as wideı 7.3 – 8.5 times as wide as circumanal ring. Outer circumanal ring with some additional pores mostly laterally ( Figure 5 View Figure 5 (f)). Marginal setae surrounding body consisting of longı denseı subacute sectasetae ( Figure 5 View Figure 5 (c)); dorsal setae smallı relatively evenly spaced sectasetae ( Figure 5 View Figure 5 (e)).
Measurements (in mm; 4 specimens). Body length 1.73 – 1.93; antennal length 0.13 – 0.15 mm; forewing pad length 0.93 – 0.95; caudal plate length 0.73 – 0.83; caudal plate width 1.38 – 1.53; width of circumanal ring 0.18 – 0.20.
Host plant
Pouteria ramiflora (Mart.) Radlk. (Sapotaceae) . Pouteria torta (Mart.) Radlk. was reported from Goiás by Santos et al. (2012) as being galled by an unspecified Hemiptera which may concern C. pouteriae based on the photos provided by the authors. The identity of the latter host is questionable and may constitute a misidentification of P. ramiflora .
Distribution
Brazil (Minas Gerais). Previously reported from Minas Gerais ( Calácio 2016 ı as Ceropsylla sp.; Naves 2017 ı as Ceropsylla sp.). The record of galls induced by an unspecified Hemiptera on Pouteria torta ( Santos et al. 2012) from Goiás may concern C. pouteriae .
Derivation of name
Named after its host Pouteria .
Comments
Ceropsylla as defined here comprises two species inducing pit galls on the leaves of Sapotaceae . The species can be separated as indicated in the key above. The immatures are similar in the structure of the dorsal surfaceı resembling many other pit gall inducing psyllidsı but differ fundamentally in the structure of the ventral side which is bulbous and weakly sclerotised in C. pouteriae but leathery and shallowly convex in C. sideroxyli . This goes together with the presence or absence of claws in the former and latterı respectively. These morphological dissimilarities are likely due to differences in behaviour. Immature of the former remain sessile in the gall whereas those of the latter spin in the gall in semicircular movements thereby spreading wax on the leaf surface ( Parsons 2012).
Apart from C. sideroxyli , Ceropsylla comprises currently three species from China ( Li 2011)ı two from Mexico (Tuthill 1945) and one from Puerto Rico (Caldwell 1942) which do not fit the diagnosis of the genus provided above and which areı thereforeı not congeneric with C. sideroxyli . The three Chinese speciesı C. angustirerta Liı C. celticola Li and C. cestolemba Li have forward directed genal processes and a short cu1 cell in the forewingı the two Mexican speciesı C. discrepans Tuthill and C. pulchra Tuthillı possess long female terminaliaı and the Puerto Rican C. martorelli Caldwell has relatively long female terminalia and is associated with Lauraceae . For this reasonı we transfer the six species to the artificial genus Trioza as Trioza angustirerta (Liı 2011) ı comb. nov.ı Trioza celticola (Liı 2011) ı comb. nov.ı Trioza cestolemba (Liı 2011) ı comb. nov.ı Trioza discrepans (Tuthillı 1945) ı comb. nov.ı Trioza martorelli (Caldwellı 1942) ı comb. nov. and Trioza pulchra (Tuthillı 1945) ı comb. nov.ı all from Ceropsylla .
General features of the gall
The leaves of Pouteria ramiflora are simpleı glabrousı oblongı alternating and spiralled ( Figure 1 View Figure 1 (a)). The gall is intralaminar and can be characterised by an intumescence facing the adaxial surface of the leaf ( Figure 1 View Figure 1 (b)) and a pit facing the abaxial face in which the galling insect is fitted being dorsally covered by thread-like waxy secretions ( Figure 1 View Figure 1 (c)). The pit bears a single psyllid immature. No parasitoids were found.
