Micropodarke fujianensis, Wang & Yang & Cai & Deng & Ke, 2023
|
publication ID |
https://doi.org/10.11646/zootaxa.5256.3.4 |
|
publication LSID |
lsid:zoobank.org:pub:17E6F4ED-5A8A-4763-8F35-25E4FCE6D9EA |
|
DOI |
https://doi.org/10.5281/zenodo.7761348 |
|
persistent identifier |
https://treatment.plazi.org/id/0FC2C74E-0B76-4142-8C3C-3991B7648D56 |
|
taxon LSID |
lsid:zoobank.org:act:0FC2C74E-0B76-4142-8C3C-3991B7648D56 |
|
treatment provided by |
Plazi |
|
scientific name |
Micropodarke fujianensis |
| status |
sp. nov. |
Micropodarke fujianensis View in CoL n. sp.
( Figures 1–4 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 , Tables 1–3 View TABLE 1 View TABLE 2 View TABLE 3 )
ZooBank URL: lsid:zoobank.org:act: 0FC2C74E-0B76-4142-8C3C-3991B7648D56
Type material. Holotype, XMU-Pol-2021-227, anterior fragment with 27 chaetigers, Dongshan , Fujian, 23°32'52.06"N, 117°26'40.78"E, subtidal, 33.9 m, sandy, 25 May 2021 GoogleMaps . Paratype #1, MBM286065 View Materials , posterior fragment, 20 chaetigers, Dongshan , Fujian, 23°32'52.06"N, 117°26'40.78"E, subtidal, 30.8 m, sandy, 25 May 2021 GoogleMaps ; Paratype #2, XMU-Pol-2021-229, complete, 34 chaetigers, Dongshan , Fujian, 23°34'31.45"N, 117°34'31.10"E, subtidal, 33.0 m, coarse sandy, 24 May 2021 GoogleMaps .
Diagnosis. Presence of a pair of lateral antennae, a pair of biarticulated palps, distally knobbed segmental ventral adhesive papillae, around 20 papillae on anterior edge of pharynx. Median neurochaetae from middle segments without basally situated long teeth (“spurs”).
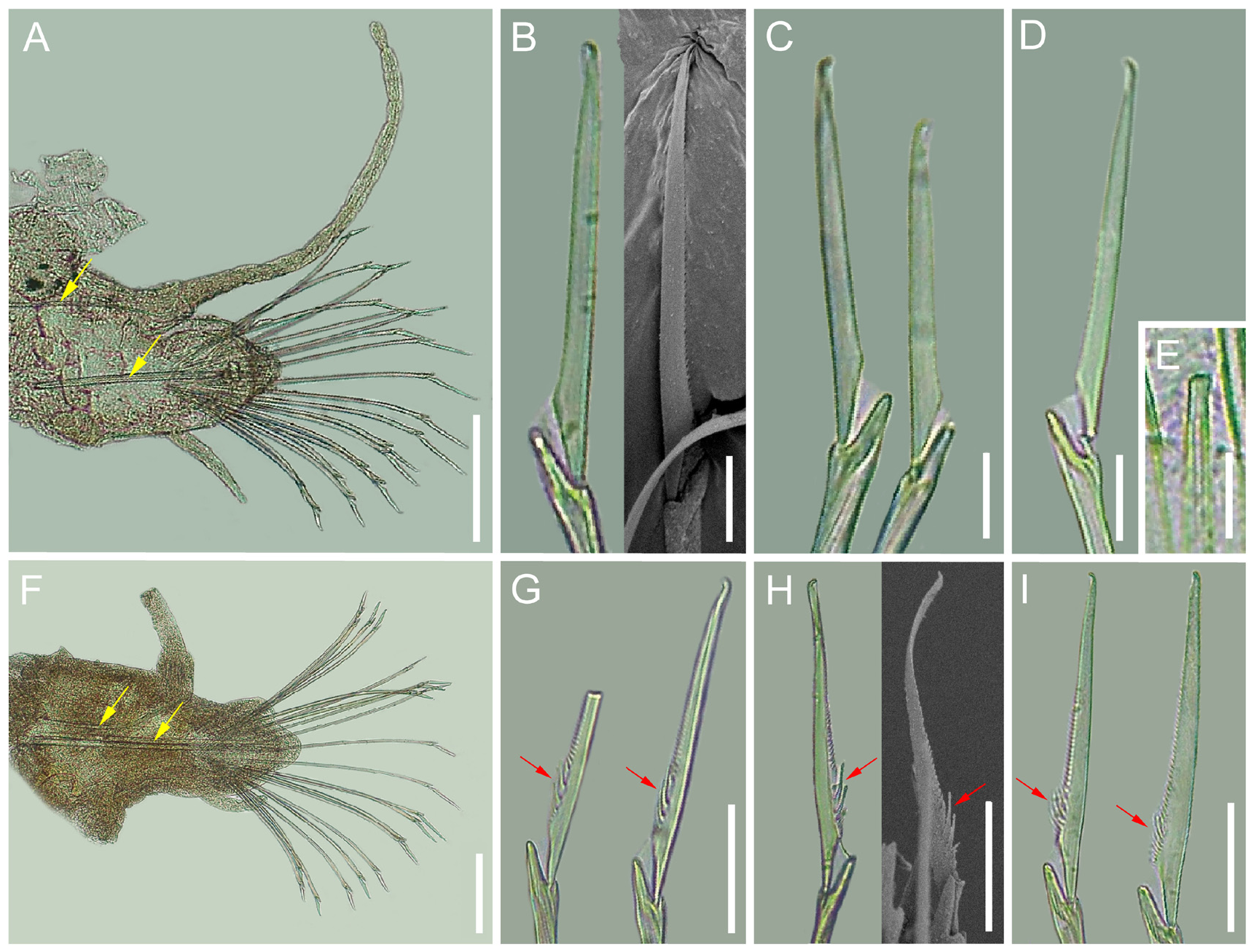
Description. Holotype (XMU-Pol-2021-227) incomplete, anterior fragment with 27 chaetigers, body width without parapodia around 0.3 mm ( Figure 2 View FIGURE 2 ). Body long and thin. Prostomium rounded quadrangular, width twice length ( Figure 2A View FIGURE 2 ). Eyes two pairs, located in posterior of prostomium, brown, similar in size; anterior pair kidneyshaped, posterior pair rounded ( Figure 2A View FIGURE 2 ). Palps biarticulated. Palpophores cylindrical, similar in diameter with base of palpostyle; thin, tapered ( Figure 2A, C View FIGURE 2 ). Lateral antennae thinner and slightly shorter than palps, tapered. Facial tubercle absent ( Figure 2C View FIGURE 2 ). Pharynx without jaws or teeth, anterior edge with a ring of around 20 papillae ( Figure 2C View FIGURE 2 ). Tentacular cirri 3 pairs, longest one reaching to chaetiger 5-6 ( Figure 2A View FIGURE 2 ).
Parapodia sub-biramous. Notoacicula single, slightly curved, extending to notopodial cirrophore; Neuroacicula single, straight, blunt ( Figure 3A, D View FIGURE 3 ). Neuropodial prechaetal lobe linguliform; postchaetal lobe semicircular, shorter than prechaetal lobe ( Figure 3A View FIGURE 3 ). Notopodial cirri biarticulated, cirrophore stout, short, cirrostyle as long as body width, tip moniliform, extending beyond chaetal tips. Neuropodial cirri digitiform, without cirrophores, distally slender, tip not extended beyond chaetal lobe ( Figure 3A View FIGURE 3 ). Adhesive papillae rounded, situated postero-laterally to parapodia ( Figure 2B View FIGURE 2 ).
Notochaetae absent; neurochaetae 13–21 heterogomph falcigers per bundle, blades unidentate, length/width ratio from 6 to 13, with 5-7 longer bladed falcigers extending from neuroacicula, and 6-7 shorter bladed falcigers from both supra- and sub-neuroacicula in middle neuropodia ( Figure 3 View FIGURE 3 A-D). Blades of median neurochaetae longer than dorsal and ventral ones ( Figure 3A View FIGURE 3 ), basally situated long teeth (“spurs”) absent ( Figure 3B, C, D View FIGURE 3 ).
Variation. The specimen of Paratype #1 has only posterior 20 chaeitgers, with 15-17 falcigers in middle chaetigers; the specimen of Paratype #2 with 34 chaetigers, with 13-14 falcigers in middle chaetigers. Due to the incomplete and small-sized specimens, no other apparent variations could be noted between the two type specimens.
Remarks. All other Micropodarke species were characterized by bearing one to several basally situated long teeth (“spurs”) in median neurochaetal blades, therefore this morphological character was included in the previous diagnosis of the genus Micropodarke ( Pleijel & Rouse 2005; Rizzo & Salazar-Vallejo 2014). M. fujianensis n. sp., however, differs from all described species in that its neurochaetae do not bear basal long teeth ( Figure 3 View FIGURE 3 B-D, G-I). In addition, M. fujianensis n. sp. differs from other Micropodarke species in a number of morphological characters. For instance, it differs from M. dubia and M. cf. dubia by having two pairs of smaller ( vs. larger) eyes ( Hessle 1925; Figure 2A, D View FIGURE 2 ), one ( vs. 1-2) notoacicula ( Uchida 2004; Figure 3A, D View FIGURE 3 ). It differs from M. pleijeli by having about 20 ( vs. about 30) papillae on the frontal edge of the pharynx, and bearing two pairs of eyes ( vs. bearing no eyes) ( Figure 2A, C View FIGURE 2 ; Rizzo & Salazar-Vallejo 2014). It differs from M. trilobata by having thin and tapered ( vs. thick and blunt) antennae, thin ( vs. medially swollen) palpostyles, one ( vs. 2) notoacicula and one ( vs. 2) neuroacicula ( Figure 2A View FIGURE 2 ; Hartmann-Schröder 1983).
Etymology. The specific epithet fujianensis refers to the type locality, Fujian Province.
Habitat. Subtidal sandy and coarse sandy sediment.
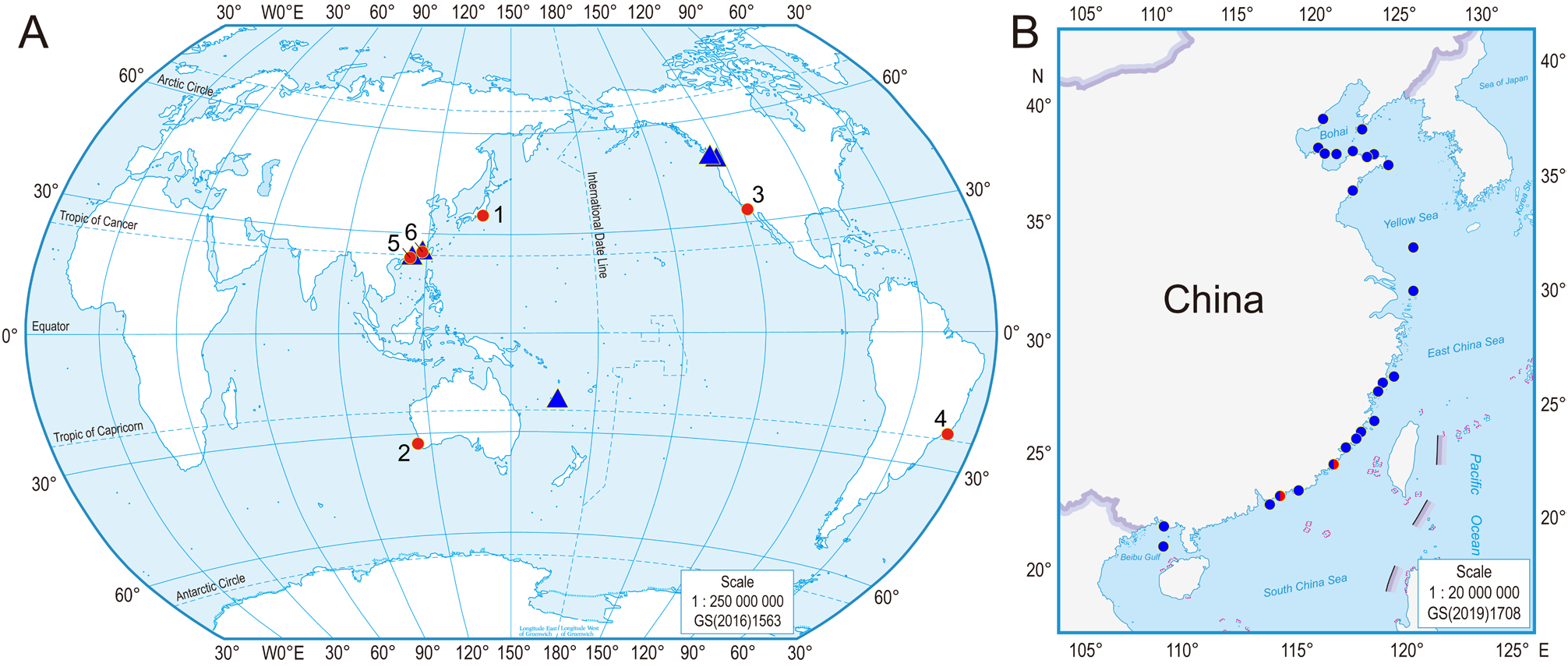
Distribution. Currently only known from coastal waters of Dongshan, Fujian, China.
Genetic distances. Table 3 View TABLE 3 showed the average K2P genetic distances among the known Micropodarke species. COI showed higher average K2P genetic distances than 16S rRNA and the other three gene markers. The average interspecific distances ranged from 14.6% ( Micropodarke sp. A & M. cf. dubia ) to 29.4% ( Micropodarke sp. A & Micropodarke sp. B ) for COI (523 bp), from 10.7% ( M. cf. dubia & M. fujianensis n. sp.) to 16.2% ( Micropodarke sp. A & M. fujianensis n. sp.) for 16S rRNA (379 bp), from 0.2% ( Micropodarke sp. A & M. cf. dubia ) to 1.2% ( Micropodarke sp. A & M. fujianensis n. sp.) for 28S rRNA (736 bp). For histone H3 (281 bp), the K2P distance was 1.1% between M. cf. dubia & M. fujianensis n. sp.; while it was 0 for 18S rRNA (934 bp) among Micropodarke sp. A , M. cf. dubia , and M. fujianensis n. sp. The average intraspecific K2P genetic distances of M. fujianensis n. sp. (0.8% for COI, 0.4% for 16S rRNA, 0 for 18S rRNA, 28S rRNA, and histone H3), M. cf. dubia (0.2% for COI, 0.4% for 16S rRNA, 0 for 18S rRNA and histone H3, and 0.1% for 28S rRNA), and Micropodarke sp. B (0.1% for COI) are much smaller than the respective interspecific distances among Micropodarke species here listed in Table 2 View TABLE 2 .
Phylogenetic trees. Phylogenetic tree reconstructed based on COI gene sequences revealed that, among the four distinct species, Micropodarke fujianensis n. sp. (Dongshan) was sister to Micropodarke sp. B ( British Columbia & Washington); together, they were sister to a clade consisting of Micropodarke sp. A (Lifou) and M. cf. dubia (Daya Bay) ( Figure 4A View FIGURE 4 ). The same sister relationship between Micropodarke sp. A (Lifou) and M. cf. dubia (Daya Bay) was also found in the trees based on 16S rRNA and 28S rRNA gene sequences ( Figure 4B, D View FIGURE 4 ). In the tree based on 18S rRNA gene sequences ( Figure 4C View FIGURE 4 ), all the 934-bp sequences were identical, therefore the relationships among Micropodarke fujianensis n. sp. (Dongshan), M. cf. dubia (Daya Bay) , and Micropodarke sp. A (Lifou) were undistinguishable. In the tree based on 28S rRNA gene sequences ( Figure 4D View FIGURE 4 ), Micropodarke sp. A (Lifou) and M. cf. dubia (Daya Bay) first formed a clade, and then it was sister to the clade of M. fujianensis n. sp. (Dongshan). The tree based on histone H3 gene sequences included only Micropodarke fujianensis n. sp. (Dongshan) and M. cf. dubia (Daya Bay) , which formed two distinct clades.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
SubFamily |
Psamathinae |
|
Genus |