Collarina balzaci ( Audouin, 1826 )
|
publication ID |
https://doi.org/ 10.5252/zoosystema2019v41a21 |
|
publication LSID |
urn:lsid:zoobank.org:pub:034A7358-CF99-4908-B047-557074A2E58E |
|
DOI |
https://doi.org/10.5281/zenodo.3729362 |
|
persistent identifier |
https://treatment.plazi.org/id/0381494A-FFF9-FF9C-FE89-3A93FCBE2AA1 |
|
treatment provided by |
Felipe |
|
scientific name |
Collarina balzaci ( Audouin, 1826 ) |
| status |
|
Collarina balzaci ( Audouin, 1826) View in CoL
( Figs 1-4 View FIG View FIG View FIG View FIG ; 7A View FIG ; 18A View FIG ; 19 View FIG ; Tables 1-3 View TABLE View TABLE View TABLE )
Flustra balzaci Audouin, 1826: 239 .
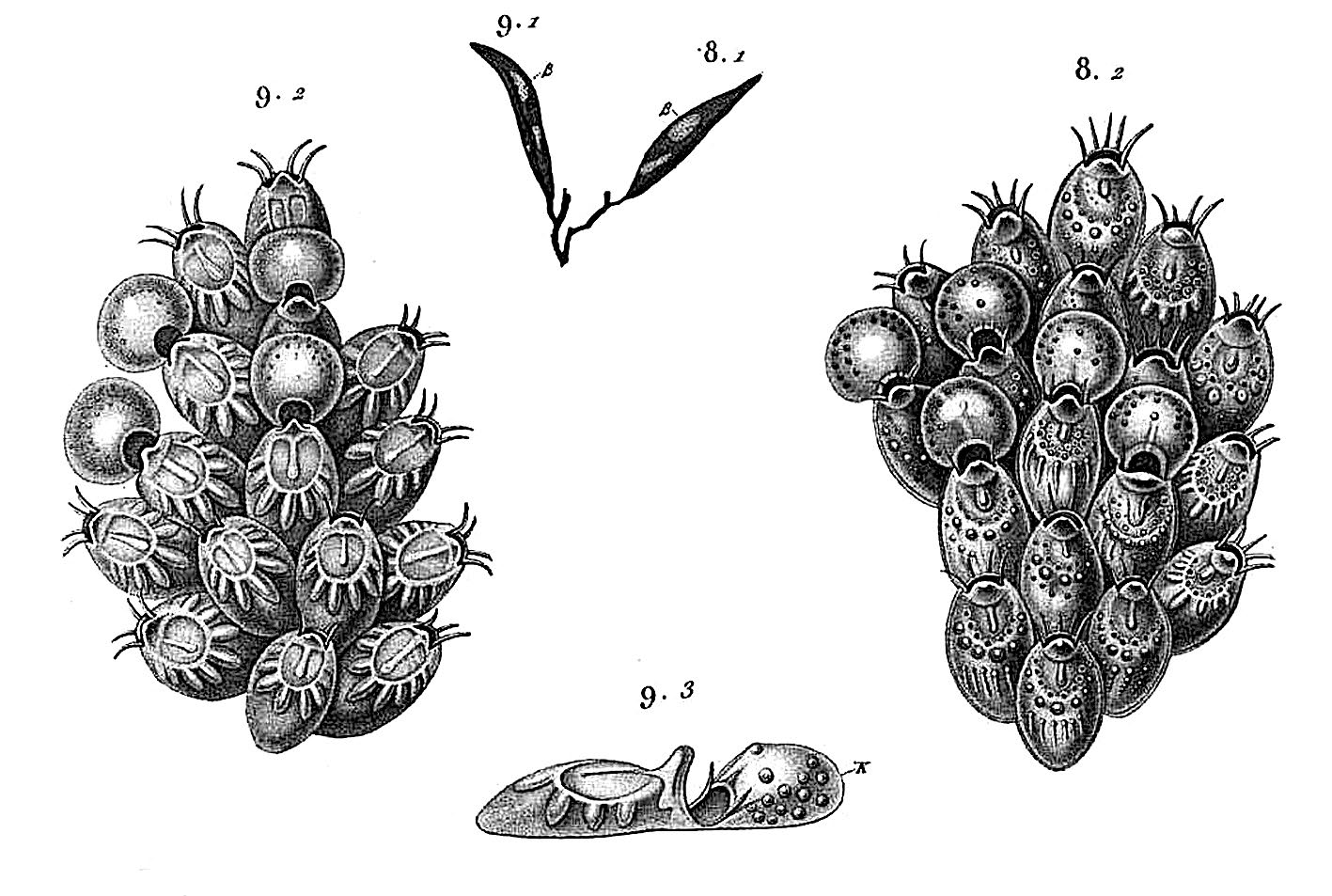
Unnamed drawings – Savigny 1817: pl. 9, figs 8.1, 8.2.