Anatomical profile
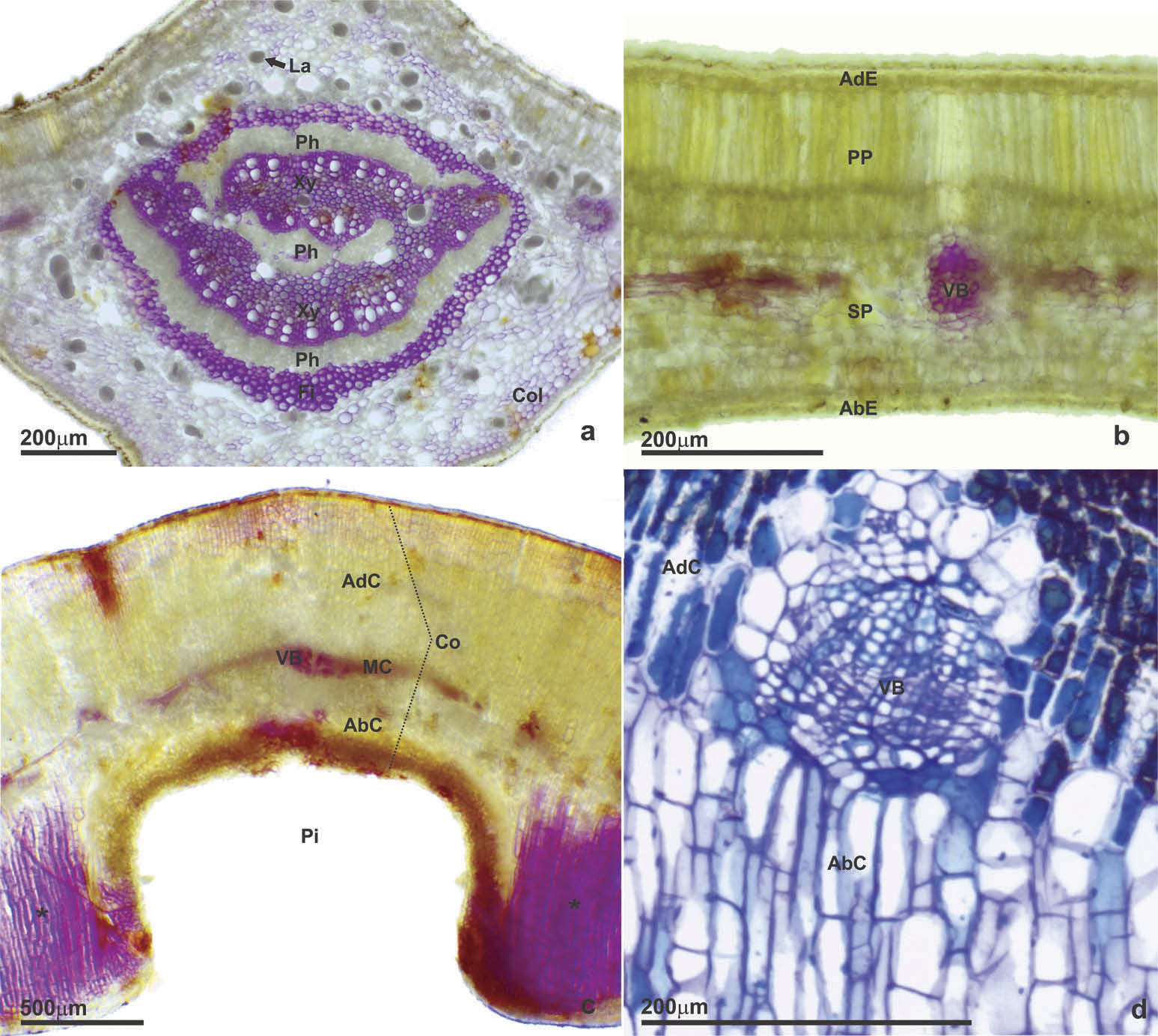
Non-galled leaf
The leaves present an uniseriate epidermis with a thick cuticle ( Figure 7 View Figure 7 (aıb)). The midrib vein has a bicollateral vascular bundle surrounded by fibres and laticifers ( Figure 7 View Figure 7 (a))ı as well as the vascular bundles with a smaller dimeter ( Figure 7 View Figure 7 (b)). Collenchyma occurs under the epidermis of the midrib vein ( Figure 7 View Figure 7 (a)). The leaf is hypostomatic and the mesophyll is dorsiventralı with two layers of palisade parenchyma and from three to five layers of spongy parenchyma ( Figure 7 View Figure 7 (b)).
