Ascarophis arctica Polyanskiy, 1952
|
publication ID |
https://doi.org/ 10.14411/fp.2018.016 |
|
publication LSID |
lsid:zoobank.org:pub:BF143B96-4194-4DF7-838C-99EB047203E6 |
|
DOI |
https://doi.org/10.5281/zenodo.8178675 |
|
persistent identifier |
https://treatment.plazi.org/id/0382671D-FFA6-D601-FF08-FA768BBAE450 |
|
treatment provided by |
Felipe |
|
scientific name |
Ascarophis arctica Polyanskiy, 1952 |
| status |
|
Ascarophis arctica Polyanskiy, 1952 View in CoL View at ENA Figs. 12–14 View Fig View Fig View Fig
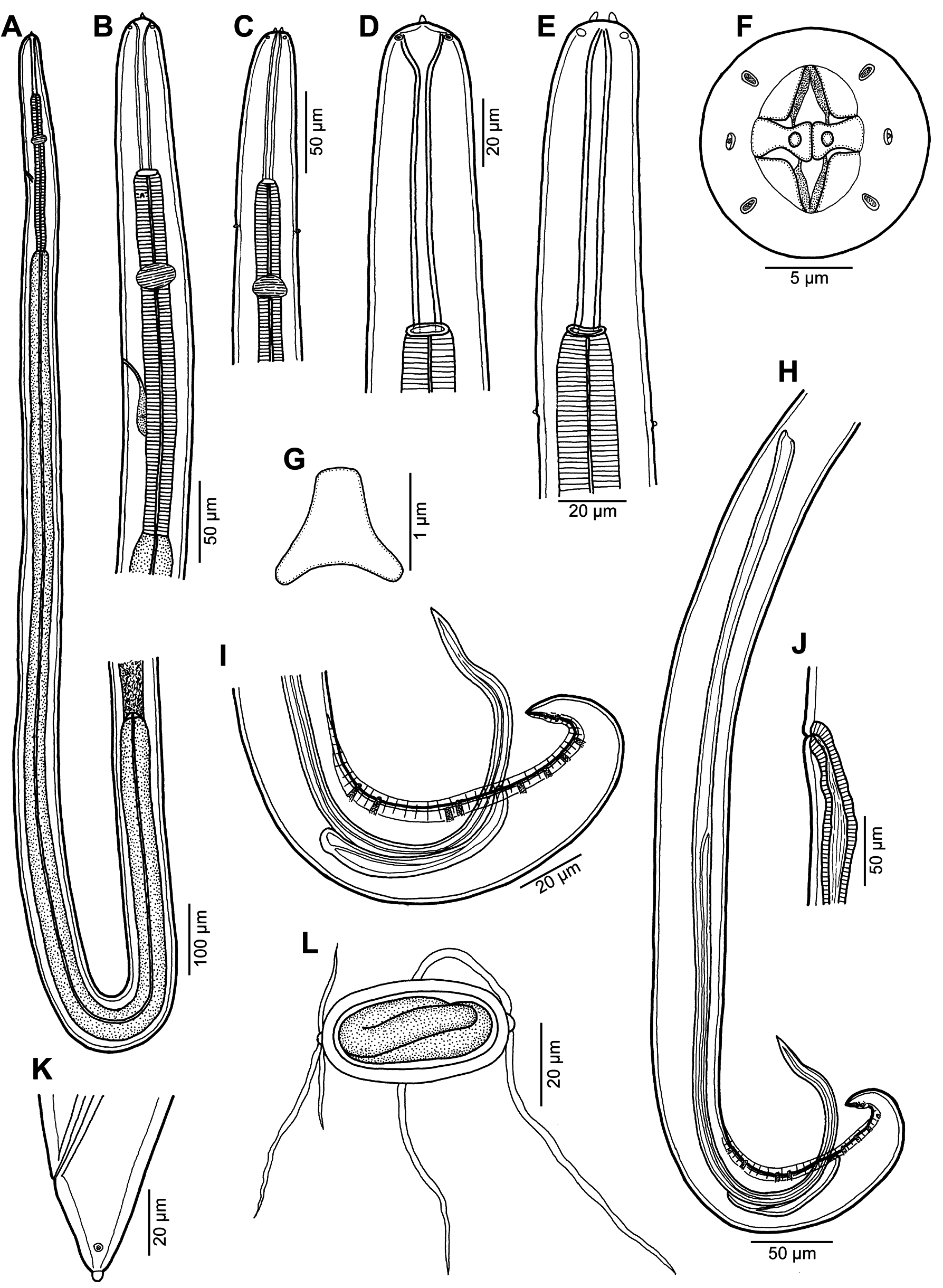
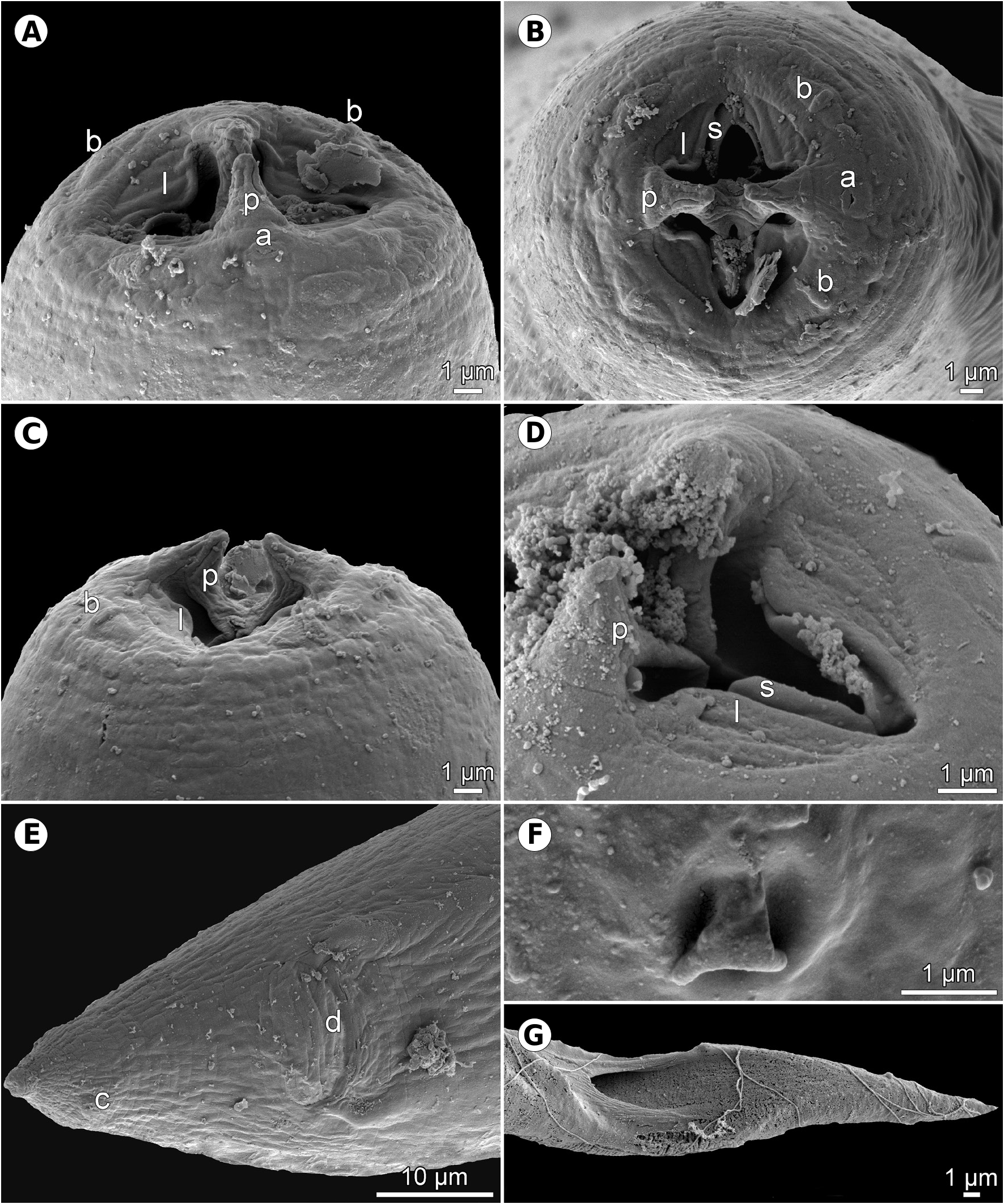
Description. Small, whitish nematodes. Cuticle thick, with fine transverse striations ( Figs. 13B,C View Fig , 14D View Fig ). Cephalic end rounded, with two conspicuous conical pseudolabial terminal protrusions ( Figs.12A–E View Fig , 