Not Lepralia cribrosa Heller, 1867:109 , pl. 2, fig. 6.
Lepralia cribrosa – Waters 1879: 36, pl. 9, fig. 4.
Collarina cribrosa Jullien 1886: 607 . — Gautier 1958a: 54; 1958b: 198. Cribrilina punctata View in CoL – Hincks 1886: 266, pl. 9, fig. 9.
Cribrilina cribrosa – Calvet 1902: 16. — Gautier 1952: 10. Puellina gattyae var. balearica Barroso, 1919: 340 , figs 1-5.
Cribrilina balzaci – Waters 1923: 561, pl. 18, fig. 8 (in part).
Collarina balzaci View in CoL – Gautier 1962: 107 (in part). — Prenant & Bobin 1966: 602 (in part), fig. 209. — Harmelin 1973: 676; 1978a: pl. 1, fig. 7 (in part). — Hayward 1974: 399, table 3 (in part). — Aristegui 1984: 229, fig. 49c; pl. 11, fig. 4-5. — Zabala 1986: 359, fig. 110. — Zabala & Maluquer 1988: 105, fig. 204. — Bishop 1988: 749, figs 1-6; 1994: 242 (in part). — Koçak et al. 2002: 237. — Moissette et al. 2007: 206, fig. 8b. — Moissette 2013: 192, fig. 13.6b. — Chimenz Gusso et al. 2014: 154 (part), figs 74ab. — Lepoint et al. 2014: 59, 61. — Harmelin et al. 2016: 423 (list).
Collarina View in CoL species B – Bishop 1986: fig. B.
? Flustra jauberti – Audouin 1826: 239 (unnamed drawings, Savigny 1817: pl. IX, figs 9.1, 9.2).
Not Collarina balzaci View in CoL – Harmelin 1978a: 189, fig. 8. — Bishop 1994: 242 (in part), figs 66-68. — Reverter et al. 1995: 125. — Fernández- Pulpeiro et al. 1996: 110. — Rosso 1996: 60. — Hayward & Ryland 1998: 318 (in part), fig. 112 a-b. — Hayward & McKinney 2002: 36, figs 16a-c. — De Blauwe 2006: 126 (list); 2009: 270, figs 282- 283; 2019: in press. — Souto et al. 2010: 1418 (list). — Chimenz Gusso et al. 2014: figs 74c-d. — WoRMS image http://www. marinespecies.org/aphia.php?p=image&tid=110891&pic=23688 (accessed on 26.IV.2018).
Not Cribrilina balzaci – Waters 1899: 9, figs 31-32. — Norman 1903: 98, pl. 9, fig. 6); 1909: 292.
TYPE LOCALITY. — Levant coast, eastern Mediterranean.
MATERIAL EXAMINED. — On Posidonia leaves. France. Marseille, Gulf of Prado (depth unknown), 22.I.1951, coll. by Y. V. Gautier, 1 colony, MNHN . — Marseille, Planier Islet , 43°11’54”N, 5°13’43”E, 7 m, 11.VI.2017, coll. by JGH, several colonies ( Fig. 3 View FIG ), MNHN, NHMUK GoogleMaps . — Port-Cros Island, Croix headland, 18 m, 18.V.1967, St. JGH- 67.12., 1 colony, MNHN . — Port-Cros Island, Tuf, 13 m, 8.II.1977, coll. by C. Eugène, several colonies including specimen BMNH 1987.1.4.1 illustrated by Bishop (1988, figs 3-6). — Spain. Catalonia, L’Estartit, Medes Islands, 42°2’43.05”N, 3°13’14.39”E, 8 m, VI.2017, coll. by TM-MZ, 1 colony ( SEM stub TM-643) GoogleMaps , MZB. — Balearic Islands, Mallorca, Punta Galinda , 18 m, 39°33’56”N, 2°20’36.5”E, VIII.2017, coll. by TM-MZ, 5 colonies, MZB. GoogleMaps — Tunisia. Kerkennah Islands, Ouled Ezzedine , c. 34°39’04”N, 11°06’31”E, 2-3 m, 26-27.VI.2018, coll. by JGH, hundreds of colonies, MNHN, NHMUK GoogleMaps .