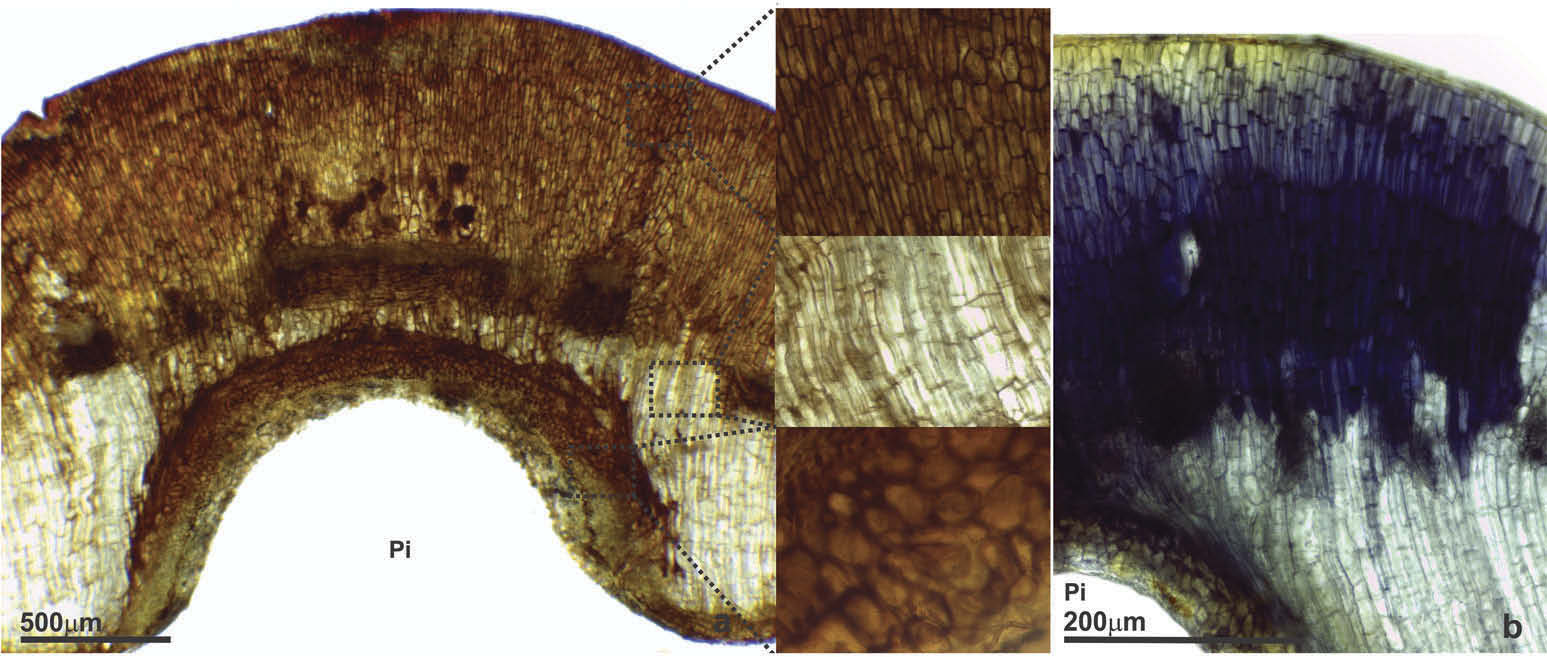
Gall
The epidermis of the gall is uniseriate and without stomata ( Figure 7 View Figure 7 (c)). The cortex presents homogeneous parenchyma and is divided into three parts: adaxial cortexı median cortex with vascular bundles ( Figure 7 View Figure 7 (d)) and abaxial cortex ( Figure 7 View Figure 7 (c)). There is no typical nutritive tissue in this gall ( Figure 7 View Figure 7 (c)). The parenchyma cells present predominantly anticlinal growth with radial distribution towards the pit bearing the immature ( Figure 7 View Figure 7 (c)). The parenchyma that occurs laterally to the vascular bundles contains cells with isotropic development ( Figure 7 View Figure 7 (cıd)). The parenchyma of the abaxial cortex presents cells with anticlinal stretchingı being composed from three to five layers ( Figure 7 View Figure 7 (c)). The orifice is open with a lignified rim ( Figure 7 View Figure 7 (c)).