13A–C View Fig ). Oral aperture oval, with lateral extensions. Four submedian labia and 4 submedian flap-like sublabia well developed. Lateral pseudolabia well developed, each provided with conspicuous terminal protrusion; in apical view, flat inner parts of pseudolabia partly cover mouth and are distinctly dorsoventrally expanded, forming 2 (1 dorsolateral and 1 ventrolateral) extensions on each; inner margins of both pseudolabia dorsoventrally straight, parallel to each other ( Figs. 12F View Fig , 13A–D View Fig ). Four elongate submedian cephalic papillae and pair of lateral amphids present ( Figs. 12F View Fig , 13A–C View Fig ). Vestibule (stoma) long, cylindrical, with small funnel-shaped prostom in lateral view ( Fig. 12A–E View Fig ). Glandular oesophagus in female 6–8 times longer than muscular; both parts of oesophagus distinctly separated from each other ( Fig. 12A View Fig ). Nerve ring encircles muscular oesophagus approximately at border of its first and second thirds; excretory pore located somewhat posterior to level of nerve ring; deirids very small, bifurcate, situated somewhat posterior to end of vestibule ( Figs. 12A–C,E,G View Fig , 13F View Fig ).
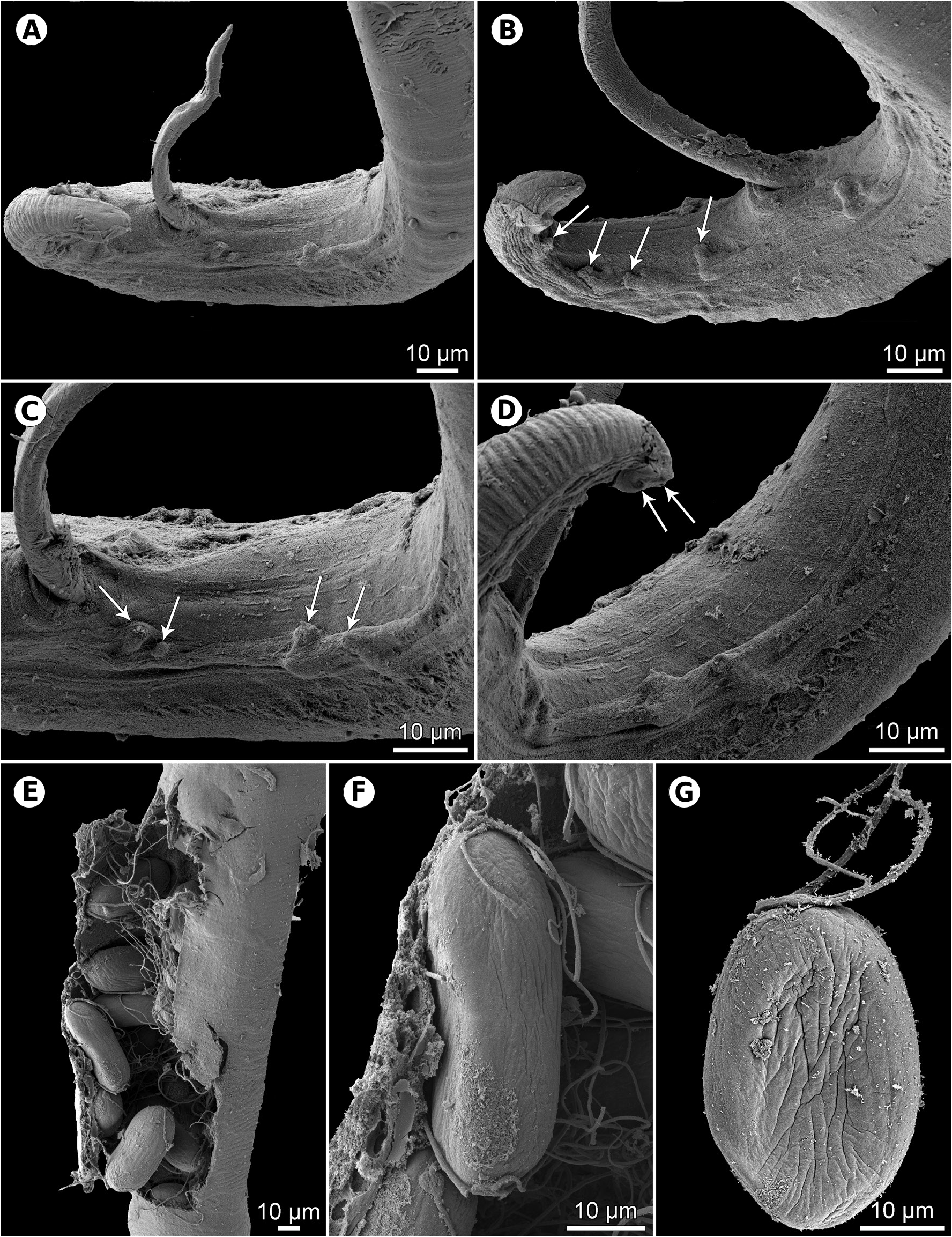
Male (1 specimen with missing anterior part of body). Length of body fragment 3.56, maximum width 51. Incomplete glandular oesophagus 653 long, maximum width 45. Posterior end of body ventrally curved, provided with narrow caudal alae. Preanal papillae: 4 pairs of subventral pedunculate papillae present, of which first and second and third and fourth are shifted in relation to each other, alternating along margins of tail ( Figs. 12I,H View Fig , 14A–D View Fig ). Postanal papillae: 6 pairs present, including 5 pairs of pedunculate subventral papillae and 1 pair of minute ventral sessile papillae located slightly posterior to level of last pair of subventrals ( Figs. 12I View Fig , 14B View Fig ). Small median ventral caudal protuberance present at tail tip ( Fig. 14D View Fig ). Ventral cuticular ridges (area rugosa) anterior to cloaca weakly developed, consisting of about 7 longitudinal tessellated ornamentations ( Fig. 14C,D View Fig ). Large (left) spicule 681 long, with pointed distal tip; its shaft 279 long, forming 41% of overall length of spicule ( Figs. 12I,H View Fig , 13G View Fig ). Small (right) spicule boat-shaped, 81 long, with rounded distal end ( Fig. 12I,H View Fig ). Length ratio of spicules 1: 8.4. Tail conical, 84 long, with rounded tip ( Fig. 14A,B,D View Fig ).