On algae. Lebanon. N Lebanon El Heri ( Beaulieu ), 1-5 m, coll. by G. Bitar, 3.VI.2000: 1 coated colony ( SEM stub JGH- 276: Fig. 2 View FIG ); MNHN-IB-2014-189 : 2 colonies kept dry; MNHN-IB- 2014-221 : 3 colonies in alcohol. — Spain. Balearic Islands, Cabrera , Cap Llebeig , 39°9’45.19”N, 2°54’48.3”E, 5 m, coll. by TM-MZ, 1 colony on Cystoseira balearica , MZB. GoogleMaps — France. Corsica, Scandola , Cala di l’Oru, 42°22’18”N, 8°32’42”E, 18 m, 31.VIII.2018, 2 small colonies on Cystoseira montagnei , coll. by JGH, MNHN GoogleMaps .
On unidentified macrophyte. France. NW Mediterranean, Saint- Raphael, NHMUK ( SEM photos: MSJ): NHMUK.1975.7.18.20. — Italy. Rapallo, NHMUK.1911.10.1.704 . — Adriatic. NHMUK.1899.5.1.437 . — NHMUK.1899.5.1.718. — Greece. Mediterranean, Chios, NHMUK.1987.1.22.1: E .
ADDITIONAL RECORDS. — France, Marseille, Jarre Island , Plateau des Chèvres , 12 m, 4.II.1977, coll. by C. Eugène, 1 colony on Posidonia . — Marseille, Frioul archipelago, Tiboulen Pomègues , 12 m, 17.VI.1977, coll. by C. Eugène, 1 colony on Posidonia . — Spain, Canaries Is., 2 small colonies on Cystoseira sp., coll. by J. Aristegui ( Fig. 4 View FIG ) .
DIAGNOSIS. — Colonies generally small, early mature; autozooids with broad gymnocyst and small costate shield with short costae relatively flat except for a peripheral bump bearing a large pelma; adventitious avicularia directed laterally perpendicular to the zooid axis, with broad semicircular opesia and short rostrum with slightly serrated edges; apertural bar in the form of a thick triangular mucro with a rounded tip bearing two pelmatidia on the proximal side; orifice poster with a straight proximal edge and two small lateral notches; 3-4 oral spines; ovicell cleithral, ooecium kenozooidal; ancestrula with 5 or 6 spines.
DESCRIPTION
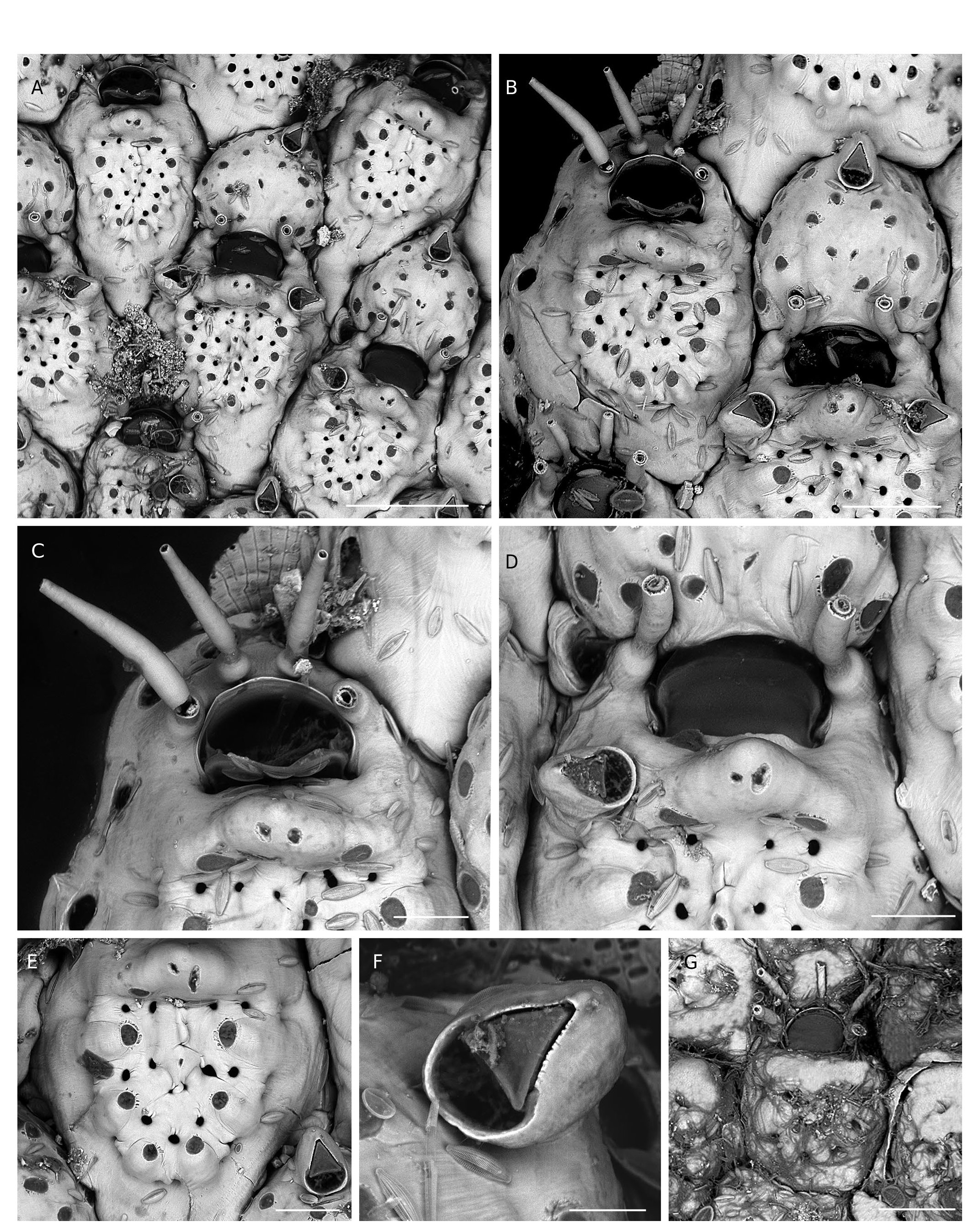
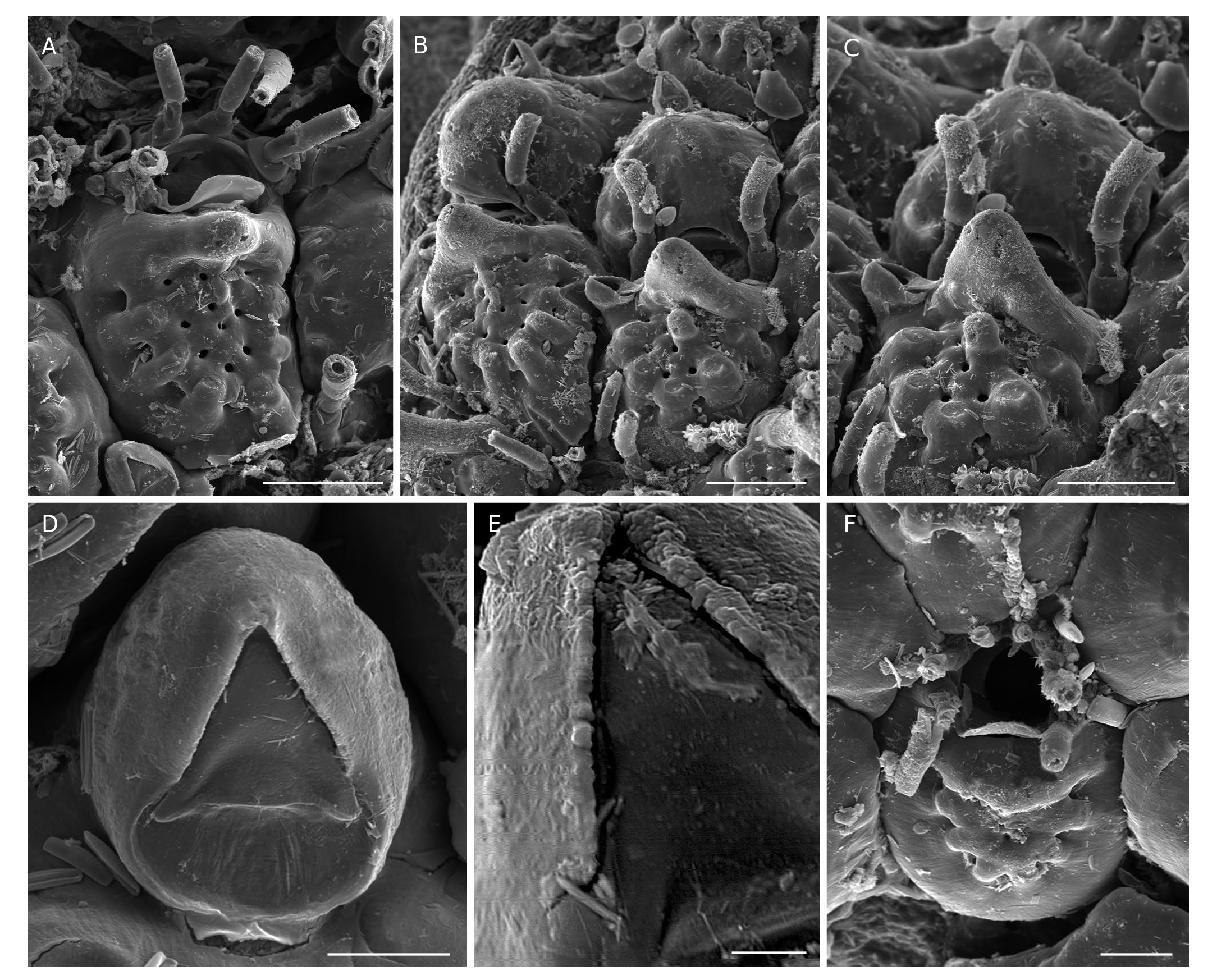
Colony encrusting, pluriserial, unilaminar, generally small (50- 100 zooids) but occasionally larger in certain environmental conditions (see below), early maturing, with rounded outline ( Fig. 2A View FIG ). Autozooids subhexagonal, longer than broad with proximal part triangular; gymnocyst forming a broad, convex border, particularly extended proximally, clearly visible in frontal view