Histochemistry profile
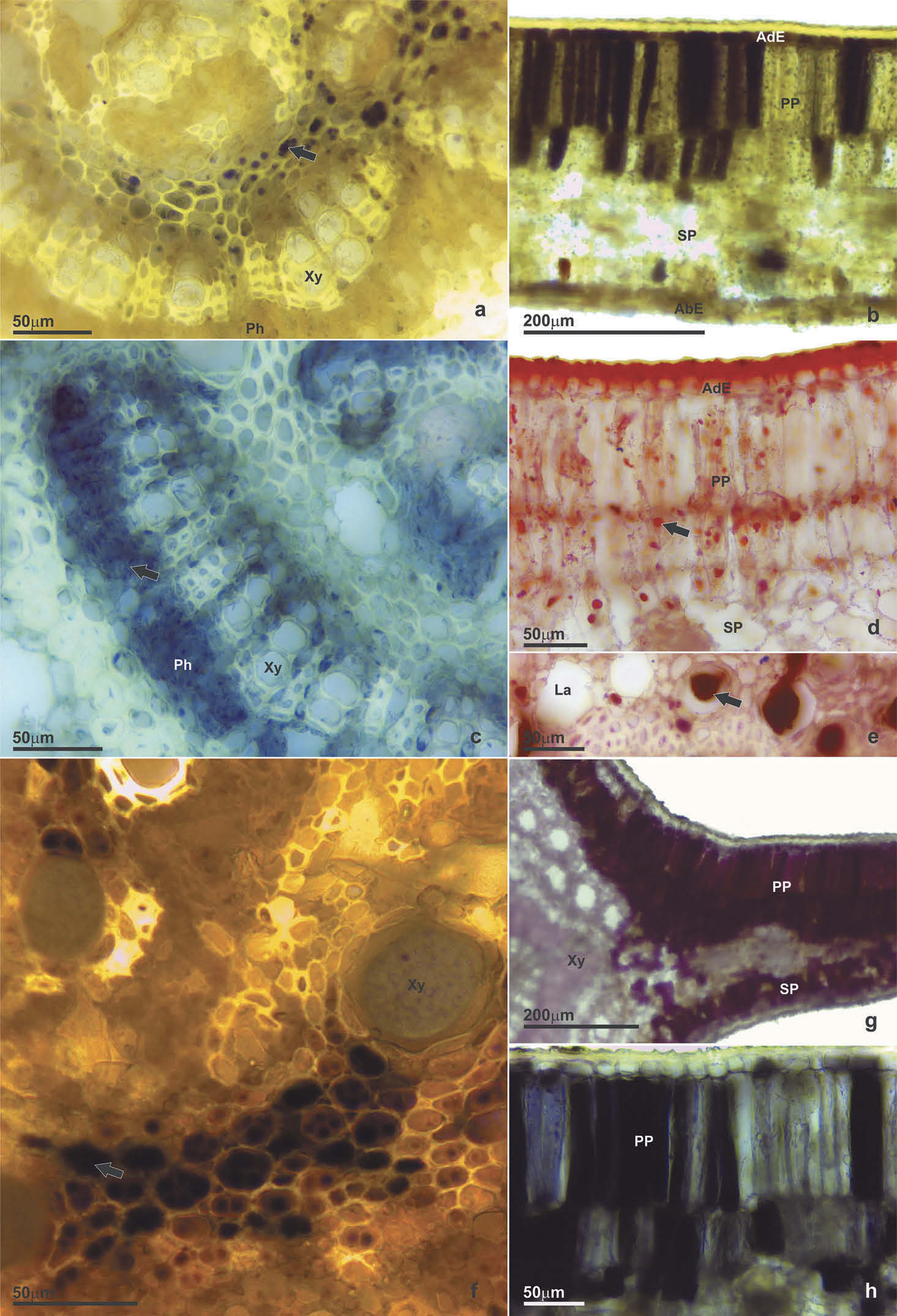
Non-galled leaf
Starches were found only in the central region of the midrib vein ( Figure 8 View Figure 8 (a)). Reducing sugars were detected in the epidermis and in the mesophyll of the intercostal region ( Figure 8 View Figure 8 (b))ı besides the subepidermal parenchyma of the midrib vein. Proteins were detected mainly in the palisade parenchyma of the mesophyll and in the phloem of the midrib vein ( Figure 8 View Figure 8 (c)). Droplets of lipids were detected in all mesophyll cells ( Figure 8 View Figure 8 (d))ı as well in the laticifers ( Figure 8 View Figure 8 (e)).
Proanthocyanidins were detected in the epidermisı in the mesophyll and in the phloem cells of the intercostal region. Alkaloids were observed in the midrib ( Figure 8 View Figure 8 (f)). Auxins occurred in the cells of the mesophyll and in the cortex of the midrib ( Figure 8 View Figure 8 (g)). Reactive oxygen species were detected at auxin-like sites: epidermisı mesophyllı phloem and cell walls of cortex of the midrib. Phenolic compounds were present in mesophyll and phloem ( Figure 8 View Figure 8 (h)).
Gall
Starches were detected in the upper portion of the abaxial cortex and near to vascular bundles ( Figure 9 View Figure 9 (a)). There is a centrifugal gradient of reducing sugars in the gallı with substances detected mainly in the adaxial cortex ( Figure 9 View Figure 9 (b)). Proteins were observed in the apoplast and symplast of adaxial and abaxial cortex cells ( Figure 9 View Figure 9 (c))ı as well in the phloem. Lipids were detected in the cortexı forming a centripetal gradient towards the vascular bundles ( Figure 9 View Figure 9 (d))ı and in the phloem.
Proanthocyanidins were detected in cells adjacent to the pit and in the adaxial cortex ( Figure 9 View Figure 9 (e)). Alkaloids were present in the cells of the parenchyma between vascular bundles. Auxins were found in the adaxial cortex and in the cells around the pit holding the immature ( Figure 9 View Figure 9 (f)). Reactive oxygen species also occurred more intensely in the cell layers of the gall that cover the pitı as well in the phloem cells ( Figure 10 View Figure 10 (a)) and in cell walls of the adaxial cortex ( Figure 10 View Figure 10 (a)). Phenolic compounds also occur mostly in the adaxial cortex ( Figure 10 View Figure 10 (b)).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
SuperFamily |
Psylloidea |
|
Family |
Ceropsylla
| Oliveira, Denis Coelho De, Burckhardt, Daniel, Calácio, Tácito De Freitas, Kuster, Vinícius Coelho & Queiroz, Dalva Luiz de 2019 |
Ceropsylla
| Calácio 2016 ı p. 15 |
| Naves 2017 ı p. 9 |