Female (6 gravid specimens with mature eggs). Length of body 8.3–10.9 mm, maximum width 81–105. Height of cephalic protrusions 3. Vestibule including prostom 90–105 long; prostom 12 long, 12–15 wide. Length of muscular oesophagus 219–264, maximum width 18–27; length of glandular oesophagus 1.22–1.89 mm, maximum width 42–57; length ratio of muscular and glandular parts of oesophagus 1: 5.6–7.6. Length of entire oesophagus and vestibule represents 17–22% of total body length. Nerve ring, excretory pore and deirids 162–198, 207–300 and 129–135, respectively, from anterior extremity. Vulva somewhat postequatorial, situated 5.07–6.09 mm from anterior end of body, at 56–69% of body length; vulval lips not elevated. Vagina directed posteriorly from vulva. Amphidelphic. Uterus filled with numerous eggs. Mature eggs (containing larvae) oval, thick-walled, size 48–54 × 24–27; thickness of egg wall 4–5. Each pole of egg mostly with small knob provided with 2–5 thread-like filaments; knob on 1 pole somewhat larger than that on opposite pole, usually bearing 0–3 short, fine filaments and 2 thicker and longer filaments; filaments growing up from smaller knob relatively short and fine, 2–3 in number ( Figs. 12L View Fig , 14E,F View Fig ); exceptionally knob and filaments present only on 1 pole of larvated egg ( Fig. 14G View Fig ). Tail conical, short, 39–57 long, with 2 lateral papilla-like phasmids near its tip, and a distinct small terminal knob-like projection ( Figs. 12K View Fig , 13E View Fig ).
Host: Three-spined stickleback, Gasterosteus aculeatus Linnaeus ( Gasterosteidae , Gasterosteiformes ).
Site of infection: Stomach.
Locality : Nishittapu River, Tomakomai, Hokkaido, Japan (collected 16 June 1996 and 19 June 2009) .
Total prevalence and intensity: 25% (3 fish infected/12 fish examined); 1–6 (mean 3) nematodes per fish. 1996: 1 fish infected, fish size not measured/1 fish examined; 3 nematodes. 2009: 18% (2 fish infected, 7.1 and 8.2 cm TL/11 fish examined, 7.9–9.1 cm TL); 1 and 6 nematodes.
Deposition of voucher specimens: IPCAS N-1153.
Remarks. The general morphology of the present nematodes from G. aculeatus indicates that they belong to the nominotypical subgenus Ascarophis van Beneden, 1870 (see Moravec and Justine 2009) of the cystidicolid genus Ascarophis van Beneden, 1870 , as defined by Ko (1986). At present, 41 species of this genus are considered to be valid ( Moravec et al. 2018).
Only the following seven species of the genus Ascarophis have filamented eggs and a large (left) spicule length similar to that of the species under study (681 µm): A. arctica (left spicule 500–980 µm long), Ascarophis draconi Muñoz et George-Nascimento, 2007 (482–640 µm), Ascarophis litoralica Zhukov, 1960 (590–670 µm), Ascarophis marina (Szidat, 1961) (405–600 µm), Ascarophis morrhuae van Beneden, 1870 (500–790 µm), Ascarophis maulensis ( Muñoz, González et George-Nascimento, 2004) (550–690 µm) and Ascarophis richeri Moravec et Justine, 2007 (663–729 µm) ( Ferrer et al. 2005, Muñoz et al. 2004, Moravec and Justine 2007, Muñoz and George-Nascimento 2007). Of these, however, A. marina , A. maulensis and A. richeri have a different structure of the mouth and they belong to the subgenus Similascarophis Muñoz, González et George-Nascimento, 2004 (see Moravec and Justine 2007). The remaining four species are representatives of the subgenus Ascarophis , as are the present specimens from G. aculeatus .
However, A. draconi has much longer males and females (males 9.9–13.3 mm, females 20.9–28.6 mm) and the eggs have two filaments on each pole; A. litoralica has also eggs with only two filaments on each pole, whereas its body measurements are similar to those of the present nematodes from Japan. Both A. draconi and A. litoralica were described from the Pacific Ocean (off Chile and in the South-Kurile shallow waters, respectively) ( Zhukov 1960, Muñoz and George-Nasciento 2007); the latter species has not yet been studied by SEM.
Two other species, A. arctica and A. morrhuae , were reported from different marine fishes (mostly Gadiformes and Scorpaeniformes ) in the Atlantic and Arctic Oceans, being separated from each other mainly by the number and arrangement of egg filaments (two filaments only on one egg pole in A. morrhuae and several filaments on both egg poles in A. arctica ) ( Polyanskiy 1952, Appy 1981, Ko 1986, Fagerholm and Berland 1988). However, as confirmed in this paper, the number and character of egg filaments may be rather variable within the same species of Ascarophis and, therefore, should not be overestimated as a taxonomic feature.