except distal to the orifice; costate shield oval to round, relatively flat except for a peripheral crown of low bumps marking the summit of the ascending basal part of each costa; 5-8 (mostly 6) short costae with a smooth surface, a large pelma on the top of each basal bump, 3-4 rounded intercostal lacunae including that between the steep basal portions of costae ( Figs 2C View FIG ; 3B, E View FIG ; 4A View FIG ). Thick apertural bar, roughly triangular, bulging, with 2 pairs of pseudopores on the proximal side of the bar: small pelmatidia near the apex of the mucro, and large pelmata near the base ( Figs 2C View FIG ; 3C, D View FIG ; 4B, C View FIG ; 7A View FIG ). Autozooidal orifice semicircular, wider than long, proximal edge straight with low lateral notches, anter forming a distinct frame ending proximally with slightly angular condyles, broader in ovicelled zooids ( Figs 2B View FIG ; 3C, D View FIG ; 7A View FIG ). Oral spines 3-4 with swollen base in non-ovicelled zooids, possibly more numerous in certain conditions (see below), 2 inwardly arched in ovicelled zooids ( Figs 2C View FIG ; 3D View FIG ; 4C View FIG ). Adventitious avicularia paired, single or both missing in the same colony, directed laterally on both sides of the apertural bar, subpedunculate with rounded proximal area, short triangular rostrum with strong condyles and slightly serrated edges, mandible in the shape of an equilateral triangle, and broad semicircular proximal opesia ( Figs 2D View FIG ; 3F View FIG ; 4D, E View FIG ). Ovicell cleithral, prominent, globose, terminal at the edge of the colony, ooecium formed by distal kenozooid, frequently bearing a distally-directed avicularium on the distal edge, ectooecium with slightly bumpy surface dotted with numerous pelmata ( Figs 2B, C View FIG ; 3B View FIG ; 4C View FIG ). Ancestrula cribrimorph with a 4-5 irregularly swollen costae and 5 or 6 oral spines ( Figs 2E View FIG ; 3G View FIG ; 4F View FIG ).
REMARKS
The species Flustra balzaci was created by Audouin (1826) without description for a specimen figured by Savigny (1817: pl. 9, fig. 8.1-2), collected during sampling surveys along the Levant coast (SE Mediterranean) during Napoleon Bonaparte’s expedition to Egypt. Unfortunately, the whole collection of bryozoans gathered during this expedition has disappeared (d’Hondt 2006). The confused history of this species and of the genus Collarina Jullien has been detailed and clarified by Bishop (1988, 1994). In short, after Audouin (1826), Waters (1899) was the first to use the name balzaci for a specimen from Madeira placed in the genus Cribrilina , considered herein to belong to a new Collarina species ( C. macaronensis Harmelin , n. sp., see below). Later, Gautier (1962) introduced the combination Collarina balzaci . The taxon Collarina balzaci was stabilized by Bishop (1988) with the designation of a neotype from Naples selected among specimens of Waters’ collection ( Waters 1879). Despite this, however, the species name C. balzaci was often attributed inappropriately with the assumption of high morphological plasticity. All specimens ascribed here to C. balzaci display the features stated above, including avicularia with broad semicircular opesia and rostrum with serrated edges ( Figs 2D View FIG ; 3F View FIG ; 4E View FIG ). This material is mostly Mediterranean, including the neotype ( Bishop 1988: figs 1-2) and specimens from Lebanon (MNHN-IB-2014-189, MNHN-IB-2014-221; Fig. 2 View FIG ), but also includes Atlantic colonies from the Canaries collected by J. Aristegui during his thesis work ( Fig. 4 View FIG ). The Lebanese material can be a valuable alternative to the neotype because of its geographical origin, similar to that of the specimen illustrated by Savigny (1817). Interestingly enough, he figured on the same plate (herewith Fig. 1 View FIG ) and on the same seaweed as Flustra balzaci another species with close features (pl. 9, fig. 9.1-2), which was named Flustra jaubertii by Audouin (1826). This species has been almost completely ignored, however, despite its clear cribrimorph features that might also be attributed to a Collarina species with well-calcified zooids (e.g. raised triangular suboral umbo with median extension, small spinocyst with bases of costae protruding widely on the gymnocyst, four oral spines). To our knowledge, this species was cited only by Waters (1879) who synonymized it doubtfully with Lepralia gattyae Landsborough, 1852 , and by d’Hondt (2006) who synonymized it with Cribrilaria radiata (Moll, 1803) .
CASE OF THE TUNISIAN MATERIAL FROM KERKENNAH ISLANDS
The recent finding by one of us (JGH, June 2018) of an extremely abundant population of C. balzaci epiphytic on Posidonia leaves at Kerkennah Islands (north of Gabes Gulf) revealed particular colonial and zooidal features. The collection consisted of some leaves taken at random from spots of
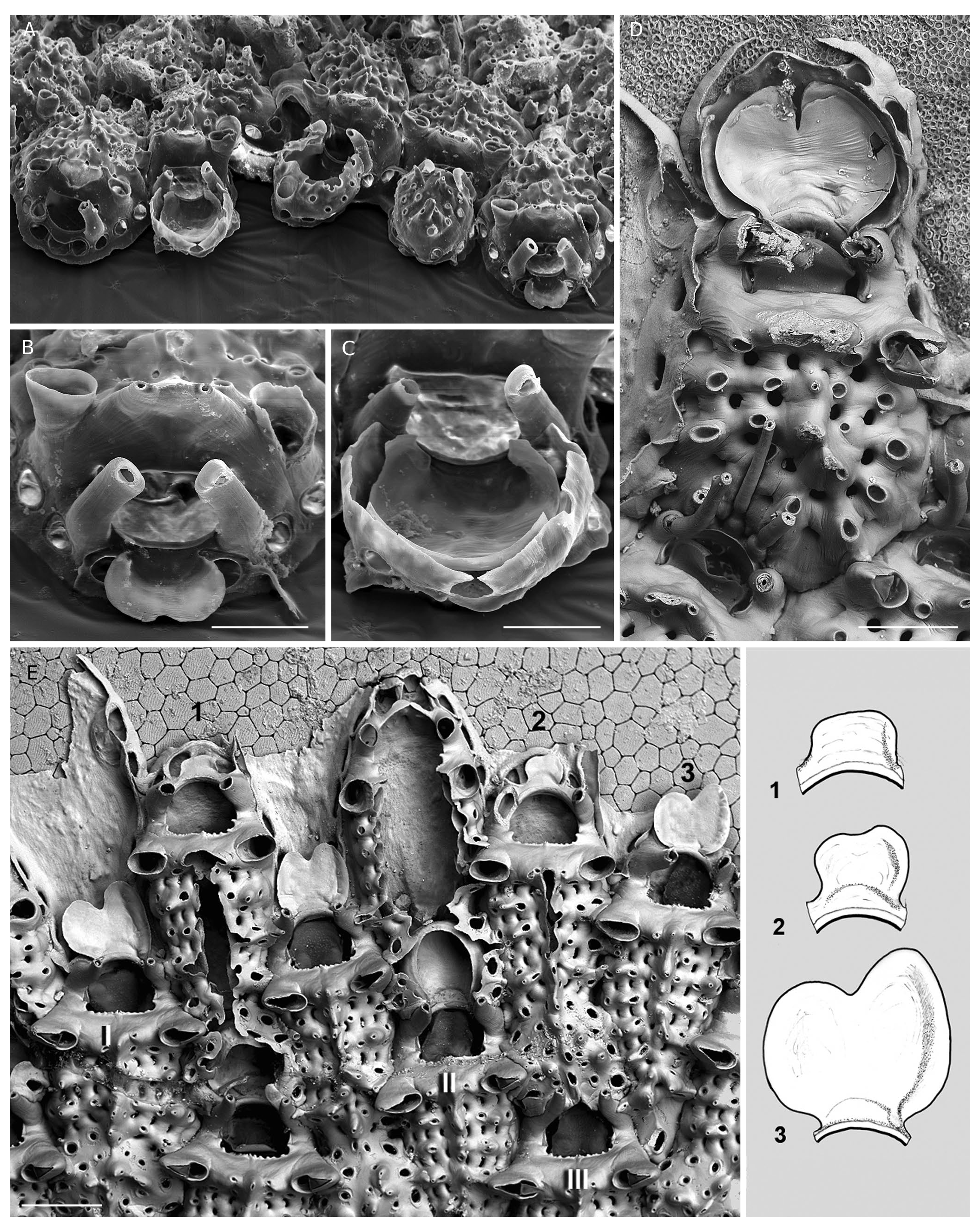
a fragmented Posidonia bed at shallow depth (2-3 m at high tide). All collected leaves were heavily colonized by C. balzaci (e.g. 89 colonies of various sizes counted on a 64 cm long leaf, predominantly on the shaded side). In contrast, other bryozoan species usually common on Posidonia leaves (e.g. Harmelin 1973; Lepoint et al. 2014) were rare or absent. Only three other species were present in the sample, with small and scarce colonies: Patinella radiata ( Audouin, 1826) , Electra posidoniae Gautier, 1954 , and Microporella sp. Preliminary investigations indicated that this population of C. balzaci presented other obvious peculiarities: large size of some colonies (> 150 zooids), autozooids also clearly larger, with more numerous oral spines, and “dirty” surfaces owing to adherent powdery deposits ( Fig. 19 View FIG ). A preliminary assessment indicated that the average length and width of non-ovicelled zooids were respectively 44% and 38% greater than the average values measured in the reference collection ( Table 1 View TABLE , LZo: 537 Μm vs 373 Μm, WZo: 322 Μm vs 233 Μm), and the commonest number of oral spines was 5 instead of 3-4, and could reach 6 or 7 in some colonies. Moreover, SEM examination of two colonies showed that zooids were densely covered with diatom frustules ( Fig. 19 View FIG ) despite ultrasonic cleaning. These atypical features are most likely related to particular local environmental conditions (see below).
GEOGRAPHIC AND HABITAT DISTRIBUTION
Collarina balzaci is present throughout the Mediterranean basin including the Adriatic. It is not endemic to the Mediterranean as noted by Koçak et al. (2002), but is also present in the near Atlantic. The only Atlantic material that has been correctly assigned to C. balzaci are the specimens from the Canaries recorded by Arístegui (1984: pl. 11, fig. 4-5). On the other hand, all other NE Atlantic specimens ascribed to C. balzaci that have been examined belong to two Collarina species described here: C. macaronensis Harmelin , n. sp. and C. gautieri Harmelin , n. sp. (see below).
As confirmed by the present reexamination of the morphological features of C. balzaci , all specimens that can be assigned to this species are epiphytic. In most cases, in the Mediterranean, they are associated to leaves of the seagrass Posidonia oceanica (L.) Delile ( Fig. 16A View FIG ). This condition was implicit in the redescription of C. balzaci by Bishop (1988) based on two epiphytic Mediterranean specimens: the neotype, from Naples, “on seaweed at slight depth” ( Waters 1879), and a specimen from Port-Cros Island on a Posidonia leaf (BMNH 1987.1.4.1, JGH leg.). It is worthwhile noting that the specimens collected at shallow depth in Lebanon ( Fig. 2 View FIG , MNHN-IB-2014-189, MNHN-IB-2014-221; Harmelin et al. 2016), where Posidonia beds are absent ( Pergent et al. 2012), encrusted seaweeds such as the specimen illustrated by Savigny (1817). As noted by Bishop (1988: 753), among the material from Chios ascribed to C. balzaci by Hayward (1974), a specimen epiphytic on red algae was correctly identified while another encrusting a stone was “a different, apparently unnamed Collarina species”, an assertion verified here (see below, Collarina sp.). On the other hand, C. balzaci was not present in samples of Posidonia leaves from Chios studied by Hayward (1975). On Posidonia leaves, C. balzaci is a characteristic component of an epiphytic assemblage including Electra posidoniae Gautier, 1954 , Fenestrulina joannae ( Calvet, 1902) and Puellina gattyae (Landsborough, 1852) ( Gautier 1962; Harmelin 1973; Eugène 1978; Fresi et al. 1982; Bal- duzzi et al. 1983; Koçak et al. 2002; Di Martino & Taylor 2014; Lepoint et al. 2014). This assemblage associated with Posidonia leaves was already present in the early Pleistocene at Rhodes ( Moissette et al. 2007, 2016; Moissette 2013). The small size of colonies with early sexual maturity (i.e., budding of ovicells soon after ancestrular settlement) and light calcification of walls are distinctive adaptations to life on Posidonia leaves, a substrate with major constraints (small size, pliability, life duration generally <1 year) ( Di Martino & Taylor 2014; Lepoint et al. 2014). In contrast, C. balzaci does not encrust the rhizomes of P. oceanica , which provide encrusters with a more rigid, perennial and shaded habitat. The occurrence of C. balzaci on other seagrasses than Posidonia leaves appears to be infrequent, at least in regions where it is present, but this potentiality should be better documented. It was recorded on another seagrass, Cymodocea nodosa (Ucria) Ascherson , in northern Croatia (fide sketch and photos by S. Bacrun, pers. com. to JGH, Nov. 2014). Its occurrence on seaweeds is attested first by Savigny’s drawings (1817: pl. 9, fig. 8) showing a specimen from the Levant coast (eastern Mediterranean) apparently encrusting pseudo-leaves of the brown seaweed Sargassum sp. Further records of C. balzaci on Sargassum in Sicily ( Gautier 1958a) and the Canary Islands ( Arístegui 1984) are worth noting. Other indications of the occurrence of C. balzaci on shallow-water seaweeds include a specimen on Cystoseira balearica Sauvageau at Cabrera Island (TM-MZ), and two colonies on Cystoseira montagnei J. Agarth (= C. spinosa Sauvageau ) at Scandola, Corsica (JGH.) which suggest that C. balzaci is frequent in this habitat (very small sample collected by chance). Some published records are not substantiated by figures or specimens in collections, for example on brown ( Gautier 1962), red ( Hayward 1974) and green algae ( Gautier 1958b). Its occurrence on Udotea petiolata (synonym of Flabellia petiolata (Turra) Nizamuddin ) noted by Gautier (1958b) was not confirmed by a recent study on bryozoans from the coralligenous of Port-Cros Island ( Harmelin 2017) in which assemblages on this alga were analysed. Mediterranean records of C. balzaci in non-epiphytic/epialgal conditions, such as on stones, shells, panels and other small substrates (e.g. Gautier 1962; Hayward 1974; Pisano & Boyer 1985; Rosso 1996) most likely correspond to other Collarina species (i.e., C. denticulata Harmelin , n. sp., C. gautieri Harmelin , n. sp. or Collarina sp.). In conclusion, pending a thorough revision of material from the whole Atlantic-Mediterranean region combining morphological and molecular criteria, we consider that C. balzaci is exclusively epiphytic/epialgal, and a better investigation of its occurrence on seaweeds in the NE Atlantic should provide a more realistic view of its distribution.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Collarina balzaci ( Audouin, 1826 )
| Harmelin, Jean-Georges, Bishop, John D. D., Madurell, Teresa, Souto, Javier, Spencer Jones, Mary E. & Zabala, Mikel 2019 |
Collarina balzaci
| SOUTO J. & GIL O. & PULPEIRO E. 2010: 1418 |
| DE BLAUWE H. 2006: 126 |
| HAYWARD P. J. & MCKINNEY F. K. 2002: 36 |
| HAYWARD P. J. & RYLAND J. S. 1998: 318 |
| PULPEIRO E. & GIL O. & BARCIA M. C. 1996: 110 |
| ROSSO A. 1996: 60 |
| REVERTER O. & D'HONDT J. - L. & FERNANDEZ E. 1995: 125 |
| BISHOP J. D. D. 1994: 242 |
| HARMELIN J. G. 1978: 189 |
Collarina balzaci
| HARMELIN J. G. & BITAR G. & ZIBROWIUS H. 2016: 423 |
| CHIMENZ GUSSO C. & NICOLETTI L. & BONDANESE C. 2014: 154 |
| LEPOINT G. & BALANCIER B. & GOBERT S. 2014: 59 |
| MOISSETTE P. & KOSKERIDOU E. & CORNEE J. J. & GUILLOCHEAU F. & LECUYER C. 2007: 206 |
| KOCAK F. & BALDUZZI A. & BENLI H. A. 2002: 237 |
| ZABALA M. & MALUQUER P. 1988: 105 |
| BISHOP J. D. D. 1988: 749 |
| ZABALA M. 1986: 359 |
| ARISTEGUI RUIZ J. 1984: 229 |
| HAYWARD P. J. 1974: 399 |
| HARMELIN J. G. 1973: 676 |
| PRENANT M. & BOBIN G. 1966: 602 |
| GAUTIER Y. V. 1962: 107 |
Cribrilina balzaci
| WATERS A. W. 1923: 561 |
Cribrilina cribrosa
| GAUTIER Y. V. 1952: 10 |
| BARROSO M. J. 1919: 340 |
| CALVET L. 1902: 16 |
Cribrilina balzaci
| NORMAN A. M. 1903: 98 |
| WATERS A. W. 1899: 9 |
Collarina cribrosa
| GAUTIER Y. V. 1958: 54 |
| GAUTIER Y. V. 1958: 198 |
| JULLIEN J. 1886: 607 |
| HINCKS T. 1886: 266 |
Lepralia cribrosa
| WATERS A. W. 1879: 36 |
Flustra balzaci
| AUDOUIN V. 1826: 239 |
Flustra jauberti
| AUDOUIN V. 1826: 239 |