The original description of A. morrhuae by van Beneden (1870) was poor and, as pointed out by Zhukov (1960), apparently more congeneric species were included under this name from different hosts by subsequent authors. Therefore, a detailed redescription of this type species of Ascarophis , based on specimens newly collected from the type host and the type locality (off the coast of Belgium) is needed.
The general morphology of the present nematodes from G. aculeatus shows that these can be assigned, according to present criteria, to A. arctica , even though, as compared with the descriptions of A. arctica from other fish hosts ( Polyanskiy 1952, Appy 1981, Fagerholm and Berland 1988), the filaments on both poles of larvated eggs of these nematodes are less numerous (2–5 vs up to18 on each pole according to Fagerholm and Berland 1988) and the knob from which filaments are growing is distictly smaller on one egg pole as compared to that on the opposite pole, and ventral precloacal ridges in the male are poorly developed (vs well developed). In addition to the presence of several filaments on each pole in the majority of larvated eggs, these nematodes possess flap-like sublabia ( Figs.12F View Fig , 13A–D View Fig ), typical of A. arctica , as visible on SEM micrographs of Appy (1981) and Fagerholm and Berland (1988). Specimens of A. morrhuae from its type host ( Gadus morhua Linnaeus ) have not yet been studied by SEM, but those from Trisopterus capellanus (Lacépède) had distinctly bi-lobed sublabia (see fig. 75 of Ko 1986), although specimens from Agonus cataphractus (Linnaeus) , also considered by Ko (1986) to be A. morrhuae , possessed flap-like sublabia.
Ascarophis arctica was originally described from the stomach of the eelpout Zoarces viviparus (Linnaeus) ( Zoarcidae , Perciformes ) in the Barents Sea ( Polyanskiy 1952) and subsequently it was also reported from different marine, mostly gadiform and scorpaeniform fishes in the North Atlantic region ( Canada, Europe) ( Appy 1981, Fagerholm and Berland 1988). The only record of A. arctica from G. aculeatus is that of Levsen (1992), who, without giving any morphological data, listed this nematode as a parasite of this fish species in western Norway (Bergen area). Therefore, the present finding of this parasite in Japan represents the first record of A. arctica in the region of the Pacific Ocean. Its host, G. aculeatus , is distributed in circumarctic and temperate regions of Northern Hemisphere ( Froese and Pauly 2018). Hanek and Threlfall (1970) reported a different species of Ascarophis , A. morrhuae, as a common parasite of G. aculeatus in Newfoundland and Labrador, Canada, but no morphological data were provided.
The Nishittapu River population of G. aculeatus is composed of both anadromous and fluvial indviduals ( Higuchi and Goto 1994, 1996; they reported this river erroneously as the Nishikitappu River): the former individuals migrate to the sea for feeding and return to the river for spawning. The infected fish found in this study are considered to have become parasitised by A. arctica at sea.
According to Nagasawa (2016), to date the only record of a species of Ascarophis from Japanese inland fishes is that of Margolis (1957), who reported Ascarophis sp. from the pink salmon Oncorhynchus gorbuscha (Walbaum) . Several years ago, we examined two damaged males of Ascarophis from the stomach of the same host species ( O. gorbuscha ) in the Shikiu River, Hokkaido (collected 14 August 1981), provided by Dr. Shigehiko Urawa (Hokkaido National Fisheries Research Institute, Sapporo, unpublished). Since their left spicule was much shorter (264 and 288 µm) than that of A. arctica , they evidently belonged to a different species. However, having no conspecific females, their identification to species was impossible. Zhukov (1960) reported seven Ascarophis spp. from marine fishes (not Salmonidae ) in the Sea of Japan and South-Kurile shallow waters, of which Ascarophis japonicus Zhukov, 1960 and Ascarophis pacificus Zhukov, 1960 have the left spicule length similar to that of Ascarophis sp. parasitising O. gorbuscha . More attention should be paid to Ascarophis sp. from salmon in Japan.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |